Metallic glasses can be a perfect alternative to conventional crystalline biomaterials (such as 316L stainless steel, Ti or Ti-based alloys, Zr or Zr-based alloys, Co-Cr alloys, etc.) when used as coatings for surgical devices and implants inside the human body. Owing to their outstanding electrocatalytic activity and durability, metallic glasses can be considered prominent candidates for energy-storage and -conversion devices, such as fuel and electrolysis cells, and batteries. Metallic-glass systems are gaining substantial momentum in the micro- and nano-imprinting of optoelectronic devices.
- metallic glass
- alloys
- glass
1. Biomedical Applications
1.1. Antibacterial Application
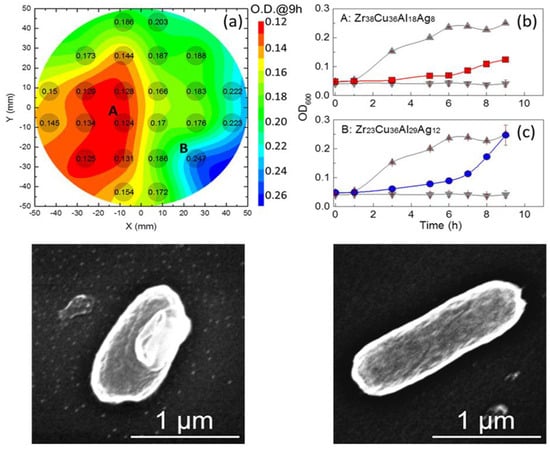
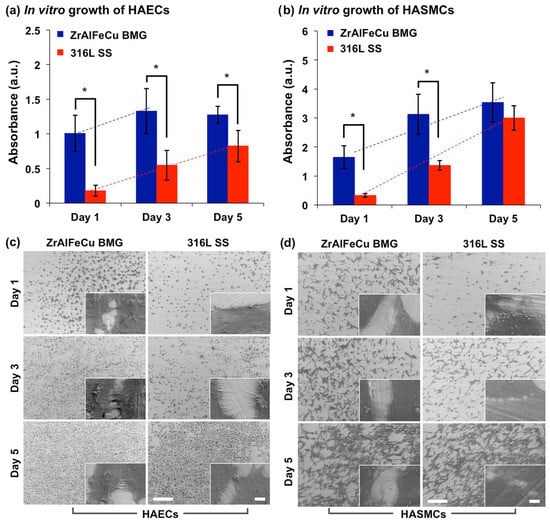
1.2. Bio-Implants
2. Electrochemical Devices
3. Optoelectronic Devices
4. Aerospace Application
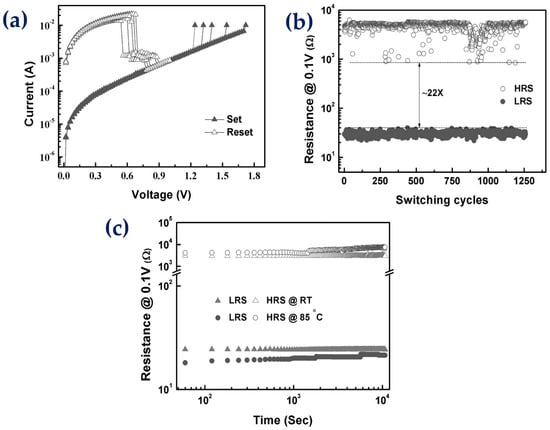
5. Memory Storage Devices
This entry is adapted from the peer-reviewed paper 10.3390/coatings13101689
References
- Etiemble, A.; Loughian, C.D.; Apreutesei, M.; Langlois, C.; Cardinal, S.; Pelletier, J.M.; Pierson, J.-F.; Steyer, P. Innovative Zr-Cu-Ag thin film metallic glass deposed by magnetron PVD sputtering for antibacterial applications. J. Alloys Compd. 2017, 707, 155–161.
- Chu, J.P.; Jang, J.S.C.; Huang, J.C.; Chou, H.S.; Yang, Y.; Ye, J.C.; Wang, Y.C.; Lee, J.W.; Liu, F.X.; Liaw, P.K.; et al. Thin film metallic glasses: Unique properties and potential applications. Thin Solid Films 2012, 520, 5097–5122.
- Chang, C.M.; Yang, C.J.; Wang, K.-K.; Liu, J.-K.; Hsu, J.H.; Huang, J.C. On the reflectivity and antibacterial/antifungal responses of Al-Ni-Y optical thin film metallic glass composites. Surf. Coat. Technol. 2017, 327, 75–82.
- Lin, B.; Mu, R.; Yang, L.; Bian, X. Antibacterial effect of metallic glasses. Chin. Sci. Bull. 2012, 57, 1069–1072.
- Chu, J.P.; Liu, T.-Y.; Li, C.-L.; Wang, C.-H.; Jang, J.S.C.; Chen, M.-J.; Chang, S.-H.; Huang, W.-C. Fabrication and characterizations of thin film metallic glasses: Antibacterial property and durability study for medical application. Thin Solid Film. 2014, 561, 102–107.
- Liu, Y.; Padmanabhan, J.; Cheung, B.; Liu, J.; Chen, Z.; Scanley, B.E.; Wesolowski, D.; Pressley, M.; Broadbridge, C.C.; Altman, S.; et al. Combinatorial development of antibacterial Zr-Cu-Al-Ag thin film metallic glasses. Sci. Rep. 2016, 6, 26950.
- Jabed, A.; Khan, M.M.; Camiller, J.; Greenlee-Wacker, M.; Haider, W.; Shabib, I. Property optimization of Zr-Ti-X (X = Ag, Al) metallic glass via combinatorial development aimed at prospective biomedical application. Surf. Coat. Technol. 2019, 372, 278–287.
- Donlan, R.M. Biofilms: Microbial life on surfaces. Emerg. Infect. Dis. 2002, 8, 881–890.
- Chu, Y.Y.Y.; Lin, Y.S.S.; Chang, C.M.M.; Liu, J.-K.K.; Chen, C.H.H.; Huang, J.C.C. Promising antimicrobial capability of thin film metallic glasses. Mater. Sci. Eng. C 2014, 36, 221–225.
- Bouala, G.I.N.; Etiemble, A.; Der Loughian, C.; Langlois, C.; Pierson, J.-F.; Steyer, P. Silver influence on the antibacterial activity of multi-functional Zr-Cu based thin film metallic glasses. Surf. Coat. Technol. 2018, 343, 108–114.
- Lemire, J.A.; Harrison, J.J.; Turner, R.J. Antimicrobial activity of metals: Mechanisms, molecular targets and applications. Nat. Rev. Microbiol. 2013, 11, 371–384.
- Chiang, P.T.; Chen, G.J.; Jian, S.R.; Shih, Y.H.; Jang, J.S.C.; Lai, C.H. Surface antimicrobial effects of Zr61Al7.5Ni10Cu17.5Si4thin film metallic glasses on Escherichia coli, Staphylococcus aureus, Pseudomonas aeruginosa, Acinetobacter baumannii and Candida albicans. Fooyin J. Health Sci. 2010, 2, 12–20.
- Tsai, P.H.H.; Lin, Y.Z.; Li, J.B.; Jian, S.R.; Jang, J.S.C.; Li, C.; Chu, J.P.; Huang, J.C. Sharpness improvement of surgical blade by means of ZrCuAlAgSi metallic glass and metallic glass thin film coating. Intermetallics 2012, 31, 127–131.
- Li, H.F.; Zheng, Y.F. Recent advances in bulk metallic glasses for biomedical applications. Acta Biomater. 2016, 36, 1–20.
- Calin, M.; Gebert, A.; Ghinea, A.C.; Gostin, P.F.; Abdi, S.; Mickel, C.; Eckert, J.H. Designing biocompatible Ti-based metallic glasses for implant applications. Mater. Sci. Eng. C 2013, 33, 875–883.
- Niinomi, M.; Nakai, M.; Hieda, J. Development of new metallic alloys for biomedical applications. Acta Biomater. 2012, 8, 3888–3903.
- Huiskes, R.I.K.; Weinans, H.; Van Rietbergen, B. The Relationship Between Stress Shielding and Bone Resorption Around Total Hip Stems and the Effects of Flexible Materials. Clin. Orthop. Relat. Res. 1992, 274, 124–134.
- Wang, J.G.; Choi, B.W.; Nieh, T.G.; Liu, C.T. Crystallization and nanoindentation behavior of a bulk Zr–Al–Ti–Cu–Ni amorphous alloy. J. Mater. Res. 2000, 15, 798–807.
- Liu, L.; Qiu, C.L.; Huang, C.Y.; Yu, Y.; Huang, H.; Zhang, S.M. Biocompatibility of Ni-free Zr-based bulk metallic glasses. Intermetallics 2009, 17, 235–240.
- Jabed, A.; Rahman, Z.U.; Khan, M.M.; Haider, W.; Shabib, I. Combinatorial Development and In Vitro Characterization of the Quaternary Zr–Ti–X–Y (X–Y = Cu–Ag/Co–Ni) Metallic Glass for Prospective Bioimplants. Adv. Eng. Mater. 2019, 21, 1900726.
- Hua, N.; Huang, L.; Wang, J.; Cao, Y.; He, W.; Pang, S.; Zhang, T. Corrosion behavior and in vitro biocompatibility of Zr–Al–Co–Ag bulk metallic glasses: An experimental case study. J. Non-Cryst. Solids 2012, 358, 1599–1604.
- Wang, Y.B.; Zheng, Y.F.; Wei, S.C.; Li, M. In vitro study on Zr-based bulk metallic glasses as potential biomaterials. J. Biomed. Mater. Res. Part B Appl. Biomater. 2011, 96B, 34–46.
- Khan, M.M.; Deen, K.M.; Haider, W. Combinatorial development and assessment of a Zr-based metallic glass for prospective biomedical applications. J. Non-Cryst. Solids 2019, 523, 119544.
- Zohdi, H.; Shahverdi, H.R.; Hadavi, S.M.M. Effect of Nb addition on corrosion behavior of Fe-based metallic glasses in Ringer’s solution for biomedical applications. Electrochem. Commun. 2011, 13, 840–843.
- Li, S.; Wei, Q.; Li, Q.; Jiang, B.; Chen, Y.; Sun, Y. Development of Fe-based bulk metallic glasses as potential biomaterials. Mater. Sci. Eng. C 2015, 52, 235–241.
- Wang, Y.B.; Li, H.F.; Zheng, Y.F.; Li, M. Corrosion performances in simulated body fluids and cytotoxicity evaluation of Fe-based bulk metallic glasses. Mater. Sci. Eng. C 2012, 32, 599–606.
- Wang, Y.B.; Li, H.F.; Cheng, Y.; Zheng, Y.F.; Ruan, L.Q. In vitro and in vivo studies on Ti-based bulk metallic glass as potential dental implant material. Mater. Sci. Eng. C 2013, 33, 3489–3497.
- Oak, J.-J.; Hwang, G.-W.; Park, Y.-H.; Kimura, H.; Yoon, S.-Y.; Inoue, A. Characterization of Surface Properties, Osteoblast Cell Culture in Vitro and Processing with Flow-Viscosity of Ni-Free Ti-Based Bulk Metallic Glass for Biomaterials. J. Biomech. Sci. Eng. 2009, 4, 384–391.
- Xie, K.-F.; Yao, K.-F.; Huang, T.-Y. A Ti-based bulk glassy alloy with high strength and good glass forming ability. Intermetallics 2010, 18, 1837–1841.
- Huang, L.; Cao, Z.; Meyer, H.M.; Liaw, P.K.; Garlea, E.; Dunlap, J.R.; Zhang, T.; He, W. Responses of bone-forming cells on pre-immersed Zr-based bulk metallic glasses: Effects of composition and roughness. Acta Biomater. 2011, 7, 395–405.
- Huang, L.; Yokoyama, Y.; Wu, W.; Liaw, P.K.; Pang, S.; Inoue, A.; Zhang, T.; He, W. Ni-free Zr-Cu-Al-Nb-Pd bulk metallic glasses with different Zr/Cu ratios for biomedical applications. J. Biomed. Mater. Res. Part B Appl. Biomater. 2012, 100B, 1472–1482.
- He, W.; Chuang, A.; Cao, Z.; Liaw, P.K. Biocompatibility Study of Zirconium-Based Bulk Metallic Glasses for Orthopedic Applications. Metall. Mater. Trans. A 2010, 41, 1726–1734.
- Blanquer, A.; Pellicer, E.; Hynowska, A.; Barrios, L.; Ibáñez, E.; Baró, M.D.; Sort, J.; Nogués, C. In vitro biocompatibility assessment of Ti40Cu38Zr10Pd12 bulk metallic glass. J. Mater. Sci. Mater. Med. 2014, 25, 163–172.
- Huang, X.B.; Lin, N.M.; Hang, R.Q.; Tang, B.; Qiao, J.W. Bio-Properties of Zr-Based BMGMC as Potential Hard Tissue Implants. Mater. Sci. Forum 2013, 745–746, 754–760.
- Liu, L.; Qiu, C.L.L.; Chen, Q.; Chan, K.C.C.; Zhang, S.M.M. Deformation behavior, corrosion resistance, and cytotoxicity of Ni-free Zr-based bulk metallic glasses. J. Biomed. Mater. Res. Part A 2008, 86A, 160–169.
- Sun, Y.; Huang, Y.; Fan, H.; Wang, Y.; Ning, Z.; Liu, F.; Feng, D.; Jin, X.; Shen, J.; Sun, J.; et al. In vitro and in vivo biocompatibility of an Ag-bearing Zr-based bulk metallic glass for potential medical use. J. Non-Cryst. Solids 2015, 419, 82–91.
- Li, H.F.; Zheng, Y.F.; Xu, F.; Jiang, J.Z. In vitro investigation of novel Ni free Zr-based bulk metallic glasses as potential biomaterials. Mater. Lett. 2012, 75, 74–76.
- Li, J.; Shi, L.-L.; Zhu, Z.; He, Q.; Ai, H.; Xu, J. Zr61Ti2Cu25Al12 metallic glass for potential use in dental implants: Biocompatibility assessment by in vitro cellular responses. Mater. Sci. Eng. C 2013, 33, 2113–2121.
- Huang, L.; Pu, C.; Fisher, R.K.; Mountain, D.J.H.; Gao, Y.; Liaw, P.K.; Zhang, W.; He, W. A Zr-based bulk metallic glass for future stent applications: Materials properties, finite element modeling, and in vitro human vascular cell response. Acta Biomater. 2015, 25, 356–368.
- Qiu, C.L.; Chen, Q.; Liu, L.; Chan, K.C.; Zhou, J.X.; Chen, P.P.; Zhang, S.M. A novel Ni-free Zr-based bulk metallic glass with enhanced plasticity and good biocompatibility. Scr. Mater. 2006, 55, 605–608.
- Ramya, M.; Sarwat, S.G.; Udhayabanu, V.; Subramanian, S.; Raj, B.; Ravi, K.R. Role of partially amorphous structure and alloying elements on the corrosion behavior of Mg–Zn–Ca bulk metallic glass for biomedical applications. Mater. Des. 2015, 86, 829–835.
- Gu, X.; Shiflet, G.J.; Guo, F.Q.; Poon, S.J. Mg–Ca–Zn Bulk Metallic Glasses with High Strength and Significant Ductility. J. Mater. Res. 2005, 20, 1935–1938.
- Zberg, B.; Uggowitzer, P.J.; Löffler, J.F. MgZnCa glasses without clinically observable hydrogen evolution for biodegradable implants. Nat. Mater. 2009, 8, 887–891.
- Gu, X.; Zheng, Y.; Zhong, S.; Xi, T.; Wang, J.; Wang, W. Corrosion of, and cellular responses to Mg-Zn-Ca bulk metallic glasses. Biomaterials 2009, 31, 1093–1103.
- Khan, M.M.; Rahman, Z.U.; Deen, K.M.; Shabib, I.; Haider, W. Sputtered Mg100-xZnx (0 ≤ x ≤ 100) systems as anode materials for a biodegradable battery aimed for transient bioelectronics. Electrochim. Acta 2020, 329, 135129.
- Cao, J.D.; Kirkland, N.T.; Laws, K.J.; Birbilis, N.; Ferry, M. Ca–Mg–Zn bulk metallic glasses as bioresorbable metals. Acta Biomater. 2012, 8, 2375–2383.
- Senkov, O.N.; Miracle, D.B.; Keppens, V.; Liaw, P.K. Development and Characterization of Low-Density Ca-Based Bulk Metallic Glasses: An Overview. Metall. Mater. Trans. A 2008, 39, 1888–1900.
- Zhao, K.; Li, J.F.; Zhao, D.Q.; Pan, M.X.; Wang, W.H. Degradable Sr-based bulk metallic glasses. Scr. Mater. 2009, 61, 1091–1094.
- Jiao, W.; Zhao, K.; Xi, X.K.; Zhao, D.Q.; Pan, M.X.; Wang, W.H. Zinc-based bulk metallic glasses. J. Non-Cryst. Solids 2010, 356, 1867–1870.
- Inoue, A.; Nishiyama, N. New Bulk Metallic Glasses for Applications as Magnetic-Sensing, Chemical, and Structural Materials. MRS Bull. 2007, 32, 651–658.
- Kim, S.C.; Yamaura, S.-I.; Shimizu, Y.; Nakashima, K.; Igarashi, T.; Makino, A.; Inoue, A. Production of Ni65Cr15P16B4 metallic glass-coated bipolar plate for fuel cell by high velocity oxy-fuel (HVOF) spray coating method. Mater. Trans. 2010, 51, 1609–1613.
- Chen, Z.; Yang, Y.; Kumar, S.; Lu, G. First-Principles Prediction of Oxygen Reduction Activity on Pd−Cu−Si Metallic Glasses. J. Phys. Chem. C 2014, 118, 48.
- Tian, R.; Qin, Z. Bulk metallic glass Zr55Cu30Al10Ni5 bipolar plates for proton exchange membrane fuel cell. Energy Convers. Manag. 2014, 86, 927–932.
- Jayalakshmi, S.; Vasantha, V.S.; Fleury, E.; Gupta, M. Characteristics of Ni–Nb-based metallic amorphous alloys for hydrogen-related energy applications. Appl. Energy 2012, 90, 94–99.
- Tkachov, V.I. Problems of hydrogen degradation of metals. Mater. Sci. 2000, 36, 481–488.
- Kirchheim, R. Hydrogen solubility and diffusivity in defective and amorphous metals. Prog. Mater. Sci. 1988, 32, 261–325.
- Huang, J.C.; Chu, J.P.; Jang, J.S.C. Recent progress in metallic glasses in Taiwan. Intermetallics 2009, 17, 973–987.
- Diyatmika, W.; Chu, J.P.; Kacha, B.T.; Yu, C.-C.C.; Lee, C.-M.M. Thin film metallic glasses in optoelectronic, magnetic, and electronic applications: A recent update. Curr. Opin. Solid State Mater. Sci. 2015, 19, 95–106.
- Kim, H.; Gilmore, C.M.; Piqué, A.; Horwitz, J.S.; Mattoussi, H.; Murata, H.; Kafafi, Z.H.; Chrisey, D.B. Electrical, optical, and structural properties of indium–tin–oxide thin films for organic light-emitting devices. J. Appl. Phys. 1999, 86, 6451.
- Hartnagel, H.L.; Dawar, A.L.; Jain, A.K. Semiconducting Transparent Thin Films. MRS Bull. 1997, 22, 66–69.
- Lee, C.J.; Lin, H.K.; Sun, S.Y.; Huang, J.C. Characteristic difference between ITO/ZrCu and ITO/Ag bi-layer films as transparent electrodes deposited on PET substrate. Appl. Surf. Sci. 2010, 257, 239–243.
- Wang, W.H.; Hsu, J.H.; Huang, J.C. Optical reflectivity improvement by upgrading metallic glass film quality. Appl. Phys. Lett. 2013, 103, 161906.
- Hu, T.T.; Hsu, J.H.; Huang, J.C.; Kuan, S.Y.; Lee, C.J.; Nieh, T.G. Correlation between reflectivity and resistivity in multi-component metallic systems. Appl. Phys. Lett. 2012, 101, 011902.
- Seddon, A.B. Chalcogenide glasses: A review of their preparation, properties and applications. J. Non-Cryst. Solids 1995, 184, 44–50.
- Axinte, E. Metallic glasses from ‘alchemy’ to pure science: Present and future of design, processing and applications of glassy metals. Mater. Des. 2012, 35, 518–556.
- Burgess, T.; Ferry, M. Nanoindentation of metallic glasses. Mater. Today 2009, 12, 24–32.
- Henao, J.; Conctustell, A.; Cano, I.G.; Dosta, S.; Cinca, N.; Guilemany, J.M.; Suhonen, T. Novel Al-based metallic glass coatings by Cold Gas Spray. Mater. Des. 2016, 94, 253–261.
- Telford, M. The case for bulk metallic glass. Mater. Today 2004, 7, 36–43.
- Peker, A.; Johnson, W.L. A highly processable metallic glass: Zr41.2Ti13.8Cu12.5Ni10.0Be22.5. Appl. Phys. Lett. 1993, 63, 2342–2344.
- Kim, C.H.; Jang, Y.H.; Hwang, H.J.; Sun, Z.H.; Moon, H.B.; Cho, J.H. Observation of bistable resistance memory switching in CuO thin films. Appl. Phys. Lett. 2009, 94, 102107.
- Sun, B.; Liu, Y.X.; Liu, L.F.; Xu, N.; Wang, Y.; Liu, X.Y.; Han, R.Q.; Kang, J.F. Highly uniform resistive switching characteristics of TiN/ZrO2/Pt memory devices. J. Appl. Phys. 2009, 105, 061630.
- Hong, S.; Long, D.X.; Hwang, I.; Kim, J.-S.; Park, Y.C.; Kang, S.-O.; Park, B.H. Unipolar resistive switching mechanism speculated from irreversible low resistance state of Cu2O films. Appl. Phys. Lett. 2011, 99, 052105.
- Kim, K.M.; Choi, B.J.; Koo, B.W.; Choi, S.; Jeong, D.S.; Hwang, C.S. Resistive switching in Pt/Al2O3/TiO2/Ru stacked structures. Electrochem. Solid-State Lett. 2006, 9, 343–346.
- Tulu, B.; Chang, W.Z.; Chu, J.P.; Wang, S.F. Forming-free resistive switching characteristics of 15 nm-thick multicomponent oxide. Appl. Phys. Lett. 2013, 103, 252904.
