Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Phosphodiesterase 5 (PDE5) inhibitors presented themselves as important players in the nitric oxide/cGMP pathway, thus exerting a profound impact on various physiological and pathological processes. Beyond their well-known efficacy in treating male erectile dysfunction (ED) and pulmonary arterial hypertension (PAH), a plethora of studies have unveiled their significance in the treatment of a myriad of other diseases, including cognitive functions, heart failure, multiple drug resistance in cancer therapy, immune diseases, systemic sclerosis and others.
- phosphodiesterase 5 inhibitors
- selectivity
- NO/cGMP
1. Introduction
PDE5 inhibitors (PDE5-Is) are groundbreaking medications for treating ED. They increase cGMP levels, causing muscle relaxation and vasodilation in the penis, leading to erections. Their therapeutic potential extends beyond ED, with clinical approval for treating PAH, BPH, and LUTS. Research highlights their potential in diseases like cancer, neurological disorders, cystic fibrosis, and diabetes. Promisingly, this has incited the development of new PDE5-Is with higher potency, selectivity, and improved pharmacokinetics for enhanced efficacy.
2. Classification of Phosphodiesterases
PDE superfamily comprises 11 families (PDE1–PDE11) that are encoded by 21 different genes, whose expression are modulated via multiple promotors and messenger RNA (mRNA) alternative splicing generating more than 50 isoforms [1][2]. It is worth noting that PDE12, which cleaves 2′,5′-phosphodiester bond linking adenosines of the 5′-triphosphorylated oligoadenylates, belongs to the C–C chemokine receptor 4 (CCR4)/nocturin family [3] and is not a member of the cyclic nucleotide PDE superfamily. PDE isoforms are classified based on their amino acid sequences, substrate specificities, catalytic and cofactor requirements, kinetic properties, regulatory mechanisms, and tissue distributions [1]. Some PDEs are selective for the hydrolysis of cAMP (PDE 4, 7, and 8) or cGMP (PDE 5, 6 and 9), while others can hydrolyze both cAMP and cGMP (PDE 1, 2, 3, 10 and 11) [4]. PDEs share a conserved catalytic domain (C domain) but differ significantly in their N-terminal regulatory domains. PDEs are mainly regulated via (i) binding of Ca2+/calmodulin (PDE1), (ii) phosphorylation/dephosphorylation events (PDE1, 3, 4 and 5) and (iii) allosteric binding of cGMP via GAF domains (PDE2, 5, 6, 10 and 11) [1]. The description of diverse tissue distribution/cell expression and functional significance of PDE isoenzymes is detailed in [5] and is beyond the scope of this discussion. Notably, such tissue/cellular compartmentalization allows selective PDE inhibitors to exert their effects almost exclusively on the target tissue.
The focus herein is on the PDE5 family, which is generated by one gene, PDE5A, and has three alternative spliced variants, PDE5A1, 5A2 and 5A3. The three human PDE5 isoforms differ only in the 5’-end of the mRNA and the corresponding N-terminal of the protein. These isoforms have similar phosphorylation sites, allosteric cGMP-binding sites, catalytic domain and cGMP binding and hydrolysis activities [6]. However, PDE5A1 was reported to be more resistant to chemical inhibition than PDE5A2 or PDE5A3. PDE5A1 and PDE5A2 are widely distributed in nearly all tissues, whereas PDE5A3 is confined to vascular smooth muscle cells [2].
3. Tissues and Organs of High Expression for PDE5
PDE5 is present in virtually all cell types, tissues and organs. PDE5 is highly expressed in the smooth muscle cells of the peripheral arteries and venous vessels and in coronary and pulmonary arteries [2]. In addition, PDE5 is expressed in the vascular smooth muscle cells of the corpora cavernosa of the penis besides spermatozoa, peritubular myoid of Leydig cells and vas deferens in males [7]. PDE5 is widely distributed in the cytoplasmic cell compartment in myometrial cells, endothelial cells and peripheral blood mononuclear cells. It is also expressed in skeletal muscles, cardiomyocytes, platelets, lung, spinal cord, cerebellum, retina, pancreas, prostate, urethra and bladder [1][2][8]. PDE5A1 and PDE5A2 are further expressed in renal vessels, glomeruli, tubular epithelial cells of the renal proximal tubule and medullary collecting duct [9]. Consequently, PDE5 isoforms exhibit diverse and numerous functions both in physiological and pathological conditions.
4. PDE5 Physiological Role
Nitric oxide (NO) is synthesized from the precursor L-arginine through the activities of different NO synthases (neuronal, inducible or endothelial NOS). Intracellularly, NO binds to and activates soluble guanylyl cyclase (sGC), promoting the conversion of guanosine triphosphate (GTP) to the second messenger cyclic guanosine monophosphate (cGMP) [10][11]. Thereafter, cGMP activates protein kinase G (PKG), whose phosphorylation mediates activities of various membrane channels/pumps, leading to decreased calcium influx through L-type calcium channels and increased calcium sequestration, resulting in smooth muscle relaxation and vascular tone modulation [12]. PKG-dependent phosphorylation of other various downstream proteins can regulate further pivotal physiological functions, such as cell differentiation and proliferation, endothelial permeability, ion transport, secretion and gene transcription [13].
Given the broad expression and the ability of PDE5 to specifically hydrolyze cGMP, controlling its cellular levels, PDE5 has been proposed as a crucial player in many NO/cGMP/PKG-dependent biological processes such as smooth muscle relaxation, heart muscle contraction, platelet activation/aggregation and immune response [14].
PDE5 inhibition was found to enhance smooth muscle relaxation and vasodilation, which in the penis corpus cavernosum favors erection, in the pulmonary vasculature decreases pulmonary vessels’ pressure, and in the systemic circulation decreases arterial blood pressure [15].
In addition, PDE5 is an important regulator of platelet function, whose inhibition increases platelet cGMP levels and augments the ability of NO to inhibit platelet aggregation and activation [16].
Furthermore, PDE5 governs fundamental physiological processes in the kidney. It can regulate renal vascular blood flow by hampering cGMP-mediated vascular relaxation. PDE5 is also a negative regulator for cGMP-dependent natriuresis. Moreover, it increases renin synthesis by degrading cGMP in juxtaglomerular cells [17].
5. PDE5 as a Drug Target for Disease Treatment
Competitive PDE5-Is reported so far exclusively bind to the catalytic domain, preventing cGMP hydrolysis and elevating its levels in cells of various tissues [18]. The subsequent activation or restoration of normal NO/cGMP/PKG signaling cascade prompted the use of these inhibitors as therapeutics for several clinical indications. Food and Drug Administration (FDA)-approved PDE5-Is (Figure 1) include (i) sildenafil (approved in 1998 for erectile dysfunction (ED) as Viagra®, and in 2005 for pulmonary arterial hypertension (PAH, WHO Group I) as Revatio®), (ii) vardenafil (approved in 2003 for ED as Levitra®), (iii) tadalafil (approved in 2003 for ED as Cialis®, in 2009 for PAH (WHO Group I) as Adcirca® and in 2011 for lower urinary tract symptoms secondary to benign prostatic hyperplasia (LUTS/BPH) with or without ED and the most recent (iv) avanafil (approved in 2012 for ED as Stendra®) [19][20].
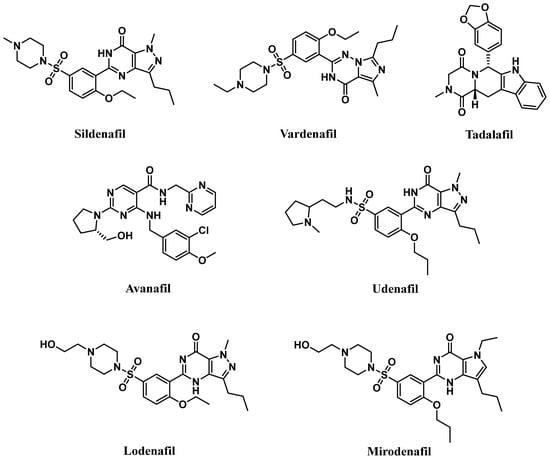
Figure 1. Chemical structures of marketed PDE5 inhibitors.
These inhibitors differ in their selectivity, potency, onset and duration of action, cost, administration considerations, precautions, and adverse effects profiles. The relative potency of vardenafil for PDE5 was reported to be the highest (PDE5 IC50 of 0.1–0.4 nM), followed by tadalafil (PDE5 IC50 of 2 nM), and then sildenafil and avanafil (PDE5 IC50 of 4 nM and 4.3–5.2 nM, respectively). They all share a quick onset time in the range of 11–16 min after oral administration, with avanafil advertised as the fastest-acting. Plasma half-lives of sildenafil and vardenafil are similar, about 4 h, and their efficacy of action lasts up to 12 h. Avanafil half-life is shorter, about 3 h, with a maximal duration of action of 6 h. Tadalafil’s half-life is the longest, 17.5 h, with an efficacy maintained for up to 36 h [18][20][21][22].
A new generation of PDE5-Is, namely lodenafil, udenafil, and mirodenafil are also available in Brazil and Korea for ED treatment (Figure 1), but none of them have been FDA-approved, yet [23].
Aside from the three FDA-approved clinical indications, PDE5-Is have been intensively investigated for their potential use in the treatment of various emerging indications, such as cancer, central nervous system (CNS) and cardiovascular system (CVS) related diseases, kidney diseases, cystic fibrosis and diabetes, all of which will be discussed herein.
5.1. Approved Clinical Uses of PDE5 Inhibitors
5.1.1. Erectile Dysfunction
In the corpora cavernosa, parasympathetic stimulation and sexual arousal induce the release of NO from endothelial cells and nitrergic neurons surrounding the arteries and sinusoids, leading to increased cGMP synthesis. PDE5-Is can slow the degradation of penile connective tissue cGMP. This leads to a drop in the intracellular Ca2+ levels in the corpus cavernosum smooth muscles, causing their relaxation and a reduction in arterial blood drainage, providing a sufficient degree of penile tumescence and sustaining penile erection (Figure 2). Accordingly, it can be deduced that the action of PDE5-Is requires normal neuronal input into the erectile tissues, as well as unimpaired cavernous endothelial structures [24][25].
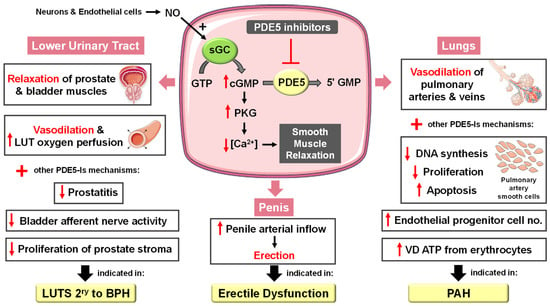
Figure 2. Approved clinical uses of PDE5 inhibitors. Nitric oxide (NO) is produced by neurons and endothelial cells. Inside smooth muscle cells, NO activates soluble guanylyl cyclase (sGC), promoting the conversion of guanosine triphosphate (GTP) to the second messenger cyclic guanosine monophosphate (cGMP). Thereafter, cGMP activates protein kinase G (PKG), whose phosphorylation mediates activities of various membrane channels/pumps, leading to decreased intracellular calcium levels resulting in smooth muscle relaxation (SMR). Phosphodiesterase 5 (PDE5) regulates cGMP levels by degrading it into inactive 5′ guanosine monophosphate (5′ GMP). PDE5-Is can thus enhance the cGMP/PKG pathway, boosting the relaxation of various smooth muscles. In the penis corpus cavernosum, SMR favors erection due to increased penile arterial inflow, and thus PDE5-Is are approved for the treatment of erectile dysfunction. In the lungs, PDE5-Is lead to vasodilation of pulmonary vasculature, which, along with other mechanisms, such as suppressed DNA synthesis and proliferation and enhanced apoptosis of pulmonary artery cells, increased endothelial progenitor cell number, and enhanced release of vasodilating adenosine triphosphate (ATP) from erythrocytes culminate in effectiveness in the treatment of pulmonary arterial hypertension (PAH). In the lower urinary tract (LUT), PDE5-Is mediate prostate and bladder SMR, vasodilation and increased LUT oxygen perfusion. In addition, PDE5-Is could suppress prostatitis, bladder afferent nerve activity and prostate stroma cell proliferation, and thus indicated in the treatment of LUT symptoms secondary to benign prostatic hyperplasia (BPH).
PDE5-Is are thus considered the first-line choice for on-demand and chronic treatment of most ED cases. The efficacy and safety of the four FDA-approved PDE5-Is (sildenafil, vardenafil, tadalafil and avanafil) have been confirmed by a multitude of worldwide clinical trials involving thousands of ED patients with diverse etiologies that were documented by several reviews [19][26][27][28][29][30].
It is worth noting that the few differences between sildenafil, tadalafil and vardenafil pharmacokinetics allow tadalafil, with a longer half-life, to be superior in a number of sexual intercourses per pill, while vardenafil and sildenafil exhibited privilege whenever duration of erection, or vascular efficacy and penile hardness are explored [31][32].
5.1.2. Pulmonary Arterial Hypertension
PAH is a disease associated with endothelial dysfunction, vascular remodeling and fibrosis that causes gradual progression of pulmonary vascular resistance, ultimately leading to right heart failure. Accordingly, PAH therapies usually aim to enhance vasodilation, suppress cellular hyperproliferation and induce apoptosis [33].
PDE5 is highly expressed in the lung vasculature [34]. The fact that lung endothelial NOS is reduced [35] and PDE5 is upregulated in the remodeled pulmonary artery during PAH has proposed PDE5-Is as a potential PAH treatment [36]. A plethora of PDE5 inhibition-mediated mechanisms have been documented (Figure 2) including (i) activation of the NO/cGMP/PKG pathway, resulting in decreased calcium influx through L-type calcium channels and increased calcium sequestration, inducing vasorelaxation [37], (ii) suppression of DNA synthesis and cell proliferation and stimulation of apoptosis of pulmonary artery smooth cells whose proliferation is involved in the pathogenesis of intimal hyperplasia and major vascular lesions in PAH [38], and (iii) increasing circulating endothelial progenitor cell (EPC) number [39].
Several clinical studies confirmed the potential of PDE5-Is to improve several hemodynamic and clinical parameters in PAH patients [40][41][42][43], such as diminishing pulmonary artery systolic and mean artery pressure, dyspnea score and gas transfer, pulmonary vascular resistance and cardiac output [44]. Furthermore, PDE5-Is could improve ventilatory efficiency and oxygen uptake kinetics and prevent exercise-induced pulmonary edema [45]. Vardenafil usually exhibits the most rapid effect on pulmonary vasorelaxation, while sildenafil and tadalafil are more selective for pulmonary circulation. Substantial enhancement of arterial oxygenation is mainly observed with sildenafil [46].
Sildenafil, in 2005, and, thereafter, tadalafil have been FDA approved and became first-line therapies for PAH [47], primary or secondary to other connective tissue diseases, such as scleroderma (SSc) or systemic lupus erythematosus (SLE) [34].
PDE5-Is can also be used as combination therapy with other PAH-targeted treatments. The combination of sildenafil and long-term intravenous epoprostenol therapy was superior to epoprostenol monotherapy regarding improved exercise capacity, hemodynamic measurements and prolonged time to clinical worsening [48]. Other combinations, such as tadalafil with the endothelin receptor antagonist ambrisentan and sildenafil with systemic nitrates [49], were proven safe and effective in potentiating vasodilation and reducing mortality in PAH patients. Moreover, combined prostacyclin analogs and PDE5-Is were reported to synergistically enhance the release of the potent vasodilator ATP from PAH erythrocytes [50].
PDE5 inhibition has also emerged as a therapeutic strategy for high-altitude PAH where sildenafil’s ability to reverse hypoxia-mediated pulmonary vasoconstriction was proved to mediate positive results on exercise performance and lung hemodynamics [51][52].
5.1.3. Lower Urinary Tract Symptoms Secondary to Benign Prostatic Hyperplasia
Several studies have established an association between ED and BPH-related LUTS where alterations in the NO/cGMP pathway, alterations in RhoA/Rho kinase/endothelin signaling, pelvic atherosclerosis, autonomic adrenergic hyperactivity, inflammatory pathways, sex hormones and psychological factors were the major contributing factors [53][54]. Accordingly, attention was drawn towards the development of a single therapy to treat both conditions.
The clinical benefits of chronic PDE5 inhibition on LUTS secondary to BPH, regardless of whether these symptoms are associated with ED, are well documented [55]. These beneficial effects have been correlated to several mechanisms (Figure 2), including (i) stromal smooth muscle relaxation of the prostate and bladder due to modulation of the NO/cGMP pathway in the nitrinergic innervated organs or enhanced generation of relaxing hydrogen sulfide, (ii) significant cGMP-mediated dilatation of local blood vessels, (iii) enhanced LUT oxygen perfusion, (iv) inhibition of afferent nerve activity of bladder, (v) down-regulation of prostate inflammation and (vi) negative regulation of proliferation and trans-differentiation of the prostate stroma [54][56][57].
Many preclinical studies of PDE5 and its inhibitors in the prostate and bladder (reviewed in [58]) could validate the role of PDE5-Is in relaxing prostatic tissue, improving the severity of urinary symptoms, reducing bladder overactivity, decreasing indicators of bladder ischemia, normalizing changes in NOS activity and preventing the accumulation of collagen [59].
Several clinical trials demonstrated that the use of PDE5-Is alone could ameliorate LUTS in the first 12 weeks of treatment, where sildenafil [60], tadalafil [61][62][63] and vardenafil [64] led to a decrease, at different degrees, in the International Prostate Symptom Score (IPPS) scale. In particular, the effects of tadalafil 5 mg once daily versus placebo on LUTS/BPH have been extensively investigated (reviewed by Gacci et al. [65]). Only tadalafil (5 mg once daily) has been licensed for the treatment of LUTS with or without ED.
The combined administration of sildenafil, tadalafil or vardenafil with the α1-adrenoceptor antagonists alfuzosin or tamsulosin for the treatment of LUTS/BPH has also been evaluated and was confirmed to often outperform either type of monotherapy [66][67][68][69][70]. Interestingly, a very recent meta-analysis of randomized clinical trials demonstrated that tadalafil could be superior to tamsulosin in treating LUTS/BPH when associated with ED [71].
5.2. Emerging and Future Uses of PDE5 Inhibitors
5.2.1. Cancer
Numerous studies have reported the role of cGMP in suppressing cell growth and inducing apoptosis and that elevated PDE5 expression is involved in the progression of various tumor types, such as chronic lymphocytic leukemia, colon adenocarcinoma, bladder squamous carcinoma, human papillary thyroid carcinomas, metastatic breast, prostate, pancreatic and lung cancers [72][73][74]. Accordingly, PDE5 has gained attention as a promising target for anticancer drug discovery. Over the last two decades, several pre-clinical and clinical studies revealed potential anti-cancer effects of PDE5-Is [75][76] that were mediated via different mechanisms of action discussed herein (Figure 3).
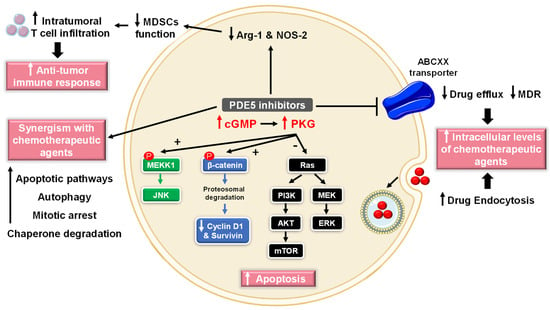
Figure 3. Anti-cancer mechanisms of PDE5 inhibitors. Via activation of the cGMP/PKG signaling cascade, PDE5-Is can induce apoptosis in cancer cells via various pathways; activation of c-Jun NH2-terminal kinase (JNK) via phosphorylation of mitogen-activated protein kinase kinase kinase 1 (MEKK1), phosphorylation of β-catenin and inducing its proteosomal degradation which leads to decreased expression of Wnt/β-catenin regulated proteins, such as cyclin D1 and survivin in addition to blocking the phosphoinositide 3-kinase (PI3K)/AKT/mammalian target of rapamycin (mTOR) and the mitogen-activated protein kinase kinase/extracellular signal-regulated kinase (MEK/ERK) signaling pathways. PDE5-Is could also increase intracellular levels of other chemotherapeutic agents via inhibition of the ATP-binding cassette (ABC) transporter-mediated drug efflux, averting multidrug resistance (MDR) in addition to increasing cellular drug uptake via enhancing endocytosis. Moreover, PDE5-Is synergize with other chemotherapeutic agents via boosting various apoptotic, autophagy, mitotic arrest and chaperone degradation pathways. PDE5-Is can also abrogate the function of myeloid-derived suppressor cells (MDSCs) via suppression of arginase-1 (Arg-1) and nitric oxide synthase–2 (NOS-2) production. This results in enhanced intratumoral T-cell infiltration and activation and restores both systemic and tumor-specific immunity. P = phosphorylation.
- (1)
-
Cell growth arrest and induction of apoptosis
Sildenafil and vardenafil were reported to induce caspase-dependent apoptosis and antiproliferative effects in B-cell chronic lymphatic leukemia [77]. Moreover, sildenafil was shown to boost intracellular reactive oxygen species (ROS) levels, induce cell cycle arrest, and suppress cell proliferation in colorectal cancer cells [78]. In addition, multiple studies have validated the proapoptotic effects of exisulind (sulindac sulfone) and sulindac sulfide (SS), two metabolites of the non-steroidal anti-inflammatory drug (NSAID) sulindac, in breast, colorectal and metastatic prostate cancers. Exisulind or SS increases the activation of cGMP-dependent PKG, triggering a series of signaling events (Figure 3), including (i) phosphorylation of β-catenin and inducing its proteosomal degradation which leads to decreased expression of Wnt/β-catenin regulated proteins, such as cyclin D1 and survivin, (ii) activation of c-Jun NH2-terminal kinase (JNK) via phosphorylation of mitogen-activated protein kinase kinase kinase 1 (MEKK1), and (iii) blocking the phosphoinositide 3-kinase (PI3K)/AKT/mammalian target of rapamycin (mTOR) and the mitogen-activated protein kinase kinase/extracellular signal-regulated kinase (MEK/ERK) signaling pathways, all of which culminate in triggering apoptosis cascade [79][80][81][82][83][84].
- (2)
-
Chemotherapy sensitization
Several studies provided evidence that PDE5-Is can increase cellular concentrations of standard chemotherapeutic drugs or even enhance their efficacy within certain tumor cells where a combination of potential agents allows the reduction of dose levels and, consequently, of toxic side effects (Figure 3) [76][85][86][87].
One of the major causes of chemotherapy failure in cancer treatment is multidrug resistance (MDR) attributed to overexpression of the ATP-binding cassette (ABC) transporters, such as P-glycoprotein (ABCB1/P-gp/MDR1), multidrug-resistance proteins (ABCCs/MRPs) and breast cancer resistant protein (ABCG2/BCRP). These transporters actively expel chemotherapeutic agents out of the cancer cell, ameliorating their cellular efficacy [88]. Vardenafil was reported to inhibit the drug efflux in ABCB1-overexpressing cells [89], while sildenafil was effective in opposing the activity of ABCB1 and ABCG2, both attenuating MDR in tumor cells [90].
Another study showed that PDE5-Is can increase cellular uptake of structurally diverse compounds into lung cancer cells both in vitro and in vivo via modulation of endocytosis [85]. Moreover, oral administration of sildenafil and vardenafil was found to actively enhance blood tumor barrier (BTB) permeability and boost the efficacy of chemotherapy in a rat brain tumor model [91]. Vardenafil could also enhance the delivery and therapeutic efficacy of herceptin monoclonal antibodies in mouse models of metastatic HER2/neu-positive brain tumors through stimulating caveolae-mediated endocytosis and micropinocytosis [92].
Besides augmenting the delivery of chemotherapeutic agents, PDE5-Is can suppress tumor growth and induce cell death by synergizing with current chemotherapy medications in treating a wide range of cancers (Figure 3).
Celecoxib and PDE5-Is synergize in a NOS-dependent cyclooxygenase (COX)-independent fashion to kill multiple tumor cell types, including human glioma cells, as well as their associated activated microglia in vitro and could suppress the growth of mammary tumors in vivo. The drug combination increased the levels of autophagy by inactivating mTOR and inducing endoplasmic reticulum (ER) stress responses in these cells [93].
A combination of sildenafil with various standard chemotherapy agents was proved effective in various gastrointestinal/genitourinary cancers, such as bladder and colon cancers [87]. A combination of the topoisomerase inhibitor doxorubicin and sildenafil resulted in increased efficacy against prostate cancer cells through ROS generation and subsequent upregulation of pro-apoptotic proteins Bad and Bax and downregulation of anti-apoptotic proteins Bcl-2 and Bcl-xL, amplifying caspase-mediated apoptotic death [94]. In a later study, sildenafil and vardenafil but not tadalafil were found to induce PDE5-independent apoptotic sensitization to doxorubicin in castration-resistant prostate cancer (CRPC) cells through impairment of both homologous recombination (HR) and non-homologous end joining (NHEJ) DNA repair pathways [95]. Furthermore, both in vitro and in vivo studies suggested that sildenafil could synergistically potentiate vincristine-induced mitotic arrest signaling and sensitize caspase-dependent apoptosis in CRPC cells via a mitochondrial damage pathway [96].
The multi-kinase inhibitors sorafenib/regorafenib in combination with sildenafil were reported to suppress xenograft tumor growth using liver and colon cancer cells in a greater than additive manner via various autophagy and intrinsic and extrinsic apoptotic pathways [97]. In multiple genetically diverse lung cancer cell lines, sildenafil increased the lethality of pemetrexed and sorafenib combination via fully inactivating signaling by multiple cytoprotective proteins, including the AKT/ERK pathways, nuclear factor-κB (NF-κB) and STAT3/STAT5 besides enhancing death receptor expression and activation [98].
Treatment of stem-like glioblastoma cells with a combination of OSU-03012 (a non-COX-2 inhibiting derivative of celecoxib) and sildenafil abolished the expression of multiple oncogenic growth factor receptors and plasma membrane drug efflux pumps and caused rapid degradation of glucose-regulated protein (GRP78) and other chaperones in tumor cells. This downregulates key oncogenic kinases, including PI3K/AKT signaling, leading to tumoricidal effects [99]. Similarly, sildenafil alone or in combination with the heat shock protein 90 (HSP90) inhibitor PU-H71 could alter the expression of HSP90 chaperone followed by degradation of the oncogenic protein kinase D2 impairing proliferation and viability of various tumor cell lines [100]. These studies suggest a combination of PDE5 and chaperone inhibitors as a novel, promising strategy for targeting cancer.
- (3)
-
Modulation of antitumor immune response
PDE5 inhibition contrasts tumor-induced immunosuppressive mechanisms and generates a measurable antitumor-immune response that significantly delays tumor progression. Both sildenafil and tadalafil could abrogate the function of myeloid-derived suppressor cells (MDSCs) via suppression of arginase-1 (Arg-1) and nitric oxide synthase–2 (NOS-2) production. This resulted in enhanced intratumoral T-cell infiltration and activation and restored both systemic and tumor-specific immunity in multiple myeloma and head and neck cancer patients (Figure 3) [101][102].
- (4)
-
Chemopreventive mechanisms
A nationwide population-based study in Sweden suggested that the use of PDE5-Is was associated with a lower risk of colorectal cancer among male patients with benign colorectal neoplasm [103]. Moreover, two very recent studies provided evidence that sildenafil was more effective than tadalafil in preventing the development and progression of aflatoxin B1-induced hepatocellular carcinoma. This beneficial effect was attributed to a plethora of mechanisms, including (i) improved enzymatic antioxidant system capacity with a concomitant decline in the level of lipid peroxidation, (ii) increase in activity of glutathione S-transferase, (iii) downregulation of glucose transporter 1 (GLUT1) restoring normal declined blood glucose levels in tumor cells, (iv) inhibition of lactate dehydrogenase dependent glycolytic machinery, (v) vasodilation of blood vessels resulting in decreased tumor hypoxia and downregulation of the angiogenesis markers; hypoxia-inducible factor 1-alpha (HIF-1α), transforming growth factor-beta 1 (TGF-β1) and vascular endothelial growth factor A (VEGFA) [104][105]. PDE5-Is have also been shown to suppress the stemness of PC3-derived cancer stem cells (PCSCs) that were confirmed essential for the initiation, progression and recurrence of prostate cancer. cGMP-dependent PKG promotes mammalian sterile 20-like kinase/large tumor suppressor (MST/LATS) kinases, leading to cytosolic degradation of the oncogenic protein Tafazzin (TAZ) and the activation of the Hippo pathway, a crucial player in modulating stemness of PCSCs [106].
5.2.2. CNS Diseases
cGMP/PKG signaling has been regarded as a central mechanism of neuroinflammation, neurodegeneration and cognitive disorders [106][107]. Accordingly, PDE5-Is have gained growing attention as potential therapeutic agents for the treatment of several CNS-related diseases, such as Alzheimer’s disease (AD), cognitive deficits, strokes, multiple sclerosis (MS), depression, noise-induced hearing loss (NIHL) and neuropathic pain that will all be discussed in this section (Figure 4).
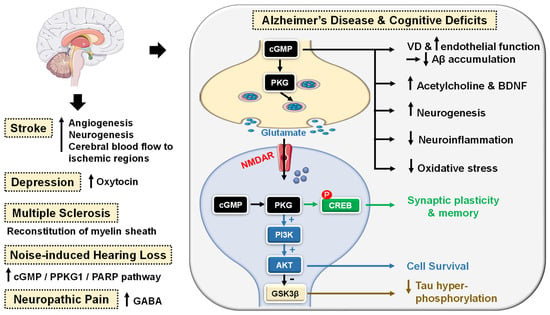
Figure 4. Emerging central nervous system (CNS)-related indications of PDE5 inhibitors. In Alzheimer’s disease (AD) and cognitive deficiency disease models, PDE5 inhibition increases presynaptic cGMP levels, which, through PKG activation, enhances the release of glutamate and activates N-methyl-D-aspartate receptors (NMDAR). On the other hand, postsynaptic PKG activates transcription factor cyclic adenosine monophosphate (cAMP) response element-binding element (CREB), promoting neurotransmission, synaptic plasticity and memory consolidation. PKG also activates the PI3K/AKT signaling pathway that mediates neuroprotection via the inhibition of apoptosis and also suppresses tau hyper-phosphorylation via inhibition of glycogen synthase kinase-3 beta (GSK3β). Elevated cGMP levels exhibit other cognitive enhancement mechanisms, such as vasodilation, which improves or maintains cerebrovascular endothelial function, preventing Aβ amyloid accumulation, rise in acetylcholine (ACh) and brain-derived neurotrophic factor (BDNF) levels in the cortex, striatum, and other areas of the brain, facilitation of neurogenesis, suppression of neuroinflammation and oxidative stress, all averting neuronal loss. In strokes, PDE5-Is could induce angiogenesis and neurogenesis and enhance cerebral blood flow to ischemic regions. PDE5-Is have anxiolytic effects in part due to enhanced oxytocin release. Moreover, PDE5-Is can promote efficient reconstitution of the myelin sheath and govern the Inflammatory processes involved in demyelination models of multiple sclerosis. PDE5-Is are also beneficial in noise-induced hearing loss via activating cGMP/protein kinase cGMP-dependent 1/poly (ADP-ribose) polymerase (cGMP/PRKG1/PARP) signaling in response to traumas in cochlea sensory cells. PDE5-Is exhibit pain-relieving effects in neuropathic pain models via enhanced release of gamma-aminobutyric acid (GABA). P = phosphorylation.
PDE5 inhibition increases presynaptic cGMP levels, which, through PKG activation, enhances the release of glutamate and activates N-methyl-D-aspartate (NMDA) receptors. On the other hand, postsynaptic PKG activates transcription factor cyclic adenosine monophosphate (cAMP) response element-binding element (CREB), promoting neurotransmission, synaptic plasticity and memory consolidation [108][109]. PKG also activates the PI3K/AKT signaling pathway that mediates neuroprotection via the inhibition of apoptosis (Figure 4) [110].
The upregulation of PDE5 expression in the brains of AD patients and the subsequent drop in cGMP levels have been linked to the elevation of Aβ amyloid peptide, whose deposition in the brain is the main hallmark of AD [111]. Sabayan et al. described PDE5-Is as disease-modifying agents against AD and proposed three main mechanisms for their action: (i) vasodilation, which improves or maintains cerebrovascular endothelial function preventing Aβ amyloid accumulation; (ii) cGMP-dependent rise in acetylcholine (ACh) levels in the cortex, striatum, and other areas of the brain, reversing low-ACh associated memory and cognitive deficits in AD, and finally (iii) inhibition of apoptosis and facilitation of neurogenesis averting neuronal loss (Figure 4) [112].
For example, chronic administration of sildenafil completely reversed cognitive impairment in Tg2576 transgenic mice without changing Aβ load. The underlying mechanism involved suppression of tau hyperphosphorylation and inhibition of glycogen synthase kinase 3β (GSK3β) and cyclin-dependent kinase 5 (CDK5) [113]. In addition, Puzzo et al. and Zhang et al. showed that chronic administration of sildenafil in amyloid precursor protein/presenilin-1 (APP/PS1) transgenic mice could reverse AD-related cognitive deficits and synaptic dysfunction via improving cGMP/PKG/CREB signaling, inhibiting neuroinflammation and reducing hippocampal Aβ levels [114][115].
Chronic treatment with tadalafil even exhibited a higher beneficial effect, probably due to its longer half-life and could improve spatial memory in the J20 mouse model of AD by decreasing tau protein via the activation of the AKT/GSK3β pathway [116]. Most recently, mirodenafil was reported to ameliorate Aβ-induced AD pathology and improve cognitive behavior in the APP-C105 mouse model through the modulation of the cGMP/PKG/CREB signaling pathway, GSK-3β activity, glucocorticoid receptor transcriptional activity and Wnt/β-catenin signaling in neuronal cells (Figure 4) [107].
Preclinical studies proved that PDE5-Is could boost memory and synaptic plasticity by augmenting the NO/cGMP/PKG pathway [107][117]. In mouse models with induced cognitive deficits, sildenafil could improve novel object recognition, ameliorate cognitive impairment and upregulate the brain-derived neurotrophic factor (BDNF), contributing to neuroprotective effects [118][119]. Another study showed the potential of sildenafil to defy neurological stress, increase neuroprotection and restore cognitive functions in the hippocampus region of noise alone-induced mice via modulation of cGMP/PKG/CREB and p25/CDK5 pathways and induction of various free radical scavengers in the brain of stressed mice [120]. A similar alleviation n of oxidative stress in the hippocampus of aged mice has been observed upon chronic tadalafil administration as well (Figure 4) [121].
Very recent reviews by Liu et al. [122] and Zuccarello et al. [123] summarized clinical trials of PDE5-Is in cognition and AD. However, none of the investigated drugs has reached the market for those indications so far.
Numerous animal models investigated the potential role of PDE5 inhibition in stroke. In these studies, PDE5-Is could induce angiogenesis, enhance cerebral blood flow to the ischemic region, increase neurogenesis and advanced functional post-stroke recovery [124][125][126]. In particular, sildenafil treatment for two weeks (25 mg daily) was proven safe in patients who suffered mild to moderate strokes [127]. Additionally, tadalafil could attenuate ischemia-induced short-term memory impairment by suppressing ischemia-induced neuronal apoptosis [128].
Further mechanisms for PDE5 inhibition-induced neurogenesis have been reported and include AKT/GSK3β phosphorylation [129] or triggering proliferation of neural stem cells (NSC) via a mitogen-activated protein kinase (MAPK) dependent signaling cascade [130].
Moreover, preclinical studies have provided further evidence of sildenafil’s neuroprotective potential observed against Aβ amyloid-induced mitochondrial toxicity [131]. Additionally, 3-nitropropionic acid-induced behavioral and biochemical toxicities in a Huntington’s disease rat model [132].
Interestingly, a clinical study showed that single-dose sildenafil could improve regional cerebrovascular reactivity deficits in chronic traumatic brain injury patients as well [133].
Sildenafil has also been reported to promote efficient reconstitution of the myelin sheath and govern the inflammatory processes involved in demyelination models of MS [134]. Sildenafil could also normalize experimental autoimmune encephalomyelitis in MS mouse models [135].
Administration of sildenafil or tadalafil could yield significant anxiolytic-like effects in rodent genetic models of depression as well due to chronic activation of the NO/cGMP system [136][137]. Another reported mechanism for the antidepressant-like effect of sildenafil involved the activation of the oxytocin [138].
Jaumann et al. unveiled a potential protective role of activated cGMP/protein kinase cGMP-dependent 1/poly (ADP-ribose) polymerase (cGMP/PRKG1/PARP) signaling in response to traumas in cochlea sensory cells of various animal models. These data suggested PDE5 as a valid target for the improvement of NIHL. In particular, treatment of rodent models with vardenafil before or 6 h after acoustic trauma was shown to diminish auditory-evoked brain stream response thresholds in all frequency ranges tested [139].
Several animal studies have also proposed a beneficial pain-relieving effect of PDE5-Is in models of lesional [140][141] or metabolic neuropathic pain [142]. Sildenafil could ameliorate neuropathic pain symptoms in patients with diabetic peripheral neuropathy [143] and showed an antinociceptive effect in Sprague–Dawley male rats’ neuropathic pain models [144]. Mechanistically, this analgesic effect has been correlated to cGMP-dependent enhanced release of gamma-aminobutyric acid (GABA) [144].
5.2.3. Cardiovascular Diseases
Cardiomyocytes normally express a minimal basal level of PDE5. However, cardiac PDE5 expression was reported to be upregulated in hypertrophic, dilated, and ischemic cardiomyopathy and in congestive heart failure [47][145][146]. The protective effects of PDE5-Is against myocardial infarction (MI), cardiac ischemic and reperfusion (I/R) injury were validated in many in vitro studies with sildenafil [147], tadalafil [148][149], and vardenafil [150]. When given either prior to occlusion or at reperfusion, these PDE5-Is could reduce infarct size, attenuate cardiac hypertrophy, improve left ventricular (LV) function and prevent progression to heart failure.
In a mouse model, sildenafil exhibited a preconditioning effect to protect the heart against necrosis and apoptosis [151]. Another study suggested that the cardioprotective effect of sildenafil in female mice is estrogen-dependent as ovariectomy suppressed its anti-hypertrophic effect [152].
Intramyocardial transplantation of human adipose stem cells (ASCs) is regarded as a potential treatment for post-ischemic heart failure. Hoke et al. showed that preconditioning of ASCs with sildenafil could trigger the release of significantly high levels of pro-angiogenic or pro-survival growth factors, which enhance ASCs survival and therapeutic efficacy in cardiac ischemic microenvironment, allowing successful cardiac regeneration [153].
Tadalafil also showed cardioprotective effects via PKG-dependent generation of hydrogen sulfide [154]. Moreover, tadalafil was suggested to be clinically beneficial in metabolic syndrome (MetS) patients who are at high risk for CVS diseases where it improved insulin sensitivity, lowered circulating lipids, improved LV diastolic dysfunction and protected against I/R injury in MetS mice [155].
PDE5-Is manifested more significant protective effects against advanced heart failure (HF) with reduced ejection fraction than in HF with preserved ejection fraction [156]. Sildenafil could suppress chamber and myocyte hypertrophy and reverse preestablished hypertrophy in mice exposed to chronic pressure overload. This anti-hypertrophic effect was mediated by the deactivation of multiple signaling pathways, including the calcineurin/nuclear factor of activated T-cells (NFAT), PI3K/AKT, and ERK1/2 signaling pathways [157]. Furthermore, several clinical studies have confirmed the potential role of sildenafil in improving cardiac output, endothelial function, muscle perfusion, and exercise ventilatory and aerobic efficiencies in systolic HF patients [158][159][160].
Moreover, prophylactic treatment with either sildenafil or tadalafil improved cardiac contractile function and survival by attenuating doxorubicin-induced apoptosis and cardiac oxidative stress without interfering with the antitumor efficacy of doxorubicin in both in vitro and in vivo tumor models [161][162].
PDE5 inhibition could govern two crucial vascular manifestations of essential hypertension as well via diminishing blood pressure and improving arterial stiffness and endothelial dysfunction [163].
In addition, sildenafil elicited a significant decrease in inducible ventricular tachycardia and ventricular fibrillation in animal models and demonstrated protection against ventricular arrhythmias associated with the early stages of cardiac ischemia or following MI [164][165].
PDE5-Is could also inhibit platelet activation and aggregation [166][167]. Sildenafil, in particular, was demonstrated to (i) improve coronary patency in an animal model [168], (ii) reduce thrombosis, thromboembolic events, and the risk of thrombotic strokes in a clinical study [169], and (iii) potentiate the anti-aggregation effect of NO donors via cGMP-dependent and independent pathways [170].
Owing to their vasoactive effects, both sildenafil and tadalafil showed advantages in minimizing skin flap necrosis and in preventing extremity and flap ischemia in patients with Raynaud’s phenomenon and with scleroderma [171][172].
Kloner et al. thoroughly investigated the cardiovascular safety profile of PDE5-Is published in the last two decades and confirmed their safety [173].
Cardio protection achieved by PDE5-Is is mainly attributed to restoring high cGMP levels in cardiomyocytes that govern diverse cardioprotective mechanisms as follows (Figure 5): (i) vascular tone regulation and release of endogenous cardioprotective molecules, such as adenosine, bradykinin and phenylephrine from endothelial cells [174], (ii) PKG-dependent opening of mitochondrial and sarcolemmal ATP-sensitive potassium channels modulating calcium homeostasis and survival of cardiomyocytes, preventing post-infarct LV remodeling and reducing infarct size [175][176], (iii) PKG-dependent suppression of adrenergic drive which reduces nerve growth factor leading to anti-arrhythmic effects [164], (iv) ischemic post-conditioning protection against MI via PKG-dependent enhancement of Na+/K+-ATPase activity [177] and inhibition of Na+/H+-exchanger, delaying normalization of pH during reperfusion [178], (v) suppression of protein kinase C (PKC) and calcineurin culminating in improved contractility and protection against HF [179], (vi) improving mitochondrial ultrastructure and function via increased sirtuin-3 (Sirt3) protein expression and decreased peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α) acetylation protecting against post-infarction HF [180], and (vii) inhibition of RhoA/Rho-kinase pathway [181].
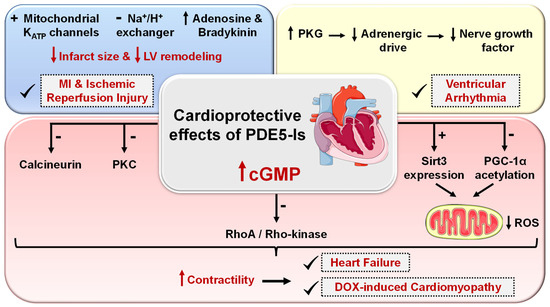
Figure 5. Cardioprotective effects of PDE5 inhibitors. PDE5-Is restore high cGMP levels in cardiomyocytes that govern diverse downstream cardioprotective mechanisms: (i) PKG-dependent opening of mitochondrial and sarcolemmal ATP-sensitive potassium channels, inhibition of Na+/H+-exchanger and release of endogenous cardioprotective molecules, such as adenosine, bradykinin from endothelial cells; resulting in reduced infarct size and hampered post-infarct left ventricular (LV) remodeling. All are beneficial for ischemic post-conditioning protection against myocardial infarction (MI) and ischemic reperfusion (I/R) injury, (ii) PKG-dependent suppression of adrenergic drive which reduces nerve growth factor leading to anti-arrhythmic effects, (iii) suppression of protein kinase C (PKC), calcineurin and RhoA/Rho-kinase pathways and (vi) suppression of oxidative stress and improving mitochondrial ultrastructure and function via increased sirtuin-3 (Sirt3) protein expression and decreased peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α) acetylation, all culminating in improved cardiac contractility and protection against heart failure (HF) and doxorubicin(dox)-induced cardiomyopathy.
5.2.4. Kidney Diseases
Coskuner and coauthor [17] and Afsar et al. [182] thoroughly investigated the renoprotective benefits of PDE5-Is in kidney-related clinical conditions, such as diabetic or nephrotoxic nephropathy, renal ischemia/reperfusion injury, renovascular hypertension and chronic kidney disease. Most reported preclinical studies highlighted a promising potential of PDE5-Is to improve renal function and histopathological changes via collaborative mechanisms, including antioxidative, anti-inflammatory, anti-apoptotic, antifibrotic pathways along with suppression of DNA damage and improving renal blood flow, NOS levels, endothelial function and mitochondrial biogenesis. Most recently, tadalafil was also reported to avert the onset of ureter inflammation and urothelial degeneration in a unilateral ureteral obstruction animal model via modulation of various histopathologic and biochemical changes [183].
5.2.5. Cystic Fibrosis
Cystic fibrosis (CF) is a disease that is caused by a mutation in the CF transmembrane conductance regulator (CFTR) gene “F508del allele” that encodes the main chloride channel expressed in epithelia, which leads to a reduced transepithelial chloride transport in multiple organs, such as pancreas, intestine, kidney, liver and most significantly lungs. This results in abnormal mucociliary clearance and endosomal hyper-acidification along with obstruction, infection and excessive proinflammatory responses that progressively damage the respective organ function and structure [184].
Several preclinical and clinical studies highlighted that PDE5-Is can correct the majority of the known pathological defects in CF, where tadalafil showed the highest efficacy, while vardenafil granted prolonged effects after a single therapeutic dose [185][186]. The efficacy of PDE5-Is in CF could be correlated to one or more of the following mechanisms: (i) correction of the mislocalization of the mutant CFTR protein, restoring normal transepithelial chloride transport [187][188][189], (ii) normalizing the excessive proinflammatory responses via downregulation of M1 markers, tumor necrosis factor (TNF)-α and inducible NOS-2 [190][191], (iii) reversing endosomal hyper-acidification via elevating cGMP levels [192], (iv) improving endothelial function via promoting NOS-3 phosphorylation in endothelial cells [193], and (v) reducing adhesion of bacterial pathogens to respiratory epithelial cells [190].
5.2.6. Diabetes
Das et al. have summarized the potential protective roles of PDE5-Is against several diabetes-related pathologies including (i) prevention of diabetic neuropathy and vasculopathy via improving endothelial function, (ii) protection against I/R injury in diabetic heart via an AMP-activated protein kinase/Sirt1/PGC-1α (AMPK/Sirt1/PGC-1α) cytoprotective signaling cascade, along with (iii) antioxidant and anti-inflammatory effects in diabetic hearts [86].
A meta-analysis of randomized controlled trials has also validated PDE5-Is as effective and safe medications for the treatment of sexual dysfunction in patients with diabetes mellitus suffering from ED [194].
Most recently, a combination of tadalafil and hydrochloroquine successfully improved several Type 2 diabetes-related clinical parameters, including a drop in fasting blood glucose and lipid levels, a rise in plasma insulin and insulin-like growth factor-1 levels and improved insulin sensitivity. Interestingly, pretreatment with the same combination showed a potential to diminish the rate and severity of COVID-19 infection in vulnerable diabetic patients [195].
5.2.7. Miscellaneous Indications
Several studies have demonstrated the efficacy of the combined administration of sildenafil with selective serotonin reuptake inhibitors (SSRIs), such as paroxetine and sertraline, for the treatment of premature ejaculation [196]. Moreover, PDE5-Is prompted penile rigidity and recovery of erections in the post-ejaculatory period [197]. Details of related preclinical and clinical trials were further elaborated by the reviews [23][198].
Long-term chronic administration of PDE5-Is could also avert the progression of fibrotic plaques and halt corporal fibrosis in animal models of Peyronie’s disease [199][200].
In addition, prolonged administration of low-dose PDE5-Is exhibited a promising beneficial effect in the treatment of male infertility. Sildenafil and vardenafil, in particular, could enhance Leydig cells’ secretory and steroidogenic functions, augmenting sperm concentration and the percentages of motile and morphologically normal sperm [201][202][203]. An increase in serum testosterone levels by both inhibitors has been reported as well [204].
Interestingly, tadalafil was proven safe to improve selective fetal growth restriction, a condition of twin pregnancy in which the development of one fetus is restricted, without severe side effects in the mothers or neonates [205]. Most recently, Isidori et al. collaborated evidence possibly linking the NO/cGMP/PDE5 axis to the pathophysiology of coronavirus disease (COVID-19) and suggested the repurposing of PDE5-Is as a treatment strategy to halt the progression of COVID-19 via diverse immunomodulatory mechanisms [206]. All reported FDA-approved and emerging uses of PDE5-Is are summarized in Figure 6.
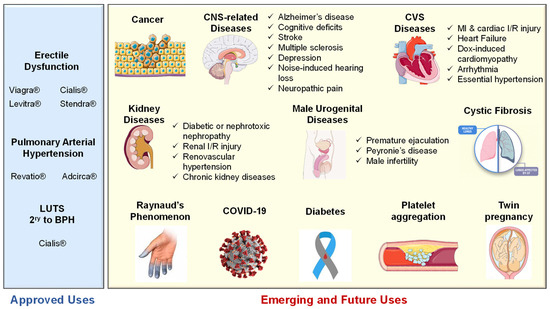
Figure 6. Summary of approved and emerging/future uses of PDE5 inhibitors.
5.3. Side Effects and Contraindications of PDE5 Inhibitors
The use of PDE5-Is is usually associated with some common side effects, which include headache, flushing, dyspepsia, visual disturbances, back pain, myalgia, tachycardia, and nasal congestion [207]. Most of these side effects are due to the inhibition of PDEs other than PDE5, visual disturbances are associated with PDE6 inhibition and back pain and myalgia are attributed to the inhibition of PDE11. Nevertheless, these side effects rarely led to discontinuation of the treatment.
Other less known, seldom encountered serious side effects have been reported concomitant to the use of PDE5-Is are highlighted in the following lines.
- (i)
-
Although PDE5 is reported as a promising target for anti-cancer therapy, as explained earlier, the prolonged use of PDE5-Is has been linked to an increased risk of melanoma. Lie and co-workers reported an association between sildenafil use and an increased risk of melanoma in a prospective cohort study conducted on 25,848 men [208]. Several other cohorts and case-control studies have also reported a correlation between the use of sildenafil and tadalafil and the increased risk of melanoma [209][210]. However, this association between the prolonged use of PDE5-Is and the development of cancer was only reported for melanoma; even the risk of other types of skin cancer, such as squamous cell carcinoma and basal cell carcinoma, was not correlated to the use of PDE5-Is [211].
- (ii)
-
Visual disturbances have been usually reported with the use of PDE5-Is because of PDE6 inhibition. However, several studies have reported more serious ophthalmologic side effects associated with the use of PDE5-Is, which include non-arteritic anterior ischemic optic neuropathy (NAION), which may eventually lead to vision loss [212]. Two case-crossover studies have shown a two-fold increase in the risk of NAION in men using PDE5-Is, and currently, all PDE5Is (Viagra®, Cialis®, Levitra® and Spedra®) mention NAION as a caution in their summary of product characteristics [213][214].
- (iii)
-
Moreover, sensorineural hearing loss (SSHL) has been associated with the prolonged use of PDE5-Is. Two in vivo studies have shown that the prolonged use of sildenafil could lead to hearing loss in mice and rats [215][216]; in addition, published trials and pharmacovigilance agencies reported 47 cases of SSHL as a result of prolonged administration of sildenafil [217], and more specifically, Maddox et al. reported two cases of SSHL due to daily use of tadalafil 10 mg and sildenafil 50 mg + tadalafil 10 mg use where both patients did not recover after a follow-up [218]. Both NAION and SSHL are of unknown pathophysiology.
- (iv)
-
Priapism (prolonged erection of the penis) is another less common side effect reported with the prolonged use of PDE5-Is, as only a few cases have been reported for priapism associated with the use of PDE5-Is [219]. The risk of priapism increases in the case of concomitant use of other ED medications along with the PDE5-Is.
Not only can these side effects potentially restrict the utilization of PDE5-Is, but PDE5-Is are also contradicted in the presence of various cardiovascular disorders. Given that approximately one out of every thirteen individuals is estimated to have a cardiovascular disorder, and considering that there are around 620 million people globally living with cardiovascular conditions, it becomes evident that this is a significant concern. Clinical guidelines dictate that the use of PDE5-Is is not recommended in cases of advanced congestive heart failure, unstable or treatment-resistant angina pectoris, recent myocardial infarction, high-risk arrhythmias, obstructive hypertrophic cardiomyopathy, and severe valve diseases, particularly aortic stenosis [220].
This entry is adapted from the peer-reviewed paper 10.3390/ph16091266
References
- Francis, S.H.; Blount, M.A.; Corbin, J.D. Mammalian cyclic nucleotide phosphodiesterases: Molecular mechanisms and physiological functions. Physiol. Rev. 2011, 91, 651–690.
- Lin, C.-S.; Lin, G.; Xin, Z.-C.; Lue, T.F. Expression, distribution and regulation of phosphodiesterase 5. Curr. Pharm. Des. 2006, 12, 3439–3457.
- Wood, E.R.; Bledsoe, R.; Chai, J.; Daka, P.; Deng, H.; Ding, Y.; Harris-Gurley, S.; Kryn, L.H.; Nartey, E.; Nichols, J. The role of phosphodiesterase 12 (PDE12) as a negative regulator of the innate immune response and the discovery of antiviral inhibitors. J. Biol. Chem. 2015, 290, 19681–19696.
- Sanchez-Cruz, J.; Cabrera-Leon, A.; Martın-Morales, A.; Fernandez, A.; Burgos, R.; Rejas, J. Male erectile dysfunction and health-related quality of life. Eur. Urol. 2003, 44, 245–253.
- Corinaldesi, C.; Di Luigi, L.; Lenzi, A.; Crescioli, C. Phosphodiesterase type 5 inhibitors: Back and forward from cardiac indications. J. Endocrinol. Investig. 2016, 39, 143–151.
- Loughney, K.; Hill, T.R.; Florio, V.A.; Uher, L.; Rosman, G.J.; Wolda, S.L.; Jones, B.A.; Howard, M.L.; McAllister-Lucas, L.M.; Sonnenburg, W.K. Isolation and characterization of cDNAs encoding PDE5A, a human cGMP-binding, cGMP-specific 3′, 5′-cyclic nucleotide phosphodiesterase. Gene 1998, 216, 139–147.
- Dimitriadis, F.; Tsounapi, P.; Saito, M.; Watanabe, T.; Sylakos, A.; Tsabalas, S.; Miyagawa, I.; Sofikitis, N. Is there a role for PDE5 inhibitors in the management of male infertility due to defects in testicular or epididymal function? Curr. Pharm. Des. 2009, 15, 3506–3520.
- Ückert, S.; Kuczyk, M.A. Cyclic nucleotide metabolism including nitric oxide and phosphodiesterase-related targets in the lower urinary tract. Urin. Tract 2011, 2011, 527–542.
- Bers, D.M. Cardiac excitation–contraction coupling. Nature 2002, 415, 198–205.
- Denninger, J.W.; Marletta, M.A. Guanylate cyclase and the⋅ NO/cGMP signaling pathway. Biochim. Biophys. Acta (BBA)-Bioenerg. 1999, 1411, 334–350.
- Murad, F. Nitric oxide and cyclic GMP in cell signaling and drug development. N. Engl. J. Med. 2006, 355, 2003–2011.
- Schlossmann, J.; Ammendola, A.; Ashman, K.; Zong, X.; Huber, A.; Neubauer, G.; Wang, G.X.; Allescher, H.D.; Korth, M.; Wilm, M.; et al. Regulation of intracellular calcium by a signalling complex of IRAG, IP3 receptor and cGMP kinase Iβ. Nature 2000, 404, 197–201.
- Hofmann, F.; Ammendola, A.; Schlossmann, J. Rising behind NO: cGMP-dependent protein kinases. J. Cell Sci. 2000, 113, 1671–1676.
- Zhu, B.; Strada, S.J. The novel functions of cGMP-specific phosphodiesterase 5 and its inhibitors in carcinoma cells and pulmonary/cardiovascular vessels. Curr. Top. Med. Chem. 2007, 7, 437–454.
- Lincoln, T.M.; Dey, N.; Sellak, H. Invited review: cGMP-dependent protein kinase signaling mechanisms in smooth muscle: From the regulation of tone to gene expression. J. Appl. Physiol. 2001, 91, 1421–1430.
- Rondina, M.T.; Weyrich, A.S. Targeting phosphodiesterases in anti-platelet therapy. Antiplatelet Agents 2012, 210, 225–238.
- Coskuner, E.R.; Ozkan, B. Reno-protective effects of Phosphodiesterase 5 inhibitors. Clin. Exp. Nephrol. 2021, 25, 585–597.
- Blount, M.A.; Beasley, A.; Zoraghi, R.; Sekhar, K.R.; Bessay, E.P.; Francis, S.H.; Corbin, J.D. Binding of tritiated sildenafil, tadalafil, or vardenafil to the phosphodiesterase-5 catalytic site displays potency, specificity, heterogeneity, and cGMP stimulation. Mol. Pharmacol. 2004, 66, 144–152.
- Bruzziches, R.; Francomano, D.; Gareri, P.; Lenzi, A.; Aversa, A. An update on pharmacological treatment of erectile dysfunction with phosphodiesterase type 5 inhibitors. Expert Opin. Pharmacother. 2013, 14, 1333–1344.
- Kedia, G.T.; Ückert, S.; Assadi-Pour, F.; Kuczyk, M.A.; Albrecht, K. Avanafil for the treatment of erectile dysfunction: Initial data and clinical key properties. Ther. Adv. Urol. 2013, 5, 35–41.
- Schellack, N.; Agoro, A. A review of phosphodiesterase type 5 inhibitors. S. Afr. Fam. Pract. 2014, 56, 96–101.
- Gupta, M.; Kovar, A.; Meibohm, B. The clinical pharmacokinetics of phosphodiesterase-5 inhibitors for erectile dysfunction. J. Clin. Pharmacol. 2005, 45, 987–1003.
- Ückert, S.; Kuczyk, M.A.; Oelke, M. Phosphodiesterase inhibitors in clinical urology. Expert Rev. Clin. Pharmacol. 2013, 6, 323–332.
- Francis, S.H.; Corbin, J.D. Phosphodiesterase-5 inhibition: The molecular biology of erectile function and dysfunction. Urol. Clin. 2005, 32, 419–429.
- Corbin, J.D.; Francis, S.H. Molecular Biology and Pharmacology of PDE-5—Inhibitor Therapy for Erectile Dysfunction. J. Androl. 2003, 24, S38–S41.
- Carson, C.C.; Burnett, A.L.; Levine, L.A.; Nehra, A. The efficacy of sildenafil citrate (Viagra®) in clinical populations: An update. Urology 2002, 60, 12–27.
- Hellstrom, W.J.; Gittelman, M.; Karlin, G.; Segerson, T.; Thibonnier, M.; Taylor, T.; Padma-Nathan, H.; Group, V.S. Sustained efficacy and tolerability of vardenafil, a highly potent selective phosphodiesterase type 5 inhibitor, in men with erectile dysfunction: Results of a randomized, double-blind, 26-week placebo-controlled pivotal trial. Urology 2003, 61, 8–14.
- Brock, G.B.; McMahon, C.G.; Chen, K.; Costigan, T.; Shen, W.; Watkins, V.; Anglin, G.; Whitaker, S. Efficacy and safety of tadalafil for the treatment of erectile dysfunction: Results of integrated analyses. J. Urol. 2002, 168, 1332–1336.
- Goldstein, I.; McCullough, A.R.; Jones, L.A.; Hellstrom, W.J.; Bowden, C.H.; DiDonato, K.; Trask, B.; Day, W.W. A randomized, double-blind, placebo-controlled evaluation of the safety and efficacy of avanafil in subjects with erectile dysfunction. J. Sex. Med. 2012, 9, 1122–1133.
- Cui, Y.-S.; Li, N.; Zong, H.-T.; Yan, H.-L.; Zhang, Y. Avanafil for male erectile dysfunction: A systematic review and meta-analysis. Asian J. Androl. 2014, 16, 472.
- Jannini, E.A.; DeRogatis, L.R.; Chung, E.; Brock, G.B. How to evaluate the efficacy of the phosphodiesterase type 5 inhibitors. J. Sex. Med. 2012, 9, 26–33.
- Mirone, V.; Fusco, F.; Rossi, A.; Sicuteri, R.; Montorsi, F. Tadalafil and vardenafil vs sildenafil: A review of patient-preference studies. BJU Int. 2009, 103, 1212–1217.
- Humbert, M.; Morrell, N.W.; Archer, S.L.; Stenmark, K.R.; MacLean, M.R.; Lang, I.M.; Christman, B.W.; Weir, E.K.; Eickelberg, O.; Voelkel, N.F. Cellular and molecular pathobiology of pulmonary arterial hypertension. J. Am. Coll. Cardiol. 2004, 43, S13–S24.
- Corbin, J.D.; Beasley, A.; Blount, M.A.; Francis, S.H. High lung PDE5: A strong basis for treating pulmonary hypertension with PDE5 inhibitors. Biochem. Biophys. Res. Commun. 2005, 334, 930–938.
- Giaid, A.; Saleh, D. Reduced expression of endothelial nitric oxide synthase in the lungs of patients with pulmonary hypertension. N. Engl. J. Med. 1995, 333, 214–221.
- Shenoy, P.; Agarwal, V. Phosphodiesterase inhibitors in the management of autoimmune disease. Autoimmun. Rev. 2010, 9, 511–515.
- Nguyen, H.; Amanullah, A.M. Therapeutic potentials of phosphodiesterase-5 inhibitors in cardiovascular disease. Rev. Cardiovasc. Med. 2014, 15, 158–167.
- Yamamura, A.; Fujitomi, E.; Ohara, N.; Tsukamoto, K.; Sato, M.; Yamamura, H. Tadalafil induces antiproliferation, apoptosis, and phosphodiesterase type 5 downregulation in idiopathic pulmonary arterial hypertension in vitro. Eur. J. Pharmacol. 2017, 810, 44–50.
- Foresta, C.; De Toni, L.; Di Mambro, A.; Garolla, A.; Ferlin, A.; Zuccarello, D. BASIC SCIENCE: The PDE5 Inhibitor Sildenafil Increases Circulating Endothelial Progenitor Cells and CXCR4 Expression. J. Sex. Med. 2009, 6, 369–372.
- Rubin, L.J.; Badesch, D.B.; Barst, R.J.; Galiè, N.; Black, C.M.; Keogh, A.; Pulido, T.; Frost, A.; Roux, S.; Leconte, I. Bosentan therapy for pulmonary arterial hypertension. N. Engl. J. Med. 2002, 346, 896–903.
- Pepke-Zaba, J.; Gilbert, C.; Collings, L.; Brown, M.C. Sildenafil improves health-related quality of life in patients with pulmonary arterial hypertension. Chest 2008, 133, 183–189.
- Oudiz, R.J.; Brundage, B.H.; Galiè, N.; Ghofrani, H.A.; Simonneau, G.; Botros, F.T.; Chan, M.; Beardsworth, A.; Barst, R.J.; PHIRST Study Group. Tadalafil for the treatment of pulmonary arterial hypertension: A double-blind 52-week uncontrolled extension study. J. Am. Coll. Cardiol. 2012, 60, 768–774.
- Pepke-Zaba, J.; Beardsworth, A.; Chan, M.; Angalakuditi, M. Tadalafil therapy and health-related quality of life in pulmonary arterial hypertension. Curr. Med. Res. Opin. 2009, 25, 2479–2485.
- Kukreja, R.C.; Salloum, F.N.; Das, A.; Koka, S.; Ockaili, R.A.; Xi, L. Emerging new uses of phosphodiesterase-5 inhibitors in cardiovascular diseases. Exp. Clin. Cardiol. 2011, 16, e30.
- Silvera, F.; Blasina, M.; Vaamonde, L.; Tellechea, S.; Godoy, C.; Zabala, S.; Mañana, G.; Martell, M.; Olivera, W. Sildenafil prevents the increase of extravascular lung water and pulmonary hypertension after meconium aspiration in newborn piglets. Braz. J. Med. Biol. Res. 2011, 44, 778–785.
- Ghofrani, H.A.; Voswinckel, R.; Reichenberger, F.; Olschewski, H.; Haredza, P.; Karadaş, B.; Schermuly, R.T.; Weissmann, N.; Seeger, W.; Grimminger, F. Differences in hemodynamic and oxygenation responses to three different phosphodiesterase-5 inhibitors in patients with pulmonary arterial hypertension: A randomized prospective study. J. Am. Coll. Cardiol. 2004, 44, 1488–1496.
- Schwartz, B.G.; Levine, L.A.; Comstock, G.; Stecher, V.J.; Kloner, R.A. Cardiac uses of phosphodiesterase-5 inhibitors. J. Am. Coll. Cardiol. 2012, 59, 9–15.
- Simonneau, G.; Rubin, L.J.; Galie, N.; Barst, R.J.; Fleming, T.R.; Frost, A.E.; Engel, P.J.; Kramer, M.R.; Burgess, G.; Collings, L. Addition of sildenafil to long-term intravenous epoprostenol therapy in patients with pulmonary arterial hypertension: A randomized trial. Ann. Intern. Med. 2008, 149, 521–530.
- Stehlik, J.; Movsesian, M.A. Combined use of PDE5 inhibitors and nitrates in the treatment of pulmonary arterial hypertension in patients with heart failure. J. Card. Fail. 2009, 15, 31–34.
- Bowles, E.A.; Moody, G.N.; Yeragunta, Y.; Stephenson, A.H.; Ellsworth, M.L.; Sprague, R.S. Phosphodiesterase 5 inhibitors augment UT-15C-stimulated ATP release from erythrocytes of humans with pulmonary arterial hypertension. Exp. Biol. Med. 2015, 240, 121–127.
- Zhao, L.; Mason, N.A.; Morrell, N.W.; Kojonazarov, B.; Sadykov, A.; Maripov, A.; Mirrakhimov, M.M.; Aldashev, A.; Wilkins, M.R. Sildenafil inhibits hypoxia-induced pulmonary hypertension. Circulation 2001, 104, 424–428.
- Richalet, J.-P.; Gratadour, P.; Robach, P.; Pham, I.; Déchaux, M.; Joncquiert-Latarjet, A.; Mollard, P.; Brugniaux, J.; Cornolo, J. Sildenafil inhibits altitude-induced hypoxemia and pulmonary hypertension. Am. J. Respir. Crit. Care Med. 2005, 171, 275–281.
- McVary, K.T.; McKenna, K.E. The relationship between erectile dysfunction and lower urinary tract symptoms: Epidemiological, clinical, and basic science evidence. Curr. Prostate Rep. 2004, 2, 71–77.
- Kerschan-Schindl, K.; Uher, E.; Wiesinger, G.; Kaider, A.; Ebenbichler, G.; Nicolakis, P.; Kollmitzer, J.; Preisinger, E.; Fialka-Moser, V. Reliability of pelvic floor muscle strength measurement in elderly incontinent women. Neurourol. Urodyn. Off. J. Int. Cont. Soc. 2002, 21, 42–47.
- Wong, P.; Lawrentschuk, N.; Bolton, D.M. Phosphodiesterase 5 inhibitors in the management of benign prostatic hyperplasia and erectile dysfunction: The best of both worlds. Curr. Opin. Urol. 2009, 19, 7–12.
- Fusco, F.; di Villa Bianca, R.D.E.; Mitidieri, E.; Cirino, G.; Sorrentino, R.; Mirone, V. Sildenafil effect on the human bladder involves the L-cysteine/hydrogen sulfide pathway: A novel mechanism of action of phosphodiesterase type 5 inhibitors. Eur. Urol. 2012, 62, 1174–1180.
- Peixoto, C.A.; dos Gomes, F.O.S. The role of phosphodiesterase-5 inhibitors in prostatic inflammation: A review. J. Inflamm. 2015, 12, 54.
- Mónica, F.Z.; De Nucci, G. Tadalafil for the treatment of benign prostatic hyperplasia. Expert Opin. Pharmacother. 2019, 20, 929–937.
- Nomiya, M.; Burmeister, D.M.; Sawada, N.; Campeau, L.; Zarifpour, M.; Keys, T.; Peyton, C.; Yamaguchi, O.; Andersson, K.-E. Prophylactic effect of tadalafil on bladder function in a rat model of chronic bladder ischemia. J. Urol. 2013, 189, 754–761.
- Sairam, K.; Kulinskaya, E.; McNicholas, T.; Boustead, G.; Hanbury, D. Sildenafil influences lower urinary tract symptoms. BJU Int. 2002, 90, 836–839.
- McVary, K.T.; Roehrborn, C.G.; Kaminetsky, J.C.; Auerbach, S.M.; Wachs, B.; Young, J.M.; Esler, A.; Sides, G.D.; Denes, B.S. Tadalafil relieves lower urinary tract symptoms secondary to benign prostatic hyperplasia. J. Urol. 2007, 177, 1401–1407.
- Roehrborn, C.G.; Kaminetsky, J.C.; Auerbach, S.M.; Montelongo, R.M.; Elion-Mboussa, A.; Viktrup, L. Changes in peak urinary flow and voiding efficiency in men with signs and symptoms of benign prostatic hyperplasia during once daily tadalafil treatment. BJU Int. 2010, 105, 502–507.
- Dmochowski, R.; Roehrborn, C.; Klise, S.; Xu, L.; Kaminetsky, J.; Kraus, S. Urodynamic effects of once daily tadalafil in men with lower urinary tract symptoms secondary to clinical benign prostatic hyperplasia: A randomized, placebo controlled 12-week clinical trial. J. Urol. 2010, 183, 1092–1097.
- Stief, C.G.; Porst, H.; Neuser, D.; Beneke, M.; Ulbrich, E. A randomised, placebo-controlled study to assess the efficacy of twice-daily vardenafil in the treatment of lower urinary tract symptoms secondary to benign prostatic hyperplasia. Eur. Urol. 2008, 53, 1236–1244.
- Gacci, M.; Andersson, K.-E.; Chapple, C.; Maggi, M.; Mirone, V.; Oelke, M.; Porst, H.; Roehrborn, C.; Stief, C.; Giuliano, F. Latest evidence on the use of phosphodiesterase type 5 inhibitors for the treatment of lower urinary tract symptoms secondary to benign prostatic hyperplasia. Eur. Urol. 2016, 70, 124–133.
- Liguori, G.; Trombetta, C.; De Giorgi, G.; Pomara, G.; Maio, G.; Vecchio, D.; Ocello, G.; Ollandini, G.; Bucci, S.; Belgrano, E. Efficacy and Safety of Combined Oral Therapy with Tadalafil andAlfuzosin: An Integrated Approach to the Management of Patientswith Lower Urinary Tract Symptoms and Erectile Dysfunction. Preliminary Report. J. Sex. Med. 2009, 6, 544–552.
- Kaplan, S.A.; Gonzalez, R.R.; Te, A.E. Combination of alfuzosin and sildenafil is superior to monotherapy in treating lower urinary tract symptoms and erectile dysfunction. Eur. Urol. 2007, 51, 1717–1723.
- Gacci, M.; Corona, G.; Salvi, M.; Vignozzi, L.; McVary, K.T.; Kaplan, S.A.; Roehrborn, C.G.; Serni, S.; Mirone, V.; Carini, M.; et al. A systematic review and meta-analysis on the use of phosphodiesterase 5 inhibitors alone or in combination with alpha-blockers for lower urinary tract symptoms due to benign prostatic hyperplasia. Eur. Urol. 2012, 61, 994–1003.
- Tuncel, A.; Nalcacioglu, V.; Ener, K.; Aslan, Y.; Aydin, O.; Atan, A. Sildenafil citrate and tamsulosin combination is not superior to monotherapy in treating lower urinary tract symptoms and erectile dysfunction. World J. Urol. 2010, 28, 17–22.
- Bechara, A.; Romano, S.; Casabé, A.; Haime, S.; Dedola, P.; Hernández, C.; Rey, H. Comparative efficacy assessment of tamsulosin vs. tamsulosin plus tadalafil in the treatment of LUTS/BPH. Pilot study. J. Sex. Med. 2008, 5, 2170–2178.
- Guo, B.; Chen, X.; Wang, M.; Hou, H.; Zhang, Z.; Liu, M. Comparative Effectiveness of Tadalafil versus Tamsulosin in Treating Lower Urinary Tract Symptoms Suggestive of Benign Prostate Hyperplasia: A Meta-Analysis of Randomized Controlled Trials. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2020, 26, e923179-1–e923179-8.
- Barone, I.; Giordano, C.; Bonofiglio, D.; Andò, S.; Catalano, S. Phosphodiesterase type 5 and cancers: Progress and challenges. Oncotarget 2017, 8, 99179.
- Catalano, S.; Campana, A.; Giordano, C.; Győrffy, B.; Tarallo, R.; Rinaldi, A.; Bruno, G.; Ferraro, A.; Romeo, F.; Lanzino, M. Expression and Function of Phosphodiesterase Type 5 in Human Breast Cancer Cell Lines and Tissues: Implications for Targeted TherapyPDE5 Enhances Breast Cancer Cell Invasive Potential. Clin. Cancer Res. 2016, 22, 2271–2282.
- Sponziello, M.; Verrienti, A.; Rosignolo, F.; De Rose, R.F.; Pecce, V.; Maggisano, V.; Durante, C.; Bulotta, S.; Damante, G.; Giacomelli, L. PDE5 expression in human thyroid tumors and effects of PDE5 inhibitors on growth and migration of cancer cells. Endocrine 2015, 50, 434–441.
- Peak, T.C.; Richman, A.; Gur, S.; Yafi, F.A.; Hellstrom, W.J. The role of PDE5 inhibitors and the NO/cGMP pathway in cancer. Sex. Med. Rev. 2016, 4, 74–84.
- Pantziarka, P.; Sukhatme, V.; Crispino, S.; Bouche, G.; Meheus, L.; Sukhatme, V.P. Repurposing drugs in oncology (ReDO)—Selective PDE5 inhibitors as anti-cancer agents. Ecancermedicalscience 2018, 12, 824.
- Sarfati, M.; Mateo, V.; Baudet, S.; Rubio, M.; Fernandez, C.; Davi, F.; Binet, J.L.; Delic, J.; Merle-Beral, H. Sildenafil and vardenafil, types 5 and 6 phosphodiesterase inhibitors, induce caspase-dependent apoptosis of B-chronic lymphocytic leukemia cells. Blood 2003, 101, 265–269.
- Mei, X.-L.; Yang, Y.; Zhang, Y.-J.; Li, Y.; Zhao, J.-M.; Qiu, J.-G.; Zhang, W.-J.; Jiang, Q.-W.; Xue, Y.-Q.; Zheng, D.-W. Sildenafil inhibits the growth of human colorectal cancer in vitro and in vivo. Am. J. Cancer Res. 2015, 5, 3311.
- Li, H.; Liu, L.; David, M.L.; Whitehead, C.M.; Chen, M.; Fetter, J.R.; Sperl, G.J.; Pamukcu, R.; Thompson, W.J. Pro-apoptotic actions of exisulind and CP461 in SW480 colon tumor cells involve β-catenin and cyclin D1 down-regulation. Biochem. Pharmacol. 2002, 64, 1325–1336.
- Tinsley, H.N.; Gary, B.D.; Keeton, A.B.; Lu, W.; Li, Y.; Piazza, G.A. Inhibition of PDE5 by sulindac sulfide selectively induces apoptosis and attenuates oncogenic Wnt/β-catenin–mediated transcription in human breast tumor cells. Cancer Prev. Res. 2011, 4, 1275–1284.
- Rice, P.L.; Beard, K.S.; Driggers, L.J.; Ahnen, D.J. Inhibition of extracellular-signal regulated kinases 1/2 is required for apoptosis of human colon cancer cells in vitro by sulindac metabolites. Cancer Res. 2004, 64, 8148–8151.
- Aono, Y.; Horinaka, M.; Iizumi, Y.; Watanabe, M.; Taniguchi, T.; Yasuda, S.; Sakai, T. Sulindac sulfone inhibits the mTORC1 pathway in colon cancer cells by directly targeting voltage-dependent anion channel 1 and 2. Biochem. Biophys. Res. Commun. 2018, 505, 1203–1210.
- Thompson, W.J.; Piazza, G.A.; Li, H.; Liu, L.; Fetter, J.; Zhu, B.; Sperl, G.; Ahnen, D.; Pamukcu, R. Exisulind induction of apoptosis involves guanosine 3′, 5′-cyclic monophosphate phosphodiesterase inhibition, protein kinase G activation, and attenuated β-catenin. Cancer Res. 2000, 60, 3338–3342.
- Soh, J.-W.; Mao, Y.; Kim, M.-G.; Pamukcu, R.; Li, H.; Piazza, G.A.; Thompson, W.J.; Weinstein, I.B. Cyclic GMP mediates apoptosis induced by sulindac derivatives via activation of c-Jun NH2-terminal kinase. Clin. Cancer Res. 2000, 6, 4136–4141.
- Li, Q.; Shu, Y. Pharmacological modulation of cytotoxicity and cellular uptake of anti-cancer drugs by PDE5 inhibitors in lung cancer cells. Pharm. Res. 2014, 31, 86–96.
- Das, A.; Durrant, D.; Salloum, F.N.; Xi, L.; Kukreja, R.C. PDE5 inhibitors as therapeutics for heart disease, diabetes and cancer. Pharmacol. Ther. 2015, 147, 12–21.
- Booth, L.; Roberts, J.; Cruickshanks, N.; Conley, A.; Durrant, D.; Das, A.; Fisher, P.; Kukreja, R.; Grant, S.; Poklepovic, A. PDE5 inhibitors enhance chemotherapy killing in gastrointestinal/genitourinary cancer cells. Mol. Pharmacol. 2014, 85, 408–419.
- Ahn, K.-S.; Sim, W.-S.; Lee, I.-K.; Seu, Y.-B.; Kim, I.-H. Decursinol angelate: A cytotoxic and protein kinase C activating agent from the root of Angelica gigas. Planta Med. 1997, 63, 360–361.
- Ding, P.-R.; Tiwari, A.K.; Ohnuma, S.; Lee, J.W.; An, X.; Dai, C.-L.; Lu, Q.-S.; Singh, S.; Yang, D.-H.; Talele, T.T. The phosphodiesterase-5 inhibitor vardenafil is a potent inhibitor of ABCB1/P-glycoprotein transporter. PLoS ONE 2011, 6, e19329.
- Shi, Z.; Tiwari, A.K.; Shukla, S.; Robey, R.W.; Singh, S.; Kim, I.-W.; Bates, S.E.; Peng, X.; Abraham, I.; Ambudkar, S.V. Sildenafil reverses ABCB1-and ABCG2-mediated chemotherapeutic drug resistance. Cancer Res. 2011, 71, 3029–3041.
- Black, K.L.; Yin, D.; Ong, J.M.; Hu, J.; Konda, B.M.; Wang, X.; Ko, M.K.; Bayan, J.-A.; Sacapano, M.R.; Espinoza, A. PDE5 inhibitors enhance tumor permeability and efficacy of chemotherapy in a rat brain tumor model. Brain Res. 2008, 1230, 290–302.
- Hu, J.; Ljubimova, J.Y.; Inoue, S.; Konda, B.; Patil, R.; Ding, H.; Espinoza, A.; Wawrowsky, K.A.; Patil, C.; Ljubimov, A.V. Phosphodiesterase type 5 inhibitors increase Herceptin transport and treatment efficacy in mouse metastatic brain tumor models. PLoS ONE 2010, 5, e10108.
- Booth, L.; Roberts, J.L.; Cruickshanks, N.; Tavallai, S.; Webb, T.; Samuel, P.; Conley, A.; Binion, B.; Young, H.F.; Poklepovic, A. PDE5 inhibitors enhance celecoxib killing in multiple tumor types. J. Cell. Physiol. 2015, 230, 1115–1127.
- Das, A.; Durrant, D.; Mitchell, C.; Mayton, E.; Hoke, N.N.; Salloum, F.N.; Park, M.A.; Qureshi, I.; Lee, R.; Dent, P. Sildenafil increases chemotherapeutic efficacy of doxorubicin in prostate cancer and ameliorates cardiac dysfunction. Proc. Natl. Acad. Sci. USA 2010, 107, 18202–18207.
- Chang, J.-F.; Hsu, J.-L.; Sheng, Y.-H.; Leu, W.-J.; Yu, C.-C.; Chan, S.-H.; Chan, M.-L.; Hsu, L.-C.; Liu, S.-P.; Guh, J.-H. Phosphodiesterase type 5 (PDE5) inhibitors sensitize topoisomerase II inhibitors in killing prostate cancer through PDE5-independent impairment of HR and NHEJ DNA repair systems. Front. Oncol. 2019, 8, 681.
- Hsu, J.-L.; Leu, W.-J.; Hsu, L.-C.; Ho, C.-H.; Liu, S.-P.; Guh, J.-H. Phosphodiesterase Type 5 Inhibitors Synergize Vincristine in Killing Castration-Resistant Prostate Cancer Through Amplifying Mitotic Arrest Signaling. Front. Oncol. 2020, 10, 1274.
- Tavallai, M.; Hamed, H.A.; Roberts, J.L.; Cruickshanks, N.; Chuckalovcak, J.; Poklepovic, A.; Booth, L.; Dent, P. Nexavar/Stivarga and viagra interact to kill tumor cells. J. Cell. Physiol. 2015, 230, 2281–2298.
- Booth, L.; Roberts, J.L.; Poklepovic, A.; Dent, P. PDE5 inhibitors enhance the lethality of . Oncotarget 2017, 8, 13464.
- Chuckalovcak, J.; Carter, J.; Poklepovic, A.; Dent, P. OSU-03012 and Viagra treatment inhibits the activity of multiple chaperone proteins and disrupts the blood brain barrier: Implications for anti-cancer therapies. J. Cell. Physiol. 2015, 230, 1982–1998.
- Chen, L.; Liu, Y.; Becher, A.; Diepold, K.; Schmid, E.; Fehn, A.; Brunner, C.; Rouhi, A.; Chiosis, G.; Cronauer, M. Sildenafil triggers tumor lethality through altered expression of HSP90 and degradation of PKD2. Carcinogenesis 2020, 41, 1421–1431.
- Serafini, P.; Meckel, K.; Kelso, M.; Noonan, K.; Califano, J.; Koch, W.; Dolcetti, L.; Bronte, V.; Borrello, I. Phosphodiesterase-5 inhibition augments endogenous antitumor immunity by reducing myeloid-derived suppressor cell function. J. Exp. Med. 2006, 203, 2691–2702.
- Califano, J.A.; Khan, Z.; Noonan, K.A.; Rudraraju, L.; Zhang, Z.; Wang, H.; Goodman, S.; Gourin, C.G.; Ha, P.K.; Fakhry, C. Tadalafil augments tumor specific immunity in patients with head and neck squamous cell carcinoma. Clin. Cancer Res. 2015, 21, 30–38.
- Huang, W.; Sundquist, J.; Sundquist, K.; Ji, J. Use of phosphodiesterase 5 inhibitors is associated with lower risk of colorectal cancer in men with benign colorectal neoplasms. Gastroenterology 2019, 157, 672–681.
- Chhonker, S.K.; Rawat, D.; Koiri, R.K. Protective and therapeutic effects of sildenafil and tadalafil on aflatoxin B1-induced hepatocellular carcinoma. Mol. Cell. Biochem. 2021, 476, 1195–1209.
- Chhonker, S.K.; Rawat, D.; Koiri, R.K. Repurposing PDE5 inhibitor tadalafil and sildenafil as anticancer agent against hepatocellular carcinoma via targeting key events of glucose metabolism and multidrug resistance. J. Biochem. Mol. Toxicol. 2022, 36, e23100.
- Liu, N.; Mei, L.; Fan, X.; Tang, C.; Ji, X.; Hu, X.; Shi, W.; Qian, Y.; Hussain, M.; Wu, J. Phosphodiesterase 5/protein kinase G signal governs stemness of prostate cancer stem cells through Hippo pathway. Cancer Lett. 2016, 378, 38–50.
- Puzzo, D.; Sapienza, S.; Arancio, O.; Palmeri, A. Role of phosphodiesterase 5 in synaptic plasticity and memory. Neuropsychiatr. Dis. Treat. 2008, 4, 371–387.
- Lu, Y.-F.; Kandel, E.R.; Hawkins, R.D. Nitric oxide signaling contributes to late-phase LTP and CREB phosphorylation in the hippocampus. J. Neurosci. 1999, 19, 10250–10261.
- Ben Aissa, M.; Lee, S.H.; Bennett, B.M.; Thatcher, G.R.J. Targeting NO/cGMP signaling in the CNS for neurodegeneration and Alzheimer’s disease. Curr. Med. Chem. 2016, 23, 2770–2788.
- Kawasaki, K.; Smith Jr, R.S.; Hsieh, C.-M.; Sun, J.; Chao, J.; Liao, J.K. Activation of the phosphatidylinositol 3-kinase/protein kinase Akt pathway mediates nitric oxide-induced endothelial cell migration and angiogenesis. Mol. Cell. Biol. 2003, 23, 5726–5737.
- García-Osta, A.; Cuadrado-Tejedor, M.; García-Barroso, C.; Oyarzabal, J.; Franco, R. Phosphodiesterases as therapeutic targets for Alzheimer’s disease. ACS Chem. Neurosci. 2012, 3, 832–844.
- Sabayan, B.; Zamiri, N.; Farshchizarabi, S.; Sabayan, B. Phoshphodiesterase-5 inhibitors: Novel weapons against Alzheimer’s disease? Int. J. Neurosci. 2010, 120, 746–751.
- Cuadrado-Tejedor, M.; Hervias, I.; Ricobaraza, A.; Puerta, E.; Pérez-Roldán, J.; García-Barroso, C.; Franco, R.; Aguirre, N.; García-Osta, A. Sildenafil restores cognitive function without affecting β-amyloid burden in a mouse model of Alzheimer’s disease. Br. J. Pharmacol. 2011, 164, 2029–2041.
- Puzzo, D.; Staniszewski, A.; Deng, S.X.; Privitera, L.; Leznik, E.; Liu, S.; Zhang, H.; Feng, Y.; Palmeri, A.; Landry, D.W. Phosphodiesterase 5 inhibition improves synaptic function, memory, and amyloid-β load in an Alzheimer’s disease mouse model. J. Neurosci. 2009, 29, 8075–8086.
- Zhang, J.; Guo, J.; Zhao, X.; Chen, Z.; Wang, G.; Liu, A.; Wang, Q.; Zhou, W.; Xu, Y.; Wang, C. Phosphodiesterase-5 inhibitor sildenafil prevents neuroinflammation, lowers beta-amyloid levels and improves cognitive performance in APP/PS1 transgenic mice. Behav. Brain Res. 2013, 250, 230–237.
- García-Barroso, C.; Ricobaraza, A.; Pascual-Lucas, M.; Unceta, N.; Rico, A.J.; Goicolea, M.A.; Sallés, J.; Lanciego, J.L.; Oyarzabal, J.; Franco, R. Tadalafil crosses the blood–brain barrier and reverses cognitive dysfunction in a mouse model of AD. Neuropharmacology 2013, 64, 114–123.
- Devan, B.D.; Sierra-Mercado, D., Jr.; Jimenez, M.; Bowker, J.L.; Duffy, K.B.; Spangler, E.L.; Ingram, D.K. Phosphodiesterase inhibition by sildenafil citrate attenuates the learning impairment induced by blockade of cholinergic muscarinic receptors in rats. Pharmacol. Biochem. Behav. 2004, 79, 691–699.
- Reneerkens, O.A.; Rutten, K.; Akkerman, S.; Blokland, A.; Shaffer, C.L.; Menniti, F.S.; Steinbusch, H.W.; Prickaerts, J. Phosphodiesterase type 5 (PDE5) inhibition improves object recognition memory: Indications for central and peripheral mechanisms. Neurobiol. Learn. Mem. 2012, 97, 370–379.
- Orejana, L.; Barros-Miñones, L.; Jordán, J.; Puerta, E.; Aguirre, N. Sildenafil ameliorates cognitive deficits and tau pathology in a senescence-accelerated mouse model. Neurobiol. Aging 2012, 33, 625.e611–625.e620.
- Sikandaner, H.E.; Park, S.Y.; Kim, M.J.; Park, S.N.; Yang, D.W. Neuroprotective effects of sildenafil against oxidative stress and memory dysfunction in mice exposed to noise stress. Behav. Brain Res. 2017, 319, 37–47.
- Al-Amin, M.M.; Hasan, S.N.; Alam, T.; Hasan, A.T.; Hossain, I.; Didar, R.R.; Alam, M.A.; Rahman, M.M. Tadalafil enhances working memory, and reduces hippocampal oxidative stress in both young and aged mice. Eur. J. Pharmacol. 2014, 745, 84–90.
- Liu, L.; Xu, H.; Ding, S.; Wang, D.; Song, G.; Huang, X. Phosphodiesterase 5 inhibitors as novel agents for the treatment of Alzheimer’s disease. Brain Res. Bull. 2019, 153, 223–231.
- Zuccarello, E.; Acquarone, E.; Calcagno, E.; Argyrousi, E.K.; Deng, S.-X.; Landry, D.W.; Arancio, O.; Fiorito, J. Development of novel phosphodiesterase 5 inhibitors for the therapy of Alzheimer’s disease. Biochem. Pharmacol. 2020, 176, 113818.
- Zhang, L.; Zhang, Z.; Zhang, R.L.; Cui, Y.; LaPointe, M.C.; Silver, B.; Chopp, M. Tadalafil, a long-acting type 5 phosphodiesterase isoenzyme inhibitor, improves neurological functional recovery in a rat model of embolic stroke. Brain Res. 2006, 1118, 192–198.
- Mendes Soares, L.; Prickaerts, J.; Milani, H.; Del Bel, E.; Wilhelm Maria Steinbusch, H.; Maria Weffort de Oliveira, R. Phosphodiesterase inhibition as a therapeutic target for brain ischemia. CNS Neurol. Disord.-Drug Targets (Former. Curr. Drug Targets-CNS Neurol. Disord.) 2015, 14, 1012–1023.
- Ölmestig, J.N.; Marlet, I.R.; Hainsworth, A.H.; Kruuse, C. Phosphodiesterase 5 inhibition as a therapeutic target for ischemic stroke: A systematic review of preclinical studies. Cell. Signal. 2017, 38, 39–48.
- Silver, B.; McCarthy, S.; Lu, M.; Mitsias, P.; Russman, A.N.; Katramados, A.; Morris, D.C.; Lewandowski, C.A.; Chopp, M. Sildenafil treatment of subacute ischemic stroke: A safety study at 25-mg daily for 2 weeks. J. Stroke Cerebrovasc. Dis. 2009, 18, 381–383.
- Ozdegirmenci, O.; Kucukozkan, T.; Akdag, E.; Topal, T.; Haberal, A.; Kayir, H.; Oter, S.; Akyol, M.; Uzbay, T. Effects of sildenafil and tadalafil on ischemia/reperfusion injury in fetal rat brain. J. Matern.-Fetal Neonatal Med. 2011, 24, 317–323.
- Wang, L.; Zhang, Z.G.; Zhang, R.L.; Chopp, M. Activation of the PI3-K/Akt pathway mediates cGMP enhanced-neurogenesis in the adult progenitor cells derived from the subventricular zone. J. Cereb. Blood Flow Metab. 2005, 25, 1150–1158.
- Santos, A.I.; Carreira, B.P.; Nobre, R.J.; Carvalho, C.M.; Araújo, I.M. Stimulation of neural stem cell proliferation by inhibition of phosphodiesterase 5. Stem Cells Int. 2014, 2014, 878397.
- Son, Y.; Kim, K.; Cho, H.-R. Sildenafil protects neuronal cells from mitochondrial toxicity induced by β-amyloid peptide via ATP-sensitive K+ channels. Biochem. Biophys. Res. Commun. 2018, 500, 504–510.
- Thakur, T.; Sharma, S.; Kumar, K.; Deshmukh, R.; Sharma, P.L. Neuroprotective role of PDE4 and PDE5 inhibitors in 3-nitropropionic acid induced behavioral and biochemical toxicities in rats. Eur. J. Pharmacol. 2013, 714, 515–521.
- Kenney, K.; Amyot, F.; Moore, C.; Haber, M.; Turtzo, L.C.; Shenouda, C.; Silverman, E.; Gong, Y.; Qu, B.X.; Harburg, L. Phosphodiesterase-5 inhibition potentiates cerebrovascular reactivity in chronic traumatic brain injury. Ann. Clin. Transl. Neurol. 2018, 5, 418–428.
- de Santana Nunes, A.K.; Rapôso, C.; de Oliveira, W.H.; Thomé, R.; Verinaud, L.; Tovar-Moll, F.; Peixoto, C.A. Phosphodiesterase-5 inhibition promotes remyelination by MCP-1/CCR-2 and MMP-9 regulation in a cuprizone-induced demyelination model. Exp. Neurol. 2016, 275, 143–153.
- Pifarre, P.; Prado, J.; Baltrons, M.A.; Giralt, M.; Gabarro, P.; Feinstein, D.L.; Hidalgo, J.; Garcia, A. Sildenafil (Viagra) ameliorates clinical symptoms and neuropathology in a mouse model of multiple sclerosis. Acta Neuropathol. 2011, 121, 499–508.
- Liebenberg, N.; Harvey, B.H.; Brand, L.; Wegener, G.; Brink, C.B. Chronic treatment with the phosphodiesterase type 5 inhibitors sildenafil and tadalafil display anxiolytic effects in Flinders Sensitive Line rats. Metab. Brain Dis. 2012, 27, 337–340.
- Otari, K.; Upasani, C. Antidepressant-like effect of tadalafil, a phosphodiesterase type 5 inhibitor, in the forced swim test: Dose and duration of treatment dependence. Neurochem. J. 2015, 9, 306–310.
- Matsushita, H.; Matsuzaki, M.; Han, X.-J.; Nishiki, T.-I.; Ohmori, I.; Michiue, H.; Matsui, H.; Tomizawa, K. Antidepressant-like effect of sildenafil through oxytocin-dependent cyclic AMP response element-binding protein phosphorylation. Neuroscience 2012, 200, 13–18.
- Jaumann, M.; Dettling, J.; Gubelt, M.; Zimmermann, U.; Gerling, A.; Paquet-Durand, F.; Feil, S.; Wolpert, S.; Franz, C.; Varakina, K. cGMP-Prkg1 signaling and Pde5 inhibition shelter cochlear hair cells and hearing function. Nat. Med. 2012, 18, 252–259.
- Huang, L.J.; Yoon, M.H.; Choi, J.I.; Kim, W.M.; Lee, H.G.; Kim, Y.O. Effect of sildenafil on neuropathic pain and hemodynamics in rats. Yonsei Med. J. 2010, 51, 82–87.
- Vieira, M.C.; de Monte, F.B.M.; Eduardo Dematte, B.; Montagnoli, T.L.; Montes, G.C.; da Silva, J.S.; Mendez-Otero, R.; Trachez, M.M.; Sudo, R.T.; Zapata-Sudo, G. Antinociceptive Effect of Lodenafil Carbonate in Rodent Models of Inflammatory Pain and Spinal Nerve Ligation-Induced Neuropathic Pain. J. Pain Res. 2021, 14, 857–866.
- Wang, L.; Chopp, M.; Szalad, A.; Liu, Z.; Bolz, M.; Ãlvarez, F.M.; Lu, M.; Zhang, L.; Cui, Y.; Zhang, R.L. Phosphodiesterase-5 is a therapeutic target for peripheral neuropathy in diabetic mice. Neuroscience 2011, 193, 399–410.
- Hackett, G. PDE5 inhibitors in diabetic peripheral neuropathy. Int. J. Clin. Pract. 2006, 60, 1123–1126.
- Finnerup, N.B.; Sindrup, S.H.; Jensen, T.S. Chronic neuropathic pain: Mechanisms, drug targets and measurement. Fundam. Clin. Pharmacol. 2007, 21, 129–136.
- Wallis, R.M.; Corbin, J.D.; Francis, S.H.; Ellis, P. Tissue distribution of phosphodiesterase families and the effects of sildenafil on tissue cyclic nucleotides, platelet function, and the contractile responses of trabeculae carneae and aortic rings in vitro. Am. J. Cardiol. 1999, 83, 3–12.
- Korkmaz-Icöz, S.; Radovits, T.; Szabó, G. Targeting phosphodiesterase 5 as a therapeutic option against myocardial ischaemia/reperfusion injury and for treating heart failure. Br. J. Pharmacol. 2018, 175, 223–231.
- du Toit, E.F.; Rossouw, E.; Salie, R.; Opie, L.H.; Lochner, A. Effect of sildenafil on reperfusion function, infarct size, and cyclic nucleotide levels in the isolated rat heart model. Cardiovasc. Drugs Ther. 2005, 19, 23–31.
- Sesti, C.; Florio, V.; Johnson, E.; Kloner, R. The phosphodiesterase-5 inhibitor tadalafil reduces myocardial infarct size. Int. J. Impot. Res. 2007, 19, 55–61.
- Ahmad, N.; Wang, Y.; Ali, A.K.; Ashraf, M. Long-acting phosphodiesterase-5 inhibitor, tadalafil, induces sustained cardioprotection against lethal ischemic injury. Am. J. Physiol.-Heart Circ. Physiol. 2009, 297, H387–H391.
- Maas, O.; Donat, U.; Frenzel, M.; Rütz, T.; Kroemer, H.; Felix, S.; Krieg, T. Vardenafil protects isolated rat hearts at reperfusion dependent on GC and PKG. Br. J. Pharmacol. 2008, 154, 25–31.
- Das, A.; Xi, L.; Kukreja, R.C. Phosphodiesterase-5 inhibitor sildenafil preconditions adult cardiac myocytes against necrosis and apoptosis: Essential role of nitric oxide signaling. J. Biol. Chem. 2005, 280, 12944–12955.
- Sasaki, H.; Nagayama, T.; Blanton, R.M.; Seo, K.; Zhang, M.; Zhu, G.; Lee, D.I.; Bedja, D.; Hsu, S.; Tsukamoto, O. PDE5 inhibitor efficacy is estrogen dependent in female heart disease. J. Clin. Investig. 2014, 124, 2464–2471.
- Hoke, N.N.; Salloum, F.N.; Kass, D.A.; Das, A.; Kukreja, R.C. Preconditioning by phosphodiesterase-5 inhibition improves therapeutic efficacy of adipose-derived stem cells following myocardial infarction in mice. Stem Cells 2012, 30, 326–335.
- Salloum, F.N.; Chau, V.Q.; Hoke, N.N.; Abbate, A.; Varma, A.; Ockaili, R.A.; Toldo, S.; Kukreja, R.C. Phosphodiesterase-5 inhibitor, tadalafil, protects against myocardial ischemia/reperfusion through protein-kinase G–dependent generation of hydrogen sulfide. Circulation 2009, 120, S31–S36.
- Koka, S.; Xi, L.; Kukreja, R.C. Chronic inhibition of phosphodiesterase 5 with tadalafil affords cardioprotection in a mouse model of metabolic syndrome: Role of nitric oxide. Mol. Cell. Biochem. 2020, 468, 47–58.
- Hutchings, D.C.; Anderson, S.G.; Caldwell, J.L.; Trafford, A.W. Phosphodiesterase-5 inhibitors and the heart: Compound cardioprotection? Heart 2018, 104, 1244–1250.
- Nagendran, J.; Archer, S.L.; Soliman, D.; Gurtu, V.; Moudgil, R.; Haromy, A.; St. Aubin, C.; Webster, L.; Rebeyka, I.M.; Ross, D.B. Phosphodiesterase type 5 is highly expressed in the hypertrophied human right ventricle, and acute inhibition of phosphodiesterase type 5 improves contractility. Circulation 2007, 116, 238–248.
- Lewis, G.D.; Lachmann, J.; Camuso, J.; Lepore, J.J.; Shin, J.; Martinovic, M.E.; Systrom, D.M.; Bloch, K.D.; Semigran, M.J. Sildenafil improves exercise hemodynamics and oxygen uptake in patients with systolic heart failure. Circulation 2007, 115, 59–66.
- Guazzi, M.; Casali, M.; Berti, F.; Rossoni, G.; D’Gennaro Colonna, V.; Guazzi, M. Endothelium-mediated modulation of ergoreflex and improvement in exercise ventilation by acute sildenafil in heart failure patients. Clin. Pharmacol. Ther. 2008, 83, 336–341.
- De Vecchis, R.; Cesaro, A.; Ariano, C.; Giasi, A.; Cioppa, C. Phosphodiesterase-5 inhibitors improve clinical outcomes, exercise capacity and pulmonary hemodynamics in patients with heart failure with reduced left ventricular ejection fraction: A meta-analysis. J. Clin. Med. Res. 2017, 9, 488.
- Fisher, P.W.; Salloum, F.; Das, A.; Hyder, H.; Kukreja, R.C. Phosphodiesterase-5 inhibition with sildenafil attenuates cardiomyocyte apoptosis and left ventricular dysfunction in a chronic model of doxorubicin cardiotoxicity. Circulation 2005, 111, 1601–1610.
- Koka, S.; Das, A.; Zhu, S.-G.; Durrant, D.; Xi, L.; Kukreja, R.C. Long-acting phosphodiesterase-5 inhibitor tadalafil attenuates doxorubicin-induced cardiomyopathy without interfering with chemotherapeutic effect. J. Pharmacol. Exp. Ther. 2010, 334, 1023–1030.
- Yaguas, K.; Bautista, R.; Quiroz, Y.; Ferrebuz, A.; Pons, H.; Franco, M.; Vaziri, N.D.; Rodriguez-Iturbe, B. Chronic sildenafil treatment corrects endothelial dysfunction and improves hypertension. Am. J. Nephrol. 2010, 31, 283–291.
- Lee, T.-M.; Chen, C.-C.; Chung, T.-H.; Chang, N.-C. Effect of sildenafil on ventricular arrhythmias in post-infarcted rat hearts. Eur. J. Pharmacol. 2012, 690, 124–132.
- Nagy, O.; Hajnal, Á.; Parratt, J.R.; Végh, Á. Sildenafil (Viagra) reduces arrhythmia severity during ischaemia 24 h after oral administration in dogs. Br. J. Pharmacol. 2004, 141, 549–551.
- De Bon, E.; Bonanni, G.; Saggiorato, G.; Bassi, P.; Cella, G. Effects of tadalafil on platelets and endothelium in patients with erectile dysfunction and cardiovascular risk factors: A pilot study. Angiology 2010, 61, 602–606.
- Toque, H.; Teixeira, C.; Priviero, F.; Morganti, R.; Antunes, E.; De Nucci, G. Vardenafil, but not sildenafil or tadalafil, has calcium-channel blocking activity in rabbit isolated pulmonary artery and human washed platelets. Br. J. Pharmacol. 2008, 154, 787–796.
- Lewis, G.D.; Witzke, C.; Colon-Hernandez, P.; Guerrero, J.L.; Bloch, K.D.; Semigran, M.J. Sildenafil improves coronary artery patency in a canine model of platelet-mediated cyclic coronary occlusion after thrombolysis. J. Am. Coll. Cardiol. 2006, 47, 1471–1477.
- Saeed, O.; Rangasamy, S.; Selevany, I.; Madan, S.; Fertel, J.; Eisenberg, R.; Aljoudi, M.; Patel, S.R.; Shin, J.; Sims, D.B. Sildenafil is associated with reduced device thrombosis and ischemic stroke despite low-level hemolysis on Heart Mate II support. Circ. Heart Fail. 2017, 10, e004222.
- Gudmundsdóttir, I.J.; McRobbie, S.J.; Robinson, S.D.; Newby, D.E.; Megson, I.L. Sildenafil potentiates nitric oxide mediated inhibition of human platelet aggregation. Biochem. Biophys. Res. Commun. 2005, 337, 382–385.
- Shenoy, P.D.; Kumar, S.; Jha, L.K.; Choudhary, S.K.; Singh, U.; Misra, R.; Agarwal, V. Efficacy of tadalafil in secondary Raynaud’s phenomenon resistant to vasodilator therapy: A double-blind randomized cross-over trial. Rheumatology 2010, 49, 2420–2428.
- Kumar, N.; Griffiths, B.; Allen, J. Thermographic and symptomatic effect of a single dose of sildenafil citrate on Raynaud’s phenomenon in patients with systemic sclerosis: A potential treatment. J. Rheumatol. 2006, 33, 1918–1919.
- Kloner, R.A.; Goldstein, I.; Kirby, M.G.; Parker, J.D.; Sadovsky, R. Cardiovascular safety of phosphodiesterase type 5 inhibitors after nearly 2 decades on the market. Sex. Med. Rev. 2018, 6, 583–594.
- Kukreja, R. Cardiovascular protection with sildenafil following chronic inhibition of nitric oxide synthase. Br. J. Pharmacol. 2007, 150, 538–540.
- Salloum, F.N.; Ockaili, R.A.; Wittkamp, M.; Marwaha, V.R.; Kukreja, R.C. Vardenafil: A novel type 5 phosphodiesterase inhibitor reduces myocardial infarct size following ischemia/reperfusion injury via opening of mitochondrial KATP channels in rabbits. J. Mol. Cell. Cardiol. 2006, 40, 405–411.
- Wang, X.; Fisher, P.W.; Xi, L.; Kukreja, R.C. Essential role of mitochondrial Ca2+-activated and ATP-sensitive K+ channels in sildenafil-induced late cardioprotection. J. Mol. Cell. Cardiol. 2008, 44, 105–113.
- Madhani, M.; Hall, A.R.; Cuello, F.; Charles, R.L.; Burgoyne, J.R.; Fuller, W.; Hobbs, A.J.; Shattock, M.J.; Eaton, P. Phospholemman Ser69 phosphorylation contributes to sildenafil-induced cardioprotection against reperfusion injury. Am. J. Physiol.-Heart Circ. Physiol. 2010, 299, H827–H836.
- Inserte, J.; Barba, I.; Poncelas-Nozal, M.; Hernando, V.; Agulló, L.; Ruiz-Meana, M.; Garcia-Dorado, D. cGMP/PKG pathway mediates myocardial postconditioning protection in rat hearts by delaying normalization of intracellular acidosis during reperfusion. J. Mol. Cell. Cardiol. 2011, 50, 903–909.
- Nagayama, T.; Hsu, S.; Zhang, M.; Koitabashi, N.; Bedja, D.; Gabrielson, K.L.; Takimoto, E.; Kass, D.A. Sildenafil stops progressive chamber, cellular, and molecular remodeling and improves calcium handling and function in hearts with pre-existing advanced hypertrophy caused by pressure overload. J. Am. Coll. Cardiol. 2009, 53, 207–215.
- Li, N.; Yuan, Y.; Li, S.; Zeng, C.; Yu, W.; Shen, M.; Zhang, R.; Li, C.; Zhang, Y.; Wang, H. PDE5 inhibitors protect against post-infarction heart failure. Front. Biosci.-Landmark 2016, 21, 1194–1210.
- Chau, V.Q.; Salloum, F.N.; Hoke, N.N.; Abbate, A.; Kukreja, R.C. Mitigation of the progression of heart failure with sildenafil involves inhibition of RhoA/Rho-kinase pathway. Am. J. Physiol.-Heart Circ. Physiol. 2011, 300, H2272–H2279.
- Afsar, B.; Ortiz, A.; Covic, A.; Gaipov, A.; Esen, T.; Goldsmith, D.; Kanbay, M. Phosphodiesterase type 5 inhibitors and kidney disease. Int. Urol. Nephrol. 2015, 47, 1521–1528.
- Köktürk, S.; Benli, E.; Ayyıldız, A.; Cırrık, S.; Çetinkol, Y.; Ayyıldız, S.N.; Noyan, T. Positive outcomes of phosphodiesterase type 5 inhibitor on histopathologic and biochemical changes induced by ureteral obstruction. Rev. Da Assoc. Médica Bras. 2019, 65, 388–393.
- Noel, S.; Dhooghe, B.; Leal, T. PDE5 inhibitors as potential tools in the treatment of cystic fibrosis. Front. Pharmacol. 2012, 3, 167.
- Lubamba, B.; Lebacq, J.; Reychler, G.; Marbaix, E.; Wallemacq, P.; Lebecque, P.; Leal, T. Inhaled phosphodiesterase type 5 inhibitors restore chloride transport in cystic fibrosis mice. Eur. Respir. J. 2011, 37, 72–78.
- Rodriguez-Miguelez, P.; Ishii, H.; Seigler, N.; Crandall, R.; Thomas, J.; Forseen, C.; McKie, K.T.; Harris, R.A. Sildenafil improves exercise capacity in patients with cystic fibrosis: A proof-of-concept clinical trial. Ther. Adv. Chronic Dis. 2019, 10, 2040622319887879.
- Dormer, R.L.; Harris, C.M.; Clark, Z.; Pereira, M.M.C.; Doull, I.J.M.; Norez, C.; Becq, F.; McPherson, M.A. Sildenafil (Viagra) corrects ΔF508-CFTR location in nasal epithelial cells from patients with cystic fibrosis. Thorax 2005, 60, 55–59.
- Lubamba, B.; Lecourt, H.; Lebacq, J.; Lebecque, P.; De Jonge, H.; Wallemacq, P.; Leal, T. Preclinical evidence that sildenafil and vardenafil activate chloride transport in cystic fibrosis. Am. J. Respir. Crit. Care Med. 2008, 177, 506–515.
- Dhooghe, B.; Noël, S.; Bouzin, C.; Behets-Wydemans, G.; Leal, T. Correction of chloride transport and mislocalization of CFTR protein by vardenafil in the gastrointestinal tract of cystic fibrosis mice. PLoS ONE 2013, 8, e77314.
- Poschet, J.F.; Timmins, G.S.; Taylor-Cousar, J.L.; Ornatowski, W.; Fazio, J.; Perkett, E.; Wilson, K.R.; Yu, H.D.; de Jonge, H.R.; Deretic, V. Pharmacological modulation of cGMP levels by phosphodiesterase 5 inhibitors as a therapeutic strategy for treatment of respiratory pathology in cystic fibrosis. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2007, 293, L712–L719.
- Noel, S.; Panin, N.; Beka, M.; Dhooghe, B.; Huaux, F.; Leal, T. Vardenafil reduces macrophage pro-inflammatory overresponses in cystic fibrosis through PDE5-and CFTR-dependent mechanisms. Clin. Sci. 2017, 131, 1107–1121.
- Poschet, J.F.; Fazio, J.A.; Timmins, G.S.; Ornatowski, W.; Perkett, E.; Delgado, M.; Deretic, V. Endosomal hyperacidification in cystic fibrosis is due to defective nitric oxide–cylic GMP signalling cascade. EMBO Rep. 2006, 7, 553–559.
- Rodriguez-Miguelez, P.; Lee, N.; Tucker, M.A.; Csányi, G.; McKie, K.T.; Forseen, C.; Harris, R.A. Sildenafil improves vascular endothelial function in patients with cystic fibrosis. Am. J. Physiol.-Heart Circ. Physiol. 2018, 315, H1486–H1494.
- Corbin, J. Mechanisms of action of PDE5 inhibition in erectile dysfunction. Int. J. Impot. Res. 2004, 16, S4–S7.
- Kukreja, R.C.; Wang, R.; Koka, S.; Das, A.; Samidurai, A.; Xi, L. Treating diabetes with combination of phosphodiesterase 5 inhibitors and hydroxychloroquine—A possible prevention strategy for COVID-19? Mol. Cell. Biochem. 2023, 479, 679–696.
- Salonia, A.; Maga, T.; Colombo, R.; Scattoni, V.; Briganti, A.; Cestari, A.; Guazzoni, G.; Rigatti, P.; Montorsi, F. A prospective study comparing paroxetine alone versus paroxetine plus sildenafil in patients with premature ejaculation. J. Urol. 2002, 168, 2486–2489.
- Gökçe, A.; Halis, F.; Demirtas, A.; Ekmekcioglu, O. The effects of three phosphodiesterase type 5 inhibitors on ejaculation latency time in lifelong premature ejaculators: A double-blind laboratory setting study. BJU Int. 2011, 107, 1274–1277.
- Mcmahon, C.G.; Mcmahon, C.N.; Leow, L.J.; Winestock, C.G. Efficacy of type-5 phosphodiesterase inhibitors in the drug treatment of premature ejaculation: A systematic review. BJU Int. 2006, 98, 259–272.
- Ferrini, M.G.; Kovanecz, I.; Nolazco, G.; Rajfer, J.; Gonzalez-Cadavid, N.F. Effects of long-term vardenafil treatment on the development of fibrotic plaques in a rat model of Peyronie’s disease. BJU Int. 2006, 97, 625–633.
- Gonzalez-Cadavid, N.F.; Rajfer, J. Treatment of Peyronie’s disease with PDE5 inhibitors: An antifibrotic strategy. Nat. Rev. Urol. 2010, 7, 215–221.
- Dimitriadis, F.; Giannakis, D.; Pardalidis, N.; Zikopoulos, K.; Paraskevaidis, E.; Giotitsas, N.; Kalaboki, V.; Tsounapi, P.; Baltogiannis, D.; Georgiou, I. Effects of phosphodiesterase 5 inhibitors on sperm parameters and fertilizing capacity. Asian J. Androl. 2008, 10, 115–133.
- Jannini, E.A.; Lombardo, F.; Salacone, P.; Gandini, L.; Lenzi, A. Treatment of sexual dysfunctions secondary to male infertility with sildenafil citrate. Fertil. Steril. 2004, 81, 705–707.
- Andric, S.A.; Janjic, M.M.; Stojkov, N.J.; Kostic, T.S. Sildenafil treatment in vivo stimulates Leydig cell steroidogenesis via the cAMP/cGMP signaling pathway. Am. J. Physiol. Endocrinol. Metab. 2010, 299, E544–E550.
- Janjic, M.M.; Stojkov, N.J.; Bjelic, M.M.; Mihajlovic, A.I.; Andric, S.A.; Kostic, T.S. Transient rise of serum testosterone level after single sildenafil treatment of adult male rats. J. Sex. Med. 2012, 9, 2534–2543.
- Magawa, S.; Nii, M.; Tanaka, H.; Furuhashi, F.; Maki, S.; Kubo, M.; Tanaka, K.; Kondo, E.; Ikeda, T. Phase-1 clinical study of tadalafil administered for selective fetal growth restriction in twin pregnancy. J. Matern.-Fetal Neonatal Med. 2021, 34, 1075–1082.
- Isidori, A.M.; Giannetta, E.; Pofi, R.; Venneri, M.A.; Gianfrilli, D.; Campolo, F.; Mastroianni, C.M.; Lenzi, A.; d’Ettorre, G. Targeting the NO-cGMP-PDE5 pathway in COVID-19 infection. The DEDALO project. Andrology 2021, 9, 33–38.
- Zurawin, J.L.; Stewart, C.A.; Anaissie, J.E.; Yafi, F.A.; Hellstrom, W.J. Avanafil for the treatment of erectile dysfunction. Expert Rev. Clin. Pharmacol. 2016, 9, 1163–1170.
- Li, W.-Q.; Qureshi, A.A.; Robinson, K.C.; Han, J. Sildenafil use and increased risk of incident melanoma in US men: A prospective cohort study. JAMA Intern. Med. 2014, 174, 964–970.
- Loeb, S.; Folkvaljon, Y.; Lambe, M.; Robinson, D.; Garmo, H.; Ingvar, C.; Stattin, P. Use of phosphodiesterase type 5 inhibitors for erectile dysfunction and risk of malignant melanoma. JAMA 2015, 313, 2449–2455.
- Pottegård, A.; Schmidt, S.A.J.; Olesen, A.B.; Achacoso, N.; Van Den Eeden, S.K.; Hallas, J.; Sørensen, H.T.; Friis, S.; Habel, L.A. Use of sildenafil or other phosphodiesterase inhibitors and risk of melanoma. Br. J. Cancer 2016, 115, 895–900.
- Gul, M.; Serefoglu, E.C. An update on the drug safety of treating erectile dysfunction. Expert Opin. Drug Saf. 2019, 18, 965–975.
- Kerr, N.M.; Danesh-Meyer, H.V. Phosphodiesterase inhibitors and the eye. Clin. Exp. Ophthalmol. 2009, 37, 514–523.
- Campbell, U.B.; Walker, A.M.; Gaffney, M.; Petronis, K.R.; Creanga, D.; Quinn, S.; Klein, B.E.; Laties, A.M.; Lewis, M.; Sharlip, I.D. Acute nonarteritic anterior ischemic optic neuropathy and exposure to phosphodiesterase type 5 inhibitors. J. Sex. Med. 2015, 12, 139–151.
- Flahavan, E.M.; Li, H.; Gupte-Singh, K.; Rizk, R.T.; Ruff, D.D.; Francis, J.L.; Kinchen, K.S. Prospective case-crossover study investigating the possible association between nonarteritic anterior ischemic optic neuropathy and phosphodiesterase type 5 inhibitor exposure. Urology 2017, 105, 76–84.
- Hong, B.N.; Yi, T.H.; Kim, S.Y.; Kang, T.H. High dosage sildenafil induces hearing impairment in mice. Biol. Pharm. Bull. 2008, 31, 1981–1984.
- Bakir, S.; Firat, U.; Gün, R.; Bozkurt, Y.; Yorgancilar, E.; Kiniş, V.; Penbegül, N.; Gökalp, O.; Topçu, İ. Histopathologic results of long-term sildenafil administration on rat inner ear. Am. J. Otolaryngol. 2012, 33, 667–672.
- Khan, A.S.; Sheikh, Z.; Khan, S.; Dwivedi, R.; Benjamin, E. Viagra deafness—Sensorineural hearing loss and phosphodiesterase-5 inhibitors. Laryngoscope 2011, 121, 1049–1054.
- Maddox, P.T.; Saunders, J.; Chandrasekhar, S.S. Sudden hearing loss from PDE-5 inhibitors: A possible cellular stress etiology. Laryngoscope 2009, 119, 1586–1589.
- Broderick, G.A.; Kadioglu, A.; Bivalacqua, T.J.; Ghanem, H.; Nehra, A.; Shamloul, R. Priapism: Pathogenesis, epidemiology, and management. J. Sex. Med. 2010, 7, 476–500.
- Nehra, A.; Jackson, G.; Miner, M.; Billups, K.L.; Burnett, A.L.; Buvat, J.; Carson, C.C.; Cunningham, G.R.; Ganz, P.; Goldstein, I. The Princeton III Consensus recommendations for the management of erectile dysfunction and cardiovascular disease. Mayo Clin. Proc. 2012, 87, 766–778.
This entry is offline, you can click here to edit this entry!
