Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
The COVID-19 pandemic challenged the medical field to rapidly identify and implement new approaches to the diagnosis, treatment and prevention of SARS-CoV-2 infections. The scientific community also needed to rapidly initiate basic, translational, clinical and epidemiological studies to understand the pathophysiology of this new family of viruses, which continues to evolve with the emergence of new genetic variants. In contrast to most other respiratory viruses, children developed less severe acute and post-acute disease compared to adults.
- pediatric COVID-19
- SARS-CoV-2 variants
- innate immunity
1. Introduction
One of the most striking observations from the beginning of the SARS-CoV-2 pandemic was the marked difference in disease severity in children compared to adults. Al-though infection rates were similar or even higher in children, most children were asymptomatic or had mild illness and rarely required hospitalization. In contrast, the morbidity and mortality in adults was substantial. Approximately, 15.6 million US children were infected with SARS-CoV-2 from March 2020 to May 2023, which accounts for ~17.9% of total cumulated cases; notably, children (<18 years) comprise ~22% of the total US population [1]. Thus, the age-related differences in disease severity are not attributable to reduced susceptibility but more likely reflect differences in host responses. Moreover, the rates of infection and clinical manifestations changed with the emergence of new variants that differed in tissue and cell tropism and the ability to elicit and evade host immune responses. Natural and vaccine-induced immunity, treatment strategies and changes in social distancing policies also impacted the clinical manifestations and host responses to new viral variants.
2. Clinical Manifestations of Acute COVID-19 in Children with Successive SARS-CoV-2 Waves
During the first wave of SARS-CoV-2 infection (March–November 2020), the ancestral virus originating from Wuhan, China was rapidly replaced by the first major variant of concern, the Alpha variant (lineage B.1.1.7), which carried multiple mutations including a major change in the spike protein (D614G) [2]. The most common symptoms reported in infants and children were fever (46.3%), cough (36.9%) and less frequently, dyspnea (6.5%) [3]. Gastrointestinal symptoms including diarrhea, vomiting and abdominal pain were also documented. Hospitalizations among children, however, were uncommon.
Clinical manifestations were similar during the second major wave through the fall of 2021, when the Delta variant (B.1.617.2) dominated. However, the Delta variant was associated with greater disease severity and increased hospitalization rates, possibly reflecting increased transmission, enhanced viral replication and immune escape [4]. Older children and adolescents with risk factors similar to those observed in adults including obesity progressed to acute respiratory distress syndrome (ARDS), although the incidence was low relative to what was observed among adults. The disease spectrum changed substantially with the emergence of Omicron variants (primarily B1.1.529) in December 2021 (Figure 1). More upper rather than lower respiratory tract diseases, including bronchiolitis and laryngotracheobronchitis (croup), were described with Omicron. Loss of taste, which was relatively uncommon in children compared to adults in the earlier waves, was almost never reported with Omicron [5][6][7]. A single-center study from New York reported an increase in the proportion of pediatric patients hospitalized with croup from 1.1% pre-Omicron to 6.6% during the Omicron wave [8]. Similar findings were observed using the US National COVID-19 Cohort Collaborative, which found that upper airway infection rates associated with SARS-CoV-2 disease increased from 1.5% to 4.1% from the pre-Omicron to Omicron period [9]. There was also an increase in febrile seizures associated with SARS-CoV-2 infections in the Omicron period, although the underlying reasons for this increase are unclear [10][11][12].
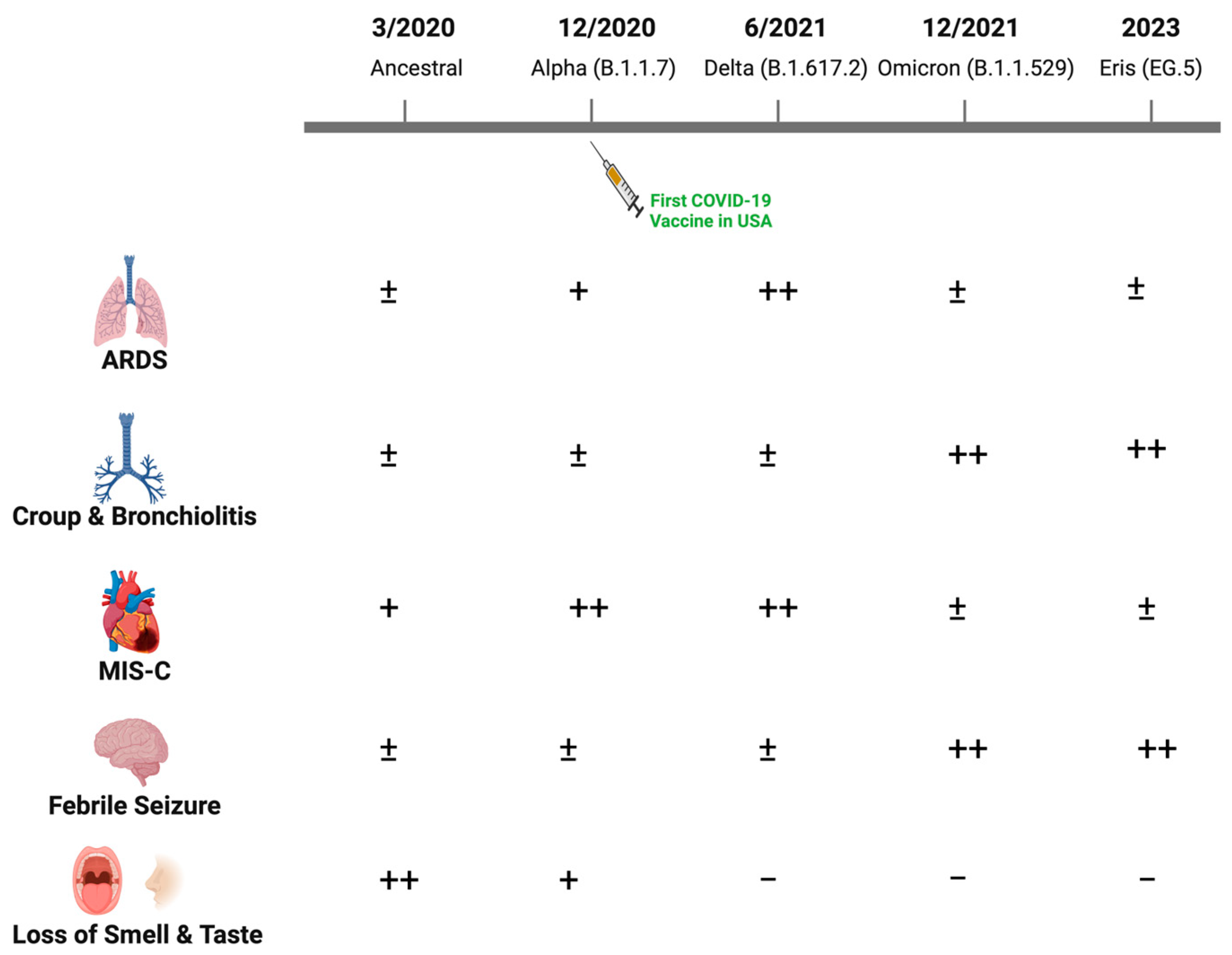
Figure 1. Changes in the clinical manifestations of SARS-CoV-2 in children with evolving viral variants. While most children infected with SARS-CoV-2 have been asymptomatic or developed self-limited fever with mild respiratory or gastrointestinal symptoms throughout the pandemic, the relative frequency of other clinical manifestations changed with emergence of new dominant viral variants as illustrated (−, ±, +, ++).
The emergence of Omicron variants resulted not only in changes in clinical manifestations but also in hospitalization patterns [12][13][14][15]. While older children and adolescents with comorbidities similar to those identified in adults including obesity and diabetes dominated pediatric hospitalizations pre-Omicron, this shifted to younger children with the emergence of Omicron variants. For example, the Center for Disease Control (CDC) surveillance data available from March 2020 to Feb 2022 found that hospitalization rates for children ≤4 years old were five times higher with the Omicron compared to the Delta surge [13]. Nearly half of these hospitalizations (n = 2562) were infants younger than 6 months of age and the majority had no underlying comorbidities [13]. Although admission rates were higher, length of stay and intensive care unit (ICU) admissions were lower. For example, Omicron was associated with significantly lower odds of moderate or severe disease compared to the Delta variant (adjusted odds ratio: 0.12) [16]. The clinical spectrum of disease with more recent Omicron subvariants, including XBB.1.16 (Arcturus) and E.G.5 (Eris), which have replaced the original Omicron variant, are not yet fully defined. However, early reports from India, where this variant emerged in April 2023, noted an increase in rates of infections in younger infants compared to older children (37.4% vs. 13.3%; p < 0.001). Mild respiratory symptoms and non-purulent conjunctivitis were the primary manifestations described in children [17].
The difference in symptoms and disease severity (greater with Delta), and the predilection for upper versus lower respiratory tract disease (more upper tract disease with Omicron variants) may be attributed, in part, to mutations in the spike protein that allow for differential tropism, entry and spread [18]. The SARS-CoV-2 spike protein is cleaved into two subunits, S1 and S2. The S1 subunit binds to angiotensin-converting enzyme-2 (ACE2), allowing for the exposure of the S2′ site. If the cell expresses transmembrane protease serine 2 (TMPRSS2) and the spike protein has affinity for this enzyme, the S2′ site is further cleaved at the plasma membrane, which promotes viral entry by direct fusion. If TMPRSS2 expression is limited and/or there is reduced affinity for the enzyme, the virus is internalized by endocytosis and S2′ is cleaved by cathepsin L within the endosomal compartment. Lower tract lung alveoli express higher levels of TMPRSS2, whereas cells in the upper respiratory tract express higher levels of cathepsin [19]. Moreover, Omicron variant spike proteins are less efficiently cleaved by TMPRSS2 compared with those of the Delta variant. This results in greater dependence on entry via endocytosis and reduced ability to infect lung cells, as evidenced by studies with pseudotype viruses [20]. These differences in cell tropism and entry mechanisms likely contribute to the differences in disease manifestations, with Delta causing more lung disease, whereas Omicron and its variants exhibit more upper respiratory tract disease. The TMPRSS2-mediated membrane fusion also promotes syncytia formation, which is associated with more severe lower tract disease. This phenotype was most pronounced with the Delta variant in which the P681R mutation in the spike protein enhanced fusogenicity and likely contributed to the increased pathogenicity [21].
3. What Makes the Acute and Post-Acute Response to SARS-CoV-2 Different in Children?
The pre-Omicron variants (Alpha and Delta) were associated with more severe acute disease and higher rates of post-acute sequelae compared to Omicron in both adults and children. However, across all waves, children consistently exhibited a milder clinical course than adults. The milder disease course cannot be attributed to differences in rates of infection, expression of ACE2 or TMPRSS2, viral loads or cross-reactivating antibodies to other coronaviruses [22][23][24] (Table 1). Rather, studies suggest that children mount a more vigorous innate response, which protects against severe disease. In a study comparing children and adults hospitalized with SARS-CoV-2 pre-Omicron and prior to the introduction of vaccines, serum levels of IL-17A and IFN-γ were higher in children versus adults and correlated significantly and inversely with age [23]. The source of these cytokines was not likely the peripheral blood because adults had more robust spike-specific T cell responses; this led to the speculation that local innate responses may differ [23]. This hypothesis was supported by a subsequent study where the transcriptional profile of nasopharyngeal cells (bulk RNA sequencing) and quantification of mucosal cytokines and antibodies in nasopharyngeal swabs obtained from children or adults with SARS-CoV-2 were compared [22]. SARS-CoV-2 RNA copies, ACE2 and TMPRSS2 gene expression were similar in children and adults, but higher expression of genes associated with IFN signaling, NLRP3 inflammasome and other innate pathways were detected in the children compared to the adults. Consistent with the RNA data, protein levels of IFN-α2, IFN-γ, IP-10, IL-8 and IL-1β proteins were also higher in the nasal fluid of children versus adults, but anti-spike IgA and IgG were detected at similar levels in the nasal fluid of both groups. The notion that children mount a stronger local innate response was further supported by a study that used single-cell profiling of nasal, airway and blood samples from pediatric and adult patients. Interferon pathways were activated in SARS-CoV-2 uninfected healthy children compared to adults and this was further increased in those with SARS-CoV-2 infection. Conversely, adults with COVID-19 exhibited a greater peripheral blood cytotoxic T cell response.
Table 1. Age-associated host features and immune responses to SARS-CoV-2 that may contribute to disease severity.
| Host Feature | Children versus Adults |
|---|---|
| Rates of infection and initial SARS-CoV-2 RNA copies | No differences |
| ACE-2 and TMPRSS2 expression | No differences |
| Innate responses nasal mucosa ↑expression IFN signaling, NLRP3 inflammasome transcripts |
Increased in children |
| Systemic cytokine inflammatory response | Increased in adults |
| SARS-CoV-2 systemic neutralizing antibodies | No differences |
| Activated T cell responses | Increased in adults |
In another study, clinically mild acute SARS-CoV-2 infection in children was associated with an increase in activated neutrophils but lower levels of circulating monocytes, dendritic cells and natural killer cells compared to blood samples obtained from adults with acute COVID-19. The authors speculated that the lower levels of these immune cells in the circulation may reflect their recruitment to mucosal sites to bolster the innate response [25]. These and other observations suggest that children may be primed at mucosal sites to respond to SARS-CoV-2, possibly because of more frequent respiratory infections. Another possible basis for the more robust pediatric innate response is increased expression of TLR2 by innate immune cells in children compared to adults, which may promote more rapid activation of interferon pathways [26].
This enhanced innate response may protect children from the vigorous systemic inflammatory response that has been shown to contribute to ARDS and other systemic disease manifestations observed in adults in the pre-Omicron era [26][27]. Importantly, this priming does not appear to interfere with the generation of SARS-CoV-2 antibody or memory T cell responses. A relatively large household study found that children had higher specific antibody levels which persisted for longer (96.2% versus 82.9% remained seropositive 11–12 months post-infection), despite being much more likely to be asymptomatic. Notably, symptomatic and asymptomatic infections induced similar antibody responses and there were no differences in the neutralization titers between adults and children [28][29]. A study on the immune cell profile of children compared to adults recovering from SARS-CoV-2 infection further supports the notion that immune activation contributes to the adverse outcomes. Adults had higher levels of activated and senescent cells, whereas children had higher T and B regulatory cell levels [30]. This may explain why children have less severe clinical manifestations attributable to immune activation and cytokine storm. A more recent study that took advantage of tonsillar and adenoidal tissue available from children with documented prior SARS-CoV-2 infection identified viral specific class-switched and somatically hypermutated B cells and expanded T cell clonotypes in the upper respiratory tract tissue [31]. These findings further support the notion that the innate response that protects children from severe disease does not prevent the development of a robust adaptive immune response.
This entry is adapted from the peer-reviewed paper 10.3390/jcm13010128
References
- Centers for Disease Control and Prevention-COVID Data Tracker. Available online: https://covid.cdc.gov/covid-data-tracker/#datatracker-home (accessed on 6 September 2023).
- Korber, B.; Fischer, W.M.; Gnanakaran, S.; Yoon, H.; Theiler, J.; Abfalterer, W.; Hengartner, N.; Giorgi, E.E.; Bhattacharya, T.; Foley, B.; et al. Tracking Changes in SARS-CoV-2 Spike: Evidence that D614G Increases Infectivity of the COVID-19 Virus. Cell 2020, 182, 812–827.e19.
- Stokes, E.K.; Zambrano, L.D.; Anderson, K.N.; Marder, E.P.; Raz, K.M.; El Burai Felix, S.; Tie, Y.; Fullerton, K.E. Coronavirus Disease 2019 Case Surveillance—United States, January 22–May 30, 2020. Morb. Mortal. Wkly. Rep. 2020, 69, 759–765.
- Wanga, V.; Gerdes, M.E.; Shi, D.S.; Choudhary, R.; Dulski, T.M.; Hsu, S.; Idubor, O.I.; Webber, B.J.; Wendel, A.M.; Agathis, N.T.; et al. Characteristics and Clinical Outcomes of Children and Adolescents Aged <18 Years Hospitalized with COVID-19—Six Hospitals, United States, July–August 2021. Morb. Mortal. Wkly. Rep. 2021, 70, 1766–1772.
- Erdede, O.; Sarı, E.; Uygur Külcü, N.; Uyur Yalçın, E.; Sezer Yamanel, R.G. An overview of smell and taste problems in paediatric COVID-19 patients. Acta Paediatr. 2020, 109, 2184–2186.
- Jelic, M.; Silveira, L.; Lang, S.; Curran-Hays, S.; Boyer, S.; Carter, B.; Choi, Y.J.; Fresia, J.; Maeda, L.C.; Nerguizian, D.; et al. Changing Characteristics of Children with COVID-19 in Colorado Admitted during Different Variant Periods. Pediatr. Infect. Dis. J. 2023, 42, 679–684.
- Sumner, M.W.; Xie, J.; Zemek, R.; Winston, K.; Freire, G.; Burstein, B.; Kam, A.; Emsley, J.; Gravel, J.; Porter, R.; et al. Comparison of Symptoms Associated with SARS-CoV-2 Variants among Children in Canada. JAMA Netw. Open 2023, 6, e232328.
- Narayanan, N.; Langer, S.; Acker, K.P.; Rosenblatt, S.D.; Simmons, W.; Wu, A.; Han, J.Y.; Abramson, E.L.; Grinspan, Z.M.; Levine, D.A. COVID-19 is Observed in Older Children during the Omicron Wave in New York City. J. Emerg. Med. 2023, 64, 195–199.
- Martin, B.; DeWitt, P.E.; Russell, S.; Sanchez-Pinto, L.N.; Haendel, M.A.; Moffitt, R.; Bennett, T.D. Acute Upper Airway Disease in Children with the Omicron (B.1.1.529) Variant of SARS-CoV-2—A Report From the US National COVID Cohort Collaborative. JAMA Pediatr. 2022, 176, 819–821.
- Iijima, H.; Kubota, M.; Ogimi, C. Change in Seizure Incidence in Febrile Children with COVID-19 in the Era of Omicron Variant of Concern. J. Pediatr. Infect. Dis. Soc. 2022, 11, 514–517.
- Choi, S.H.; Choi, J.H.; Lee, J.K.; Eun, B.W.; Song, S.H.; Ahn, B.; Kim, Y.K.; Yun, K.W. Clinical Characteristics and Outcomes of Children with SARS-CoV-2 Infection during the Delta and Omicron Variant-Dominant Periods in Korea. J. Korean Med. Sci. 2023, 38, e65.
- Han, M.S.; Kim, K.M.; Oh, K.J.; Chang, J.Y.; Lee, S.Y.; Choi, J.E.; Shin, S.M.; Sun, J. Distinct Clinical and Laboratory Features of COVID-19 in Children during the Pre-Delta, Delta and Omicron Wave. Pediatr. Infect. Dis. J. 2023, 42, 423–428.
- Marks, K.J.; Whitaker, M.; Agathis, N.T.; Anglin, O.; Milucky, J.; Patel, K.; Pham, H.; Kirley, P.D.; Kawasaki, B.; Meek, J.; et al. Hospitalization of Infants and Children Aged 0–4 Years with Laboratory-Confirmed COVID-19—COVID-NET, 14 States, March 2020–February 2022. Morb. Mortal. Wkly. Rep. 2022, 71, 429–436.
- Shoji, K.; Akiyama, T.; Tsuzuki, S.; Matsunaga, N.; Asai, Y.; Suzuki, S.; Iwamoto, N.; Funaki, T.; Ohmagari, N. Clinical characteristics of COVID-19 in hospitalized children during the Omicron variant predominant period. J. Infect. Chemother. 2022, 28, 1531–1535.
- Shi, D.S.; Whitaker, M.; Marks, K.J.; Anglin, O.; Milucky, J.; Patel, K.; Pham, H.; Chai, S.J.; Kawasaki, B.; Meek, J.; et al. Hospitalizations of Children Aged 5–11 Years with Laboratory-Confirmed COVID-19—COVID-NET, 14 States, March 2020–February 2022. Morb. Mortal. Wkly. Rep. 2022, 71, 574–581.
- Butt, A.A.; Dargham, S.R.; Loka, S.; Shaik, R.M.; Chemaitelly, H.; Tang, P.; Hasan, M.R.; Coyle, P.V.; Yassine, H.M.; Al-Khatib, H.A.; et al. Coronavirus Disease 2019 Disease Severity in Children Infected with the Omicron Variant. Clin. Infect. Dis. 2022, 75, e361–e367.
- Vashishtha, V.M.; Kumar, P. Conjunctival Involvement in Infants as an Unusual Symptom of Omicron XBB.1.16 Driven Surge. Indian Pediatr. 2023, 60, 861–862.
- Li, C.; Huang, J.; Yu, Y.; Wan, Z.; Chiu, M.C.; Liu, X.; Zhang, S.; Cai, J.P.; Chu, H.; Li, G.; et al. Human airway and nasal organoids reveal escalating replicative fitness of SARS-CoV-2 emerging variants. Proc. Natl. Acad. Sci. USA 2023, 120, e2300376120.
- Gili, R.; Burioni, R. SARS-CoV-2 before and after Omicron: Two different viruses and two different diseases? J. Transl. Med. 2023, 21, 251.
- Meng, B.; Abdullahi, A.; Ferreira, I.; Goonawardane, N.; Saito, A.; Kimura, I.; Yamasoba, D.; Gerber, P.P.; Fatihi, S.; Rathore, S.; et al. Altered TMPRSS2 usage by SARS-CoV-2 Omicron impacts infectivity and fusogenicity. Nature 2022, 603, 706–714.
- Saito, A.; Irie, T.; Suzuki, R.; Maemura, T.; Nasser, H.; Uriu, K.; Kosugi, Y.; Shirakawa, K.; Sadamasu, K.; Kimura, I.; et al. Enhanced fusogenicity and pathogenicity of SARS-CoV-2 Delta P681R mutation. Nature 2022, 602, 300–306.
- Pierce, C.A.; Sy, S.; Galen, B.; Goldstein, D.Y.; Orner, E.; Keller, M.J.; Herold, K.C.; Herold, B.C. Natural mucosal barriers and COVID-19 in children. JCI Insight 2021, 6, e148694.
- Pierce, C.A.; Preston-Hurlburt, P.; Dai, Y.; Aschner, C.B.; Cheshenko, N.; Galen, B.; Garforth, S.J.; Herrera, N.G.; Jangra, R.K.; Morano, N.C.; et al. Immune responses to SARS-CoV-2 infection in hospitalized pediatric and adult patients. Sci. Transl. Med. 2020, 12, eabd5487.
- McLean, H.Q.; Grijalva, C.G.; Hanson, K.E.; Zhu, Y.; Deyoe, J.E.; Meece, J.K.; Halasa, N.B.; Chappell, J.D.; Mellis, A.M.; Reed, C.; et al. Household Transmission and Clinical Features of SARS-CoV-2 Infections. Pediatrics 2022, 149, e2021054178.
- Neeland, M.R.; Bannister, S.; Clifford, V.; Dohle, K.; Mulholland, K.; Sutton, P.; Curtis, N.; Steer, A.C.; Burgner, D.P.; Crawford, N.W.; et al. Innate cell profiles during the acute and convalescent phase of SARS-CoV-2 infection in children. Nat. Commun. 2021, 12, 1084.
- Loske, J.; Rohmel, J.; Lukassen, S.; Stricker, S.; Magalhaes, V.G.; Liebig, J.; Chua, R.L.; Thurmann, L.; Messingschlager, M.; Seegebarth, A.; et al. Pre-activated antiviral innate immunity in the upper airways controls early SARS-CoV-2 infection in children. Nat. Biotechnol. 2022, 40, 319–324.
- Yoshida, M.; Worlock, K.B.; Huang, N.; Lindeboom, R.G.H.; Butler, C.R.; Kumasaka, N.; Dominguez Conde, C.; Mamanova, L.; Bolt, L.; Richardson, L.; et al. Local and systemic responses to SARS-CoV-2 infection in children and adults. Nature 2022, 602, 321–327.
- Renk, H.; Dulovic, A.; Seidel, A.; Becker, M.; Fabricius, D.; Zernickel, M.; Junker, D.; Gross, R.; Muller, J.; Hilger, A.; et al. Robust and durable serological response following pediatric SARS-CoV-2 infection. Nat. Commun. 2022, 13, 128.
- Garrido, C.; Hurst, J.H.; Lorang, C.G.; Aquino, J.N.; Rodriguez, J.; Pfeiffer, T.S.; Singh, T.; Semmes, E.C.; Lugo, D.J.; Rotta, A.T.; et al. Asymptomatic or mild symptomatic SARS-CoV-2 infection elicits durable neutralizing antibody responses in children and adolescents. JCI Insight 2021, 6, e150909.
- Petrara, M.R.; Bonfante, F.; Costenaro, P.; Cantarutti, A.; Carmona, F.; Ruffoni, E.; Di Chiara, C.; Zanchetta, M.; Barzon, L.; Donà, D.; et al. Asymptomatic and Mild SARS-CoV-2 Infections Elicit Lower Immune Activation and Higher Specific Neutralizing Antibodies in Children Than in Adults. Front. Immunol. 2021, 12, 741796.
- Xu, Q.; Milanez-Almeida, P.; Martins, A.J.; Radtke, A.J.; Hoehn, K.B.; Oguz, C.; Chen, J.; Liu, C.; Tang, J.; Grubbs, G.; et al. Adaptive immune responses to SARS-CoV-2 persist in the pharyngeal lymphoid tissue of children. Nat. Immunol. 2023, 24, 186–199.
This entry is offline, you can click here to edit this entry!
