The World Health Organization 2021 (WHO) manual defines the evaluation of sperm parameters, such as sperm count, viability, motility, morphology, and seminal fluid composition as the principal approach for semen quality assessment, as these are crucial to male fertility [
23,
24]. HPV infection has been linked in several studies to a decrease in progressive sperm motility and changes in immotile sperm rate, sperm morphology and concentration, although these correlations remain controversial [
8,
14,
25,
26]. The first work to report HPV-related alterations in sperm motility was conducted in 1997 by Lai et al. [
27], who observed a lower curvilinear and straight-line velocity and a reduced mean amplitude of lateral head displacement of hrHPV-infected sperm cells (
Table 1).
Table 1. Summary of the HPV-related effects on sperm.
| Type of Infection |
Effect |
Reference |
| HPV positivity |
Increased risk of DFI > 30%, asthenozoospermia, ASAs production, and negative ART outcome (alteration in fertilization, implantation, and development of the embryo). |
[28,29,30] |
| hrHPV genotype |
Reduced sperm count and motility alterations. |
[27,31] |
Multiple HPV
infections |
Hypospermia, abnormal viscosity, and increased seminal pH. |
[24] |
| HPV16 or HPV31 |
Sperm genomic DNA breaks and increased apoptotic events. |
[32] |
HPV16 and
HPV18 |
Exonic modification of p53 gene. |
[33] |
In the following years, some studies confirmed these observations [
18,
32], whilst others [
34,
35] rejected these hypotheses, arguing that there is no association between HPV semen infection and impaired semen function. The detection of HPV-DNA in semen would therefore only be a secondary effect of the desquamation of penile-HPV-infected keratinocytes [
36]. Damke et al. [
24] conducted a study which demonstrated a correlation between HPV semen infection, mostly with multiple HPV types, and the detection of hypospermia, abnormal viscosity, and increased seminal pH (
Table 1). In contrast, several works [
25,
34] found no clear relationship between HPV infection and alteration in seminal volume or concentration. Such disagreement could be related to the different sample sizes or methodologies used in the various studies. For instance, Damke et al. [
24] based their study on 229 semen samples, whereas Rintala et al. [
34] based theirs on 65 semen samples. From a methodological point of view, both studies based the semen analysis on the WHO criteria, but they used different genotyping techniques. Rintala et al. [
34] used hybridization with digoxigenin-labeled high-risk HPV oligoprobes, identifying 12 genotypes; Damke et al. [
24] used PCR-RFLP, which allowed the identification of a higher number of genotypes (39 genotypes). The lower number of samples and genotypes analyzed by Rintala et al. [
34] compared to Damke et al. [
24] may explain the lack of correlation found by the latter.
In terms of semen composition, semen generally has a pH of around 7.72 and must contain appropriate concentrations of citrate, potassium, sodium, calcium, magnesium, zinc, glucose, fructose, albumin, and proteins [
37]. Since the volume of seminal vesicles and prostate secretions affects the concentration of sperm in the ejaculate, it also affects fertility and pregnancy rates [
23]. Particularly, seminal vesicles produce fructose, the main sugar involved in the metabolic processes and motility of sperm, which has also a fundamental role in zinc chelation, fertilization, and sperm chromatin condensation. Meanwhile, the prostate gland is bound to the production of zinc and citric acid. The latter, whose levels are regulated by testosterone, is thought to be the main ligand of zinc, a key element in the regulation of sperm motility, germ cell maintenance, and spermatogenesis progression [
38]. It has been suggested that in HPV-infected males, zinc production is not enough to condense sperm, which may be correlated with male infertility [
10]. Indeed, Damke et al. [
24] showed that men with HPV-positive semen may have altered proportions of prostate and seminal vesicle secretions, both associated with glandular dysfunction, which would have negative effects on fertility. Moreover, the detection in a 2013 study [
39] of HPV-DNA in both epithelial and non-epithelial semen cells, as well as in semen leukocytes, supported the idea that HPV does not exclusively infect sperm cells. It can be concluded that the heterogeneous results regarding the relationship between seminal HPV-positivity and sperm quality could be due to the different targets of infection [
40].
3. Correlation between HPV and Sperm DNA Fragmentation
Sperm DNA fragmentation (SDF) is a form of sperm nucleic acid damage that occurs before or after ejaculation and consists of double-stranded or single-stranded breaks [
41]. At present, three main mechanisms are considered responsible for sperm DNA damage, namely impaired sperm chromatin maturation, unnatural sperm cell apoptosis, and oxidative stress [
42,
43].
The delivery of an undamaged paternal genome from the spermatozoa to the egg cell is essential for the correct development of the embryo [
43,
44]. Thus, there is a correlation between sperm DNA damage and impaired fertilization, lower embryo quality, decreased pregnancy rates, and high rates of pregnancy loss following in vitro fertilization (IVF) [
41,
45,
46,
47,
48].
In light of this evidence, for the first time, the latest edition of the WHO laboratory manual for the examination/processing of human semen has included methods for evaluating the DNA fragmentation index (DFI) as an indicator of the measure of sperm DNA integrity [
23]. Based on the data available in the literature, a DFI > 30% is considered predictive of infertility, while a value below 20% is considered physiological, although the WHO guidelines do not specify cut-off values [
10,
49]. Several techniques are considered suitable for assessing SDF, including terminal deoxynucleotidyl transferase (dUTP) nick-end-labeling (TUNEL) assay, single cell gel electrophoresis (Comet) assay, acridine orange flow cytometry (AO FCM) assay, and sperm chromatin dispersion (SCD) test (
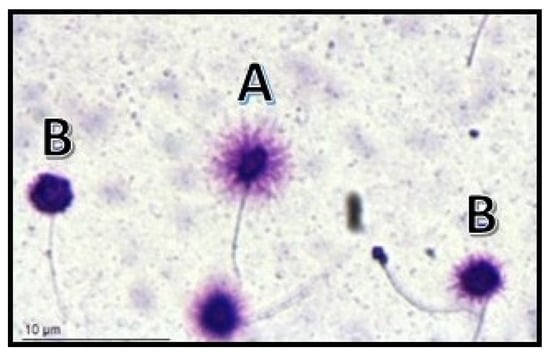
Figure 1) [
23].
Figure 1. Visualization of sperm DNA fragmentation using sperm chromatin dispersion test (SCD). (A) Normal sperms. (B) Sperms with fragmented DNA.
To the best of our knowledge, the integrity of sperm DNA is constantly affected by endogenous and exogenous elements. For example, double-strand breaks are physiologically induced during spermatogenesis to facilitate meiotic crossover and during spermiogenesis to facilitate histone-protamine replacement [
49,
50]. Nonetheless, other factors that are not always physiological, such as lifestyle (e.g., alcohol, smoking, drugs, diet), exposure to pollutants, diseases, aging, and infections can increase levels of SDF [
8,
43]. Specifically, infections of the genital tract in males lead to leukocytospermia and elevate oxidative stress in semen, resulting in DNA breakage in sperm [
43]. Several studies have described the disruptive effect of infection with certain pathogens on sperm DNA integrity. These include mycoplasma, Chlamydia trachomatis, hepatitis B virus, and HPV, although for the latter, the literature’s data are not in agreement with each other [
32,
51,
52]. Connelly and colleagues [
32] showed that DNA fragmentation driven by fragments of the E6–E7 genes of HPV16 or 31 caused genomic DNA breaks and increased sperm apoptosis (
Table 1). This finding was subsequently confirmed by Lee et al. [
33], who demonstrated exonic modification of the p53 gene by the E6–E7 DNA fragments of HPV16 and 18, and Moreno-Sepulveda and Rajmil [
28], who showed an increased risk of DFI > 30% in patients with HPV seminal infection (
Table 1).
Despite this, some studies in the current literature have failed to find an association between a DFI value greater than 30% and the detection of HPV DNA in semen [
17,
53]. Furthermore, some years later, a correlation between the expression of an isoform of the E6 protein and the enhancement of oxidative stress-induced cellular DNA damage was highlighted [
54]. In this context, Kato et al. [
55] reported higher levels of superoxide dismutase (SOD), an antioxidant enzyme, in the seminal plasma of HPV-positive men compared to HPV-negative ones, suggesting the possible involvement of reactive oxygen species (ROS) in sperm DNA damage. ROS are physiologically produced in the cell microenvironment, and their correct production is crucial for different stages of fertilization [
56]. However, the overproduction and imbalance of ROS causes oxidative stress, which is highly damaging to proteins, lipids, and DNA [
55,
56]. When the sperm cell fails to counteract effectively such effects, damaged DNA could promote HPV DNA integration [
54,
56]. Oxidative stress can be favored by genital tract infections, which induce an increase in seminal fluid leukocytes, capable of producing ROS [
57]. Recently, it has been hypothesized to monitor the effect of various antioxidants based on SDF, but further studies are needed to propose it as a potential solution [
56].