1. Introduction
Diabetic kidney disease is the most common type of chronic kidney disease (CKD) and end-stage kidney disease (ESKD), with substantial morbidity and mortality worldwide [
1,
2]. Chronic kidney disease and congestive heart failure frequently coexist in patients with underlying diabetes mellitus (DM). The association between diabetes mellitus and heart failure (HF) has been well documented. Chronic kidney disease is considered an independent risk factor for cardiovascular disease, and the likelihood of cardiovascular mortality increases with a decline in renal function [
3].
Traditional medical management for CKD and CHF includes blood pressure control, tight glycemic management, and renin–angiotensin–aldosterone system (RAAS) blockade. In practice, angiotensin-converting enzyme inhibitors (ACE inhibitors) or angiotensin receptor blockers (ARBs) are the first line of agents in patients with hypertension and DKD, even in the absence of proteinuria. ACE inhibitors/ARBs have been shown to decrease the risk of progression of CKD in patients with proteinuria, although these agents have not been shown to reduce the progression to ESKD in nonproteinuric patients [
4,
5,
6].
The introduction of SGLT-2 inhibitors has radically changed the therapeutic landscape for CKD and CHF patients. This has added new opportunities for improving outcomes in these patients. SGLT-2 inhibitors were initially developed as glucose-lowering medication. Still, during the earlier cardiovascular outcome trials (CVOTs), it became clear that there is a significant advantage for kidney protection and cardioprotection.
Currently, four SGLT-2i agents are available in North America: canagliflozin, dapagliflozin, empagliflozin, and ertugliflozin, whereas the fifth agent, sotagliflozin, is a combined SGLT-1 and SGLT-2 inhibitor that the FDA recently approved in May 2023 for use in the USA.
2. Mechanism of Action
SGLT-2 inhibitors work by inhibiting the SGLT-2 channel found in the proximal convoluted tubules. This channel is primarily responsible for absorbing most of the filtered glucose, which is >90%. This leads to glucosuria, which is more pronounced in individuals with diabetes than in non-diabetic individuals. The extent of glucose reduction is influenced by kidney function, and it declines when the eGFR falls below 60 mL/min per 1.73 m
2. This effect is almost nonexistent with an eGFR below 30 mL/min per 1.73 m
2 [
7].
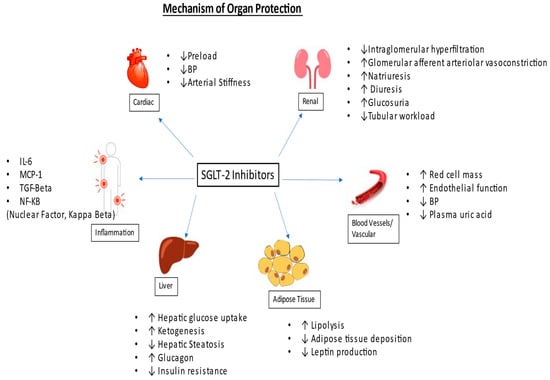
The renoprotective benefits of SGLT-2 inhibitors are independent of the glycosuria effect. It is proposed that SGLT-2 inhibitors reduce sodium reabsorption, leading to increased sodium delivery to the macula densa, which causes negative feedback through the tubuloglomerular feedback mechanism. This results in decreased intraglomerular pressure, causing lowered glomerular hyperfiltration [
8]. Refer to
Figure 1.
Figure 1. Mechanism of organ protection.
It has also been proposed that SGLT-2 inhibitors exert renal protection by improving tubular hypoxia by blocking proximal tubular reabsorption of glucose, reducing the workload of the proximal tubules [
9]. Chronic tubular hypoxia has been attributed to the progression of underlying chronic kidney disease in patients with diabetes mellitus [
10].
Beyond glucosuria, they also contribute to modest bp control, kaliuresis, and reduction in oxidative stress, as well as reducing markers of inflammation and fibrosis such as interleukin-6, monocyte chemoattractant protein 1 (MCP-1), nuclear factor-kB, urinary IL-6 as well as serum tumor necrosis factor receptor 1 (TNFR1) [
11,
12].
These kidney protective benefits lead directly to an improved metabolic profile and indirectly to beneficial effects on cardiovascular disease. Cardioprotective benefits include a decline in arterial stiffness and improvement in endothelial function. SGLT-2 inhibitors also cause diuresis and natriuresis, which may cause a reduction in preload [
13,
14].
3. Effectiveness
3.1. Effectiveness in CKD
Initial cardiovascular outcome trials (CVOTs), including EMPA-REG OUTCOME and CANVAS, were the first to demonstrate the kidney benefit as a secondary outcome. Subsequently, CREDENCE, DAPA-CKD, and EMPA-KIDNEY are the major randomized clinical trials that have solidified prior findings that SGLT-2 inhibitors are effective in slowing the progression to ESKD, reducing albuminuria, and preventing adverse outcomes, including the need for dialysis and cardiovascular mortality. All three trials were terminated early due to strong evidence of efficacy.
The CREDENCE trial included patients with an eGFR 30–90 mL/min/1.73 m
2 and albuminuria (microalbumin-to-creatinine ratio (ACR) of 300–5000 mg/g) and 3.9% of patients had an eGFR less than 30 mL/min per 1.73 m
2. It showed that canagliflozin at a dose of 100 mg once daily was effective in reducing the incidence of ESKD, heart failure hospitalization, all-cause mortality, and cardiovascular mortality, as well as doubling serum creatinine by approximately 30–35% [
15]. In a separate subgroup analysis of patients with an eGFR less than 30 mL/min per 1.73 m
2, it was found that it was equally effective in slowing down the progression of kidney disease as compared to patients with an eGFR greater than 30 mL/min/1.73 m
2 [
16].
DAPA-CKD included patients with an eGFR of 25 to 75 mL/min per 1.73 m
2, proteinuria of 200 to 5000 mg/g, and one-third of the participants in this study had nondiabetic CKD. This trial showed that dapagliflozin 10 mg once daily was able to reduce the incidence of ESKD, all-cause mortality, and cardiac mortality by approximately 35–45%, doubling serum creatinine. The DAPA-CKD trial demonstrated that the protective benefit increases with an increasing hemoglobin A1c level as well as with increased proteinuria and decreased eGFR [
17,
18].
The EMPA-KIDNEY trial is the most recent, published in November 2022, which included patients with an eGFR of 20 to 44 mL/min/1.73 m
2 (regardless of albuminuria) or an eGFR of 45 to 89 mL/min/1.73 m
2 (if albuminuria > 200 mg/g). This was the first trial that included patients with no albuminuria; most patients in this trial (54%) had known chronic diabetic kidney disease. It demonstrated that empagliflozin 10 mg daily reduces the incidence of ESKD, cardiovascular mortality, and all-cause mortality by 25–30%, doubling serum creatinine. The most significant benefit was seen in patients with albuminuria ≥ 300 mg/g and less effect in patients with lower levels of proteinuria [
19]. In this trial with a subgroup of normal albuminuria, the primary endpoint showed no improvement. It is unclear whether this was a false negative or a true negative result. One reason could be that this study was terminated early, after two years, so maybe the duration was not sufficiently long enough to see a difference. However, more studies are needed to evaluate the effect of SGLT-2 inhibitors in patients with normal albuminuria. (
Table 1).
Table 1. Summary of major clinical Trials.
DM-2—Type 2 Diabetes mellitus; CAD—coronary artery disease; CHF—congestive heart failure; CVD—cardiovascular disease; ESKD—end-stage kidney disease; MI—myocardial infarction; NNT—number needed to treat; UACR—urinary albumin-to-creatinine ratio.
3.2. Effectiveness in CHF
The DAPA-HF, EMPEROR-Reduced, and EMPEROR-Preserved trials showed improvement in cardiovascular outcomes and a reduction in death and heart failure hospitalizations [
20,
21,
22].
The DAPA-HF and EMPEROR-Reduced trials focused on patients with congestive heart failure with reduced left ventricular ejection fraction, which was at or below 40%. On the other hand, the EMPEROR-Preserved trial involved patients with a left ventricular ejection fraction greater than 40% [
20,
21,
22].
The DAPA-HF trial included patients with chronic heart failure with LVEF < 40% and an eGFR ≥ 30 mL/min per 1.73 m
2, and it showed significant improvements in composite primary outcomes, including reducing cardiovascular death and heart failure hospitalization by 24% [
20]. The DAPA-HF trial indicated that dapagliflozin may prevent diabetes, and this study showed a decrease in the incidence of new diabetes mellitus [
21].
The EMPEROR-Reduced trial enrolled patients with an eGFR >20 mL/min per 1.73 m and lower mean LVEF of up to 27% compared to up to 31% in the DAPA-HF trial. Empagliflozin reduced the combined risk of primary outcomes, including cardiovascular death or heart failure hospitalization, in patients with or without diabetes by 22% [
22].
The EMPEROR-Preserved clinical trial was groundbreaking as it included patients with heart failure, regardless of whether they had mildly reduced ejection fraction or preserved ejection fraction or whether they had diabetes or not. Empagliflozin decreased the incidence of heart failure hospitalization and cardiovascular death by approximately 20%. Although, in this study, 23% of patients discontinued treatment, which was on the higher side, this outcome was similar in other treatment groups as well [
23].
Similarly, the DELIVER trial was published in September 2022 and included patients with LVEF >40% with medical therapy. In this study, dapagliflozin showed a reduction in the primary composite endpoint by 18% [
24].
3.3. Effectiveness in Stroke
SGLT-2 inhibitors have been very successful in improving outcomes in chronic kidney disease and cardiovascular disease. However, when it comes to preventing strokes, the available data are not yet as compelling.
Chronic kidney disease frequently coexists with other complex medical problems, including cardiovascular disease and stroke. Many experts believe that chronic kidney disease is an independent risk factor for stroke, and chronic kidney disease is present in over 40% of patients with stroke [
25]
Stroke and chronic kidney disease have many risk factors in common, such as hypertension, diabetes mellitus, and atrial fibrillation. In the DECLARE-TIMI 58 trial, dapagliflozin was shown to decrease the incidence of atrial fibrillation [
26].
In theory, one would believe that as SGLT-2 inhibitors improved outcomes in diabetes, kidney disease, and cardiovascular disease, they would also reduce the risk of stroke, but that has not been the case so far.
Multiple meta-analysis trials from recent cardiovascular outcome trials (CVOTs) showed no effect of SGLT-2 inhibitors in preventing strokes [
27]. A meta-analysis of the credence trial showed that the SGLT-2 inhibitor showed no overall positive impact on total stroke prevention but had a slight benefit in terms of preventing hemorrhagic stroke [
28].
However, most recently, in May 2023, an observational study from Taiwan published in the
Journal of the American Heart Association suggested that an SGLT-2 inhibitor can decrease the incidence of new stroke by 20% in patients with atrial fibrillation [
29]. This was an observational study to evaluate the impact of SGLT-2 inhibitors in preventing embolic stroke in patients with atrial fibrillation and diabetes mellitus. One of the drawbacks of this study was that it was not a randomized control trial. Nonetheless, this brings a ray of hope regarding stroke outcomes and provides direction for further studies.
This entry is adapted from the peer-reviewed paper 10.3390/jcm12196202