Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Nanobubbles also known as ultra-fine bubbles or sub-micron bubbles, are extremely small gas-filled spherical bodies enclosed by an interface (gas–liquid or gas–solid) with a diameter of less than 1000 nm (i.e., ranging in size from tens to hundreds of nanometers in diameter). In other words, nanobubbles are composed of gas, such as oxygen or nitrogen, encapsulated within a liquid, typically water, and stabilized by a thin layer of molecules at the gas–liquid interface.
- oxygen
- nanobubbles
- hypoxia
- mitochondria
- metabolism
1. Introduction
Hypoxia is a medical condition characterized by an inadequate/insufficient oxygen supply to tissues and organs, leading to potential harm or dysfunction. It results from various pathological phenomena, including diminished cardiac output, reduced hemoglobin concentration, and an atypical vasculature network. Currently, there is a widely acknowledged consensus that restricted oxygen availability jeopardizes not only cell viability but also cellular phenotype and functionality [1]. Furthermore, inadequate oxygen supply (hypoxia: different levels of oxygen deprivation, from mild to severe; hypoxic stress: in cellular or molecular biology research, this specifically refers to a situation where cells or tissues are exposed to lower-than-optimal oxygen levels, causing them to adapt or respond to this oxygen deficiency, where they may activate specific molecular pathways, like the hypoxia-inducible factor or HiF pathway to adapt to the low oxygen environment) may disturb numerous biological processes, particularly those requiring high energy demands, such as tissue repair, restoration, and regeneration. Upon establishing hypoxia, it could lead to a grave metabolic crisis, especially in individuals with metabolic syndromes, cardiovascular diseases, acute traumas, chronic diseases, and cancer, to list a few [2][3][4][5].
Today, it is widely recognized that exposing human blood to free oxygen gas bubbles causes hemolysis [6]. Nonetheless, various approaches have been employed to increase oxygen levels and overcome hypoxia, including the utilization of oxygen microparticles, hydrogen peroxide solutions, hyperbaric oxygenation, and inhalation of gas mixtures such as carbogen (composed of 5% CO2 and 95% O2) [7]. However, they are considered ineffective due to clinical and logistical limitations. These approaches include limited gas solubility, cytotoxicity, instability, low gas pay-load, ill-defined gas-release profiles, and low cellular uptake, rendering them unsuitable for gas delivery. Consequently, an imminent need for novel methodologies and strategies arises that can offer and/or provide oxygen to tissues and organs in a manner that is both safe/biocompatible as well as cost-effective whilst being highly biodegradable and easily deployable or user-friendly too [7].
Herein, nanobubbles, also known as ultra-fine bubbles or sub-micron bubbles, are extremely small gas-filled spherical bodies enclosed by an interface (gas–liquid or gas–solid) with a diameter of less than 1000 nm (i.e., ranging in size from tens to hundreds of nanometers in diameter) [8][9]. In other words, nanobubbles are composed of gas, such as oxygen or nitrogen, encapsulated within a liquid, typically water, and stabilized by a thin layer of molecules at the gas–liquid interface. Nanobubbles have gained significant attention in various fields, including medicine, materials science, and environmental science due to their unique properties and potential applications. Indeed, several remarkable properties of these nanobubbles have been widely studied, such as their long-term stability and longevity [10][11][12], enhanced dissolution of gases, and increased gas interfacial diffusion [13][14]. In addition, nanobubbles can be biocompatible and have demonstrated interesting potential as suitable candidates for drug delivery purposes (drugs, genes, or other therapeutic agents, with improved release and delivery to specific tissues or cells) [7][15]. Given that an optimal oxygen-release material must demonstrate compatibility with biological systems, strategies for oxygen delivery, such as nanobubbles, have garnered substantial attention as robust mechanisms to alleviate the hypoxic stress response(s) [16][17], which is an exciting and growing area of research, development, and innovation in various scientific, medical, and industrial fields, in laboratories World-wide.
Notably, during the past decade, there has been a remarkable increase in scientific (and technological) interest and a surge in publications in this emerging field (Figure 1), particularly concerning oxygen nanobubbles (OnB) and OnB-mediated oxygenation.
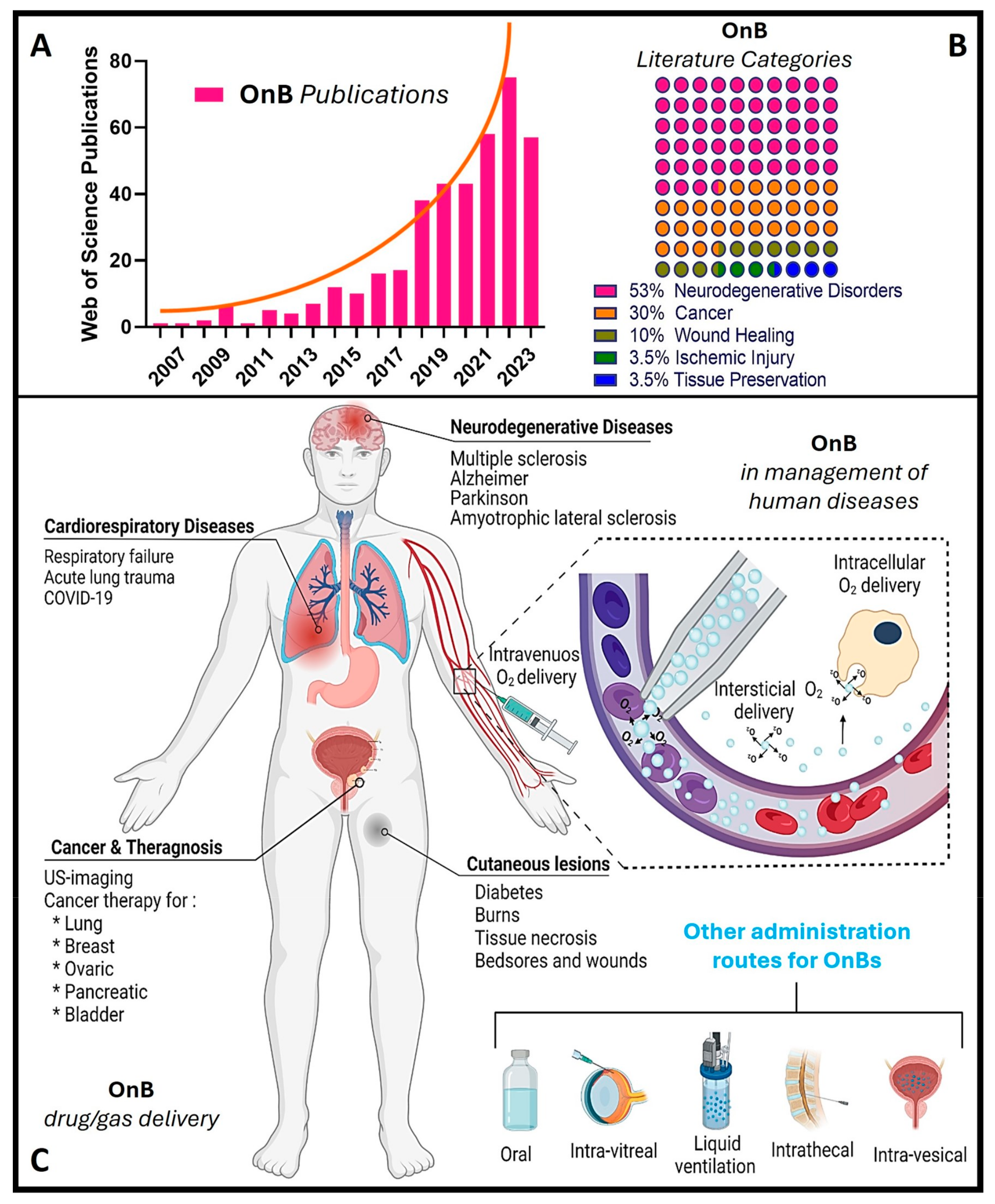
Figure 1. OnB (oxygen nanobubbles) and biomedicine research. (A) The number of articles published each year retrieved through a scientific search engine (Web of Science) using the term “oxygen nanobubble” followed by filtration/classification for application(s) in biomedicine. (B) The percentage of OnB articles published categorized by indication. (C) Illustration of drug/gas delivery based on OnB and their commonly investigated administration route(s) for the management of disease(s).
Nonetheless, it is widely accepted that metabolism and oxygen levels act as intricately interlinked factors that determine the physiological behavior of cells, the onset of pathologies, and the progression of chronic diseases. Among the advantages of developing oxygenation systems, selective metabolic reprogramming can target (1) tumor suppression in cancer by disrupting their ability to adapt to low oxygen conditions [18], (2) enhanced wound healing by stimulating angiogenesis and oxygen utilization in affected tissues [19], and (3) restoration of mitochondrial function in neurodegenerative diseases through promoting the alternative energy production pathways and reducing oxidative stress, i.e., most likely slowing disease progression and enhancing neuronal survival [20].
2. Hypoxia and Mitochondrial Dysfunction: Interplay and Implications in Pathology(ies)
The interplay between hypoxia (oxygen deficiency) and mitochondrial dysfunction (perturbing the cellular energy-producing organelles) is pivotal in the development and progression of various human diseases. These intertwined processes, once considered distinct, have emerged as key players in conditions spanning cancer, cardiovascular diseases, neuro-degenerative disorders, and more. Hypoxia disrupts the balance of oxygen supply to tissues, triggering a cascade of cellular responses. Simultaneously, mitochondrial dysfunction, characterized by impaired energy production and oxidative stress, contributes to cellular damage and dysfunction. The dynamic interaction between these two phenomena amplifies disease mechanisms and influences treatment outcomes.
The normal partial pressure of oxygen (PaO2) in arterial blood typically falls within the range of 75 to 100 mm of mercury (mmHg) when measured at sea level and standard atmospheric pressure, exhibiting variability across various tissues. Normal tissue oxygen levels can range from 0 to around 60 mmHg, but these values can fluctuate significantly depending on the specific tissue type and its physiological conditions. Measuring PaO2 directly within cells can be challenging and typically requires specialized techniques, such as microelectrode oxygen sensors or imaging methods. These measurements are valuable for understanding tissue oxygenation in various physiological and pathological conditions and can guide treatment strategies in clinical settings. In most cases, the physiological range spans from 14 to 65 mmHg, corresponding to atmospheric oxygen levels of 2–9% [21]. Distinct PaO2 gradients are also evident across both superficial and deep cellular layers, displaying diverse characteristics from vascular lumen to perivascular tissues (Figure 2A) [21][22]. On the other hand, cellular hypoxia occurs when the oxygen level is in the range of 10–16 mmHg (1–2%), and it depends on the type of tissue [23]. Once the demand for oxygen exceeds its supply, the oxygen level decreases (herein known as hypoxia) in local tissues and/or peripherical tissues, leading to a metabolic crisis [24][25] (Figure 2B,C).
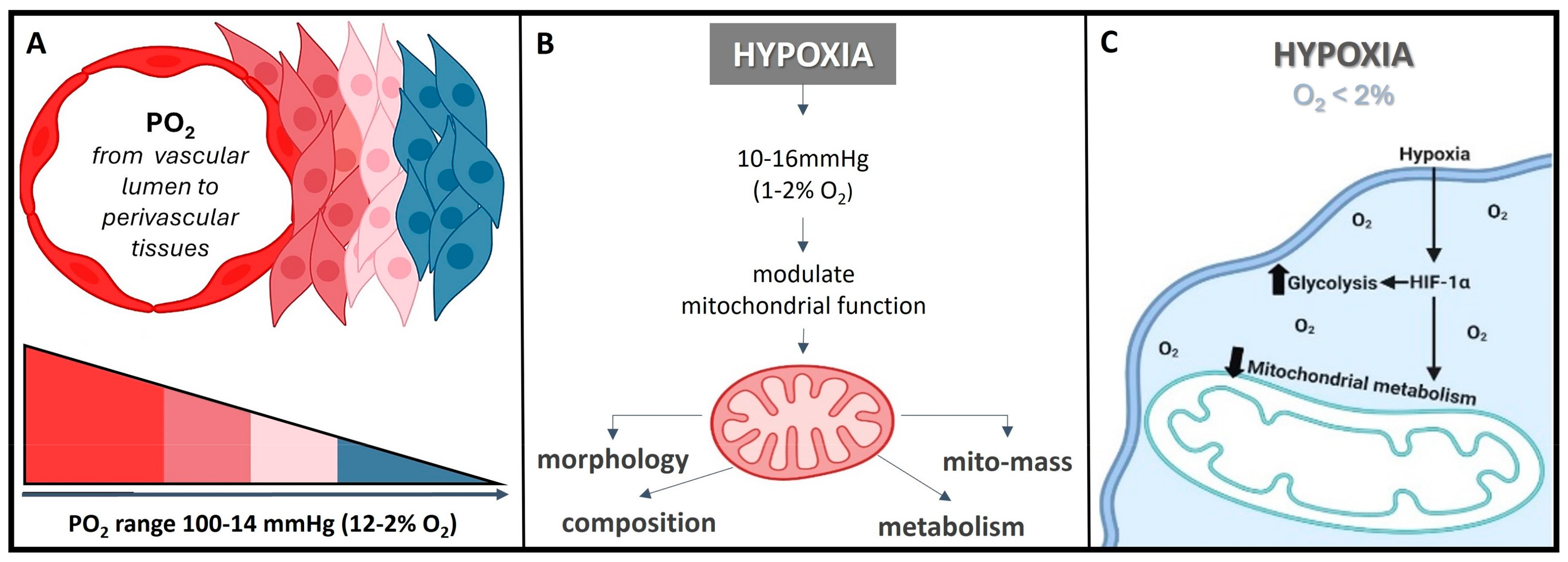
Figure 2. O2 need depends on cell niche amongst other demands. (A) Sites that are distant from major blood vessels are usually kept at a lower O2 level. However, low PO2 might still be higher than the PO2 of truly hypoxic conditions <16 mmHg (<2% O2). (B) Hypoxia acts on mitochondrial function, affecting the protein composition of the electron transport chain, mitochondrial morphology, and mitochondrial mass. (C) Hypoxia, besides playing a crucial role in influencing the cellular signaling pathways, gene expression, and the overall microenvironment, it regulates metabolic adaptation, promotes glycolysis, and decreases mitochondrial respiration, including HiF stabilization.
Herein, the hypoxia-inducible factor or HiF is a heterodimeric transcription factor that plays a central role in cellular adaptation to low oxygen levels. It is considered a master regulator of the cellular response to hypoxia and plays a crucial role in maintaining oxygen homeostasis in various tissues and cells [26]. Briefly, the HiF is composed of two sub-units: HiF-α (alpha) and HiF-β (beta). The alpha subunit is oxygen-sensitive and exists in different isoforms, with HiF-1α and HiF-2α being the most well-known. In normoxic (oxygen-rich) conditions, prolyl hydroxylase enzymes modify HiF-α, marking it for degradation. However, in hypoxic conditions, this degradation is inhibited, allowing HiF-α to accumulate and form a heterodimer with HiF-β. This active HiF complex then translocates to the cell nucleus, where it binds to specific DNA sequences known as hypoxia-response elements or HREs and initiates the transcription of genes involved in various adaptive responses to low oxygen, including angiogenesis, erythropoiesis, glycolysis, and more. It is noteworthy to mention that the HiF ability to regulate the expression of genes involved in oxygen homeostasis and adaptation to hypoxia makes it a critical factor in various physiological and pathological processes, including embryogenesis, ischemic diseases, cancer, and metabolic disorders. Understanding the role of HiF and its downstream targets is essential for gaining insights into how cells and organisms respond to changing oxygen levels and for developing therapeutic strategies to address related diseases and conditions [26]. Remember that once the oxygen decreases below physiological levels, cells trigger a hypoxic cell response, characterized by the HiF-1α stabilization and its nuclear transcription (Figure 3A). Consequently, the translocation of HiF-1α leads to the activation of approximately 400 target genes. This activation influences metabolic processes by upregulating proteins associated with glycolysis while concurrently suppressing oxygen-dependent pathways, particularly those related to mitochondrial metabolism [27][28]. These findings establish a direct connection between mitochondria and HiF pathway, highlighting interdependence in response to changes in oxygen supply and demand.
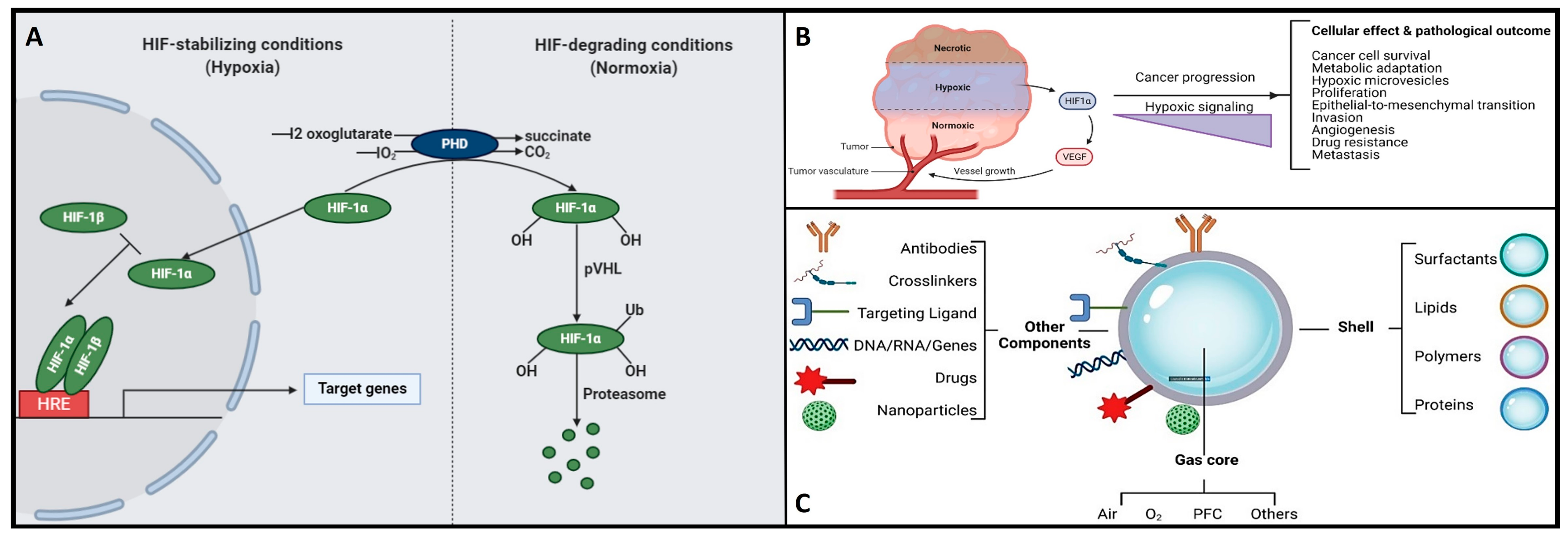
Figure 3. (A) The functional interplay of the HiF pathway in response to different oxygen concentrations. Herein, under hypoxic conditions, PHD is inhibited, and HiF-α is stabilized, which then translocates to the nucleus, dimerizes with its corresponding β-sub-unit, and recognizes the HREs motifs to induce the transcription of several target genes. On the other hand, when under normoxia, HiF1α-subunit prolyl residues are continuously hydroxylated by PHD. Following hydroxylation, pVHL binds and targets the HiF-1 α-subunit for ubiquitination and proteasomal degradation. (B) Hypoxic signaling and HiF-1α over-expression: a key hallmark in cancer progression. (C) Schematic representation of the structure and composition of engineering shelled micro/nanobubble systems.
Hypoxia exerts diverse effects on mitochondria, but mitochondria also play a role in modulating the cellular response to hypoxia [24][29]. Mitochondria serve as the primary oxygen sensors within cells, and their function is profoundly impacted by decreased oxygen levels. Concurrently, hypoxia influences various mitochondrial processes, including fusion, fission, mitophagy, and oxidative phosphorylation (OXPHOS) [24]. Intriguingly, the inhibition of mitochondrial function can reverse the activation of the HiF pathway induced by hypoxia [29]. Such observations prompted researchers to propose a role for the mitochondria in metabolic re-programing and in modulating HiF activation. The use of liposomes loaded with an anti-tumor drug to inhibit mitochondrial complexes I and II showed a significant reduction in OXPHOS metabolism and oxygen consumption [18]. Consequently, higher oxygen levels inside the cell are critical to reverse HiF activation. In general, these strategies have been reported to sensitize radiotherapy or photodynamic therapy (PDT) in tumors via enhancing selective DNA damage [18][30] rather than endogenous O2 delivery (perfluorocarbon emulsion or nanobubbles) or inhibitors of mitochondrial complexes, which may even complement each other, implicating O2 re-distribution, facilitating the degradation of HiF, inhibiting DNA repair, and facilitating anti-tumoral immunological response. In essence, mitochondria and the cellular response to hypoxia are two inter-connected systems sensitive to oxygen levels. When HiF-1α is degraded, it promotes mitochondrial metabolism, typically observed in normoxic conditions [29]. Conversely, when HiF-1α is stabilized, it triggers the expression of proteins that, in turn, suppress the mitochondrial metabolism, a characteristic response in a hypoxic condition.
On the other hand, hypoxia is also implicated in the development and severity of numerous age-related and chronic diseases, including cancer and neurodegenerative conditions [5][7][31][32][33]. In the context of cancer, cellular responses to hypoxia play a pivotal role in various aspects of tumor progression, encompassing cancer cell survival, proliferation, epithelial-to-mesenchymal transition (EMT), invasion, angiogenesis, drug resistance, and metastasis [34][35]. Additionally, hypoxic tumors display greater resistance to chemotherapy, radiotherapy, and photodynamic therapy, ultimately resulting in a poorer prognosis and increased patient mortality [34][35]. Please refer to Figure 3B for further insights. Conversely, the central nervous system (CNS) is particularly susceptible to acute hypoxia and can become compromised rapidly, triggering an ischemic cascade. The brain is highly vulnerable to oxidative stress and mitochondrial impairment in hypoxic conditions. Consequently, it experiences neurodegeneration due to energy deficits. Recent research has shed light on significant alterations in mitochondrial balance within the CNS of individuals suffering from chronic neurodegenerative disorders [33][36][37][38][39][40]. As a result, neurodegeneration is characterized by mitochondrial dysfunction, including reduced activity of complex I, cytochrome oxidase, the electron transport chain (ETC), and the occurrence of mitochondrial DNA mutations. This neuroinflammatory degeneration has been extensively studied in the context of conditions such as Parkinson’s disease (PD), Alzheimer’s disease (AD), Huntington’s disease, and hereditary neuropathy [33][36][37][38][39][40]. Herein, as various neurodegenerative diseases share a common association with mitochondrial dysfunction, there is a pressing need to address, repair, and enhance mitochondrial bio-genesis. Within this context, strategies centered on oxygen delivery, such as nanobubbles, have attracted significant attention due to their potential to mitigate hypoxic stress responses and rejuvenate mitochondrial function. To gain a good understanding of the mechanisms and potential applications of nanobubbles in fields like biomedicine, neuroscience, and oncology, it is essential to discover the fundamental principles governing the hypoxia-inducible factor (HiF) pathway and mitochondrial metabolism. Likewise, nanobubbles hold promise in various applications, primarily due to their stability in oxygenation processes, enabling efficient gas exchange within a defined volume. This capacity has the potential to enhance mitochondrial metabolism, alleviate hypoxia, and augment oxygen supply, which is particularly valuable in chronic conditions like cancer, neurodegenerative diseases, and chronic inflammation, amongst others. Herein, to unlock the maximum potential and grasp the mechanism underlying the behavior of OnB in biomedicine, the next section presents foundational principles that govern their bio-performance.
3. Nanobubbles in Biomedicine: Bridging Basic Fundamentals to Practical Application(s)
Nanobubbles are minuscule gas-filled spherical entities enclosed by an interface (gas–liquid or gas–solid) with diameters typically in the nanometer range, spanning from tens to hundreds of nanometers [8][9]. They represent a distinct class of bubbles, significantly smaller than conventional microbubbles, and are characterized by their unique properties at the nanoscale. In the realm of nanotechnology, nanobubbles have recently garnered considerable attention, mainly attributed to their exceptional stability and longevity. This remarkable stability arises from the presence of a thin molecular layer at the gas–liquid interface, which effectively confines the gas within the liquid medium. As a result, nanobubbles exhibit prolonged lifetimes, making them pertinent for biomedicine in various scientific and technological applications, including drug delivery [41][42], gene delivery [43], cancer immunotherapy and chemotherapy [34][35], wound healing, and tissue regeneration, through the intravenous delivery of oxygen carriers such as OnB [44]. Furthermore, nanobubbles have displayed proficiency in surface modification and cleaning at the nanoscale. Their presence can facilitate the detachment of nanosized contaminants or particles from solid surfaces, offering potential applications in surface engineering, nanofabrication, and precision cleaning processes. In the field of medical imaging, nanobubbles serve as contrast agents for advanced ultrasound imaging techniques. Their nanoscale dimensions allow for improved resolution and specificity in visualizing biological tissues, vasculature, and cellular structures. Beyond the biomedical realm, it is perhaps worth mentioning to the interested reader that nanobubbles have promising environmental applications, including groundwater remediation and wastewater treatment, where their ability to facilitate gas transfer and enhance chemical reactions is leveraged to address various environmental challenges. Henceforth, the multi-faceted properties and applications of nanobubbles will continue to be further explored, fueling ongoing research endeavors and innovation across diverse scientific and technological disciplines [41][42][43][44].
Various techniques enable the synthesis of nanobubbles, including hydrodynamic cavitation, acoustic cavitation, microfluidics, intense mechanical agitation, and electrolysis, among others. What sets nanobubbles apart from other bubbles are several defining parameters, with bubble diameter being the most critical classification criterion, as outlined by the ISO/TC 281 [45] and ISO 20480-1:2017 [46] standards for terminology and definitions in the R&D&I area of fine bubble technology(ies). The growing interest in nanobubbles across numerous fields stems not only from their expansive surface area and reactivity when compared to macrobubbles and microbubbles but also from their remarkable attributes, including a heightened gas diffusion rate, enhanced cellular uptake, and robust stability against coalescence, collapse, or rupture. These qualities enable nanobubbles to persist in liquid environments for extended periods, spanning several weeks [12][47].
3.1. Bubble Size and Physico-Chemico-Mechanical Properties
As mentioned previously, the size of nanobubbles, typically falling within the range of 100 to 250 nm, serves as a fundamental parameter that imparts them with inherent and advantageous physico-chemical characteristics [12][47]. This size distribution proves highly advantageous in nanobubble preparations, as it aids in overcoming infiltration limitations and facilitates the disruption of the blood–brain barrier [48]. A noteworthy feature of nanobubbles is their ability to effectively target sites of inflammation, such as those observed in cancer. This efficacy is attributed to the pore cutoff size of the fenestrated endothelium, which ranges from 380 to 780 nm. This mechanism, known as the enhanced permeability and retention effect (EPR effect basically describes the abnormal characteristics of the blood vessels associated with pathological conditions, particularly in cancer and inflammation), represents a passive targeting strategy that capitalizes on the efficient accumulation of nanobubbles within tissues featuring elevated vascular permeability [7][15]. To simplify, in the context of EPR (leaky blood vessels and the unique properties of the TME to allow therapeutic agents to accumulate preferentially in cancerous tissue while sparing healthy tissue) and nanobubbles, passive targeting means that nanobubbles, due to their size and other properties, can effectively accumulate in areas of disease, such as tumors or inflamed tissues, without the need for active targeting mechanisms. This passive accumulation can enhance the delivery of therapeutic agents or imaging agents to the specific site of interest, contributing to the effectiveness of various medical interventions.
Furthermore, their mechanical properties also contribute significantly to their unique characteristics and potential applications. One notable mechanical property is the elevated internal pressure within nanobubbles. Due to their extremely small size, nanobubbles can contain gas at significantly higher pressures compared to larger bubbles. This heightened internal pressure is a result of the Laplace overpressure, which is inversely proportional to the radius of the bubble. Despite this theoretical thermos-dynamic instability, nanobubbles often exhibit impressive stability, remaining intact for extended periods. The mechanical robustness of nanobubbles, particularly their resistance to coalescence, collapse, or rupture, is another noteworthy trait. This resilience against external forces makes them suitable for various applications where durability is crucial. Indeed, understanding the stability of nanosized bubbles can be partially elucidated through their underlying physical behavior, as elaborated below. When any bubble forms within a solution, it creates an interface defined by the surface tension (γ) [49]. The existence of nanobubbles has generated extensive discussion, partly due to the predicted Laplace overpressure associated with them, which can reach several atmospheres, theoretically rendering them thermos-dynamically unstable [49]. However, in contrast to macro- or microbubbles, nanobubbles often exhibit robust persistence characterized by reduced buoyancy, enabling them to remain intact for days, weeks, or even months [12][50]. Additionally, nanobubbles exhibit unique acoustic properties, which have implications in diagnostic imaging and therapeutic ultrasound. Their response to acoustic waves differs from that of microbubbles, opening new opportunities for innovative ultrasound-based techniques. The remarkable attributes defining nanobubbles, including their diminutive size, high surface-to-volume ratio, elevated internal pressure, prolonged stability, electrostatic charge properties, acoustic characteristics, and biocompatibility, have spurred intensive research into their potential across various biomedical applications [51]. In the realm of biomedicine, the combination of nanobubbles’ mechanical attributes, such as their size, stability, and response to external forces, along with their biocompatibility, has led to intensive research into their potential across diverse applications. These mechanical properties, along with their other afore-mentioned physico-chemico-mechanical characteristics, position nanobubbles as a promising platform for advancing various fields, from drug delivery to medical imaging.
3.2. Structural Composition and Electrostatic Charge of OnB Affects Gas Core and Diffusion
The gas core of a nanobubble is the central part filled with a gas, typically oxygen or nitrogen [6]. This gas core is encapsulated by a thin layer of liquid, often water, which forms the bubble’s shell. The gas core plays a crucial role in the unique properties and applications of nanobubbles. (1) High internal pressure: Nanobubbles exhibit significantly higher internal pressure compared to their larger counterparts, such as microbubbles. This increased pressure is a consequence of the Laplace overpressure, which is inversely proportional to the radius of the bubble. The small size of nanobubbles results in elevated internal pressures, which can have implications for their stability and behavior. (2) Gas diffusion: The gas core of nanobubbles allows for enhanced gas diffusion in liquids. This property is particularly relevant in applications where gas delivery, such as oxygen, to specific biological targets is essential. Nanobubbles can serve as carriers for gases, facilitating their transport to desired locations. The composition of the gas core is another critical factor influencing the stability of nanobubbles [6]. The introduction of gases like perfluorocarbons (PFCs), especially when combined with oxygen, leads to a reduced efflux from the nanobubble, resulting in a prolonged residence time [52]. In parallel research efforts, studies have explored combinations of PFCs, sulfur hexafluoride (SF6), and nitric oxide (NO) with oxygen to formulate micro-nanobubble systems [53][54][55]. On a different note, within the realm of nanodroplets (i.e., extremely tiny liquid droplets significantly smaller than conventional microdroplets and hence, due to their very small size, can offer more advantages such as increased surface area-to-volume ratios and the ability to penetrate biological barriers or tissues more effectively; they are increasingly being considered as valuable tools in challenging applications including drug delivery, medical imaging, and nanomedicine), liquid PFCs have found application as O2 carriers, capitalizing on their higher O2 release capability [50][56].
Nanobubbles have a unique structural composition that sets them apart from larger bubbles. While their precise composition can vary depending on the preparation method and the surrounding medium, the following is a general overview. (1) Gas core: At the center of a nanobubble is the gas core, typically composed of gases like oxygen, nitrogen, or a mixture of gases. The choice of gas can influence the bubble’s behavior and applications. (2) Liquid shell: Surrounding the gas core is a thin shell of liquid, which acts as a stabilizing layer. The liquid is often water but can include other liquids or solutions depending on the specific application. (3) Surface molecules: The interface between the gas core and the liquid shell is defined by surface molecules. These molecules play a critical role in stabilizing the nanobubble and preventing its premature collapse or coalescence. Surface molecules can be surfactants or other stabilizing agents that reduce surface tension. (4) Electrostatic charge: Nanobubbles may carry a charge on their surface due to interactions with the surrounding medium or the presence of charged molecules. This surface charge can influence behavior, stability, and interactions with other particles and surfaces.
Henceforth, understanding the structural composition of nanobubbles is crucial for customizing their properties to suit specific applications. Researchers and engineers can manipulate these structural elements to craft nanobubbles with tailored characteristics, catering to a wide array of fields like medicine, materials science, and environmental science. For instance, uncoated nanobubbles, lacking a shell structure, have primarily found utility in applications like water treatment and select human applications such as oral drinks and liquid ventilation (Table 1, Table 2, Table 3 and Table 4). These uncoated nanobubbles maintain stability through charge stabilization, interacting with electrolytes in water, ultrapure water, distilled water, or saline solutions. Conversely, most engineered nanobubbles adopt a core–shell structure, where the core predominantly contains the chosen gas content strategically selected for its intended purpose. The shell component exhibits varying compositions and structures, which may include lipids, proteins, polymers, or surfactants. This core–shell configuration has traditionally been employed in the realm of biomedical applications, serving as a contrast agent for ultrasound or as a platform for oxygenation [57]. The outer shell of nanobubbles functions as a protective layer surrounding the gas core, exerting control over crucial characteristics such as stiffness, elasticity, half-life, and the rate of gas exchange. By encapsulating the gas core, the shell effectively prevents direct contact between the gas and the surrounding solvent, minimizing premature interfacial gas diffusion [50][58]. In earlier studies, it was argued that a shell structure was not necessarily a fundamental requirement for bubble formation [11][59]. Consequently, both uncoated and coated nanobubbles or unshelled and shelled nanobubbles have been utilized in the development of therapeutic strategies for various medical applications. However, increased attention has been directed toward coated nanobubbles, primarily because of their ability to incorporate a versatile array of core and shell materials, manipulation options, and bio-conjugation. This versatility extends beyond gases and drugs, allowing for the inclusion of a diverse range of molecules such as proteins, DNA, and ligands [60], enabling precise in vivo targeted delivery (Figure 3C). It is perhaps worth mentioning for the reader herein that shelled micro/nanobubbles (tiny gas-filled bubbles with a protective thin outer shell or coating that is typically composed of various materials, including lipids, proteins, polymers, surfactants, or other biocompatible substances) exhibit a more gradual external diffusion of internal gases [7][58][61][62][63], contributing their unique characteristics.
Table 1. OnB application in cancer studies.
| Clinical Indication |
Study Type |
Oxygen Release Strategy | Delivery | Main Results and Conclusions |
|---|---|---|---|---|
| Lung cancer | in vitro | Diffusion | Cell Culture Media | Uncoated OnB reduce hypoxia-induced resistance in cancer cells [64]. |
| Pancreatic cancer |
in vitro/in vivo | UltraSound mediated |
Oral | Surfactant-stabilized OnB reduce the transcriptional and protein levels of HIF1α [65]. Lipid-stabilized oxygen micro-bubbles and US reveal a 45% reduction in tumor volume five days after treatment [66]. |
| Ovarian cancer |
in vivo | UltraSound mediated |
Intraperitoneal | Oxygen and Paclitaxel (PTX)-loaded lipid micro-bubbles downregulate HiF-1α and increase PTX effectivity [67]. |
| Breast cancer | in vitro | Diffusion | Cell Culture Media | OnB revert hypoxia, downregulates HiF-1a, and improve cellular conditions, leading to further medical applications [7]. |
| in vivo | UltraSound mediated |
Intraperitoneal | Oxygen micro-bubbles strongly enhance echo intensity in tumor and significantly enhance PO2 after US irradiation [53]. | |
| Chorio- carcinoma |
in vitro | Diffusion | Cell Culture Media | Human JEG-3 cells showed a reduction (2-fold decrease—50%) in HiF-1α transcript levels at 3% O2 incubation [68]. |
| NasoPharyngeal carcinoma | in vitro/in vivo | pH-responsive | Intravenous | pH-responsive OnB increase the intra-tumoral oxygen concentration six-fold, suggesting great potential for overcoming hypoxia-induced resistance [69]. |
| Bladder/Colon Tumor Cell lines | in vitro/in vivo | UltraSound mediated |
Intravesical Intravascular |
OnB are a promising multi-modal and multifunctional strategy for imaging and targeting the hypoxic tumoral micro-environment [70][71]. The bladder tumoral PO2 increased by around 140% after the injection of OnB [72]. Biogenic nanobubbles (gas vesicles) enhance US contrast signal when compared to/with synthetic nanobubbles, enhancing tumor penetration with a range size of 2–200 nm [73]. |
| Glioma | in vitro/in vivo | Photodynamic (PDT) | Intravenous | OnB, stimulated by function as an oxygen self-supplement agent, enhance the survival rate in the glioma-bearing mice model [30]. |
Table 2. OnB application in neuroscience studies.
| Clinical Indication |
Study Type |
Oxygen Release Strategy |
Delivery | Main Results and Conclusions |
|---|---|---|---|---|
| Amyotrophic Lateral Sclerosis (ALS) |
in vitro/in vivo | Diffusion | Intraperitoneal | RNS60 protects neurons, decreasing ALS progression [37][74] and demonstrating the feasibility, safety, and tolerability of long-term administration of RNS60 in patients with ALS [75]. |
| Clinical Trial | Diffusion | Intravenous | The effect of RNS60 treatment on selected pharmacodynamic biomarkers in ALS patients was concurrently treated with riluzole (NCT03456882). | |
| Multiple Sclerosis (MS) | in vitro/in vivo | Diffusion | Intraperitoneal and Nebulization |
RNS60 induced the activation of PI3K, promoting myelin gene transcription in oligodendrocytes (OL) and glial cells [76]. RNS60 enhanced OL spare respiratory capacity (SRC) in response to metabolic stress (glucose-nutrient deprivation) [5]. RNS60 led to the enrichment of anti-autoimmune regulatory T cells (Tregs) suppression of autoimmune Th17 cells [77]. |
| Alzheimer’s disease (AD) | in vivo | Diffusion | Intraperitoneal | RNS60 suppressed the hippocampus neuronal apoptosis and attenuated Tau phosphorylation and the burden of Ab [33]. RNS60 upregulated the plasticity-related proteins (PSD95 and NR2A) and NMDA-dependent hippocampal calcium influx [31]. |
| Parkinson’s disease (PD) | in vitro/in vivo | Diffusion | Intraperitoneal | RNS60 enhance mitochondrial bio-genesis via PI3K/CREB and PGC1alpha in PD model [78]. Moreover, RNS60 inhibited the activation of NF-κB in the SNpc of MPTP-intoxicated mice [78]. |
| Spinal Cord Diseases, Injuries, and Compression | Clinical Trial | US Contrast |
Intrathecal | The use of OnB and US improves the identification of discrete areas of perfusion changes in the spinal cord in subjects undergoing spinal cord decompression (NCT05530798). |
Table 3. OnB application in cardio–respiratory studies.
| Clinical Indication |
Study Type |
Oxygen Release Strategy | Delivery | Main Results and Conclusions |
|---|---|---|---|---|
| Respiratory Failure and Acute Lung Trauma/Injury | in vitro/in vivo | Diffusion | Intraperitoneal Intravenous |
Peritoneal microbubble oxygenation (PMO) provides extrapulmonary ventilation after complete tracheal occlusion [79]. PMO is also a promising strategy for other pulmonary diseases [6] as it oxygenates blood within 4 sec and does not cause hemolysis or complement activation in hypoxic rabbits [80]. |
| Blood Oxygenation |
in vitro/in vivo | Diffusion | Cell Culture Media |
Oxygen microbubble-containing dextran solutions were effective for improving blood oxygenation [81]. |
| in vivo | Diffusion | Intravenous | Oxygen microbubbles are safe and effective in delivering more oxygen than human red blood cells (per gram) after being injected in vivo [82]. | |
| ExtraCorporeal Membrane Oxygenation (ECMO) |
in vitro | Diffusion | De-oxygenated PBS | Protein-encapsulated oxygen microbubbles rapidly equilibrate hypoxia by releasing their oxygen core into an oxygen-depleted saline solution [83]. |
| Anti-Thrombotic Effect | in vitro | US-mediated | Intravascular | PLA-combined Fe3O4-GO-ASA nanobubbles improve the anti-thrombin parameters and significantly inhibit thrombosis within rabbit blood [84]. |
| MicroFluidic Device for Hypoxemia | in vitro/in vivo | Diffusion | Intravascular | OnB from the device was infused into the femoral vein, in vivo, wherein ∼20% of baseline VO2 can be delivered intravenously in real time [85]. |
| Ischemic Stroke Re-Perfusion |
in vivo | US-mediated | Intravascular | OnB with US stimulation provide sono-perfusion and local oxygen for the reduction in brain infarct size and neuroprotection after stroke re-perfusion [86]. |
Table 4. OnB application in metabolic diseases, regenerative medicine, and molecular imaging studies.
| Clinical Indication |
Study Type |
Oxygen Release Strategy | Delivery | Main Results and Conclusions |
|---|---|---|---|---|
| Diabetes, Burns, Tissue necrosis, Bedsores, and Wounds | in vitro/in vivo | US-mediated | Cell Culture Media | Dextran and chitosan nanobubbles might be proposed for the delivery of oxygen, which is enhanced by US with a frequency of 45 kHz in hypoxic-related diseases [2][87]. |
| Clinical Trial (recruiting) |
Diffusion | Irrigation | This micro-/nano-bubble solution is suggested as an irrigation solution to improve wound oxygenation in ischemic tissues (NCT05169814). | |
| Diabetic Retinopathy |
in vitro/in vivo | Diffusion | Cell Culture Intravitreal |
Dextran-OnB release 74.06 µg of O2 after 12 h at 37 °C and mitigate hypoxia during ischemic conditions in the eye upon timely administration [88]. |
| Tissue Cutaneous Lesions |
in vitro/in vivo | US-mediated | Cell Culture Media/Gel Formulation Topically Applied | Oxygen-loaded nano-droplets (OLNDs) are more effective than former oxygen-loaded nanobubbles enhancing oxy-hemoglobin levels by photoacoustic [89]. US-activated chitosan-shelled/DFP-cored OLNDs might be a novel, suitable, and cost-effective way to treat several hypoxia-associated pathologies of the cutaneous tissues [3]. |
| Transdermal Drug Release |
in vitro/in vivo | US-mediated | Transdermal Micro-needle Gel | The nanobubbles added into a micro-needle patch and used in addition to US cause better penetration and diffusion of drugs [90]. |
| Cytocompatibility of OnB | in vitro | US-mediated | Cell Culture Media | Lipid-shelled/coated OnB show US imaging-responsiveness and enhance cell viability in several cell lines [15]. |
| Drug Delivery in Fluids | in vitro | US-mediated | Hypoxic Solution |
Oxygen release from polysaccharides–peptides, Pingxiao, and chitosan is 94.6%, 75.1%, and 40.2% (respectively) higher than water, especially under the US stimulus [91]. |
| On-chip Contrast Agent for US Imaging |
in vitro | US-mediated | Microfluidic | Micron-sized lipid shell-based perfluorocarbon gas microbubbles enhance the gas composition for US contrast agents with new shell materials [92]. |
4. NanoBubbles as a Platform for O2 Delivery: Innovative OnB-Mediated Oxygenation
In the ever-evolving landscape of medical science and biotechnology, the emergence of nanobubbles as a platform for oxygen delivery has sparked innovation and excitement. These minuscule nano-scaled bubbles have paved the way for groundbreaking approaches to oxygenation, offering a promising solution to address critical challenges in various fields, from medicine to environmental science. Indeed, the rising incorporation of nanobubbles into biomedicine is underpinned by a range of highly beneficial characteristics, as detailed above. To re-emphasize, these versatile physical–chemical–mechanical properties encompass stability, the ability to provide a larger surface area for interaction, the availability of uniform or varied size distributions, increased payload capacity, efficient interfacial gas diffusion for precise gas delivery, biodegradability, biocompatibility, accelerated cellular uptake, and the strategic utilization of the EPR effect. The potential of harnessing these nanobubble platforms within clinical settings to encapsulate and deliver oxygen with exceptional precision (targeted oxygen delivery) is increasingly feasible. Indeed, their diminutive size allows them to navigate intricate biological pathways and reach specific cellular or tissue targets with unparalleled efficiency. These advancements are driven by the development of enhanced formulations of stable and versatile OnB that can be tailored to a myriad of applications, ranging from enhancing oxygen levels in biological systems to serving as contrast agents in medical imaging. Development and innovation efforts continue to advance the practical integration of nanobubbles into clinical application, which is becoming increasingly plausible with the translation of novel formulations, including coated and core–shell nanobubbles, which have been and are being developed, including in the labs, to enhance gas solubility and extend oxygen retention durations, all while avoiding the formation of problematic macro-bubbles [12][47]. In this era of innovation, the exploration of OnB as a platform for oxygen delivery represents a promising frontier. Their ability to navigate the complexities of biological systems and precisely deliver oxygen opens new avenues for addressing hypoxia-related conditions, improving medical diagnostics, and revolutionizing drug delivery and release strategies.
O2 release strategies involving OnB technologies: The efficient release of oxygen from nanobubbles into a liquid is a spontaneous process driven by the pre-existing concentration gradient between the gas core and the surrounding liquid phase. Moreover, OnB serves a dual purpose as contrast-enhancement agents for ultrasound (US) imaging, offering the distinct advantage of enabling guided and real-time monitoring of oxygen delivery [53]. In scenarios involving an ultra-sound (US) -triggered release mechanism, the emission of high-intensity US waves gives rise to distinct high and low-pressure resonance zones within the propagating wave. These zones result from resonant phenomena, ultimately leading to the disruption of bubbles and the subsequent liberation of the encapsulated gas core. A current summary of studies and potential applications of both uncoated and coated OnB in the field of biomedicine is presented (Table 1, Table 2, Table 3 and Table 4). The intravascular (IV) delivery of oxygen using OnB has emerged as a prominent administration route in cancer therapies and for conditions characterized by microcirculation dysfunction [44]. Additionally, OnB shows promise as a secure platform for blood oxygenation, achieving oxygen saturation levels of up to 95% without activating the blood complement system or causing hemolysis. Moreover, their minimal IV fluid injection volume requirement underscores their practicality and feasibility [6]. Research indicates that the cellular uptake of OnB involves complex endocytosis processes, thus preserving cellular functionality and supporting the achievement of desired therapeutic outcomes [93]. In essence, oxygen release from OnB can occur through gentle methods such as US cavitation or diffusion mechanisms. While alternative oxygen release mechanisms have been explored, such as those involving OnB assisted by photodynamic processes [30] or pH variations [69], a significant gap persists in the molecular understanding of how OnB behave within cells, extending to the intricate processes by which mitochondria and HiF-α sense OnB presence.
This entry is adapted from the peer-reviewed paper 10.3390/nano13233060
References
- Magliaro, C.; Mattei, G.; Iacoangeli, F.; Corti, A.; Piemonte, V.; Ahluwalia, A. Oxygen Consumption Characteristics in 3D Constructs Depend on Cell Density. Front. Bioeng. Biotechnol. 2019, 7, 251.
- Cavalli, R.; Bisazza, A.; Giustetto, P.; Civra, A.; Lembo, D.; Trotta, G.; Guiot, C.; Trotta, M. Preparation and Characterization of Dextran Nanobubbles for Oxygen Delivery. Int. J. Pharm. 2009, 381, 160–165.
- Magnetto, C.; Prato, M.; Khadjavi, A.; Giribaldi, G.; Fenoglio, I.; Jose, J.; Gulino, G.R.; Cavallo, F.; Quaglino, E.; Benintende, E.; et al. Ultrasound-Activated Decafluoropentane-Cored and Chitosan-Shelled Nanodroplets for Oxygen Delivery to Hypoxic Cutaneous Tissues. RSC Adv. 2014, 4, 38433–38441.
- Wigerup, C.; Påhlman, S.; Bexell, D. Therapeutic Targeting of Hypoxia and Hypoxia-Inducible Factors in Cancer. Pharmacol. Ther. 2016, 164, 152–169.
- Rao, V.T.S.; Khan, D.; Jones, R.G.; Nakamura, D.S.; Kennedy, T.E.; Cui, Q.-L.; Rone, M.B.; Healy, L.M.; Watson, R.; Ghosh, S.; et al. Potential Benefit of the Charge-Stabilized Nanostructure Saline RNS60 for Myelin Maintenance and Repair. Sci. Rep. 2016, 6, 30020.
- Kheir, J.N.; Polizzotti, B.D.; Thomson, L.M.; O’Connell, D.W.; Black, K.J.; Lee, R.W.; Wilking, J.N.; Graham, A.C.; Bell, D.C.; McGowan, F.X. Bulk Manufacture of Concentrated Oxygen Gas-Filled Microparticles for Intravenous Oxygen Delivery. Adv. Healthc. Mater. 2013, 2, 1131–1141.
- Khan, M.S.; Hwang, J.; Seo, Y.; Shin, K.; Lee, K.; Park, C.; Choi, Y.; Hong, J.W.; Choi, J. Engineering Oxygen Nanobubbles for the Effective Reversal of Hypoxia. Artif. Cells Nanomed. Biotechnol. 2018, 46, S318–S327.
- Uchida, T.; Oshita, S.; Ohmori, M.; Tsuno, T.; Soejima, K.; Shinozaki, S.; Take, Y.; Mitsuda, K. Transmission Electron Microscopic Observations of Nanobubbles and Their Capture of Impurities in Wastewater. Nanoscale Res. Lett. 2011, 6, 1–9.
- Zimmerman, W.B.; Tesař, V.; Bandulasena, H.C.H. Towards Energy Efficient Nanobubble Generation with Fluidic Oscillation. Curr. Opin. Colloid Interface Sci. 2011, 16, 350–356.
- Agarwal, A.; Ng, W.J.; Liu, Y. Principle and Applications of Microbubble and Nanobubble Technology for Water Treatment. Chemosphere 2011, 84, 1175–1180.
- Matsuki, N.; Ichiba, S.; Ishikawa, T.; Nagano, O.; Takeda, M.; Ujike, Y.; Yamaguchi, T. Blood Oxygenation Using Microbubble Suspensions. Eur. Biophys. J. 2012, 41, 571–578.
- Takahashi, M.; Chiba, K.; Li, P. Free-Radical Generation from Collapsing Microbubbles in the Absence of a Dynamic Stimulus. J. Phys. Chem. B 2007, 111, 1343–1347.
- Eriksson, J.C.; Ljunggren, S. On the Mechanically Unstable Free Energy Minimum of a Gas Bubble Which Is Submerged in Water and Adheres to a Hydrophobic Wall. Colloids Surf. A Physicochem. Eng. Asp. 1999, 159, 159–163.
- Ljunggren, S.; Eriksson, J.C. The Lifetime of a Colloid-Sized Gas Bubble in Water and the Cause of the Hydrophobic Attraction. Colloids Surf. A Physicochem. Eng. Asp. 1997, 129–130, 151–155.
- Khan, M.S.; Hwang, J.; Lee, K.; Choi, Y.; Jang, J.; Kwon, Y.; Hong, J.W.; Choi, J. Surface Composition and Preparation Method for Oxygen Nanobubbles for Drug Delivery and Ultrasound Imaging Applications. Nanomaterials 2019, 9, 48.
- Bitterman, H. Bench-to-Bedside Review: Oxygen as a Drug. Crit. Care 2009, 13, 205.
- Guo, S.; DiPietro, L.A. Factors Affecting Wound Healing. J. Dent. Res. 2010, 89, 219–229.
- Wang, S.; Zhou, Z.; Hu, R.; Dong, M.; Zhou, X.; Ren, S.; Zhang, Y.; Chen, C.; Huang, R.; Zhu, M.; et al. Metabolic Intervention Liposome Boosted Lung Cancer Radio-Immunotherapy via Hypoxia Amelioration and PD-L1 Restraint. Adv. Sci. 2023, 10, 2207608.
- Wu, Y.; Chen, M.; Jiang, J. Mitochondrial Dysfunction in Neurodegenerative Diseases and Drug Targets via Apoptotic Signaling. Mitochondrion 2019, 49, 35–45.
- Manchanda, M.; Torres, M.; Inuossa, F.; Bansal, R.; Kumar, R.; Hunt, M.; Wheelock, C.E.; Bachar-Wikstrom, E.; Wikstrom, J.D. Metabolic Reprogramming and Reliance in Human Skin Wound Healing. J. Investig. Dermatol. 2023, 143, 2039–2051.
- Mas-Bargues, C.; Sanz-Ros, J.; Roman-Dominguez, A.; Inglés, M.; Gimeno-Mallench, L.; Alami, M.; Viña-Almunia, J.; Gambini, J.; Viña, J.; Borras, C. Relevance of Oxygen Concentration in Stem Cell Culture for Regenerative Medicine. Int. J. Mol. Sci. 2019, 20, 1195.
- Hirai, D.; Colburn, T.; Craig, J.; Hotta, K.; Kano, Y.; Musch, T.; Poole, D. Skeletal Muscle Interstitial O2 Pressures: Bridging the Gap between the Capillary and Myocyte. Microcirculation 2019, 26, e12497.
- Chan, D.A.; Giaccia, A.J. Hypoxia, Gene Expression, and Metastasis. Cancer Metastasis Rev. 2007, 26, 333–339.
- Fuhrmann, D.C.; Brüne, B. Mitochondrial Composition and Function under the Control of Hypoxia. Redox Biol. 2017, 12, 208–215.
- Solaini, G.; Baracca, A.; Lenaz, G.; Sgarbi, G. Hypoxia and Mitochondrial Oxidative Metabolism. Biochim. Biophys. Acta Bioenerg. 2010, 1797, 1171–1177.
- Schödel, J.; Oikonomopoulos, S.; Ragoussis, J.; Pugh, C.W.; Ratcliffe, P.J.; Mole, D.R. High-Resolution Genome-Wide Mapping of HIF-Binding Sites by ChIP-Seq. Blood 2011, 117, e207–e217.
- Schofield, C.J.; Ratcliffe, P.J. Oxygen Sensing by HIF Hydroxylases. Nat. Rev. Mol. Cell Biol. 2004, 5, 343–354.
- Bhattacharya, S.; Ratcliffe, P.J. ExCITED about HIF. Nat. Struct. Mol. Biol. 2003, 10, 501–503.
- Eui-Ju, Y. Hypoxia and Aging. Exp. Mol. Med. 2019, 51, 1–15.
- Song, R.; Hu, D.; Chung, H.Y.; Sheng, Z.; Yao, S. Lipid-Polymer Bilaminar Oxygen Nanobubbles for Enhanced Photodynamic Therapy of Cancer. ACS Appl. Mater. Interfaces 2018, 10, 36805–36813.
- Chandra, G.; Kundu, M.; Rangasamy, S.B.; Dasarathy, S.; Ghosh, S.; Watson, R.; Pahan, K. Increase in Mitochondrial Biogenesis in Neuronal Cells by RNS60, a Physically-Modified Saline, via Phosphatidylinositol 3-Kinase-Mediated Upregulation of PGC1α. J. Neuroimmune Pharmacol. 2018, 13, 143–162.
- Qin, H.-Y.; Mukherjee, R.; Lee-Chan, E.; Ewen, C.; Bleackley, C.; Singh, B. A Novel Mechanism of Regulatory T Cell-Mediated down-Regulation of Autoimmunity. Int. Immunol. 2006, 18, 1001–1015.
- Modi, K.K.; Jana, A.; Ghosh, S.; Watson, R.; Pahan, K. A Physically-Modified Saline Suppresses Neuronal Apoptosis, Attenuates Tau Phosphorylation and Protects Memory in an Animal Model of Alzheimer’s Disease. PLoS ONE 2014, 9, e103606.
- Lukianova-Hleb, E.Y.; Ren, X.; Zasadzinski, J.A.; Wu, X.; Lapotko, D.O. Plasmonic Nanobubbles Enhance Efficacy and Selectivity of Chemotherapy against Drug-Resistant Cancer Cells. Adv. Mater. 2012, 24, 3831–3837.
- Um, W.; Ko, H.; You, D.G.; Lim, S.; Kwak, G.; Shim, M.K.; Yang, S.; Lee, J.; Song, Y.; Kim, K.; et al. Necroptosis-Inducible Polymeric Nanobubbles for Enhanced Cancer Sonoimmunotherapy. Adv. Mater. 2020, 32, e1907953.
- Mondal, S.; Martinson, J.A.; Ghosh, S.; Watson, R.; Pahan, K. Protection of Tregs, Suppression of Th1 and Th17 Cells, and Amelioration of Experimental Allergic Encephalomyelitis by a Physically-Modified Saline. PLoS ONE 2012, 7, e51869.
- Khasnavis, S.; Jana, A.; Roy, A.; Mazumder, M.; Bhushan, B.; Wood, T.; Ghosh, S.; Watson, R.; Pahan, K. Suppression of Nuclear Factor-ΚB Activation and Inflammation in Microglia by Physically Modified Saline. J. Biol. Chem. 2012, 287, 29529–29542.
- Choi, J.C.; Mega, T.L.; German, S.; Wood, A.B.; Watson, R.L. Electrokinetically Altered Normal Saline Modulates Ion Channel Activity. Biophys. J. 2012, 102, 683a.
- German, S.; Ghosh, S.; Mega, T.; Burke, L.; Diegel, M.; Wood, A.; Watson, R. Isotonic Saline Subjected to Taylor-Couette-Poiseuille Demonstrates Anti-Inflammatory Activity In Vitro. J. Allergy Clin. Immunol. 2011, 127, AB260.
- Kalmes, A.A.; Ghosh, S.; Watson, R.L. A saline-based therapeutic containing charge-stabilized nanostructures protects against cardiac ischemia/reperfusion injury. J. Am. Coll. Cardiol. 2013, 61, E106.
- Batchelor, D.; Hassan-Abou-Saleh, R.; Coletta, L.; McLaughlan, J.; Peyman, S.; Evans, S. Nested-Nanobubbles for Ultrasound Triggered Drug Release. ACS Appl. Mater. Interfaces 2020, 12, 29085–29093.
- Prabhakar, A.; Banerjee, R. Nanobubble Liposome Complexes for Diagnostic Imaging and Ultrasound-Triggered Drug Delivery in Cancers: A Theranostic Approach. ACS Omega 2019, 4, 15567–15580.
- Tayier, B.; Deng, Z.; Wang, Y.; Wang, W.; Mu, Y.; Yan, F. Biosynthetic Nanobubbles for Targeted Gene Delivery by Focused Ultrasound. Nanoscale 2019, 11, 14757–14768.
- Sayadi, L.R.; Banyard, D.A.; Ziegler, M.E.; Obagi, Z.; Prussak, J.; Klopfer, M.J.; Evans, G.R.D.; Widgerow, A.D. Topical Oxygen Therapy & Micro/Nanobubbles: A New Modality for Tissue Oxygen Delivery. Int. Wound J. 2018, 15, 363–374.
- ISO/TC 281; Fine Bubble Technology. Japanese Industrial Standards Committee: Tokyo, Japan, 2013.
- ISO 20480-1:2017; General Principles for Usage and Measurement of Fine Bubbles, Part 1: Terminology. ISO: Geneve, Switzerland, 2017.
- Ushikubo, F.Y.; Furukawa, T.; Nakagawa, R.; Enari, M.; Makino, Y.; Kawagoe, Y.; Shiina, T.; Oshita, S. Evidence of the Existence and the Stability of Nano-Bubbles in Water. Colloids Surf. A Physicochem. Eng. Asp. 2010, 361, 31–37.
- Huang, H.Y.; Liu, H.L.; Hsu, P.H.; Chiang, C.S.; Tsai, C.H.; Chi, H.S.; Chen, S.Y.; Chen, Y.Y. A Multitheragnostic Nanobubble System to Induce Blood-Brain Barrier Disruption with Magnetically Guided Focused Ultrasound. Adv. Mater. 2015, 27, 655–661.
- Hernandez, C.; Nieves, L.; de Leon, A.C.; Advincula, R.; Exner, A.A. Role of Surface Tension in Gas Nanobubble Stability under Ultrasound. ACS Appl. Mater. Interfaces 2018, 10, 9949–9956.
- Khan, M.S.; Hwang, J.; Lee, K.; Choi, Y.; Kim, K.; Koo, H.-J.; Hong, J.W.; Choi, J. Oxygen-Carrying Micro/Nanobubbles: Composition, Synthesis Techniques and Potential Prospects in Photo-Triggered Theranostics. Mol. J. Synth. Chem. Nat. Product. Chem. 2018, 23, 2210.
- Lohse, D. Bubble Puzzles: From Fundamentals to Applications. Phys. Rev. Fluids 2018, 3, 110504.
- Burkard, M.E.; Van-Liew, H.D. Oxygen Transport to Tissue by Persistent Bubbles: Theory and Simulations. J. Appl. Physiol. 1994, 77, 2874–2878.
- Yang, C.; Xiao, H.; Sun, Y.; Zhu, L.; Gao, Y.; Kwok, S.; Wang, Z.; Tang, Y. Lipid Microbubbles as Ultrasound-Stimulated Oxygen Carriers for Controllable Oxygen Release for Tumor Reoxygenation. Ultrasound Med. Biol. 2018, 44, 416–425.
- Cavalieri, F.; Finelli, I.; Tortora, M.; Mozetic, P.; Chiessi, E.; Polizio, F.; Brismar, T.B.; Paradossi, G. Polymer Microbubbles as Diagnostic and Therapeutic Gas Delivery Device. Chem. Mater. 2008, 20, 3254–3258.
- Kwan, J.J.; Kaya, M.; Borden, M.A.; Dayton, P.A. Theranostic Oxygen Delivery Using Ultrasound and Microbubbles. Theranostics 2012, 2, 1174–1184.
- Riess, J.G. Understanding the Fundamentals of Perfluorocarbons and Perfluorocarbon Emulsions Relevant to In Vivo Oxygen Delivery. Artif. Cells Blood Substit. Immobil. Biotechnol. 2005, 33, 47–63.
- Stride, E.; Edirisinghe, M. Novel Microbubble Preparation Technologies. Soft Matter 2008, 4, 235–2359.
- Park, B.; Yoon, S.; Choi, Y.; Jang, J.; Park, S.; Choi, J. Stability of Engineered Micro or Nanobubbles for Biomedical Applications. Pharmaceutics 2020, 12, 1089.
- Ebina, K.; Shi, K.; Hirao, M.; Hashimoto, J.; Kawato, Y.; Kaneshiro, S.; Morimoto, T.; Koizumi, K.; Yoshikawa, H. Oxygen and Air Nanobubble Water Solution Promote the Growth of Plants, Fishes, and Mice. PLoS ONE 2013, 8, e65339.
- Unger, E.C.; Hersh, E.; Vannan, M.; Matsunaga, T.O.; McCreery, T. Local Drug and Gene Delivery through Microbubbles. Prog. Cardiovasc. Dis. 2001, 44, 45–54.
- Kiessling, F.; Huppert, J.; Palmowski, M. Functional and Molecular Ultrasound Imaging: Concepts and Contrast Agents. Curr. Med. Chem. 2009, 16, 627–642.
- Bjerknes, K.; Sontum, P.C.; Smistad, G.; Agerkvist, I. Preparation of Polymeric Microbubbles: Formulation Studies and Product Characterisation. Int. J. Pharm. 1997, 158, 129–136.
- Unger, E.C.; Porter, T.; Culp, W.; Labell, R.; Matsunaga, T.; Zutshi, R. Therapeutic Applications of Lipid-Coated Microbubbles. Adv. Drug Deliv. Rev. 2004, 56, 1291–1314.
- Iijima, M.; Gombodorj, N.; Tachibana, Y.; Tachibana, K.; Yokobori, T.; Honma, K.; Nakano, T.; Asao, T.; Kuwahara, R.; Aoyama, K.; et al. Development of Single Nanometer-Sized Ultrafine Oxygen Bubbles to Overcome the Hypoxia-Induced Resistance to Radiation Therapy via the Suppression of Hypoxia-Inducible Factor-1α. Int. J. Oncol. 2018, 52, 679–686.
- Owen, J.; McEwan, C.; Nesbitt, H.; Bovornchutichai, P.; Averre, R.; Borden, M.; McHale, A.P.; Callan, J.F.; Stride, E. Reducing Tumour Hypoxia via Oral Administration of Oxygen Nanobubbles. PLoS ONE 2016, 11, e0168088.
- McEwan, C.; Owen, J.; Stride, E.; Fowley, C.; Nesbitt, H.; Cochrane, D.; Coussios, C.C.; Borden, M.; Nomikou, N.; McHale, A.P.; et al. Oxygen Carrying Microbubbles for Enhanced Sonodynamic Therapy of Hypoxic Tumours. J. Control Release 2015, 203, 51–56.
- Luo, T.; Sun, J.; Zhu, S.; He, J.; Hao, L.; Xiao, L.; Zhu, Y.; Wang, Q.; Pan, X.; Wang, Z.; et al. Ultrasound-Mediated Destruction of Oxygen and Paclitaxel Loaded Dual-Targeting Microbubbles for Intraperitoneal Treatment of Ovarian Cancer Xenografts. Cancer Lett. 2017, 391, 1–11.
- Bisazza, A.; Giustetto, P.; Rolfo, A.; Caniggia, I.; Balbis, S.; Guiot, C.; Cavalli, R. Microbubble-Mediated Oxygen Delivery to Hypoxic Tissues as a New Therapeutic Device. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2008, 2008, 2067–2070.
- Song, R.; Peng, S.; Lin, Q.; Luo, M.; Chung, H.Y.; Zhang, Y.; Yao, S. PH-Responsive Oxygen Nanobubbles for Spontaneous Oxygen Delivery in Hypoxic Tumors. Langmuir 2019, 35, 10166–10172.
- Bhandari, P.; Novikova, G.; Goergen, C.J.; Irudayaraj, J. Ultrasound Beam Steering of Oxygen Nanobubbles for Enhanced Bladder Cancer Therapy. Sci. Rep. 2018, 8, 3110–3112.
- Zhang, J.; Chen, Y.; Deng, C.; Zhang, L.; Sun, Z.; Wang, J.; Yang, Y.; Lv, Q.; Han, W.; Xie, M. The Optimized Fabrication of a Novel Nanobubble for Tumor Imaging. Front. Pharmacol. 2019, 10, 610.
- Bhandari, P.N.; Cui, Y.; Elzey, B.D.; Goergen, C.J.; Long, C.M.; Irudayaraj, J. Oxygen Nanobubbles Revert Hypoxia by Methylation Programming. Sci. Rep. 2017, 7, 9214–9268.
- Feng, Y.; Hao, Y.; Wang, Y.; Song, W.; Zhang, S.; Ni, D.; Yan, F.; Sun, L. Ultrasound Molecular Imaging of Bladder Cancer via Extradomain B Fibronectin-Targeted Biosynthetic GVs. Int. J. Nanomed. 2023, 18, 4871–4884.
- Vallarola, A.; Sironi, F.; Tortarolo, M.; Gatto, N.; de Gioia, R.; Pasetto, L.; de Paola, M.; Mariani, A.; Ghosh, S.; Watson, R.; et al. RNS60 Exerts Therapeutic Effects in the SOD1 ALS Mouse Model through Protective Glia and Peripheral Nerve Rescue. J. Neuroinflammation 2018, 15, 65–1835.
- Paganoni, S.; Alshikho, M.J.; Luppino, S.; Chan, J.; Pothier, L.; Schoenfeld, D.; Andres, P.L.; Babu, S.; Zürcher, N.R.; Loggia, M.L.; et al. A Pilot Trial of RNS60 in Amyotrophic Lateral Sclerosis. Muscle Nerve 2019, 59, 303–308.
- Jana, M.; Jana, M.; Ghosh, S.; Ghosh, S.; Pahan, K.; Pahan, K. Upregulation of Myelin Gene Expression by a Physically-Modified Saline via Phosphatidylinositol 3-Kinase-Mediated Activation of CREB: Implications for Multiple Sclerosis. Neurochem. Res. 2018, 43, 407–419.
- Mondal, S.; Rangasamy, S.B.; Ghosh, S.; Watson, R.L.; Pahan, K. Nebulization of RNS60, a Physically-Modified Saline, Attenuates the Adoptive Transfer of Experimental Allergic Encephalomyelitis in Mice: Implications for Multiple Sclerosis Therapy. Neurochem. Res. 2017, 42, 1555–1570.
- Khasnavis, S.; Roy, A.; Ghosh, S.; Watson, R.; Pahan, K. Protection of Dopaminergic Neurons in a Mouse Model of Parkinson’s Disease by a Physically-Modified Saline Containing Charge-Stabilized Nanobubbles. J. Neuroimmune Pharmacol. 2014, 9, 218–232.
- Legband, N.D.; Feshitan, J.A.; Borden, M.A.; Terry, B.S. Evaluation of Peritoneal Microbubble Oxygenation Therapy in a Rabbit Model of Hypoxemia. IEEE Trans. Biomed. Eng. 2015, 62, 1376–1382.
- Kheir, J.N.; Scharp, L.A.; Borden, M.A.; Swanson, E.J.; Loxley, A.; Reese, J.H.; Black, K.J.; Velazquez, L.A.; Thomson, L.M.; Walsh, B.K.; et al. Oxygen Gas-Filled Microparticles Provide Intravenous Oxygen Delivery. Sci. Transl. Med. 2012, 4, 140ra88.
- Matsuki, N.; Ishikawa, T.; Ichiba, S.; Shiba, N.; Ujike, Y.; Yamaguchi, T. Oxygen Supersaturated Fluid Using Fine Micro/Nanobubbles. Int. J. Nanomed. 2014, 9, 4495–4505.
- Seekell, R.P.; Lock, A.T.; Peng, Y.; Cole, A.R.; Perry, D.A.; Kheir, J.N.; Polizzotti, B.D. Oxygen Delivery Using Engineered Microparticles. Proc. Natl. Acad. Sci. USA 2016, 113, 12380–12385.
- Swanson, E.J.; Borden, M.A. Injectable oxygen delivery based on protein-shelled microbubbles. Nano Life 2010, 1, 215–218.
- Zhang, J.; Liu, Z.; Chang, C.; Hu, M.; Teng, Y.; Li, J.; Zhang, X.; Chi, Y. Ultrasound Imaging and Antithrombotic Effects of PLA-Combined Fe3O4-GO-ASA Multifunctional Nanobubbles. Front. Med. 2021, 8, 6422.
- Kumar Vutha, A.; Patenaude, R.; Cole, A.; Kumar, R.; Kheir, J.N.; Polizzotti, B.D. A Microfluidic Device for Real-Time on-Demand Intravenous Oxygen Delivery. Proc. Natl. Acad. Sci. USA 2022, 119, e2115276119.
- Ho, Y.-J.; Cheng, H.-L.; Liao, L.-D.; Lin, Y.-C.; Tsai, H.-C.; Yeh, C.-K. Oxygen-Loaded Microbubble-Mediated Sonoperfusion and Oxygenation for Neuroprotection after Ischemic Stroke Reperfusion. Biomater. Res. 2023, 27, 65.
- Cavalli, R.; Bisazza, A.; Rolfo, A.; Balbis, S.; Madonnaripa, D.; Caniggia, I.; Guiot, C. Ultrasound-Mediated Oxygen Delivery from Chitosan Nanobubbles. Int. J. Pharm. 2009, 378, 215–217.
- Messerschmidt, V.; Ren, W.; Tsipursky, M.; Irudayaraj, J. Characterization of Oxygen Nanobubbles and In Vitro Evaluation of Retinal Cells in Hypoxia. Transl. Vis. Sci. Technol. 2023, 12, 16.
- Prato, M.; Magnetto, C.; Jose, J.; Khadjavi, A.; Cavallo, F.; Quaglino, E.; Panariti, A.; Rivolta, I.; Benintende, E.; Varetto, G.; et al. 2H,3H-Decafluoropentane-Based Nanodroplets: New Perspectives for Oxygen Delivery to Hypoxic Cutaneous Tissues. PLoS ONE 2015, 10, e0119769.
- Shao, S.; Wang, S.; Ren, L.; Wang, J.; Chen, X.; Pi, H.; Sun, Y.; Dong, C.; Weng, L.; Gao, Y.; et al. Layer-by-Layer Assembly of Lipid Nanobubbles on Microneedles for Ultrasound-Assisted Transdermal Drug Delivery. ACS Appl. Bio Mater. 2022, 5, 562–569.
- Zhao, W.; Hu, X.; Duan, J.; Liu, T.; Liu, M.; Dong, Y. Oxygen Release from Nanobubbles Adsorbed on Hydrophobic Particles. Chem. Phys. Lett. 2014, 608, 224–228.
- Hettiarachchi, K.; Talu, E.; Longo, M.L.; Dayton, P.A.; Lee, A.P. On-Chip Generation of Microbubbles as a Practical Technology for Manufacturing Contrast Agents for Ultrasonic Imaging. Lab Chip 2007, 7, 463–468.
- Khan, M.S.; Hwang, J.; Lee, K.; Choi, Y.; Seo, Y.; Jeon, H.; Hong, J.W.; Choi, J. Anti-Tumor Drug-Loaded Oxygen Nanobubbles for the Degradation of HIF-1α and the Upregulation of Reactive Oxygen Species in Tumor Cells. Cancers 2019, 11, 1464.
This entry is offline, you can click here to edit this entry!
