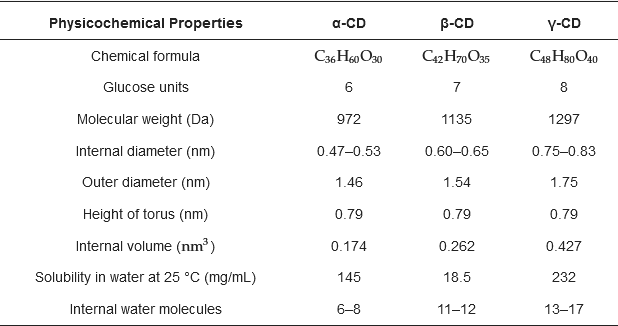
Natural cyclodextrins (CDs) can be formed by 6, 7, or 8 glucose molecules (α-, β-, and γ-, respectively) linked in a ring, creating a cone shape. Its interior has an affinity for hydrophobic molecules, while the exterior is hydrophilic and can interact with water molecules. This feature has been used to develop active packaging applied to food, interacting with the product or its environment to improve one or more aspects of its quality or safety. It also provides monitoring information when food is optimal for consumption, as intelligent packaging is essential for the consumer and the merchant.
- cyclodextrin
- smart packaging
- intelligent packaging
- active packaging
- food additives
- biopolymers
- polysaccharide
1. Introduction
2. Cyclodextrins
3. Solubility and Toxicological Considerations
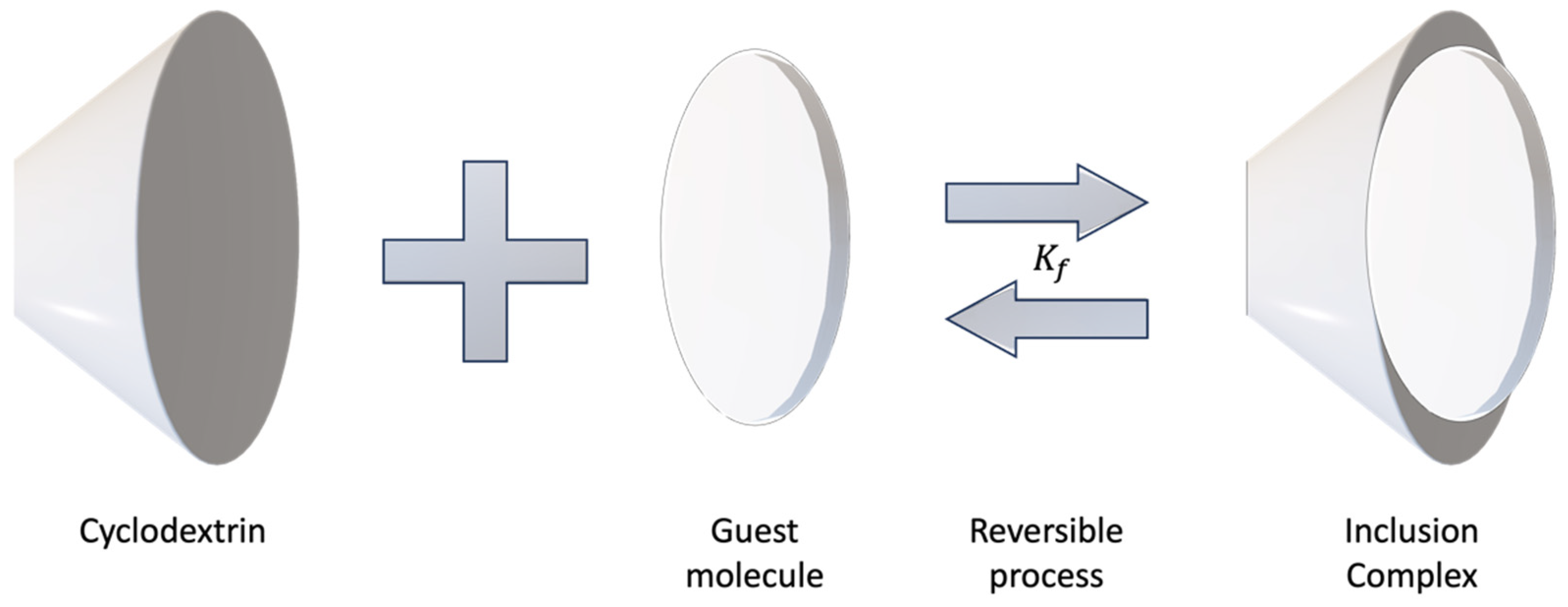
4. Formation of Inclusion Complexes
5. Cyclodextrins in Food Packaging
This entry is adapted from the peer-reviewed paper 10.3390/polym15214317
References
- Accorsi, R.; Manzini, R. Modeling inclusive food supply chains toward sustainable ecosystem planning. In Sustainable Food Supply Chains; Academic Press: Dordrecht, The Netherlands, 2019; pp. 1–21.
- Haji, M.; Kerbache, L.; Al-Ansari, T. Food Quality, Drug Safety, and Increasing Public Health Measures in Supply Chain Management. Processes 2022, 10, 1715.
- Huang, J.; Zhang, M.; Fang, Z. Perspectives on Novel Technologies of Processing and Monitoring the Safety and Quality of Prepared Food Products. Foods 2023, 12, 3052.
- Han, J.W.; Ruiz-Garcia, L.; Qian, J.P.; Yang, X.T. Food Packaging: A Comprehensive Review and Future Trends. In Comprehensive Reviews in Food Science and Food Safety; Blackwell Publishing Inc.: Oxford, UK, 2018; Volume 17, pp. 860–877.
- Alam, A.U.; Rathi, P.; Beshai, H.; Sarabha, G.K.; Deen, M.J. Fruit Quality Monitoring with Smart Packaging. Sensors 2021, 21, 1509.
- Stramarkou, M.; Boukouvalas, C.; Koskinakis, S.E.; Serifi, O.; Bekiris, V.; Tsamis, C.; Krokida, M. Life Cycle Assessment and Preliminary Cost Evaluation of a Smart Packaging System. Sustainability 2022, 14, 7080.
- Hu, Q.D.; Tang, G.P.; Chu, P.K. Cyclodextrin-based host-guest supramolecular nanoparticles for delivery: From design to applications. Acc. Chem. Res. 2014, 47, 2017–2025.
- Matencio, A.; Navarro-Orcajada, S.; García-Carmona, F.; López-Nicolás, J.M. Applications of cyclodextrins in food science. A review. Trends Food Sci. Technol. 2020, 104, 132–143.
- Astray, G.; Mejuto, J.C.; Simal-Gandara, J. Latest developments in applying cyclodextrin host-guest complexes in beverage technology processes. Food Hydrocoll. 2020, 106, 105882.
- Liu, H.-N.; Jiang, X.-X.; Naeem, A.; Chen, F.-C.; Wang, L.; Liu, Y.-X.; Li, Z.; Ming, L.-S. Fabrication and Characterization of β-Cyclodextrin/Mosla Chinensis Essential Oil Inclusion Complexes: Experimental Design and Molecular Modeling. Molecules 2023, 28, 37.
- Bortolus, P.; Grabner, G.; Köhler, G.; Monti, S. Photochemistry of cyclodextrin host-guest complexes. Coord. Chem. Rev. 1993, 125, 261–268.
- Cram, D.J.; Cram, J.M. Host-Guest Chemistry. Science 1974, 183, 803–809.
- Lehn, J.M. Supramolecular chemistry: Receptors, catalysts, and carriers. Science 1985, 227, 849–856.
- Aiassa, V.; Garnero, C.; Zoppi, A.; Longhi, M.R. Cyclodextrins and Their Derivatives as Drug Stability Modifiers. Pharmaceuticals 2023, 16, 1074.
- Mazurek, A.H.; Szeleszczuk, Ł. A Review of Applications of Solid-State Nuclear Magnetic Resonance (ssNMR) for the Analysis of Cyclodextrin-Including Systems. Int. J. Mol. Sci. 2023, 24, 3648.
- Stella, V.J.; Rajewski, R.A. Sulfobutylether-β-cyclodextrin. Int. J. Pharm. 2020, 583, 119396.
- González Pereira, A.; Carpena, M.; García Oliveira, P.; Mejuto, J.C.; Prieto, M.A.; Simal Gandara, J. Principales aplicaciones de las ciclodextrinas en la industria alimentaria como los compuestos de elección para formar complejos huésped-invitado. Int. J. Mol. Sci. 2021, 22, 1339.
- Zhou, J.; Jia, J.; He, J.; Li, J.; Cai, J. Cyclodextrin Inclusion Complexes and Their Application in Food Safety Analysis: Recent Developments and Future Prospects. Foods 2022, 11, 3871.
- Jansook, P.; Ogawa, N.; Loftsson, T. Cyclodextrins: Structure, physicochemical properties, and pharmaceutical applications. Int. J. Pharm. 2018, 535, 272–284.
- Munoz-Shuguli, C.; Vidal, C.P.; Cantero-Lopez, P.; Lopez-Polo, J. Encapsulation of plant extract compounds using cyclodextrin inclusion complexes, liposomes, electrospinning and their combinations for food purposes. Trends Food Sci. Technol. 2021, 108, 177–186.
- Chizhov, A.O.; Tsvetkov, Y.E.; Nifantiev, N.E. Gas-Phase Fragmentation of Cyclic Oligosaccharides in Tandem Mass Spectrometry. Molecules 2019, 24, 2226.
- Szente, L.; Szejtli, J. Cyclodextrins as food ingredients. Trends Food Sci. Technol. 2004, 15, 137–142.
- Van der Veen, B.A.; Uitdehaag, J.C.M.; Penninga, D.; van Alebeek, G.; Smith, L.M.; Dijkstra, B.W.; Dijkhuizen, L. Rational design of cyclodextrin glycosyltransferase from Bacillus circulans strain 251 to increase alpha-cyclodextrin production. J. Mol. Biol. 2000, 296, 1027–1038.
- Messner, M.; Kurkov, S.V.; Flavia-Piera, R.; Brewster, M.E.; Loftsson, T. Self-assembly of cyclodextrins: The effect of the guest molecule. Int. J. Pharm. 2011, 408, 235–247.
- Wang, X.; Luo, Z.; Xiao, Z. Preparation, Characterization, and Thermal Stability of β-Cyclodextrin/Soybean Lecithin Inclusion Complex. Carbohydr. Polym. 2014, 101, 1027–1032.
- Braithwaite, M.C.; Kumar, P.; Choonara, Y.E.; du Toit, L.C.; Tomar, L.K.; Tyagi, C.; Pillay, V. A novel multi-tiered experimental approach unfolding the mechanisms behind cyclodextrin-vitamin inclusion complexes for enhanced vitamin solubility and stability. Int. J. Pharm. 2017, 532, 90–104.
- Szente, L.; Fenyvesi, É. Cyclodextrin-Enabled Polymer Composites for Packaging. Molecules 2018, 23, 1556.
- Li, Z.; Chen, S.; Gu, Z.; Chen, J.; Wu, J. Alpha-cyclodextrin: Enzymatic production and food applications. Trends Food Sci. Technol. 2014, 35, 151–160.
- Samuelsen, L.; Holm, R.; Schönbeck, C. Simultaneous Determination of Cyclodextrin Stability Constants as a Function of pH and Temperature—A Tool for Drug Formulation and Process Design. J. Drug Deliv. Sci. Technol. 2021, 65, 102675.
- Wei, X.; Yu, C.-Y.; Wei, H. Application of Cyclodextrin for Cancer Immunotherapy. Molecules 2023, 28, 5610.
- Murrieta-Rico, F.N.; Yocupicio-Gaxiola, R.I.; Antúnez-Garcia, J.; Reyes-Serrato, A.; Sánchez, P.; Petranovskii, V. Textile Functionalization Using LTA and FAU Zeolitic Materials. Polymers 2022, 15, 99.
- Sadjadi, S.; Koohestani, F. Composite of β-cyclodextrin and bentonite clay: A promising support for Pd immobilization and developing a catalyst for hydrogenation of nitroarenes under mild reaction condition. J. Phys. Chem. Solids 2021, 151, 109894.
- Loftsson, T.; Brewster, M.E. Pharmaceutical Applications of Cyclodextrins: Basic Science and Product Development. J. Pharm. Pharmacol. 2010, 62, 1607–1621.
- Păduraru, D.N.; Niculescu, A.G.; Bolocan, A.; Andronic, O.; Grumezescu, A.M.; Bîrlă, R. An Updated Overview of Cyclodextrin-Based Drug Delivery Systems for Cancer Therapy. Pharmaceutics 2022, 14, 1748.
- Crini, G.; Fourmentin, S.; Fenyvesi, É.; Torri, G.; Fourmentin, M.; Morin-Crini, N. Fundamentals and applications of cyclodextrins. In Cyclodextrin Fundamentals, Reactivity and Analysis; Springer: Berlin/Heidelberg, Germany, 2018; pp. 1–55.
- Řezanka, M. Synthesis of cyclodextrin derivatives. In Cyclodextrin Fundamentals, Reactivity and Analysis; Springer: Berlin/Heidelberg, Germany, 2018; pp. 57–103.
- Leclercq, L. Interactions between cyclodextrins and cellular components: Towards greener medical applications? Beilstein J. Org. Chem. 2016, 12, 2644–2662.
- Liu, Y.; Chen, Y.N.; Gao, X.L.; Fu, J.J.; Hu, L.D. Application of cyclodextrin in food industry. Crit. Rev. Food Sci. Nutr. 2022, 62, 2627–2640.
- Szejtli, J. Introduction and general overview of cyclodextrin chemistry. Chem. Rev. 1998, 98, 1743–1754.
- Del Valle, E.M.M. Cyclodextrins and their uses: A review. Process Biochem. 2004, 39, 1033–1046.
- Rehman, Q.; Akash, M.S.H.; Imran, I.; Rehman, K. Stability of Pharmaceutical Products. In Drug Stability and Chemical Kinetics; Akash, M.S.H., Rehman, K., Eds.; Springer: Singapore, 2020; pp. 147–154. ISBN 978-981-15-6426-0.
- Loftsson, T. Introduction. In Drug Stability for Pharmaceutical Scientists; Loftsson, T., Ed.; Academic Press: New York, NY, USA, 2014; pp. 1–3. ISBN 9780124115484.
- Przybyla, M.A.; Yilmaz, G.; Becer, C.R. Natural cyclodextrins and their derivatives for polymer synthesis. Polym. Chem. 2020, 11, 7582–7602.
- Jambhekar, S.S.; Breen, P. Cyclodextrins in pharmaceutical formulations II: Solubilization, binding constant, and complexation efficiency. Drug Discov. Today 2016, 21, 363–368.
- Lachowicz, M.; Stańczak, A.; Kołodziejczyk, M. Characteristic of Cyclodextrins: Their Role and Use in the Pharmaceutical Technology. Curr. Drug Targets 2020, 21, 1495–1510.
- Saokham, P.; Muankaew, C.; Jansook, P.; Loftsson, T. Solubility of cyclodextrins and drug/cyclodextrin complexes. Molecules 2018, 23, 1161.
- De Marco, I.; Franco, P. Effect of the Carrier on the Coprecipitation of Curcumin through Supercritical-Assisted Atomization. ChemEngineering 2021, 5, 59.
- Linde, G.A.; Laverde, A.; Colauto, N.B. Changes to Taste Perception in the Food Industry: Use of Cyclodextrins. In Handbook of Behavior, Food and Nutrition; Preedy, V.R., Watson, R.R., Martin, C.R., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 99–118.
- Blanch, G.P.; Ruiz del Castillo, M.L.; del Mar Caja, M.; Pérez-Méndez, M.; Sánchez-Cortés, S. Stabilization of all-trans-lycopene from tomato by encapsulation using cyclodextrins. Food Chem. 2007, 105, 1335–1341.
- Andreu-Sevilla, A.J.; López-Nicolás, J.M.; Carbonell-Barrachina, Á.A.; García-Carmona, F. Comparative Effect of the Addition of α-, β-, or γ-Cyclodextrin on Main Sensory and Physico-Chemical Parameters. J. Food Sci. 2011, 76, S347–S353.
- Dos Santos, C.; Buera, P.; Mazzobre, F. Novel trends in cyclodextrins encapsulation. Applications in food science. Curr. Opin. Food Sci. 2017, 16, 106–113.
- Irie, T.; Uekama, K. Pharmaceutical applications of cyclodextrins. III. Toxicological issues and safety evaluation. J. Pharm. Sci. 1997, 86, 147–162.
- Krawczyk, K.; Silvestri, D.; Nguyen, N.H.; Ševců, A.; Łukowiec, D.; Padil, V.V.; Řezanka, M.; Černík, M.; Dionysiou, D.D.; Wacławek, S. Enhanced degradation of sulfamethoxazole by a modified nano zero-valent iron with a β-cyclodextrin polymer: Mechanism and toxicity evaluation. Sci. Total Environ. 2022, 817, 152888.
- Jumah, M.N.B.; Eid, M.H.; AL-Huqail, A.A.; Mohammad, M.A.; Bin-Murdhi, N.S.; Abu-Taweel, G.M.; Altoom, N.; Allam, A.A.; AbuKhadra, M.R. Enhanced remediation of As (V) and Hg (II) ions from aqueous environments using β-cyclodextrin/MCM-48 composite: Batch and column studies. J. Water Process. Eng. 2021, 42, 102118.
- Paramita, V.; Novia, S.F.; Ariyanto, H.D.; Pramudono, B.; Yoshii, H.; Kusumayanti, H.; Amalia, R. The influence of α-cyclodextrin on the stability and fatty acids of medium-chain triglycerides high oil load nanoemulsion. Mater. Today Proc. 2022, 63, S312–S317.
- Cid-Samamed, A.; Rakmai, J.; Mejuto, J.C.; Simal-Gandara, J.; Astray, G. Cyclodextrins inclusion complex: Preparation methods, analytical techniques and food industry applications. Food Chem. 2022, 384, 132467.
- Astray, G.; Gonzalez-Barreiro, C.; Mejuto, J.C.; Rial-Otero, R.; Simal-Gandara, J. A review on the use of cyclodextrins in foods. Food Hydrocoll. 2009, 23, 1631–1640.
- Mortensen, A.; Aguilar, F.; Crebelli, R.; Di Domenico, A.; Dusemund, B.; Frutos, M.J.; Galtier, P.; Gott, D.; Gundert-Remy, U.; Leblanc, J.; et al. Re-evaluation of β-cyclodextrin (E 459) as a food additive. EFSA J. 2016, 14, e04628.
- Munro, I.C.; Newberne, P.M.; Young, V.R.; Bär, A. Safety assessment of γ-cyclodextrin. Regul. Toxicol. Pharmacol. 2004, 39, 3–13.
- Mura, P. Analytical Techniques for Characterization of Cyclodextrin Complexes in Aqueous Solution: A Review. J. Pharm. Biomed. Anal. 2014, 101, 238–250.
- Jiang, Y.; Abukhadra, M.R.; Refay, N.M.; Sharaf, M.F.; El-Meligy, M.A.; Awwad, E.M. Synthesis of chitosan/MCM-48 and β-cyclodextrin/MCM-48 composites as bio-adsorbents for environmental removal of Cd2+ ions; kinetic and equilibrium studies. React. Funct. Polym. 2020, 154, 104675.
- Chen, L.; Dong, Q.; Shi, Q.; Du, Y.; Zeng, Q.; Zhao, Y.; Wang, J.J. Novel 2,3-Dialdehyde Cellulose-Based Films with Photodynamic Inactivation Potency by Incorporating the β-Cyclodextrin/Curcumin Inclusion Complex. Biomacromolecules 2021, 22, 2790–2801.
- Muankaew, C.; Loftsson, T. Cyclodextrin-based formulations: A non-invasive platform for targeted drug delivery. Basic Clin. Pharmacol. Toxicol. 2018, 122, 46–55.
- Salazar Sandoval, S.; Bruna, T.; Maldonado-Bravo, F.; Bolaños, K.; Adasme-Reyes, S.; Riveros, A.; Caro, N.; Yutronic, N.; Silva, N.; Kogan, M.J.; et al. β-Cyclodextrin Nanosponges Inclusion Compounds Associated with Silver Nanoparticles to Increase the Antimicrobial Activity of Quercetin. Materials 2023, 16, 3538.
- Farouk, A.; Sharaf, S.; Refaie, R.; Abd El-Hady, M.M. Highly Durable Antibacterial Properties of Cellulosic Fabric via β-Cyclodextrin/Essential Oils Inclusion Complex. Polymers 2022, 14, 4899.
- Jia, S.; Jiang, S.; Chen, Y.; Wei, Y.; Shao, X. Comparison of Inhibitory Effects of Cinnamic Acid, β-Cyclodextrin, L-Cysteine, and Ascorbic Acid on Soluble and Membrane-Bound Polyphenol Oxidase in Peach Fruit. Foods 2023, 12, 167.
- Wu, J.; Li, J.; Xu, F.; Zhou, A.; Zeng, S.; Zheng, B.; Lin, S. Effective Preservation of Chilled Pork Using Photodynamic Antibacterial Film Based on Curcumin-β-Cyclodextrin Complex. Polymers 2023, 15, 1023.
- Wu, K.; Zhang, T.; Chai, X.; Duan, X.; He, D.; Yu, H.; Liu, X.; Tao, Z. Encapsulation Efficiency and Functional Stability of Cinnamon Essential Oil in Modified β-cyclodextrins: In Vitro and In Silico Evidence. Foods 2023, 12, 45.
- Zhang, N.; Bi, F.; Xu, F.; Yong, H.; Bao, Y.; Jin, C.; Liu, J. Structure and functional properties of active packaging films prepared by incorporating different flavonols into chitosan based matrix. Int. J. Biol. Macromol. 2020, 165, 625–634.
- Zhang, Y.; Zhang, H.; Wang, F.; Wang, L.-X. Preparation and Properties of Ginger Essential Oil β-Cyclodextrin/Chitosan Inclusion Complexes. Coatings 2018, 8, 305.
- Beshai, H.; Sarabha, G.K.; Rathi, P.; Alam, A.U.; Deen, M.J. Freshness Monitoring of Packaged Vegetables. Appl. Sci. 2020, 10, 7937.
- Wang, H.; Hu, L.; Peng, L.; Du, J.; Lan, M.; Cheng, Y.; Ma, L.; Zhang, Y. Dual encapsulation of β-carotene by β-cyclodextrin and chitosan for 3D printing application. Food Chem. 2022, 378, 132088.
- Bangalore, D.V.; McGlynn, W.; Scott, D.D. Effect of beta-cyclodextrin in improving the correlation between lycopene concentration and ORAC values. J. Agric. Food Chem. 2005, 53, 1878–1883.
- Choi, M.-J.; Ruktanonchai, U.; Min, S.-G.; Chun, J.-Y.; Soottitantawat, A. Physical characteristics of fish oil encapsulated by β-cyclodextrin using an aggregation method or polycaprolactone using an emulsion-diffusion method. Food Chem. 2010, 119, 1694–1703.
- Li, J.; Xiong, S.; Wang, F.; Regenstein, J.M.; Liu, R. Optimization of Microencapsulation of Fish Oil with Gum Arabic/Casein/Beta-Cyclodextrin Mixtures by Spray Drying. J. Food Sci. 2015, 80, C1445–C1452.
