Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Prostate cancer (PCa) remains a significant global health concern, being a major cause of cancer morbidity and mortality worldwide. Furthermore, profound understanding of the disease is needed. Prostate inflammation caused by external or genetic factors is a central player in prostate carcinogenesis.
- prostate cancer
- inflammation
- immune response
- leukocytes
1. Neutrophils
Neutrophils, originating from hematopoietic stem cells, are among the first immune cells recruited after an insult. They possess a short lifespan to prevent excessive tissue damage, owing to their high plasticity and robust effector response [1]. When recruited to a damaged area, neutrophils release proteases, including neutrophil elastase, neutrophil extracellular traps (NETs), and reactive oxygen species (ROS), which exacerbate damage and contribute to the development of chronic inflammation [2]. Under normal circumstances, neutrophils can shift their function towards immunosuppression, thus regulating the production of pro-inflammatory mediators. However, in disease states, this shift may not occur correctly, leading to the development of carcinogenesis [3]. Therefore, neutrophils serve as a crucial link between inflammation and cancer. A study has observed a correlation between low neutrophil counts and a positive PCa biopsy, while elevated neutrophil counts may indicate a benign prostate biopsy [4]. These results can predict the progression from an acute response, characterized by increased neutrophil levels, to a carcinogenic phenotype dominated by chronic inflammation [5]. Tumour associated neutrophils (TANs) have been reported in cancer-affected regions. TANs, along with regular neutrophils, secrete substantial amounts of matrix metalloproteinase (MMP)-9, which play a role in the degradation of the extracellular matrix and cancer progression [6].
TANs are a complex population in the tumour microenvironment, associated with poor outcomes in some PCa studies [7] and demonstrating antitumoral effects in others [8]. In vitro assays showed that coculture of human PCa cells in the presence of neutrophils leads to a reduction of cell growth via caspase activation [9]. These findings suggest that, as tumours progress, neutrophil cytotoxicity diminishes, allowing PCa to avoid neutrophil cytotoxic effects. Studies have linked neutrophils as crucial cells in PCa prevention. In bone metastatic PCa, there is an increased formation of neutrophils and NETs to limit the spread of infection and control metastasis [9]. The role of different inflammatory mediators produced by neutrophils and its role in cancer progression is summarized on Table 1.
Table 1. Involvement of different leukocytes and their associated cytokines in cancer and prostate cancer progression.
| Leukocyte | Inflammatory Mediator Produced |
Effect | Refs. |
|---|---|---|---|
| Neutrophils | MMP-9 | MMP-9 produced by TANs and neutrophils degrade ECM leading to cancer progression in human xenografts and Mmp9-knockout mice | [6] |
| GM-CSF and CXCL8 | KRAS stimulated the expression of GM-CSF and CXCL8 in neutrophils which modulates the tumour microenvironment towards cancer progression in mouse models of ovarian cancer | [10] | |
| IL-8/CXCR2 | Overexpression of CXCR2 in neutrophils promotes their attachment in lung cancer regions in a K-RAS mutant mouse model of lung cancer | [11] | |
| NDE, ROS, RNE | NDE, ROS, and RNE release from neutrophils lead to hMSH2-dependent G2/M checkpoint arrest and for the presence of replication errors in a co-culture model that mimics intestinal inflammation in ulcerative colitis | [12] | |
| ARG-1 | Release of ARG-1 from neutrophils inhibit CD3-mediated T cell activation and proliferation leading to cancer progression in classic Hodgkin Lymphoma patients | [13] | |
| NET | Increased neutrophil and NET formation intended attenuate the rate of metastatic PCa in bones in vitro and an in vivo mouse model | [14] | |
| IL-1 | IL-1RA from neutrophils leads to inhibition of senescence promoting cancer progression | [15] | |
| TNF-α | TNFR1 KO mice with depletion of TNF receptor 1 developed smaller tumours with attenuated proliferation and absence of metastasis | [16] | |
| Cathepsin G | Cathepsin G release from neutrophils increases cancer cell adhesion, and aggregation, and metastasis in breast cancer cells | [17] | |
| Basophils | IL-4 | Basophils from pancreatic ductal adenocarcinomas secrete IL-4 which induce GATA-3 expression in Th2 cells in patient samples and Mcpt8Cre mice | [18] |
| CCL3 | Basophils express CCL3 to negatively regulate the normal hematopoietic process in MCPT8-DTR mice and bone marrow samples from patients with CML | [19] | |
| CCL3/CCL4 | Basophil plays a role in tumour rejection by increasing CD8+ T cell infiltration promoted by CCL3 and CCL4 in HCmel12-, B16-, and 616-OVA-induced transgenic FOXP3.LuciDTR-4 mice melanoma | [20] | |
| VEGFA | Immunologic activation by VEGF-2 of basophils induced the release of VEGF-A which induce basophil chemotaxis | [21] | |
| HGF | HGF is expressed in CML basophils in KU812-induced CML cell line | [22] | |
| ANGPT | Basophils express ANGPT1 and ANGPT2 mRNAs | [23] | |
| Eosinophils | IL-2 | IL-2 activate Tregs and Th17 cells involved in the promotion of cancer in a mouse model of PCa, and fibrosarcoma and head and neck human cancer tissues | [24] |
| IL-4 | IL-4 production promotes tumour growth and interaction with TAMs in a pancreatic-induced cancer mouse model | [25] | |
| IL-6 | Increase of IL-6 correlated in patients with metastatic PCa compared with localized PCa | [26] | |
| IL-5 and CCL17 | Eosinophils increase after Sipuleucel-T treatment of patients with metastatic castration-resistant PCa correlated with increase of IL-5 and CCL17, survival and maximal T-cell proliferation responses | [27] | |
| IFN-γ | IFN-γ induced CD4+ T cells to eliminate MHC II-negative cancer cells | [28] | |
| TNF-α | TNF-α correlated with increased extension of PCa in samples from PCa patients | [26] | |
| TGF-α | Overexpression of TGF-α decreased latency, increased growth, and tumour size of bladder cancer rat model | [29] | |
| VEGF | VEGF associated with poor prognosis of human small-cell lung carcinoma | [30] | |
| GM-CSF | Expression of GM-CSF correlated with NF-κB activation in bone-metastatic tumour tissues from individuals with metastatic breast cancer | [31] | |
| Mast cells | Chymase | Chymase released from human mast cell release latent TGF-β-binding protein from the matrix | [32] |
| Histamine | Histamine inhibition from mast cells inactivate EMT and cholangiocarcinoma growth via inhibition of c-Kit signalling in Mz-ChA-1-induced cholangiocarcinoma mouse model and human Mz-ChA-1 cells | [33] | |
| TNF-α | TNF-α released from mast cells amplifies and activates the functionality of CD8+ dendritic cells in Mcpt5-CreTNFfl/fl mice | [34] | |
| IL-1β | Overexpression of IL-1β promoted tumour invasiveness and metastasis by inducing the expression of angiogenic genes and growth factors | [35] | |
| IL1, IL-4, IL-6 | Decreased cell growth and participates in tumour rejection in breast cancer cells | [36] | |
| IL-8, IL-10 | Mast cell-derived IL-8 and IL-10 act as tumour suppressors contributing to tumour cell growth | [37][38] | |
| PGD2 | PGD2 secretion from mast cells attenuates angiogenesis in a Lewis lung carcinoma mouse model | [39] | |
| Macrophages | IL-1β | IL-1β induced Snail stabilization in Snail/MCF7 cells and this effect was dependent on cell types and IL-1β concentration | [40] |
| IL-8 | IL-8 produced by macrophages induce EMT in hepatocellular carcinoma samples via JAK2/STAT3/Snail pathway | [41] | |
| TNF-α | TNF-α induces the stabilization of Snail in a non-phosphorylated, functional form and thus enhances cell migration and invasion dependent on NF-κB activation | [40] | |
| TGF-β | TGF-β induced EMT phenotypes in A549 cells, including changes in cell morphology and induction of mesenchymal marker expression in part by NF-κB signalling | [42] | |
| MMP-2, MMP-9 | Macrophage-derived MMP-9 and MMP-2 related with fibrous capsule leading led to the migration and invasion of hepatocellular carcinoma cells in human samples | [43] | |
| CHI3L1 | M2 macrophage-secreted CHI3L1 promoted metastasis of gastric and breast cancer cells in vitro and in vivo; CHI3L1 interaction with IL-13Rα2 upregulates MMPs | [44] | |
| IL-23/IL-17 | Upregulation of IL-23 leads to tumour growth and progression and development of a tumoral IL-17 response which promote tumorigenesis in a mouse model of colorectal cancer | [45] | |
| IL-6 | TAM-derived IL-6 highly expressed in Hepatocellular carcinoma patients, which is correlated with disease grades and tumour progression | [46] | |
| PDGF | PDGF release from macrophages mediates the recruitment of pericytes in human melanoma cell lines and OCM-1-induced melanoma mouse model | [47] | |
| T cells | IL17-A | Inhibition of IL17-A release by Th17 cells prevent development of microinvasive PCa in mouse models | [48] |
| IL-17 | IL-17-producing T cells can promote PCa progression by enhancing inflammation and angiogenesis | [49] | |
| PD-1 | A high percentage of CD8+ T cells express PD-1 in PCa samples, which impair an effective immune response by these cells | [50] | |
| IL-3 | IL-3 expressed by T cells increase the recruitment of basophils and immune cells into the tumour microenvironment, which is linked with a poor survival | [18] | |
| IFN-γ | IFN-γ can enhance antigen presentation and contribute to immune surveillance in PCa | [51] | |
| TNF-α | TNF-α produced by activated T cells regulated apoptosis, angiogenesis, and inflammation in PCa | [52] | |
| TGF-β | TGF-β produced by T cells can suppress and promote tumour growth in PCa depending on the signal it receives | [53] | |
| B cells | Lymphotoxin | Lymphotoxin lead to CXCL13/IKKa/STAT3/E2F1/BMI1 (RNF51) activation, ubiquitination of histones within PCa cell nuclei and proliferation of androgen-deprived PCa cells in castration-resistant PCa in mice | [54] |
| TGF-β | Secretion of TGF-β by B-cells leads to anergy of CD8+ T cells | [55] | |
| IL-2 | IL-2 and IL-4 produced by B cells regulate the Th2 memory responses to Heligmosomoides polygyrus (Hp) in chimeric mice lacking AID infected with Hp | [56] | |
| IL-6 | Chimera’s mice with B cell lack IL-6 have impaired Th1 and Th17 responses to Salmonella | [57] | |
| GABA | B cell-derived GABA promotes monocyte differentiation into anti-inflammatory macrophages that secrete IL-10 and inhibit CD8+ T cell killer function in mice | [58] |
ANGPT: Angiopoietin, ARG: Arginase, CCL: CC chemokine ligand, CML: Chronic myeloid leukaemia, CXCL: Chemokine (C-X-C motif) ligand, ECM: Extracellular matrix, EMT: Epithelial-mesenchymal transition, FOXP3: Forkhead box subfamily 3, GABA: γ-amino butyric acid, GM-CSF: Granulocyte-macrophage colony-stimulating factor, HGF: Hepatocyte growth factor, IFN-γ: Interferon-gamma, IL: Interleukin, IL-13Rα2: Interleukin-13 receptor α2 chain, IL-1RA: IL-1 receptor antagonist, MHC: Major histocompatibility complex, MMP: Matrix metalloproteinase, NDE: Neutrophil-derived elastase, NET: Neutrophil extracellular traps, PD: Programmed death, PDGF: Platelet-derived growth factor, PGD: Prostaglandin, RNE: Reactive nitrogen species, ROS: Reactive oxygen species, TAM: Tumour-associated macrophages, TAN: Tumour-associated neutrophils, TGF: Transforming growth factor, TNF: Tumour necrosis factor, VEGF: Vascular endothelial growth factor.
2. Basophils
Basophils constitute approximately 1% of circulating white blood cells and serve as protectors against allergens, pathogens, and parasites. In an inflammatory context, basophils can migrate to inflammatory regions and promote M2-like macrophage polarization, highlighting the disparity in function between circulating and resident basophils [59].
Elevated basophil and basophil-to-lymphocyte ratio were associated with a poor outcome in metastatic hormone sensitive PCa [60]. Epithelial-derived pro-inflammatory cytokines including interleukin (IL)-33, IL-18, granulocyte-macrophage colony-stimulating factor (GM-CSF), and growth factors including IL-3, IL-7, transforming growth factor-beta (TGF-β), vascular endothelial growth factor A (VEGF) promote activation of basophils [61]. Several studies demonstrated that activated basophils can secrete different cytokines involved in PCa including IL-4, which promotes tumour-promoting Th2 inflammation [18][62] and M2 macrophage polarization related to a poor prognosis [63], IL-13 [59], and tumour necrosis factor-alpha (TNF-α) [64]. Studies also suggested the role of basophils in angiogenesis. Basophils release high amount of VEGFA, a potent proangiogenic molecule [21]. Basophils are a source of hepatocyte growth factor (HGF), a powerful angiogenic factor in tumours [22]. Human basophils also express angiopoietins (ANGPT) 1 and ANGPT2 mRNAs which are involved in vascular permeability [23]. Other studies showed the protective role of basophils in cancer development [20]. Low levels of circulating basophils correlated with higher size and extend of the tumour, higher number of lymph nodes and poor survival in colorectal cancer patients [65]. The effects of different inflammatory molecules produced by basophils in cancer are described in Table 1.
While most data on the role of basophils in cancer progression pertains to cancers other than PCa, additional studies are needed to elucidate the mechanisms by which basophils influence PCa.
3. Eosinophils
Eosinophils constitute 1–4% of white blood cells and play a vital role in maintaining homeostasis and defending the host against infectious agents. They originate from multipotent CD34+ progenitors in the bone marrow [66]. Under normal conditions they are located in spleen, lymph nodes and thymus. When activated, they have the capacity to modulate the immune response, including the phenotype of T cells. [67]. The migration and recruitment of eosinophils to the tumour microenvironment are orchestrated by eotaxins, namely CC chemokine ligand (CCL)11, CCL24, CCL26, and CCL5 which activate the CCR3 receptor, highly expressed on eosinophils [68]. Eosinophils secrete cytotoxic granules including eosinophil cationic protein (ECP), major basic protein (MBP), eosinophil derived neurotoxin (EDN) and eosinophil peroxidase (EPO). Additionally, they release pro-inflammatory mediators such as IL-2, IL-4, IL-5, TGF-β, TNF-α, GM-CSF, and interferon-gamma (IFN-γ) [69]. IL-5 is a key mediator for eosinophil growth, differentiation, and activation [70]. Moreover, eosinophils express adhesion molecules CD11a/CD18, allowing them to interact with tumour cells, indicating their role in cancer progression [71]. Histological analysis of PCa samples revealed an increase in eosinophils compared to healthy controls in correlation with age and Gleason score [72].
On the other hand, activated eosinophils inhibited PCa cell growth through upregulation of E-cadherin, a metastasis suppressor molecule [73]. Evidence demonstrated that incubation of PCa cell lines with activated eosinophils inhibited cell growth [74]. Treatment of patients with metastatic castration-resistant PCa with Sipuleucel-T led to an increase in eosinophil counts, correlated with improved survival and enhanced maximal T-cell proliferation responses [27]. The role of different cytokines and chemokines produced by eosinophils are labelled in Table 1.
To advance the understanding of eosinophils in the tumour microenvironment and their interactions with other immune cells, it is crucial to improve the technological detection of eosinophils and discover novel biomarkers for defining eosinophil subpopulations. This will provide insights into their ability to modulate various cells in different cancers, including PCa, and their role in cancer progression.
4. Mast Cells
Mast cells derive from CD34+/CD117+ hematopoietic stem cells in the bone marrow and they undergo maturation within target tissues [75]. Besides KIT activation, which is essential for mast cell development, several cytokines, including IL-3, IL-4, IL-9, IL-10, IL-33, and TGF-β, influence their growth and survival [76]. Mast cells exhibit significant plasticity and can adopt various phenotypes depending on the host’s genetic background and local or systemic factors [77]. These cells are characterized by the presence of numerous granules rich in histamine and heparin. Upon activation, mast cells can degranulate and release inflammatory mediators to combat pathogens [78]. This response leads to the synthesis of specific cytokines, including anti-inflammatory TGF-β, IL-10, as well as the proinflammatory associated cytokines IL-4, IL-6, and IFN-γ [38]. Evidence demonstrated that mast cells are present in several tumours [79][80]. Zadvornyi et al. [81] demonstrated that increased mast cell infiltration and degranulation were associated with malignancy of PCa. Another study demonstrated the potential of mast cells to promote PCa cell proliferation and epithelial mesenchymal transition which is linked with invasion and metastasis [82]. A study dissected that infiltrating mast cells in PCa suppress androgen receptor-MMP signalling promoting PCa cell invasion [83]. Intratumoral mast cell tryptase+/chymase+/CD117+ phenotype was founded in malignant PCa samples [84]. Additionally, a high extratumoral mast cell count was linked with a high risk of biochemical recurrence and PCa metastasis [85].
Release of IL-1, IL-4 and IL-6 from mast cells was associated with elimination of tumour cells and rejection of tumours [36]. Other studies demonstrated that IL-1 is linked with tumour growth, angiogenesis, macrophage recruitment and metastasis [35]. Another study in human PCa samples associated higher mast cell infiltrates with a better PCa prognosis [86]. Moreover, mast cells can contribute to angiogenesis inhibition through secretion of prostaglandin D2 (PGD2) [39]. Table 1 dissects the role of each cytokine released by mast cells on cancer progression.
5. Macrophages
Macrophages are vital phagocytic cells integral to the innate immune response. The primary sources of macrophages are monocytes, which circulate in the blood, and tissue-resident macrophages originating from the yolk sac. These cells are recruited and activated by the specific microenvironment in which they operate. In the context of the tumour microenvironment, macrophage activation plays a significant role in influencing tumour development, progression, metastasis, immune regulation, and angiogenesis [87].
Activated macrophages can be classified into two main categories: M1-like macrophages, which promote inflammation to combat pathogen invasion and cancer, and M2-like macrophages, which are associated with tissue repair and support tumour progression [88]. M1-like macrophages secrete proinflammatory mediators including IL-12, TNF-α, chemokine (C-X-C motif) ligand (CXCL)-10, IFN-γ, and nitric oxide synthase (NOS), whereas M2-like macrophages produce anti-inflammatory IL-10, IL-13, and IL-4, arginase-1, the mannose receptor CD206, and scavenger receptors [89][90]. The polarization of macrophages into M1 or M2 phenotypes depends on the signals present in the microenvironment.
Research has demonstrated that tumour-associated macrophages (TAMs) often acquire a tumour-suppressive M2-like phenotype, contributing to the development of carcinogenesis [91]. The release of IL-1β, IL-8, TNF-α, TGF-β [40][41][42], MMP-2, and MMP-9 [43] by macrophages is involved in epithelial-mesenchymal transition (EMT), which promotes cancer cell invasion and metastasis. It is believed that metastatic processes could not be a late event in tumour progression. The primary tumours could prime the metastatic organ before tumour cell arrival. Macrophages are involved in the formation of this pre-metastatic niches. They are mobilized to bloodstream and are then clustered in these regions by CCL2, CSF-1, VEGF, platelet-derived growth factor (PDGF), TNF-α, and TGF-β [92][93]. The role of inflammatory mediators produced by macrophages in cancer is discussed in Table 2.
Given their ability to modulate the tumour microenvironment, the strategic targeting of macrophages has emerged as a promising approach in the development of new strategies for treating PCa. This approach holds the potential to provide novel and effective therapeutic alternatives for PCa patients.
6. T Cells
T cells play a crucial function in the adaptive immune response and were identified as key cells in the PCa tumour microenvironment [94]. These cells originate from the bone marrow and comprise various subtypes, including CD8+ T cells, CD4+ T cells, Th17 and regulatory T cells (Tregs). CD8+ T cells, known as cytotoxic T cells, exert their effects directly on infected cells. They predominantly secrete immune mediators such as IFN-γ, TNF-α, IL-2, granzyme, and perforin. In contrast, CD4+ T cells, or T helper cells, orchestrate immune responses by activating B cells and CD8+ T cells. They can be further categorized into Th1, Th2, Th17, and regulatory T cell (Treg) subsets. Th1 cells release proinflammatory cytokines, including IFN-γ, TNF-α, and IL-2, while Th2 cells secrete IL-4, IL-5, IL-13, IL-25, and IL-10, driving an anti-inflammatory response [95]. Th17 cells, characterized by secretion of IL-17A, IL-17F, IL-21, and IL-22, have been implicated in PCa metastasis. Studies have shown that the loss of Th17 function can hinder the development of microinvasive PCa in murine models [48]. Tregs were firstly defined as CD4+CD25high cells and were found to be increased in PCa patients [96]. These cells play a vital role in discriminating self from foreign antigens and can either activate or suppress immune responses. Tregs release immunosuppressive cytokines, including IL-10, TGF-β, IL-35, CD39, CD73, and indoleamine 2,3-dioxygenase (IDO) [97]. The immunosuppressive functions of Tregs favour tumour progression, and elevated Treg levels in PCa patients have been associated with poorer survival outcomes [98]. CD8+ T cells have been associated with a good prognosis and a study identified CD8+CD44+ population as important for reducing the tumour burden [99]. On the other hand, accumulation of CD4+ T cells in the PCa tumour microenvironment was associated with a poor survival [100]. In fact, this population is increased in PCa patients in comparison to healthy controls. An increase of CD4+ T cells was also associated with increased chemo-resistance to docetaxel in PCa cells [101]. Later, Kaur et al. [102] showed an association between increased transcription factor FOXP3+ Treg cells and risk of metastasis. Another study identified CD4+FOXP3+ Tregs and CD8+FOXP3+ Tregs increased in PCa samples and associated with increased risk of death [103][104].
Understanding the intricate roles of T cells, including their subtypes and cytokine profiles (Table 1), is essential for deciphering the complex immune landscape within the PCa microenvironment.
7. B Cells
B cells originate in the bone marrow and have the ability to migrate to the spleen and lymph nodes. Naïve B cells undergo activation into plasma cells in response to specific antigens during their development, leading to proliferation and differentiation. The maturation of B cells results in changes to their epitopes, and their characterization relies on CD markers such as CD19, CD20, CD21, CD40, and CD79b [105]. B cells can influence the tumour microenvironment through various mechanisms, including antibody presentation, antibody production, and cytokine secretion [106]. Studies have demonstrated that B cells activate CD4+ T cells, resulting in the accumulation of T cells in the tumour microenvironment and the differentiation of CD4+ and CD8+ T cells into distinct phenotypes [107]. Interactions between CD20+ B cells and T cells in the tumour microenvironment have been shown to impact the protective function of T cells [108]. Regulatory B cells (Bregs) are frequently found in advanced hepatocellular, gastric, and PCa, suggesting their potential influence on tumour development and progression [109][110][111]. Bregs are associated with an anti-immune function, as they release immunosuppressive molecules such as IL-10, IL-35, and TGF-β, which hinder the activity of T cells [55][112][113][114].
Literature data suggest that the infiltration of B cells increases the risk of adverse events in prostate carcinogenesis and malignancies. The effect of inflammatory modulators produced by B cells on tumour progression is described in Table 1.
8. Overall Remarks
Overall, IL-1, IL-6, IL-2, IL-4, IL-7, IL-8, IL-10, IL-17, IL-23, TNF-α, TGF-β, IFN-γ, VEGF, and GM-CSF are the main inflammatory mediators involved in PCa (Figure 1).
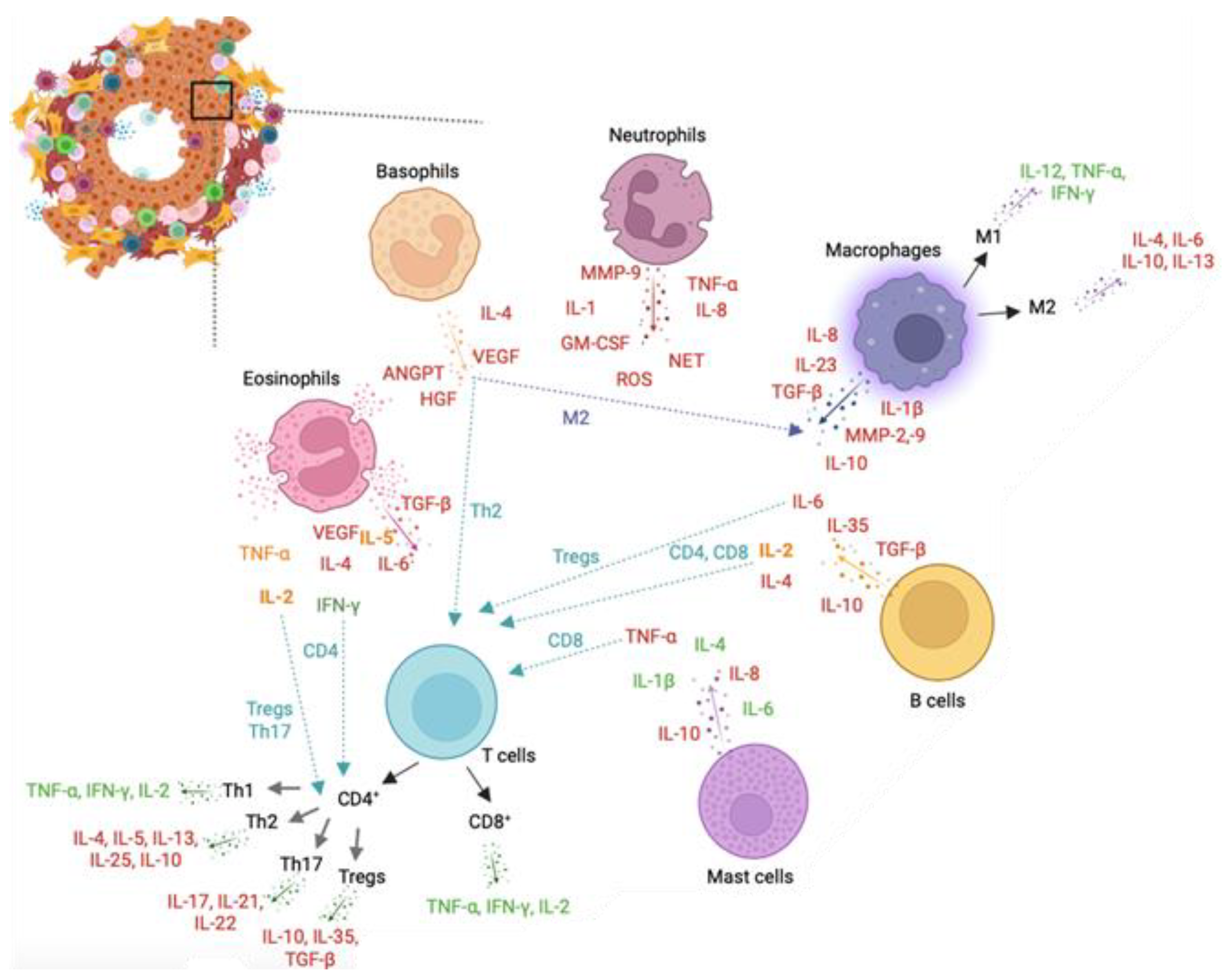
Figure 1. Leukocytes and leukocyte-mediated cytokines involved in prostate cancer (PCa). Representation of key cells in the prostate tumour microenvironment. Neutrophils mainly release matrix metalloproteinase (MMP)-9, interleukin (IL)-1, granulocyte-macrophage colony-stimulating factor (GM-CSF), reactive oxygen species (ROS), neutrophil extracellular traps (NETs), IL-8, and tumour necrosis factor (TNF)-α, all promoting PCa progression. Basophils promote activation of the M2-like phenotype in macrophages and Th2 response in T cells and release pro-tumour IL-4, VEGF, ANGPTs, and HGF cytokines. Eosinophils secrete pro-tumoral transforming growth factor (TGF)-β, IL-6, vascular endothelial growth factor (VEGF), and IL-4 cytokines and anti-tumoral interferon (IFN)-γ cytokine. Release of TNF-α, IL-2, and IL-5 from eosinophils can modulate a pro-tumoral or anti-tumoral activity depending on cell signals. IL-2 and IFN-γ stimulates activation of, respectively, Tregs/Th17 and CD4+ T cells. Mast cells secrete pro-tumoral TNF-α, IL-8, and IL-10 and anti-tumoral IL-1β, IL-4, and IL-6 cytokines. Tumour-associated macrophages secrete IL-1β, IL-8, IL-10, IL-23, MMP-2, MMP-9, and TGF-β, impacting tumour progression. Macrophages can shift to pro-inflammatory M1-like or anti-inflammatory M2-like phenotypes, influencing tumour outcomes. M1-like macrophages release IL-12, TNF-α, and IFN-γ, while M2-like macrophages secrete IL-4, IL-6, IL-10, and IL-13. T cells encompass CD4+ T cells (Th1, Th2, Th17, and Tregs) and CD8+ T cells, with Th1 having a pro-inflammatory response. Th2, Th17, and Tregs contribute to tumour progression. CD8+ T cells secrete IFN-γ, TNF-α, and IL-2, associated with a favourable prognosis in PCa. B cells release pro-tumoral IL-4, IL-6, IL-10, and TGF-β cytokines and the intermediate IL-2 cytokine. IL-2 and IL-4 stimulate CD4 and CD8 responses, while IL-6 activates Tregs in PCa. Green-coloured cytokines support tumour resolution and a positive prognosis. Red-coloured cytokines promote tumour growth, proliferation, and metastasis. Orange-coloured cytokines can trigger a pro- or anti-tumoral response in PCa. ANGPT: Angiopoietin, GM-CSF: Granulocyte-macrophage colony-stimulating factor, HGF: Hepatocyte growth factor, IFN: Interferon, IL- Interleukin, MMP: Matrix metalloproteinase, NETs: Neutrophil extracellular traps, PCa: Prostate cancer, ROS: Reactive oxygen species, TGF: Transforming growth factor, TNF: Tumour necrosis factor, Tregs: Regulatory T cells, VEGF: Vascular endothelial growth factor.
IL-1 and IL-6 promote cancer growth, proliferation, and progression [115][116]. IL-1 is increased in PCa and induces immunosuppressive function of mesenchymal stem cells [117][118]. IL-6 is increased in PCa, induces EMT and metastasis, increases the expression of androgen receptor, and induces infiltration of T cells into the tumour microenvironment [119][120]. IL-2 has been found to stimulate Tregs, with some studies associating it with tumour growth and progression, while others suggest its potential anti-tumour activity [121]. IL-4 increases the expression of androgens, activates the JNK pathway, and promotes tumour progression [122]. IL-7 induces EMT and cancer metastasis [123]. IL-8 stimulates proliferation of prostate stromal cells, regulates the expression of MMPs, promotes PCa progression, angiogenesis, and metastasis [124]. IL-10 inhibits anti-tumour responses and regulates androgen signalling, promoting cancer metastasis [125]. IL-17 promotes PCa growth, angiogenesis, and metastasis [126], increases the expression of programmed death-ligand 1 (PD-L1) and COX-2 and induces the release of IL-6 and IL-8 [127]. IL-23 regulates the androgen response and Th17 survival [125]. TNF-α and TGF-β are able to promote PCa progression and metastasis [128]. TNF-α upregulates the expression of PD-L1, and its control is indicative of tumour cell behaviour [127][129]. TGF-β induces EMT, inhibition of anti-tumour activity, reduces the expression of major histocompatibility complex (MHC)-I, regulates angiogenesis, the formation of the premetastatic niche, and metastasis in bone [130][131][132]. IFN-γ induces the release of IL-6 and IL-8 and promotes anti-tumour response [133]. VEGF contributes to angiogenesis, formation of premetastatic niche, tumour microenvironment remodelling, tumour invasion, and metastasis [132][134]. GM-CSF stimulates leukocytes and increases tumour antigen presentation to effector T cells [135][136].
The activity of interleukins is primarily modulated through the Janus Kinase/signal transducers and activators of transcription (JAK/STAT) pathway. This signalling pathway is integral to normal development, cellular homeostasis, cell proliferation, differentiation, and apoptosis [137]. Ligand binding initiates the multimerization of receptor subunits, leading to the activation of the JAK/STAT pathway and the transmission of signals through the phosphorylation of receptor-associated JAK tyrosine kinases. Consequently, activated JAKs induce the phosphorylation and activation of STATs. This phosphorylation prompts the dimerization of STATs via their conserved SH2 domain, subsequently allowing them to enter the nucleus. Within the nucleus, STATs bind to specific DNA sequences, either stimulating or suppressing the transcription of target genes [138]. It was reported that JAK/STAT3 inhibition suppress PCa cell growth and increases apoptosis [139]. BRCA1 via JAK1/2 and STAT3 phosphorylation can induce cell proliferation and inhibit cancer cell death [140]. The androgen receptor could also activate JAK/STAT3 and stimulate cell proliferation and antiapoptotic effect increasing tumour invasion [141][142].
NF-κB is a transcription factor predominantly activated by cytokines such as TNF-α in PCa. In androgen-dependent PCa, IL-6 and VEGF stimulates the increase of the expression of NF-κB [143]. NF-κB targets a transcription regulatory element of PSA and correlates with cancer progression, chemoresistance, and PSA recurrence [144].
Growth factors including VEGF, epidermal growth factor (EGF), insulin-like growth factor (IGF)-1, HGF, and TGF-β are key players in the receptor tyrosine kinase (RTK) signalling pathway. These growth factors activate the extracellular signal-regulated kinases (ERK)/MAPK or PI3K/AKT/mTOR mechanisms [145]. Growth factor receptors possess RTK activity, and their binding to ligands leads to the activation of transcription factors, resulting in the altered expression of genes associated with cell growth, proliferation, and survival [146]. IGF-1 functions as a positive growth-promoting signal transduction pathway, while FGF plays a dual role as a positive growth factor and an angiogenic growth factor. On the other hand, TGF-β serves as a negative growth factor, regulating cell differentiation and proliferation [147]. Studies demonstrated that alterations on the expression of TGF-β, EGF and their receptors correlates with PCa progression and biochemical recurrence [148][149]. The phosphoinositide 3-kinase (PI3K)/protein kinase B (AKT) pathway is often upregulated due to the loss of the tumour suppressor PTEN, which negatively regulates the PI3K/AKT pathway [150]. It has been demonstrated that the aberrant PI3K/AKT pathway disturbs the action of ERKs, thereby supporting androgen receptor-independent growth in PCa [151]. Overexpression of growth factors promotes the activation of Ras and MAPK pathways [152]. Upon activation, MAPKs phosphorylate transcription factors such as c-Jun, c-Fos, ATF2, and p53. Additionally, ERK or p38 MAPKs can activate MAPK interacting protein kinases 1 and 2 (MNK1 and MNK2), which controls signals involved in mRNA translation [153]. Interestingly, MNKs have been found to be overexpressed in PCa [154].
Inflammatory signalling plays a significant role in the development and progression of PCa. Considering these findings, therapeutic strategies targeting inflammatory signalling pathways in PCa may help manage the disease and potentially improve outcomes.
Ongoing research is exploring new treatments and strategies, especially those utilizing natural bioactive compounds, to mitigate the severe side effects, radiotherapy resistance, and recurrence of PCa.
This entry is adapted from the peer-reviewed paper 10.3390/biomedicines11123140
References
- Ballesteros, I.; Rubio-Ponce, A.; Genua, M.; Lusito, E.; Kwok, I.; Fernández-Calvo, G.; Khoyratty, T.E.; van Grinsven, E.; González-Hernández, S.; Nicolás-Ávila, J.Á.; et al. Co-Option of Neutrophil Fates by Tissue Environments. Cell 2020, 183, 1282–1297.e18.
- El-Benna, J.; Hurtado-Nedelec, M.; Marzaioli, V.; Marie, J.C.; Gougerot-Pocidalo, M.A.; Dang, P.M.C. Priming of the Neutrophil Respiratory Burst: Role in Host Defense and Inflammation. Immunol. Rev. 2016, 273, 180–193.
- Venet, F.; Monneret, G. Advances in the Understanding and Treatment of Sepsis-Induced Immunosuppression. Nat. Rev. Nephrol. 2018, 14, 121–137.
- Fujita, K.; Imamura, R.; Tanigawa, G.; Nakagawa, M.; Hayashi, T.; Kishimoto, N.; Hosomi, M.; Yamaguchi, S. Low Serum Neutrophil Count Predicts a Positive Prostate Biopsy. Prostate Cancer Prostatic Dis. 2012, 15, 386–390.
- Fujita, K.; Hosomi, M.; Nakagawa, M.; Tanigawa, G.; Imamura, R.; Uemura, M.; Nakai, Y.; Takayama, H.; Yamaguchi, S.; Nonomura, N. White Blood Cell Count Is Positively Associated with Benign Prostatic Hyperplasia. Int. J. Urol. 2014, 21, 308–312.
- Deryugina, E.I.; Zajac, E.; Juncker-Jensen, A.; Kupriyanova, T.A.; Welter, L.; Quigley, J.P. Tissue-Infiltrating Neutrophils Constitute the Major In Vivo Source of Angiogenesis-Inducing MMP-9 in the Tumor Microenvironment. Neoplasia 2014, 16, 771–788.
- Shaul, M.E.; Fridlender, Z.G. Tumour-Associated Neutrophils in Patients with Cancer. Nat. Rev. Clin. Oncol. 2019, 16, 601–620.
- Hedrick, C.C.; Malanchi, I. Neutrophils in Cancer: Heterogeneous and Multifaceted. Nat. Rev. Immunol. 2022, 22, 173–187.
- Alsamraae, M.; Costanzo-Garvey, D.; Teply, B.A.; Boyle, S.; Sommerville, G.; Herbert, Z.T.; Morrissey, C.; Dafferner, A.J.; Abdalla, M.Y.; Fallet, R.W.; et al. Androgen receptor inhibition suppresses anti-tumor neutrophil response against bone metastatic prostate cancer via regulation of TβRI expression. Cancer Letters 2023, 28, 216468.
- Yoshida, M.; Taguchi, A.; Kawana, K.; Adachi, K.; Kawata, A.; Ogishima, J.; Nakamura, H.; Fujimoto, A.; Sato, M.; Inoue, T.; et al. Modification of the Tumor Microenvironment in KRAS or C-MYC-Induced Ovarian Cancer-Associated Peritonitis. PLoS ONE 2016, 11, e0160330.
- Gong, L.; Cumpian, A.M.; Caetano, M.S.; Ochoa, C.E.; De La Garza, M.M.; Lapid, D.J.; Mirabolfathinejad, S.G.; Dickey, B.F.; Zhou, Q.; Moghaddam, S.J. Promoting Effect of Neutrophils on Lung Tumorigenesis Is Mediated by CXCR2 and Neutrophil Elastase. Mol. Cancer 2013, 12, 154.
- Campregher, C.; Luciani, M.G.; Gasche, C. Activated Neutrophils Induce an HMSH2-Dependent G2/M Checkpoint Arrest and Replication Errors at a (CA)13-Repeat in Colon Epithelial Cells. Gut 2008, 57, 780–787.
- Romano, A.; Parrinello, N.L.; Vetro, C.; Tibullo, D.; Giallongo, C.; La Cava, P.; Chiarenza, A.; Motta, G.; Caruso, A.L.; Villari, L.; et al. The Prognostic Value of the Myeloid-Mediated Immunosuppression Marker Arginase-1 in Classic Hodgkin Lymphoma. Oncotarget 2016, 7, 67333–67346.
- Costanzo-Garvey, D.L.; Keeley, T.; Case, A.J.; Watson, G.F.; Alsamraae, M.; Yu, Y.; Su, K.; Heim, C.E.; Kielian, T.; Morrissey, C.; et al. Neutrophils Are Mediators of Metastatic Prostate Cancer Progression in Bone. Cancer Immunol. Immunother. 2020, 69, 1113–1130.
- Nielsen, S.R.; Strøbech, J.E.; Horton, E.R.; Jackstadt, R.; Laitala, A.; Bravo, M.C.; Maltese, G.; Jensen, A.R.D.; Reuten, R.; Rafaeva, M.; et al. Suppression of Tumor-Associated Neutrophils by Lorlatinib Attenuates Pancreatic Cancer Growth and Improves Treatment with Immune Checkpoint Blockade. Nat. Commun. 2021, 12, 3414.
- Rodriguez, Y.I.; Campos, L.E.; Castro, M.G.; Bannoud, N.; Blidner, A.G.; Filippa, V.P.; Croci, D.O.; Rabinovich, G.A.; Alvarez, S.E. Tumor Necrosis Factor Receptor-1 (P55) Deficiency Attenuates Tumor Growth and Intratumoral Angiogenesis and Stimulates CD8+ T Cell Function in Melanoma. Cells 2020, 9, 2469.
- Morimoto-Kamata, R.; Yui, S. Insulin-like Growth Factor-1 Signaling Is Responsible for Cathepsin G-Induced Aggregation of Breast Cancer MCF-7 Cells. Cancer Sci. 2017, 108, 1574–1583.
- De Monte, L.; Wörmann, S.; Brunetto, E.; Heltai, S.; Magliacane, G.; Reni, M.; Paganoni, A.M.; Recalde, H.; Mondino, A.; Falconi, M.; et al. Basophil Recruitment into Tumor-Draining Lymph Nodes Correlates with Th2 Inflammation and Reduced Survival in Pancreatic Cancer Patients. Cancer Res. 2016, 76, 1792–1803.
- Baba, T.; Tanabe, Y.; Yoshikawa, S.; Yamanishi, Y.; Morishita, S.; Komatsu, N.; Karasuyama, H.; Hirao, A.; Mukaida, N. MIP-1a/CCL3-Expressing Basophil-Lineage Cells Drive the Leukemic Hematopoiesis of Chronic Myeloid Leukemia in Mice. Blood 2016, 127, 2607–2617.
- Sektioglu, I.M.; Carretero, R.; Bulbuc, N.; Bald, T.; Tüting, T.; Rudensky, A.Y.; Hämmerling, G.J. Basophils Promote Tumor Rejection via Chemotaxis and Infiltration of CD8+ T Cells. Cancer Res. 2017, 77, 291–302.
- de Paulis, A.; Prevete, N.; Fiorentino, I.; Rossi, F.W.; Staibano, S.; Montuori, N.; Ragno, P.; Longobardi, A.; Liccardo, B.; Genovese, A.; et al. Expression and Functions of the Vascular Endothelial Growth Factors and Their Receptors in Human Basophils. J. Immunol. 2006, 177, 7322–7331.
- Cerny-Reiterer, S.; Ghanim, V.; Hoermann, G.; Aichberger, K.J.; Herrmann, H.; Muellauer, L.; Repa, A.; Sillaber, C.; Walls, A.F.; Mayerhofer, M.; et al. Identification of Basophils as a Major Source of Hepatocyte Growth Factor in Chronic Myeloid Leukemia: A Novel Mechanism of BCR-ABL1-Independent Disease Progression. Neoplasia 2012, 14, 572–584.
- Prevete, N.; Staiano, R.I.; Granata, F.; Detoraki, A.; Necchi, V.; Ricci, V.; Triggiani, M.; De Paulis, A.; Marone, G.; Genovese, A. Expression and Function of Angiopoietins and Their Tie Receptors in Human Basophils and Mast Cells. J. Biol. Regul. Homeost. Agents 2013, 27, 827–839.
- Kryczek, I.; Wei, S.; Zou, L.; Altuwaijri, S.; Szeliga, W.; Kolls, J.; Chang, A.; Zou, W. Cutting Edge: Th17 and Regulatory T Cell Dynamics and the Regulation by IL-2 in the Tumor Microenvironment. J. Immunol. 2007, 178, 6730–6733.
- Gocheva, V.; Wang, H.W.; Gadea, B.B.; Shree, T.; Hunter, K.E.; Garfall, A.L.; Berman, T.; Joyce, J.A. IL-4 Induces Cathepsin Protease Activity in Tumor-Associated Macrophages to Promote Cancer Growth and Invasion. Genes Dev. 2010, 24, 241–255.
- Michalaki, V.; Syrigos, K.; Charles, P.; Waxman, J. Serum Levels of IL-6 and TNF-α Correlate with Clinicopathological Features and Patient Survival in Patients with Prostate Cancer. Br. J. Cancer 2004, 90, 2312–2316.
- McNeel, D.G.; Gardner, T.A.; Higano, C.S.; Kantoff, P.W.; Small, E.J.; Wener, M.H.; Sims, R.B.; DeVries, T.; Sheikh, N.A.; Dreicer, R. A Transient Increase in Eosinophils Is Associated with Prolonged Survival in Men with Metastatic Castration-Resistant Prostate Cancer Who Receive Sipuleucel-T. Cancer Immunol. Res. 2014, 2, 988–999.
- Mumberg, D.; Monach, P.A.; Wanderling, S.; Philip, M.; Toledano, A.Y.; Schreiber, R.D.; Schreiber, H.; Rowley, J.D. CD4 T Cells Eliminate MHC Class II-Negative Cancer Cells in Vivo by Indirect Effects of IFN-gamma. Proc. Natl. Acad. Sci. USA 1999, 96, 8633–8638.
- Kawamata, H.; Kameyama, S.; Oyasu’, R. In Vitro and In Vivo Acceleration of the Neoplastic Phenotype of a Low-Tumorigenicity Rat Bladder Carcinoma Cell Line by Transfected Transforming Growth Factor-a. Mol. Carcinog. 1994, 9, 210–219.
- Salven, P.; Ruotsalainen, T.; Mattson, K.; Joensuu, H. High pre-treatment serum level of vascular endothelial growth factor (vegf) is associated with poor outcome in small-cell lung cancer. Int. J. Cancer 1998, 79, 144–146.
- Park, B.K.; Zhang, H.; Zeng, Q.; Dai, J.; Keller, E.T.; Giordano, T.; Gu, K.; Shah, V.; Pei, L.; Zarbo, R.J.; et al. NF-ΚB in Breast Cancer Cells Promotes Osteolytic Bone Metastasis by Inducing Osteoclastogenesis via GM-CSF. Nat. Med. 2007, 13, 62–69.
- Taipale, J.; Lohi, J.; Saarinen, J.; Kovanen, P.T.; Keski-Oja, J. Human Mast Cell Chymase and Leukocyte Elastase Release Latent Transforming Growth Factor-PI from the Extracellular Matrix of Cultured Human Epithelial and Endothelial Cells. J. Biol. Chem. 1995, 270, 4689–4696.
- Johnson, C.; Huynh, V.; Hargrove, L.; Kennedy, L.; Graf-Eaton, A.; Owens, J.; Trzeciakowski, J.P.; Hodges, K.; Demorrow, S.; Han, Y.; et al. Inhibition of Mast Cell-Derived Histamine Decreases Human Cholangiocarcinoma Growth and Differentiation via c-Kit/Stem Cell Factor-Dependent Signaling. Am. J. Pathol. 2016, 186, 123–133.
- Dudeck, J.; Ghouse, S.M.; Lehmann, C.H.K.; Hoppe, A.; Schubert, N.; Nedospasov, S.A.; Dudziak, D.; Dudeck, A. Mast-Cell-Derived TNF Amplifies CD8+ Dendritic Cell Functionality and CD8+ T Cell Priming. Cell Rep. 2015, 13, 399–411.
- Litmanovich, A.; Khazim, K.; Cohen, I. The Role of Interleukin-1 in the Pathogenesis of Cancer and Its Potential as a Therapeutic Target in Clinical Practice. Oncol. Ther. 2018, 6, 109–127.
- Cimpean, A.M.; Tamma, R.; Ruggieri, S.; Nico, B.; Toma, A.; Ribatti, D. Mast Cells in Breast Cancer Angiogenesis. Crit. Rev. Oncol. Hematol. 2017, 115, 23–26.
- Ribatti, D.; Ranieri, G. Tryptase, a Novel Angiogenic Factor Stored in Mast Cell Granules. Exp. Cell Res. 2015, 332, 157–162.
- da Silva, E.Z.M.; Jamur, M.C.; Oliver, C. Mast Cell Function: A New Vision of an Old Cell. J. Histochem. Cytochem. 2014, 62, 698–738.
- Murata, T.; Aritake, K.; Matsumoto, S.; Kamauchi, S.; Nakagawa, T.; Hori, M.; Momotani, E.; Urade, Y.; Ozaki, H. Prostagladin D 2 Is a Mast Cell-Derived Antiangiogenic Factor in Lung Carcinoma. Proc. Natl. Acad. Sci. USA 2011, 108, 19802–19807.
- Wu, Y.; Deng, J.; Rychahou, P.G.; Qiu, S.; Evers, B.M.; Zhou, B.P. Stabilization of Snail by NF-ΚB Is Required for Inflammation-Induced Cell Migration and Invasion. Cancer Cell 2009, 15, 416–428.
- Fu, X.T.; Dai, Z.; Song, K.; Zhang, Z.J.; Zhou, Z.J.; Zhou, S.L.; Zhao, Y.M.; Xiao, Y.S.; Sun, Q.M.; Ding, Z.; et al. Macrophage-Secreted IL-8 Induces Epithelial-Mesenchymal Transition in Hepatocellular Carcinoma Cells by Activating the JAK2/STAT3/Snail Pathway. Int. J. Oncol. 2015, 46, 587–596.
- Kawata, M.; Koinuma, D.; Ogami, T.; Umezawa, K.; Iwata, C.; Watabe, T.; Miyazono, K. TGF-β-Induced Epithelial-Mesenchymal Transition of A549 Lung Adenocarcinoma Cells Is Enhanced by pro-Inflammatory Cytokines Derived from RAW 264.7 Macrophage Cells. J. Biochem. 2012, 151, 205–216.
- Cui, Q.; Wang, X.; Zhang, Y.; Shen, Y.; Qian, Y. Macrophage-Derived MMP-9 and MMP-2 Are Closely Related to the Rupture of the Fibrous Capsule of Hepatocellular Carcinoma Leading to Tumor Invasion. Biol. Proced. Online 2023, 25, 8.
- Chen, Y.; Zhang, S.; Wang, Q.; Zhang, X. Tumor-Recruited M2 Macrophages Promote Gastric and Breast Cancer Metastasis via M2 Macrophage-Secreted CHI3L1 Protein. J. Hematol. Oncol. 2017, 10, 36.
- Grivennikov, S.I.; Wang, K.; Mucida, D.; Stewart, C.A.; Schnabl, B.; Jauch, D.; Taniguchi, K.; Yu, G.Y.; Österreicher, C.H.; Hung, K.E.; et al. Adenoma-Linked Barrier Defects and Microbial Products Drive IL-23/IL-17-Mediated Tumour Growth. Nature 2012, 491, 254–258.
- Kong, L.; Zhou, Y.; Bu, H.; Lv, T.; Shi, Y.; Yang, J. Deletion of Interleukin-6 in Monocytes/Macrophages Suppresses the Initiation of Hepatocellular Carcinoma in Mice. J. Exp. Clin. Cancer Res. 2016, 35, 131.
- Thijssen, V.L.J.L.; Paulis, Y.W.J.; Nowak-Sliwinska, P.; Deumelandt, K.L.; Hosaka, K.; Soetekouw, P.M.M.B.; Cimpean, A.M.; Raica, M.; Pauwels, P.; van den Oord, J.J.; et al. Targeting PDGF-Mediated Recruitment of Pericytes Blocks Vascular Mimicry and Tumor Growth. J. Pathol. 2018, 246, 447–458.
- Zhang, Q.; Liu, S.; Ge, D.; Cunningham, D.M.; Huang, F.; Ma, L.; Burris, T.P.; You, Z. Targeting Th17-IL-17 Pathway in Prevention of Micro-Invasive Prostate Cancer in a Mouse Model. Prostate 2017, 77, 888–899.
- Sfanos, K.S.; de Marzo, A.M. Prostate Cancer and Inflammation: The Evidence. Histopathology 2012, 60, 199–215.
- Sfanos, K.S.; Bruno, T.C.; Meeker, A.K.; De Marzo, A.M.; Isaacs, W.B.; Drake, C.G. Human Prostate-Infiltrating CD8+ T Lymphocytes Are Oligoclonal and PD-1+. Prostate 2009, 69, 1694–1703.
- Adekoya, T.O.; Richardson, R.M. Cytokines and Chemokines as Mediators of Prostate Cancer Metastasis. Int. J. Mol. Sci. 2020, 21, 4449.
- Osawa, Y.; Nagaki, M.; Banno, Y.; Brenner, D.A.; Asano, T.; Nozawa, Y.; Moriwaki, H.; Nakashima, S. Tumor Necrosis Factor Alpha-Induced Interleukin-8 Production via NF-ΚB and Phosphatidylinositol 3-Kinase/Akt Pathways Inhibits Cell Apoptosis in Human Hepatocytes. Infect. Immun. 2002, 70, 6294–6301.
- SMith, P.; Hobisch, A.; Lin, D.; Culig, Z.; Keller, E. Interleukin-6 and Prostate Cancer Progression. Cytokine Growth Factor Rev. 2001, 12, 33–40.
- Ammirante, M.; Luo, J.L.; Grivennikov, S.; Nedospasov, S.; Karin, M. B-Cell-Derived Lymphotoxin Promotes Castration-Resistant Prostate Cancer. Nature 2010, 464, 302–305.
- Parekh, V.V.; Prasad, D.V.R.; Banerjee, P.P.; Joshi, B.N.; Kumar, A.; Mishra, G.C. B Cells Activated by Lipopolysaccharide, But Not by Anti-Ig and Anti-CD40 Antibody, Induce Anergy in CD8+ T Cells: Role of TGF-Β1. J. Immunol. 2003, 170, 5897–5911.
- Wojciechowski, W.; Harris, D.P.; Sprague, F.; Mousseau, B.; Makris, M.; Kusser, K.; Honjo, T.; Mohrs, K.; Mohrs, M.; Randall, T.; et al. Cytokine-Producing Effector B Cells Regulate Type 2 Immunity to H. Polygyrus. Immunity 2009, 30, 421–433.
- Barr, T.A.; Brown, S.; Mastroeni, P.; Gray, D. TLR and B Cell Receptor Signals to B Cells Differentially Program Primary and Memory Th1 Responses to Salmonella Enterica. J. Immunol. 2010, 185, 2783–2789.
- Zhang, B.; Vogelzang, A.; Miyajima, M.; Sugiura, Y.; Wu, Y.; Chamoto, K.; Nakano, R.; Hatae, R.; Menzies, R.J.; Sonomura, K.; et al. B Cell-Derived GABA Elicits IL-10+ Macrophages to Limit Anti-Tumour Immunity. Nature 2021, 599, 471–476.
- Cohen, M.; Giladi, A.; Gorki, A.D.; Solodkin, D.G.; Zada, M.; Hladik, A.; Miklosi, A.; Salame, T.M.; Halpern, K.B.; David, E.; et al. Lung Single-Cell Signaling Interaction Map Reveals Basophil Role in Macrophage Imprinting. Cell 2018, 175, 1031–1044.e18.
- Hadadi, A.; Smith, K.E.; Wan, L.; Brown, J.R.; Russler, G.; Yantorni, L.; Caulfield, S.; Lafollette, J.; Moore, M.; Kucuk, O.; et al. Baseline Basophil and Basophil-to-Lymphocyte Status Is Associated with Clinical Outcomes in Metastatic Hormone Sensitive Prostate Cancer. Urol. Oncol. Semin. Orig. Investig. 2022, 40, e9–e271.
- Pellefigues, C.; Mehta, P.; Chappell, S.; Yumnam, B.; Old, S.; Camberis, M.; Le Gros, G. Diverse Innate Stimuli Activate Basophils through Pathways Involving Syk and IκB Kinases. Proc. Natl. Acad. Sci. USA 2021, 118, 2019524118.
- Galeotti, C.; Stephen-Victor, E.; Karnam, A.; Das, M.; Gilardin, L.; Maddur, M.S.; Wymann, S.; Vonarburg, C.; Chevailler, A.; Dimitrov, J.D.; et al. Intravenous Immunoglobulin Induces IL-4 in Human Basophils by Signaling through Surface-Bound IgE. J. Allergy Clin. Immunol. 2019, 144, 524–535.e8.
- He, X.; Cao, Y.; Gu, Y.; Fang, H.; Wang, J.; Liu, X.; Lv, K.; Yu, K.; Fei, Y.; Lin, C.; et al. Clinical Outcomes and Immune Metrics in Intratumoral Basophil-Enriched Gastric Cancer Patients. Ann. Surg. Oncol. 2021, 28, 6439–6450.
- Falkencrone, S.; Poulsen, L.K.; Bindslev-Jensen, C.; Woetmann, A.; Odum, N.; Poulsen, B.C.; Blom, L.; Jensen, B.M.; Gibbs, B.F.; Yasinska, I.M.; et al. IgE-Mediated Basophil Tumour Necrosis Factor Alpha Induces Matrix Metalloproteinase-9 from Monocytes. Allergy Eur. J. Allergy Clin. Immunol. 2013, 68, 614–620.
- Liu, Q.; Luo, D.; Cai, S.; Li, Q.; Li, X. Circulating Basophil Count as a Prognostic Marker of Tumor Aggressiveness and Survival Outcomes in Colorectal Cancer. Clin. Transl. Med. 2020, 9, 6.
- Fulkerson, P.C. Transcription Factors in Eosinophil Development and as Therapeutic Targets. Front. Med. 2017, 4, 115.
- Hogan, S.P.; Rosenberg, H.F.; Moqbel, R.; Phipps, S.; Foster, P.S.; Lacy, P.; Kay, A.B.; Rothenberg, M.E. Eosinophils: Biological Properties and Role in Health and Disease. Clin. Exp. Allergy 2008, 38, 709–750.
- Ponath, P.D.; Qin, S.; Postyl, T.W.; Wang, J.; Wu, L.; Gerardyl, N.P.; Newman, W.; Gerard, C.; Mackay, C.R. Molecular Cloning and Characterization of a Human Eotaxin Receptor Expressed Selectively on Eosinophils. J. Exp. Med. 1996, 183, 2437–2448.
- Sakkal, S.; Miller, S.; Apostolopoulos, V.; Nurgali, K. Eosinophils in Cancer: Favourable or Unfavourable? Curr. Med. Chem. 2016, 23, 650–666.
- Varricchi, G.; Bagnasco, D.; Borriello, F.; Heffler, E.; Canonica, G.W. Interleukin-5 Pathway Inhibition in the Treatment of Eosinophilic Respiratory Disorders: Evidence and Unmet Needs. Curr. Opin. Allergy Clin. Immunol. 2016, 16, 186–200.
- Caruso, R.A.; Parisi, A.; Quattrocchi, E.; Scardigno, M.; Branca, G.; Parisi, C.; Lucianò, R.; Paparo, D.; Fedele, F. Ultrastructural Descriptions of Heterotypic Aggregation between Eosinophils and Tumor Cells in Human Gastric Carcinomas. Ultrastruct. Pathol. 2011, 35, 145–149.
- Rodríguez Bustos, H.; Cortés Chau, F.; Cortés Pino, F.; Aguirre, P.; Bravo, G.; Gallegos Méndez, I.; Arriaza Onel, C.; Cuellar Godoy, C.; Aguayo González, F.; Espinoza Navarro, O. Morphological Changes of the Cellularity in the Prostatic Gland from Patients with Confirmed Cancer: Gleason Level and Presence of Eosinophils and Mast Cells: Cellular Bioindicators. Int. J. Morphol. 2020, 38, 882–887.
- Furbert-Harris, P.M.; Parish-Gause, D.; Hunter, K.A.; Vaughn, T.R.; Howland, C.; Okomo-Awich, J.; Forrest, K.; Laniyan, I.; Abdelnaby, A.; Oredipe, O.A. Activated Eosinophils Upregulate the Metastasis Suppressor Molecule E-Cadherin on Prostate Tumor Cells. Cell Mol. Biol. 2003, 49, 1009–1016.
- Furbert-Harris, P.; Parish-Gause, D.; Laniyan, I.; Hunter, K.A.; Okomo-Awich, J.; Vaughn, T.R.; Forrest, K.C.; Howland, C.; Abdelnaby, A.; Oredipe, O.A. Inhibition of Prostate Cancer Cell Growth by Activated Eosinophils. Prostate 2003, 57, 165–175.
- Moon, T.C.; St Laurent, C.D.; Morris, K.E.; Marcet, C.; Yoshimura, T.; Sekar, Y.; Befus, A.D. Advances in Mast Cell Biology: New Understanding of Heterogeneity and Function. Mucosal Immunol. 2010, 3, 111–128.
- Gurish, M.F.; Austen, K.F. Developmental Origin and Functional Specialization of Mast Cell Subsets. Immunity 2012, 37, 25–33.
- Dwyer, D.F.; Barrett, N.A.; Austen, K.F.; Kim, E.Y.; Brenner, M.B.; Shaw, L.; Yu, B.; Goldrath, A.; Mostafavi, S.; Regev, A.; et al. Expression Profiling of Constitutive Mast Cells Reveals a Unique Identity within the Immune System. Nat. Immunol. 2016, 17, 878–887.
- Elieh Ali Komi, D.; Wöhrl, S.; Bielory, L. Mast Cell Biology at Molecular Level: A Comprehensive Review. Clin. Rev. Allergy Immunol. 2020, 58, 342–365.
- Varricchi, G.; Galdiero, M.R.; Loffredo, S.; Marone, G.; Iannone, R.; Marone, G.; Granata, F. Are Mast Cells MASTers in Cancer? Front. Immunol. 2017, 8, 424.
- Johansson, A.; Rudolfsson, S.; Hammarsten, P.; Halin, S.; Pietras, K.; Jones, J.; Stattin, P.; Egevad, L.; Granfors, T.; Wikström, P.; et al. Mast Cells Are Novel Independent Prognostic Markers in Prostate Cancer and Represent a Target for Therapy. Am. J. Pathol. 2010, 177, 1031–1041.
- Zadvornyi, T.; Lukianova, N.; Borikun, T.; Tymoshenko, A.; Mushii, O.; Voronina, O.; Vitruk, I.; Stakhovskyi, E.; Chekhun, V. Mast Cells as a Tumor Microenvironment Factor Associated with the Aggressiveness of Prostate Cancer. Neoplasma 2022, 69, 1490–1498.
- Ma, Z.; Yue, L.; Xu, Z.; Zeng, S.; Ma, Y.; Li, Z.; Li, W.; Wang, D. The Effect of Mast Cells on the Biological Characteristics of Prostate Cancer Cells. Cent. Eur. J. Immunol. 2018, 43, 1–8.
- Li, L.; Dang, Q.; Xie, H.; Yang, Z.; He, D.; Liang, L.; Song, W.; Yeh, S.; Chang, C. Infiltrating Mast Cells Enhance Prostate Cancer Invasion via Altering LncRNA-HOTAIR/PRC2-Androgen Receptor (AR)-MMP9 Signals and Increased Stem/Progenitor Cell Population. Oncotarget 2015, 6, 14179–14190.
- Globa, T.; Şptefrţi, L.; Ceauşu, R.A.; Gaje, P.; Cimpean, A.M.; Raica, M. Mast Cell Phenotype in Benign and Malignant Tumors of the Prostate. Pol. J. Pathol. 2014, 65, 147–153.
- Sullivan, H.H.; Heaphy, C.M.; Kulac, I.; Cuka, N.; Lu, J.; Barber, J.R.; de Marzo, A.M.; Lotan, T.L.; Joshu, C.E.; Sfanos, K.S. High Extratumoral Mast Cell Counts Are Associated with a Higher Risk of Adverse Prostate Cancer Outcomes. Cancer Epidemiol. Biomark. Prev. 2020, 29, 668–675.
- Nonomura, N.; Takayama, H.; Nishimura, K.; Oka, D.; Nakai, Y.; Shiba, M.; Tsujimura, A.; Nakayama, M.; Aozasa, K.; Okuyama, A. Decreased Number of Mast Cells Infiltrating into Needle Biopsy Specimens Leads to a Better Prognosis of Prostate Cancer. Br. J. Cancer 2007, 97, 952–956.
- Lin, Y.; Xu, J.; Lan, H. Tumor-Associated Macrophages in Tumor Metastasis: Biological Roles and Clinical Therapeutic Applications. J. Hematol. Oncol. 2019, 12, 76.
- Biswas, S.K.; Mantovani, A. Macrophage Plasticity and Interaction with Lymphocyte Subsets: Cancer as a Paradigm. Nat. Immunol. 2010, 11, 889–896.
- Qian, B.Z.; Pollard, J.W. Macrophage Diversity Enhances Tumor Progression and Metastasis. Cell 2010, 141, 39–51.
- Movahedi, K.; Laoui, D.; Gysemans, C.; Baeten, M.; Stangé, G.; Den Van Bossche, J.; Mack, M.; Pipeleers, D.; In’t Veld, P.; De Baetselier, P.; et al. Different Tumor Microenvironments Contain Functionally Distinct Subsets of Macrophages Derived from Ly6C(High) Monocytes. Cancer Res. 2010, 70, 5728–5739.
- Laoui, D.; Movahedi, K.; van Overmeire, E.; van den Bossche, J.; Schouppe, E.; Mommer, C.; Nikolaou, A.; Morias, Y.; de Baetselier, P.; van Ginderachter, J.A. Tumor-Associated Macrophages in Breast Cancer: Distinct Subsets, Distinct Functions. Int. J. Dev. Biol. 2011, 55, 861–867.
- Kaplan, R.N.; Riba, R.D.; Zacharoulis, S.; Bramley, A.H.; Vincent, L.; Costa, C.; MacDonald, D.D.; Jin, D.K.; Shido, K.; Kerns, S.A.; et al. VEGFR1-Positive Haematopoietic Bone Marrow Progenitors Initiate the Pre-Metastatic Niche. Nature 2005, 438, 820–827.
- Sceneay, J.; Smyth, M.J.; Möller, A. The Pre-Metastatic Niche: Finding Common Ground. Cancer Metastasis Rev. 2013, 32, 449–464.
- Dai, J.; Lu, Y.; Roca, H.; Keller, J.M.; Zhang, J.; McCauley, L.K.; Keller, E.T. Immune Mediators in the Tumor Microenvironment of Prostate Cancer. Chin. J. Cancer 2017, 36, 29.
- Cenerenti, M.; Saillard, M.; Romero, P.; Jandus, C. The Era of Cytotoxic CD4 T Cells. Front. Immunol. 2022, 13, 867189.
- Miller, A.M.; Lundberg, K.; Özenci, V.; Banham, A.H.; Hellström, M.; Egevad, L.; Pisa, P. CD4+CD25high T Cells Are Enriched in the Tumor and Peripheral Blood of Prostate Cancer Patients. J. Immunol. 2006, 177, 7398–7405.
- Barkin, J.; Rodriguez-Suarez, R.; Betito, K. Association between Natural Killer Cell Activity and Prostate Cancer: A Pilot Study. Can. J. Urol. 2017, 24, 8708–8713.
- Erlandsson, A.; Carlsson, J.; Lundholm, M.; Fält, A.; Andersson, S.O.; Andrén, O.; Davidsson, S. M2 Macrophages and Regulatory T Cells in Lethal Prostate Cancer. Prostate 2019, 79, 363–369.
- Strasner, A.; Karin, M. Immune Infiltration and Prostate Cancer. Front. Oncol. 2015, 5, 128.
- Hu, S.; Li, L.; Yeh, S.; Cui, Y.; Li, X.; Chang, H.-C.; Jin, J.; Chang, C. Infiltrating T Cells Promote Prostate Cancer Metastasis via Modulation of FGF11→miRNA-541→androgen Receptor (AR)→MMP9 Signaling. Mol. Oncol. 2015, 9, 44–57.
- Xiang, P.; Jin, S.; Yang, Y.; Sheng, J.; He, Q.; Song, Y.; Yu, W.; Hu, S.; Jin, J. Infiltrating CD4+ T Cells Attenuate Chemotherapy Sensitivity in Prostate Cancer via CCL5 Signaling. Prostate 2019, 79, 1018–1031.
- Kaur, H.B.; Guedes, L.B.; Lu, J.; Maldonado, L.; Reitz, L.; Barber, J.R.; De Marzo, A.M.; Tosoian, J.J.; Tomlins, S.A.; Schaeffer, E.M.; et al. Association of Tumor-Infiltrating T-Cell Density with Molecular Subtype, Racial Ancestry and Clinical Outcomes in Prostate Cancer. Mod. Pathol. 2018, 31, 1539–1552.
- Kiniwa, Y.; Miyahara, Y.; Wang, H.Y.; Peng, W.; Peng, G.; Wheeler, T.M.; Thompson, T.C.; Old, L.J.; Wang, R.-F. CD8+ Foxp3+ Regulatory T Cells Mediate Immunosuppression in Prostate Cancer. Clin. Cancer Res. 2007, 13, 6947–6958.
- Sfanos, K.S.; Bruno, T.C.; Maris, C.H.; Xu, L.; Thoburn, C.J.; DeMarzo, A.M.; Meeker, A.K.; Isaacs, W.B.; Drake, C.G. Phenotypic Analysis of Prostate-Infiltrating Lymphocytes Reveals TH17 and Treg Skewing. Clin. Cancer Res. 2008, 14, 3254–3261.
- Lebien, T.W.; Thomas, T.F. B Lymphocytes: How They Develop and Function. Blood 2008, 112, 1570–1580.
- Hegde, P.S.; Chen, D.S. Top 10 Challenges in Cancer Immunotherapy. Immunity 2020, 52, 17–35.
- Cui, C.; Wang, J.; Fagerberg, E.; Chen, P.M.; Connolly, K.A.; Damo, M.; Cheung, J.F.; Mao, T.; Askari, A.S.; Chen, S.; et al. Neoantigen-Driven B Cell and CD4 T Follicular Helper Cell Collaboration Promotes Anti-Tumor CD8 T Cell Responses. Cell 2021, 184, 6101–6118.e13.
- Chen, Z.; Huang, Y.; Hu, Z.; Zhao, M.; Li, M.; Bi, G.; Zheng, Y.; Liang, J.; Lu, T.; Jiang, W.; et al. Landscape and Dynamics of Single Tumor and Immune Cells in Early and Advanced-stage Lung Adenocarcinoma. Clin. Transl. Med. 2021, 11, e350.
- Roya, N.; Fatemeh, T.; Faramarz, M.A.; Milad, S.G.; Mohammad-Javad, S.; Najmeh, S.V.; Yousef, M.; Nader, B. Frequency of IL-10+CD19+ B Cells in Patients with Prostate Cancer Compared to Patients with Benign Prostatic Hyperplasia. Afr. Health Sci. 2020, 20, 1264–1272.
- Shao, Y.; Lo, C.M.; Ling, C.C.; Liu, X.B.; Ng, K.T.P.; Chu, A.C.Y.; Ma, Y.Y.; Li, C.X.; Fan, S.T.; Man, K. Regulatory B Cells Accelerate Hepatocellular Carcinoma Progression via CD40/CD154 Signaling Pathway. Cancer Lett. 2014, 355, 264–272.
- Murakami, Y.; Saito, H.; Shimizu, S.; Kono, Y.; Shishido, Y.; Miyatani, K.; Matsunaga, T.; Fukumoto, Y.; Ashida, K.; Sakabe, T.; et al. Increased Regulatory B Cells Are Involved in Immune Evasion in Patients with Gastric Cancer. Sci. Rep. 2019, 9, 13083.
- Carter, N.A.; Rosser, E.C.; Mauri, C. Interleukin-10 Produced by B Cells Is Crucial for the Suppression of Th17/Th1 Responses, Induction of T Regulatory Type 1 Cells and Reduction of Collagen-Induced Arthritis. Arthritis Res. Ther. 2012, 14, R32.
- Shen, P.; Roch, T.; Lampropoulou, V.; O’Connor, R.A.; Stervbo, U.; Hilgenberg, E.; Ries, S.; Dang, V.D.; Jaimes, Y.; Daridon, C.; et al. IL-35-Producing B Cells Are Critical Regulators of Immunity during Autoimmune and Infectious Diseases. Nature 2014, 507, 366–370.
- Tian, J.; Zekzer, D.; Hanssen, L.; Lu, Y.; Olcott, A.; Kaufman, D.L. Lipopolysaccharide-Activated B Cells Down-Regulate Th1 Immunity and Prevent Autoimmune Diabetes in Nonobese Diabetic Mice. J. Immunol. 2001, 167, 1081–1089.
- Voronov, E.; Shouval, D.S.; Krelin, Y.; Cagnano, E.; Benharroch, D.; Iwakura, Y.; Dinarello, C.A.; Apte, R.N. IL-1 Is Required for Tumor Invasiveness and Angiogenesis. Proc. Natl. Acad. Sci. USA 2003, 100, 2645–2650.
- Nguyen, D.P.; Li, J.; Tewari, A.K. Inflammation and Prostate Cancer: The Role of Interleukin 6 (IL-6). BJU Int. 2014, 113, 986–992.
- Bouraoui, Y.; Ricote, M.; García-Tuñón, I.; Rodriguez-Berriguete, G.; Touffehi, M.; Rais, N.B.; Fraile, B.; Paniagua, R.; Oueslati, R.; Royuela, M. Pro-Inflammatory Cytokines and Prostate-Specific Antigen in Hyperplasia and Human Prostate Cancer. Cancer Detect. Prev. 2008, 32, 23–32.
- Cheng, J.; Li, L.; Liu, Y.; Wang, Z.; Zhu, X.; Bai, X. Interleukin-1α Induces Immunosuppression by Mesenchymal Stem Cells Promoting the Growth of Prostate Cancer Cells. Mol. Med. Rep. 2012, 6, 955–960.
- Adler, H.L.; McCURDY, M.A.; Kattan, M.W.; Timme, T.L.; Scardino, P.T.; Thompson, T.C. Elevated levels of circulating interleukin-6 and transforming growth factor-p1 in patients with metastatic prostatic carcinoma. J. Urol. 1999, 161, 182–187.
- Nakashima, J.; Tachibana, M.; Horiguchi, Y.; Oya, M.; Ohigashi, T.; Asakura, H.; Murai, M. Serum Interleukin 6 as a Prognostic Factor in Patients with Prostate Cancer. Clin. Cancer Res. 2000, 6, 2702–2706.
- Sugimoto, Y.; Hirota, M.; Yoshikawa, K.; Sumitomo, M.; Nakamura, K.; Ueda, R.; Niwa, R.; Suzawa, T.; Yamasaki, M.; Shitara, K.; et al. The Therapeutic Potential of a Novel PSMA Antibody and Its IL-2 Conjugate in Prostate Cancer. Anticancer Res. 2014, 34, 89–97.
- Wise, G.J.; Marella, V.K.; Talluri, G.; Shirazian, D. Cytokine variations in patients with hormone treated prostate cancer. J. Urol. 2000, 164, 722–725.
- Seol, M.A.; Kim, J.H.; Oh, K.; Kim, G.; Seo, M.W.; Shin, Y.K.; Sim, J.H.; Shin, H.M.; Seo, B.Y.; Lee, D.S.; et al. Interleukin-7 Contributes to the Invasiveness of Prostate Cancer Cells by Promoting Epithelial–Mesenchymal Transition. Sci. Rep. 2019, 9, 6917.
- Liu, L.; Li, Q.; Han, P.; Li, X.; Zeng, H.; Zhu, Y.; Wei, Q. Evaluation of Interleukin-8 in Expressed Prostatic Secretion as a Reliable Biomarker of Inflammation in Benign Prostatic Hyperplasia. Urology 2009, 74, 340–344.
- Culig, Z. Response to Androgens and Androgen Receptor Antagonists in the Presence of Cytokines in Prostate Cancer. Cancers 2021, 13, 2944.
- Steiner, G.E.; Newman, M.E.; Paikl, D.; Stix, U.; Memaran-Dagda, N.; Lee, C.; Marberger, M.J. Expression and Function of Pro-Inflammatory Interleukin IL-17 and IL-17 Receptor in Normal, Benign Hyperplastic, and Malignant Prostate. Prostate 2003, 56, 171–182.
- Wang, X.; Yang, L.; Huang, F.; Zhang, Q.; Liu, S.; Ma, L.; You, Z. Inflammatory Cytokines IL-17 and TNF-α up-Regulate PD-L1 Expression in Human Prostate and Colon Cancer Cells. Immunol. Lett. 2017, 184, 7–14.
- Park, J.I.; Lee, M.G.; Cho, K.; Park, B.J.; Chae, K.S.; Byun, D.S.; Ryu, B.K.; Park, Y.K.; Chi, S.G. Transforming Growth Factor-Β1 Activates Interleukin-6 Expression in Prostate Cancer Cells through the Synergistic Collaboration of the Smad2, P38-NF-ΚB, JNK, and Ras Signaling Pathways. Oncogene 2003, 22, 4314–4332.
- Wang, H.; Fang, R.; Wang, X.F.; Zhang, F.; Chen, D.Y.; Zhou, B.; Wang, H.S.; Cai, S.H.; Du, J. Stabilization of Snail through AKT/GSK-3β Signaling Pathway Is Required for TNF-α-Induced Epithelial-Mesenchymal Transition in Prostate Cancer PC3 Cells. Eur. J. Pharmacol. 2013, 714, 48–55.
- Mariathasan, S.; Turley, S.J.; Nickles, D.; Castiglioni, A.; Yuen, K.; Wang, Y.; Kadel, E.E.; Koeppen, H.; Astarita, J.L.; Cubas, R.; et al. TGFβ Attenuates Tumour Response to PD-L1 Blockade by Contributing to Exclusion of T Cells. Nature 2018, 554, 544–548.
- Chen, X.H.; Liu, Z.C.; Zhang, G.; Wei, W.; Wang, X.X.; Wang, H.; Ke, H.P.; Zhang, F.; Wang, H.S.; Cai, S.H.; et al. TGF-β and EGF Induced HLA-I Downregulation Is Associated with Epithelial-Mesenchymal Transition (EMT) through Upregulation of Snail in Prostate Cancer Cells. Mol. Immunol. 2015, 65, 34–42.
- Zhang, F.; Lee, J.; Lu, S.; Pettaway, C.A.; Dong, Z. Blockade of Transforming Growth Factor-B Signaling Suppresses Progression of Androgen-Independent Human Prostate Cancer in Nude Mice. Clin. Cancer Res. 2005, 11, 4512–4520.
- Kramer, G.; Steiner, G.E.; Handisurya, A.; Stix, U.; Haitel, A.; Knerer, B.; Gessl, A.; Lee, C.; Marberger, M. Increased Expression of Lymphocyte-Derived Cytokines in Benign Hyperplastic Prostate Tissue, Identification of the Producing Cell Types, and Effect of Differentially Expressed Cytokines on Stromal Cell Proliferation. Prostate 2002, 52, 43–58.
- Tuxhorn, J.A.; McAlhany, S.J.; Yang, F.; Dang, T.D.; Rowley, D.R. Inhibition of Transforming Growth Factor-Beta Activity Decreases Angiogenesis in a Human Prostate Cancer-Reactive Stroma Xenograft Model. Cancer Res. 2002, 62, 6021–6025.
- Gillessen, S.; Naumov, Y.N.; Nieuwenhuis, E.E.S.; Exley, M.A.; Lee, F.S.; Mach, N.; Luster, A.D.; Blumberg, R.S.; Taniguchi, M.; Balk, S.P.; et al. CD1d-Restricted T Cells Regulate Dendritic Cell Function and Antitumor Immunity in a Granulocyte-Macrophage Colony-Stimulating Factor-Dependent Fashion. Proc. Natl. Acad. Sci. USA 2003, 100, 8874–8879.
- Mo, L.; Zhang, X.; Shi, X.; Wei, L.; Zheng, D.; Li, H.; Gao, J.; Li, J.; Hu, Z. Norcantharidin Enhances Antitumor Immunity of GM-CSF Prostate Cancer Cells Vaccine by Inducing Apoptosis of Regulatory T Cells. Cancer Sci. 2018, 109, 2109–2118.
- Kiu, H.; Nicholson, S.E. Biology and Significance of the JAK/STAT Signalling Pathways. Growth Factors 2012, 30, 88–106.
- Li, W.X. Canonical and Non-Canonical JAK-STAT Signaling. Trends Cell Biol. 2008, 18, 545–551.
- Liu, X.; He, Z.; Li, C.H.; Huang, G.; Ding, C.; Liu, H. Correlation Analysis of JAK-STAT Pathway Components on Prognosis of Patients with Prostate Cancer. Pathol. Oncol. Res. 2012, 18, 17–23.
- Gao, B.; Shen, X.; Kunos, G.; Meng, Q.; Goldberg, I.D.; Rosen, E.M.; Fan, S. Constitutive Activation of JAK-STAT3 Signaling by BRCA1 in Human Prostate Cancer Cells. FEBS Lett. 2001, 488, 179–184.
- Zhu, M.L.; Kyprianou, N. Androgen Receptor and Growth Factor Signaling Cross-Talk in Prostate Cancer Cells. Endocr. Relat. Cancer 2008, 15, 841–849.
- Bishop, J.L.; Thaper, D.; Zoubeidi, A. The Multifaceted Roles of STAT3 Signaling in the Progression of Prostate Cancer. Cancers 2014, 6, 829–859.
- Xiao, W.; Hodge, D.R.; Wang, L.; Yang, X.; Zhang, X.; Farrar, W.L. Co-Operative Functions between Nuclear Factors NFκB and CCAT/Enhancer-Binding Protein-β (C/EBP-β) Regulate the IL-6 Promoter in Autocrine Human Prostate Cancer Cells. Prostate 2004, 61, 354–370.
- Jin, R.; Yi, Y.; Yull, F.E.; Blackwell, T.S.; Clark, P.E.; Koyama, T.; Smith, J.A.; Matusik, R.J. Nf-Kb Gene Signature Predicts Prostate Cancer Progression. Cancer Res. 2014, 74, 2763–2772.
- Ramalingam, S.; Ramamurthy, V.P.; Njar, V.C.O. Dissecting Major Signaling Pathways in Prostate Cancer Development and Progression: Mechanisms and Novel Therapeutic Targets. J. Steroid Biochem. Mol. Biol. 2017, 166, 16–27.
- Lemmon, M.A.; Schlessinger, J. Cell Signaling by Receptor Tyrosine Kinases. Cell 2010, 141, 1117–1134.
- Reynolds, A.R.; Kyprianou, N. Growth Factor Signalling in Prostatic Growth: Significance in Tumour Development and Therapeutic Targeting. Br. J. Pharmacol. 2006, 147, 144–152.
- Derynck, R.; Akhurst, R.J.; Balmain, A. TGF-β Signaling in Tumor Suppression and Cancer Progression. Nat. Genet. 2001, 29, 117–129.
- Peraldo-Neia, C.; Migliardi, G.; Mello-Grand, M.; Montemurro, F.; Segir, R.; Pignochino, Y.; Cavalloni, G.; Torchio, B.; Mosso, L.; Chiorino, G.; et al. Epidermal Growth Factor Receptor (EGFR) Mutation Analysis, Gene Expression Profiling and EGFR Protein Expression in Primary Prostate Cancer. BMC Cancer 2011, 11, 31.
- Fresno Vara, J.Á.; Casado, E.; de Castro, J.; Cejas, P.; Belda-Iniesta, C.; González-Barón, M. P13K/Akt Signalling Pathway and Cancer. Cancer Treat. Rev. 2004, 30, 193–204.
- Gao, N.; Zhang, Z.; Jiang, B.H.; Shi, X. Role of PI3K/AKT/MTOR Signaling in the Cell Cycle Progression of Human Prostate Cancer. Biochem. Biophys. Res. Commun. 2003, 310, 1124–1132.
- Steiner, H.; Godoy-Tundidor, S.; Rogatsch, H.; Berger, A.P.; Fuchs, D.; Comuzzi, B.; Bartsch, G.; Hobisch, A.; Culig, Z. Accelerated in Vivo Growth of Prostate Tumors That Up-Regulate Interleukin-6 Is Associated with Reduced Retinoblastoma Protein Expression and Activation of the Mitogen-Activated Protein Kinase Pathway. Am. J. Pathol. 2003, 162, 655–663.
- Cargnello, M.; Roux, P.P. Activation and Function of the MAPKs and Their Substrates, the MAPK-Activated Protein Kinases. Microbiol. Mol. Biol. Rev. 2011, 75, 50–83.
- Mbatia, H.W.; Ramalingam, S.; Ramamurthy, V.P.; Martin, M.S.; Kwegyir-Afful, A.K.; Njar, V.C.O. Novel C-4 Heteroaryl 13- Cis -Retinamide Mnk/AR Degrading Agents Inhibit Cell Proliferation and Migration and Induce Apoptosis in Human Breast and Prostate Cancer Cells and Suppress Growth of MDA-MB-231 Human Breast and CWR22Rv1 Human Prostate Tumor Xenografts in Mice. J. Med. Chem. 2015, 58, 1900–1914.
This entry is offline, you can click here to edit this entry!
