Marine tunicates are identified as a potential source of marine natural products (MNPs), demonstrating a wide range of biological properties, like antimicrobial and anticancer activities. The symbiotic relationship between tunicates and specific microbial groups has revealed the acquisition of microbial compounds by tunicates for defensive purpose. For instance, yellow pigmented compounds, “tambjamines”, produced by the tunicate, Sigillina signifera (Sluiter, 1909), primarily originated from their bacterial symbionts, which are involved in their chemical defense function, indicating the ecological role of symbiotic microbial association with tunicates.
- tunicates
- symbiotic microbes
- pigments
- bioactive compounds
- alkaloids & peptides
1. Introduction
2. Ecological Importance of Tunicates
3. Profile of MNPs from Tunicates and Associated Microbes
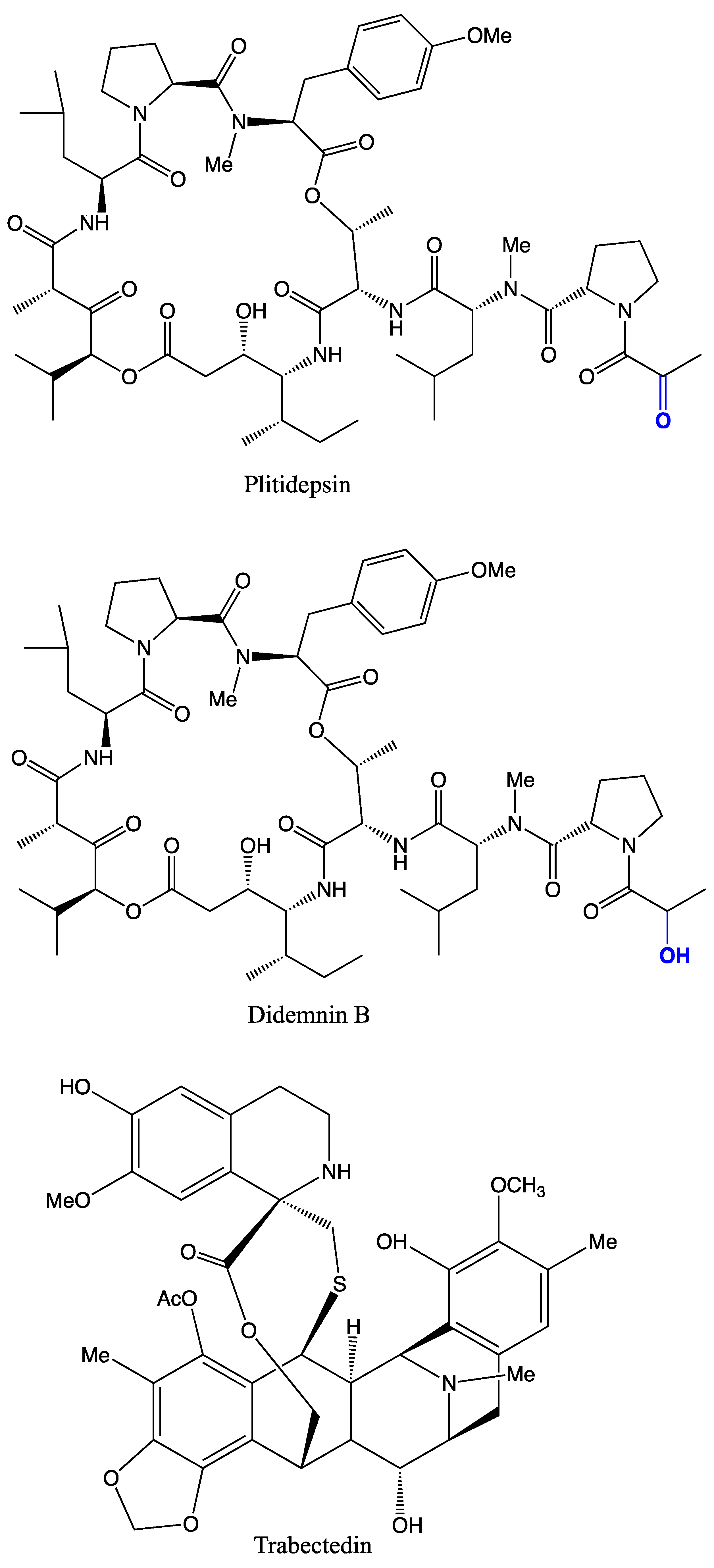
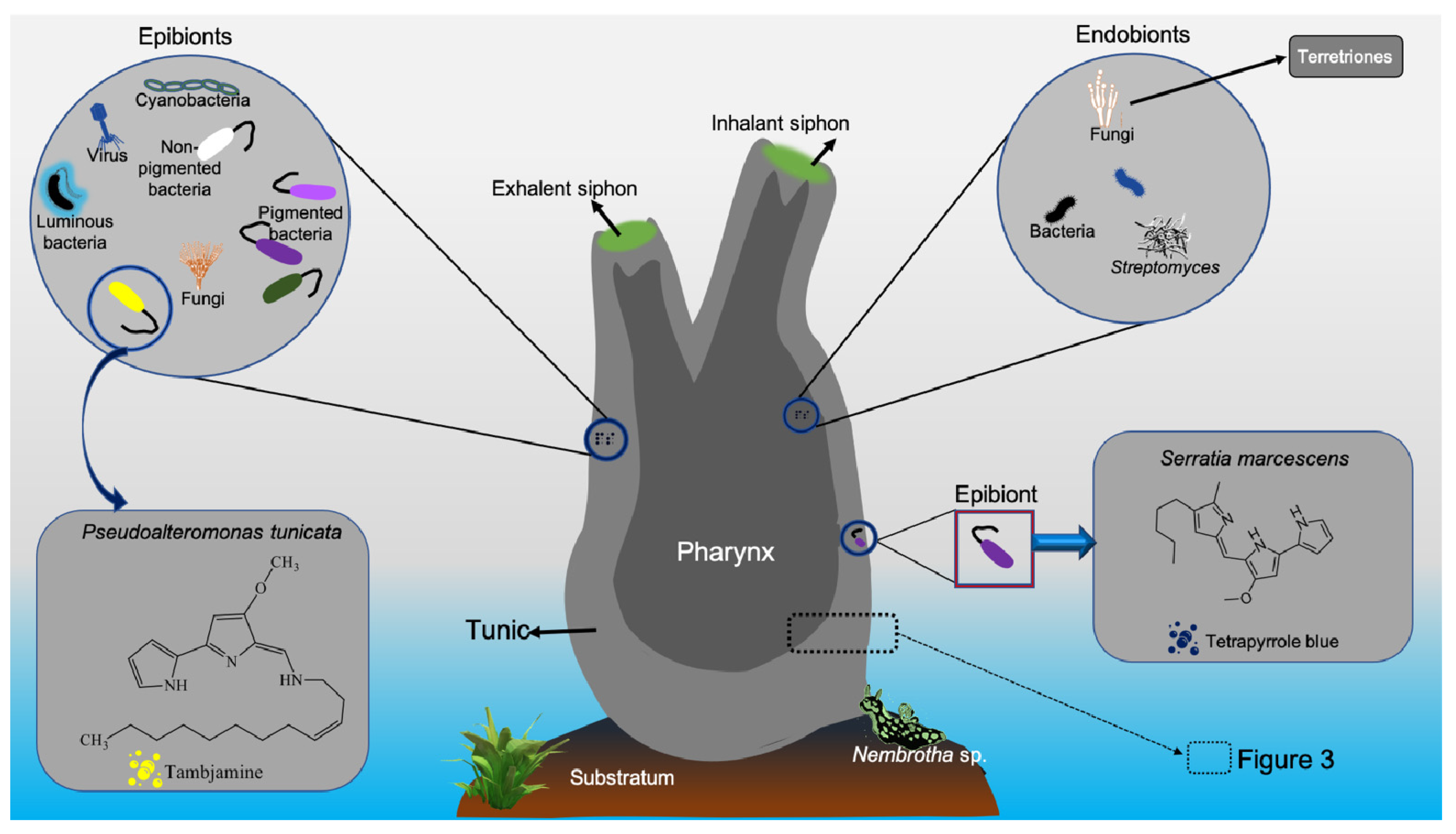
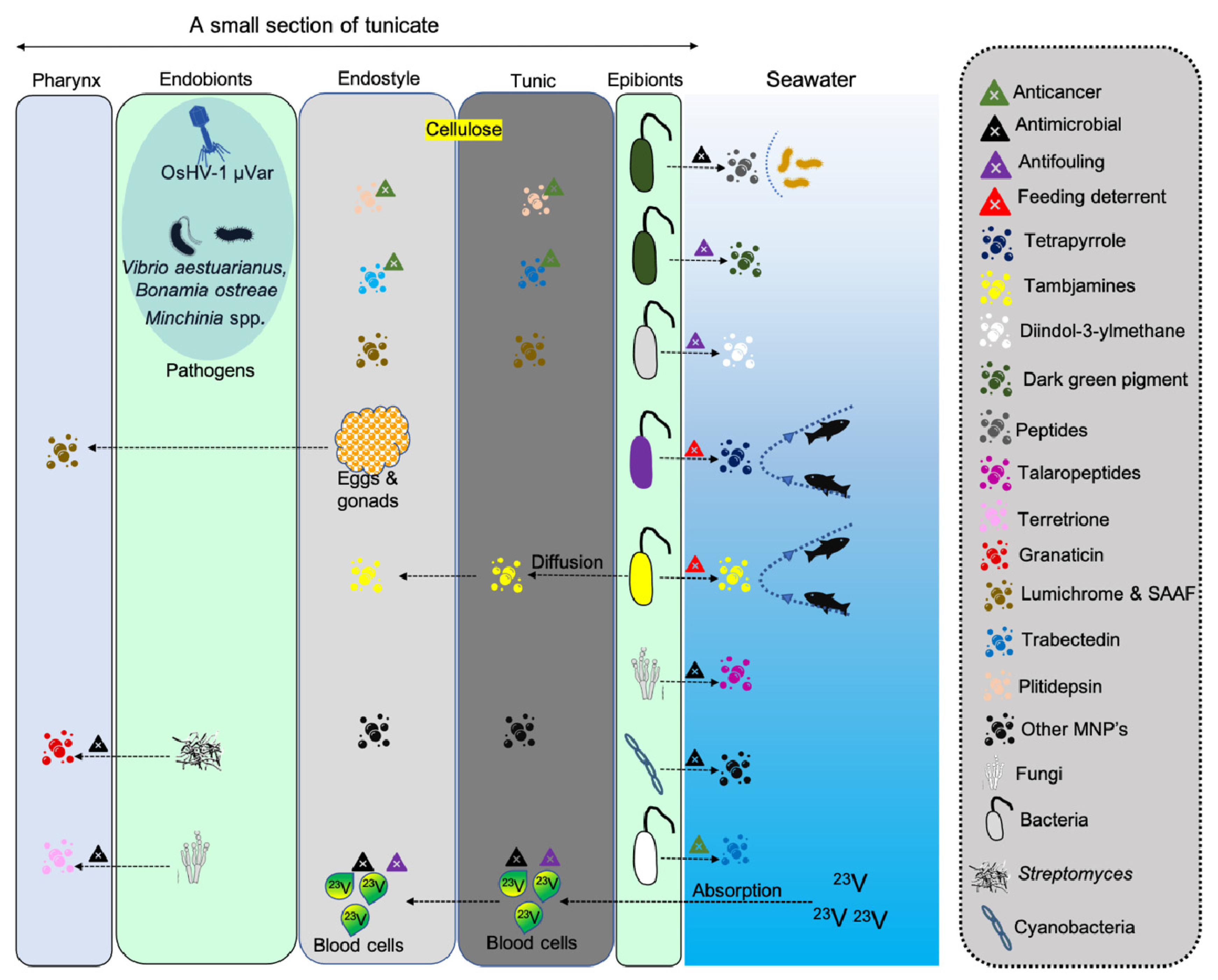
4. Antimicrobial Applications
5. Anticancer and Antitumor Applications
6. Antifouling and Anti-Deterrent Activities
7. Miscellaneous Applications
This entry is adapted from the peer-reviewed paper 10.3390/md19060308
References
- Shenkar, N.; Swalla, B.J. Global diversity of Ascidiacea. PLoS ONE 2011, 6, e20657.
- Holland, L.Z. Tunicates. Curr. Biol. 2016, 26, R141–R156.
- Gasparini, F.; Ballarin, L. Reproduction in Tunicates. In Encyclopedia of Reproduction, 2nd ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2018; Volume 6, pp. 546–553.
- Bone, Q.; Carré, C.; Chang, P. Tunicate feeding filters. J. Mar. Biol. Assoc. UK 2003, 83, 907–919.
- Delsuc, F.; Philippe, H.; Tsagkogeorga, G.; Simion, P.; Tilak, M.K.; Turon, X.; López-Legentil, S.; Piette, J.; Lemaire, P.; Douzery, E.J.P. A phylogenomic framework and timescale for comparative studies of tunicates. BMC Biol. 2018, 16, 39.
- Berna, L.; Alvarez-Valin, F. Evolutionary genomics of fast evolving tunicates. Genome Biol. Evol. 2014, 6, 1724–1738.
- Swalla, B.J. Building divergent body plans with similar genetic pathways. Heredity 2006, 97, 235–243.
- Jeffery, W.R. Tunicates: Models for Chordate Evolution and Development at Low Genomic Complexity. In Comparative Genomics; Clark, M.S., Ed.; Springer Science + Business Media: New York, NY, USA, 2000; pp. 43–69.
- Lemaire, P. Evolutionary crossroads in developmental biology: The tunicates. Development 2011, 138, 2143–2152.
- Zhan, A.; Briski, E.; Bock, D.G.; Ghabooli, S.; MacIsaac, H.J. Ascidians as models for studying invasion success. Mar. Biol. 2015, 162, 2449–2470.
- Watters, D.J. Ascidian toxins with potential for drug development. Mar. Drugs 2018, 16, 162.
- Gouiffes, D.; Juge, M.; Grimaud, N.; Welin, L.; Sauviat, M.P.; Barbin, Y.; Laurent, D.; Roussakis, C.; Henichart, J.P.; Verbist, J.F. Bistramide A, a new toxin from the urochordata Lissoclinum bistratum Sluiter: Isolation and preliminary characterization. Toxicon 1988, 26, 1129–1136.
- Oh, K.-S.; Kim, J.-S.; Heu, M.-S. Food Constituents of Edible Ascidians Halocynthia roretzi and Pyura michaelseni. Korean J. Food Sci. Technol. 1997, 29, 955–962.
- Ali, A.J.H.; Tamilselvi, M. Ascidians in Coastal Water: A Comprehensive Inventory of Ascidian Fauna from the Indian Coast; Springer Nature: Cham, Switzerland, 2016; ISBN 9783319291185.
- DeFilippo, J.; Beck, G. Tunicate Immunology. In Reference Module in Life Sciences; Elsevier: Amsterdam, The Netherlands, 2018; pp. 1–10.
- Ayuningrum, D.; Liu, Y.; Riyanti; Sibero, M.T.; Kristiana, R.; Asagabaldan, M.A.; Wuisan, Z.G.; Trianto, A.; Radjasa, O.K.; Sabdono, A.; et al. Tunicate-associated bacteria show a great potential for the discovery of antimicrobial compounds. PLoS ONE 2019, 14, e0213797.
- Franchi, N.; Ballarin, L. Immunity in protochordates: The tunicate perspective. Front. Immunol. 2017, 8, 674.
- Walters, T.L.; Gibson, D.M.; Frischer, M.E. Cultivation of the marine pelagic tunicate Dlioletta gegenbauri (Uljanin 1884) for experimental studies. J. Vis. Exp. 2019, 150, e59832.
- Fusetani, N. Drugs from the Sea; Krager: Basel, Switzerland, 2000.
- Michibara, H.; Uyama, T.; Ueki, T.; Kanamori, K. The mechanism of accumulation and reduction of vanadium by ascidians. In The Biology of Ascidians; Sawada, H., Yokosawa, H., Lambert, C.C., Eds.; Springer: Tokyo, Japan, 2001; pp. 363–373.
- Shen, G.Q.; Baker, B.J. Biosynthetic studies of the eudistomins in the tunicate Eudistoma olivaceum. Tetrahedron Lett. 1994, 35, 1141–1144.
- Le, V.H.; Inai, M.; Williams, R.M.; Kan, T. Ecteinascidins. A review of the chemistry, biology and clinical utility of potent tetrahydroisoquinoline antitumor antibiotics. Nat. Prod. Rep. 2015, 32, 328–347.
- Xu, Y.; Kersten, R.D.; Nam, S.J.; Lu, L.; Al-Suwailem, A.M.; Zheng, H.; Fenical, W.; Dorrestein, P.C.; Moore, B.S.; Qian, P.Y. Bacterial biosynthesis and maturation of the didemnin anti-cancer agents. J. Am. Chem. Soc. 2012, 134, 8625–8632.
- Dewapriya, P.; Khalil, Z.G.; Prasad, P.; Salim, A.A.; Cruz-Morales, P.; Marcellin, E.; Capon, R.J. Talaropeptides A-D: Structure and biosynthesis of extensively N-methylated linear peptides from an Australian marine tunicate-derived Talaromyces sp. Front. Chem. 2018, 6, 394.
- Dou, X.; Dong, B. Origins and bioactivities of natural compounds derived from marine ascidians and their symbionts. Mar. Drugs 2019, 17, 670.
- Casertano, M.; Menna, M.; Imperatore, C. The ascidian-derived metabolites with antimicrobial properties. Antibiotics 2020, 9, 510.
- Palanisamy, S.K.; Rajendran, N.M.; Marino, A. Natural products diversity of marine ascidians (Tunicates; Ascidiacea) and successful drugs in clinical development. Nat. Prod. Bioprospect. 2017, 7, 1–111.
- Petersen, J.K.; Riisgard, H.U. Filtration capacity of the ascidian Ciona intestinalis and its grazing impact in a shallow fjord. Mar. Ecol. Prog. Ser. 1992, 88, 9–17.
- Morris, R.J.; Bone, Q.; Head, R.; Braconnot, J.C.; Nival, P. Role of salps in the flux of organic matter to the bottom of the Ligurian Sea. Mar. Biol. 1988, 97, 237–241.
- Pomeroy, L.R.; Deibel, D. Aggregation of organic matter by pelagic tunicates. Limnol. Oceanogr. 1980, 25, 643–652.
- Gorsky, G.; Da Silva, N.L.; Dallot, S.; Laval, P.; Braconnot, J.C.; Prieur, L. Midwater tunicates: Are they related to the permanent front of the Ligurian Sea (NW Mediterranean)? Mar. Ecol. Prog. Ser. 1991, 74, 195–204.
- Sutherland, K.R.; Madin, L.P.; Stocker, R. Filtration of submicrometer particles by pelagic tunicates. Proc. Natl. Acad. Sci. USA 2010, 107, 15129–15134.
- Hirose, E.; Nozawa, Y. Latitudinal difference in the species richness of photosymbiotic ascidians along the east coast of Taiwan. Zool. Stud. 2020, 59, e19.
- Davis, A.R. Alkaloids and ascidian chemical defense: Evidence for the ecological role of natural products from Eudistoma olivaceum. Mar. Biol. 1991, 111, 375–379.
- Zhao, Y.; Li, J. Excellent chemical and material cellulose from tunicates: Diversity in cellulose production yield and chemical and morphological structures from different tunicate species. Cellulose 2014, 21, 3427–3441.
- Menna, M.; Aiello, A. The Chemistry of Marine Tunicates. In Handbook of Marine Natural Products; Fattorusso, E., Gerwick, W.H., Taglialatela-Scafati, O., Eds.; Springer Science + Business Media B.V.: Berlin/Heidelberg, Germany, 2012; pp. 295–385.
- Franchi, N.; Ballarin, L. Cytotoxic cells of compound Ascidians. In Lessons in Immunity: From Single-Cell Organisms to Mammals; Ballarin, L., Cammarata, M., Eds.; Elsevier Inc.: London, UK, 2016; pp. 193–199.
- Parrinello, N. Cytotoxic activity of tunicate hemocytes. In Invertebrate Immunology; Rinkevich, B., Müller, W.E.G., Eds.; Springer: Berlin/Heidelberg, Germany, 1996; pp. 190–217.
- Liu, L.; Zheng, Y.-Y.; Shao, C.-L.; Wang, C.-Y. Metabolites from marine invertebrates and their symbiotic microorganisms: Molecular diversity discovery, mining, and application. Mar. Life Sci. Technol. 2019, 1, 60–94.
- Leal, M.C.; Sheridan, C.; Osinga, R.; Dionísio, G.; Rocha, R.; Silva, B.; Rosa, R.; Calado, C. Marine Microorganism-Invertebrate Assemblages: Perspectives to Solve the “Supply Problem” in the Initial Steps of Drug Discovery. Mar. Drugs 2014, 12, 3929–3952.
- Chen, L.; Hu, J.S.; Xu, J.L.; Shao, C.L.; Wang, G.Y. Biological and chemical diversity of ascidian-associated microorganisms. Mar. Drugs 2018, 16, 362.
- Evans, J.S.; Erwin, P.M.; Shenkar, N.; López-Legentil, S. Introduced ascidians harbor highly diverse and host-specific symbiotic microbial assemblages. Sci. Rep. 2017, 7, 11033.
- Tianero, M.D.B.; Kwan, J.C.; Wyche, T.P.; Presson, A.P.; Koch, M.; Barrows, L.R.; Bugni, T.S.; Schmidt, E.W. Species specificity of symbiosis and secondary metabolism in ascidians. ISME J. 2015, 9, 615–628.
- Flood, P. Yellow-stained oikopleurid appendicularians are caused by bacterial parasitism. Mar. Ecol. Prog. Ser. 1991, 71, 291–295.
- Karthikeyan, M.M.; Ananthan, G.; Balasubramanian, T. Antimicrobial activity of crude extracts of some ascidians (Urochordata: Ascidiacea), from Palk Strait, (Southeast Coast of India). World J. Fish. Mar. Sci. 2009, 1, 262–267.
- Ayuningrum, D.; Kristiana, R.; Nisa, A.A.; Radjasa, S.K.; Muchlissin, S.I.; Radjasa, O.K.; Sabdono, A.; Trianto, A. Bacteria associated with tunicate, Polycarpa aurata, from Lease sea, Maluku, Indonesia exhibiting anti-multidrug resistant bacteria. Biodiversitas 2019, 20, 956–964.
- Litaay, M.; Christine, G.; Gobel, R.B.; Dwyana, Z. Bioactivity of endo-symbiont bacteria of tunicate Polycarpa aurata as antimicrobial. In Proceedings of the 23 National Seminar of Indonesia Biology Society, Jayapura, Indonesia, 18 September 2015.
- Menna, M.; Fattorusso, E.; Imperatore, C. Alkaloids from marine ascidians. Molecules 2011, 16, 8694–8732.
- Franks, A.; Haywood, P.; Holmström, C.; Egan, S.; Kjelleberg, S.; Kumar, N. Isolation and structure elucidation of a novel yellow pigment from the marine bacterium Pseudoalteromonas tunicata. Molecules 2005, 10, 1286–1291.
- Franks, A.; Egan, S.; Holmström, C.; James, S.; Lappin-Scott, H.; Kjelleberg, S. Inhibition of fungal colonization by Pseudoalteromonas tunicata provides a competitive advantage during surface colonization. Appl. Environ. Microbiol. 2006, 72, 6079–6087.
- Egan, S.; James, S.; Holmström, C.; Kjelleberg, S. Correlation between pigmentation and antifouling compounds produced by Pseudoalteromonas tunicata. Environ. Microbiol. 2002, 4, 433–442.
- Matz, C.; Webb, J.S.; Schupp, P.J.; Phang, S.Y.; Penesyan, A.; Egan, S.; Steinberg, P.; Kjelleberg, S. Marine biofilm bacteria evade eukaryotic predation by targeted chemical defense. PLoS ONE 2008, 3, e2744.
- Holmström, C.; James, S.; Neilan, B.A.; White, D.C.; Kjelleberg, S. Pseudoalteromonas tunicata sp. nov., a bacterium that produces antifouling agents. Int. J. Syst. Bacteriol. 1998, 48, 1205–1212.
- Kumaran, N.S.; Bragadeeswaran, S.; Meenakshi, V.K.; Balasubramanian, T. Bioactivity potential of extracts from ascidian Lissoclinum fragile. Afr. J. Pharm. Pharmacol. 2012, 6, 1854–1859.
- Devi, S.; Rajasekharan, K.; Padmakumar, K.; Tanaka, J.; Higa, T. Biological activity and chemistry of the compound ascidian Eusynstyela tineta. In The Biology of Ascidians; Sawada, H., Yokosawa, H., Lambert, C.C., Eds.; Springer: Tokyo, Japan, 2001; pp. 341–354.
- James, S.G.; Holmström, C.; Kjelleberg, S. Purification and characterization of a novel antibacterial protein from the marine bacterium D2. Appl. Environ. Microbiol. 1996, 62, 2783–2788.
- Lee, I.H.; Zhao, C.; Cho, Y.; Harwig, S.S.L.; Cooper, E.L.; Lehrer, R.I. Clavanins, α-helical antimicrobial peptides from tunicate hemocytes. FEBS Lett. 1997, 400, 158–162.
- Jang, W.S.; Kim, K.N.; Lee, Y.S.; Nam, M.H.; Lee, I.H. Halocidin: A new antimicrobial peptide from hemocytes of the solitary tunicate, Halocynthia aurantium. FEBS Lett. 2002, 521, 81–86.
- Galinier, R.; Roger, E.; Sautiere, P.E.; Aumelas, A.; Banaigs, B.; Mitta, G. Halocyntin and papillosin, two new antimicrobial peptides isolated from hemocytes of the solitary tunicate, Halocynthia papillosa. J. Pept. Sci. 2009, 15, 48–55.
- Azumi, K.; Yokosawa, H.; Ishii, S.I. Halocyamines: Novel antimicrobial tetrapeptide-like substances isolated from the hemocytes of the solitary ascidian Halocynthia roretzi. Biochemistry 1990, 29, 159–165.
- Menzel, L.P.; Lee, I.H.; Sjostrand, B.; Lehrer, R.I. Immunolocalization of clavanins in Styela clava hemocytes. Dev. Comp. Immunol. 2002, 26, 505–515.
- Fedders, H.; Michalek, M.; Grötzinger, J.; Leippe, M. An exceptional salt-tolerant antimicrobial peptide derived from a novel gene family of haemocytes of the marine invertebrate Ciona intestinalis. Biochem. J. 2008, 416, 65–75.
- Odate, S.; Pawlik, J.R. The role of vanadium in the chemical defense of the solitary tunicate, Phallusia nigra. J. Chem. Ecol. 2007, 33, 643–654.
- Sung, A.A.; Gromek, S.M.; Balunas, M.J. Upregulation and identification of antibiotic activity of a marine-derived Streptomyces sp. via co-cultures with human pathogens. Mar. Drugs 2017, 15, 250.
- Nurfadillah, A.; Litaay, M.; Gobel, R.B.; Haedar, N. Potency of tunicate Polycarpa aurata as inoculum source of sebagai sumber endosimbyotic fungi that produce antimicrobe. J. Alam Lingkung. 2015, 6, 10–16.
- Tahir, E.; Litaay, M.; Gobel, R.B.; Haedar, N.; Al, E. Potency of tunicate Rhopalaea crassa as inoculum source of endosymbiont fungi that produce antimicrobe. Spermonde 2016, 2, 33–37.
- Shaala, L.A.; Youssef, D.T.A. Identification and bioactivity of compounds from the fungus Penicillium sp. CYE-87 isolated from a marine tunicate. Mar. Drugs 2015, 13, 1698–1709.
- Rinehart, K.L.; Gloer, J.B.; Hughes, R.G.; Renis, H.E.; Patrick McGovren, J.; Swynenberg, E.B.; Stringfellow, D.A.; Kuentzel, S.L.; Li, L.H. Didemnins: Antiviral and antitumor depsipeptides from a Caribbean tunicate. Science 1981, 212, 933–935.
- Canonico, P.G.; Pannier, W.L.; Huggins, J.W.; Rienehart, K.L. Inhibition of RNA viruses in vitro and in Rift Valley fever-infected mice by didemnins A and B. Antimicrob. Agents Chemother. 1982, 22, 696–697.
- Kobayashi, J.; Harbour, G.C.; Gilmore, J.; Rinehart, K.L. Eudistomins A, D, G, H, I, J, M, N, O, P, and Q, Bromo-, Hydroxy-, Pyrrolyl-, and 1-Pyrrolinyl-β-carbolines from the antiviral Caribbean tunicate Eudistoma olivaceum. J. Am. Chem. Soc. 1984, 106, 1526–1528.
- Mitchell, S.S.; Rhodes, D.; Bushman, F.D.; Faulkner, D.J. Cyclodidemniserinol trisulfate, a sulfated serinolipid from the Palauan ascidian Didemnum guttatum that inhibits HIV-1 integrase. Org. Lett. 2000, 2, 1605–1607.
- Smith, T.E.; Pond, C.D.; Pierce, E.; Harmer, Z.P.; Kwan, J.; Zachariah, M.M.; Harper, M.K.; Wyche, T.P.; Matainaho, T.K.; Bugni, T.S.; et al. Accessing chemical diversity from the uncultivated symbionts of small marine animals. Nat. Chem. Biol. 2018, 14, 179–185.
- Mayer, A.M.S.; Glaser, K.B.; Cuevas, C.; Jacobs, R.S.; Kem, W.; Little, R.D.; McIntosh, J.M.; Newman, D.J.; Potts, B.C.; Shuster, D.E. The odyssey of marine pharmaceuticals: A current pipeline perspective. Trends Pharmacol. Sci. 2010, 31, 255–265.
- Zelek, L.; Yovine, A.; Brain, E.; Turpin, F.; Taamma, A.; Riofrio, M.; Spielmann, M.; Jimeno, J.; Misset, J.L. A phase II study of Yondelis® (trabectedin, ET-743) as a 24-h continuous intravenous infusion in pretreated advanced breast cancer. Br. J. Cancer 2006, 94, 1610–1614.
- Atmaca, H.; Bozkurt, E.; Uzunoglu, S.; Uslu, R.; Karaca, B. A diverse induction of apoptosis by trabectedin in MCF-7 (HER2−/ER+) and MDA-MB-453 (HER2+/ER−) breast cancer cells. Toxicol. Lett. 2013, 221, 128–136.
- Grosso, F.; Jones, R.L.; Demetri, G.D.; Judson, I.R.; Blay, J.-Y.; Cesne, A.L.; Lippo, R.S.; Casieri, P.; Collini, P.; Dileo, P.; et al. Effi cacy of trabectedin (ecteinascidin-743) in advanced pretreated myxoid liposarcomas: A retrospective study. Lancet Oncol. 2007, 8, 595–602.
- Sessa, C.; De Braud, F.; Perotti, A.; Bauer, J.; Curigliano, G.; Noberasco, C.; Zanaboni, F.; Gianni, L.; Marsoni, S.; Jimeno, J.; et al. Trabectedin for women with ovarian carcinoma after treatment with platinum and taxanes fails. J. Clin. Oncol. 2005, 23, 1867–1874.
- Krasner, C.N.; McMeekin, D.S.; Chan, S.; Braly, P.S.; Renshaw, F.G.; Kaye, S.; Provencher, D.M.; Campos, S.; Gore, M.E. A Phase II study of trabectedin single agent in patients with recurrent ovarian cancer previously treated with platinum-based regimens. Br. J. Cancer 2007, 97, 1618–1624.
- Monk, B. A randomized phase III study of trabectedin with pegylated liposomal doxorubicin (PLD) versus PLD in relapsed, recurrent ovarian cancer (OC). Eur. J. Cancer Suppl. 2008, 19, viii1–viii4.
- Rath, C.M.; Janto, B.; Earl, J.; Ahmed, A.; Hu, F.Z.; Hiller, L.; Dahlgren, M.; Kreft, R.; Yu, F.; Wolff, J.J.; et al. Meta-omic characterization of the marine invertebrate microbial consortium that produces the chemotherapeutic natural product ET-743. ACS Chem. Biol. 2011, 6, 1244–1255.
- Tohme, R.; Darwiche, N.; Gali-Muhtasib, H. A journey under the sea: The quest for marine anti-cancer alkaloids. Molecules 2011, 16, 9665–9696.
- González-Santiago, L.; Suárez, Y.; Zarich, N.; Muñoz-Alonso, M.J.; Cuadrado, A.; Martínez, T.; Goya, L.; Iradi, A.; Sáez-Tormo, G.; Maier, J.V.; et al. Aplidin® induces JNK-dependent apoptosis in human breast cancer cells via alteration of glutathione homeostasis, Rac1 GTPase activation, and MKP-1 phosphatase downregulation. Cell Death Differ. 2006, 13, 1968–1981.
- Cuadrado, A.; García-Fernández, L.F.; González, L.; Suárez, Y.; Losada, A.; Alcaide, V.; Martínez, T.; Máa Fernández-Sousa, J.; Sánchez-Puelles, J.M.; Muñoz, A. AplidinTM induces apoptosis in human cancer cells via glutathione depletion and sustained activation of the epidermal growth factor receptor, Src, JNK, and p38 MAPK. J. Biol. Chem. 2003, 278, 241–250.
- Mitsiades, C.S.; Ocio, E.M.; Pandiella, A.; Maiso, P.; Gajate, C.; Garayoa, M.; Vilanova, D.; Montero, J.C.; Mitsiades, N.; McMullan, C.J.; et al. Aplidin, a marine organism-derived compound with potent antimyeloma activity in vitro and in vivo. Cancer Res. 2008.
- Rinehart, K.L. Antitumor compounds from tunicates. Med. Res. Rev. 2000, 20, 1–27.
- Rajesh, R.P.; Annappan, M. Anticancer effects of brominated indole alkaloid eudistomin H from marine ascidian Eudistoma viride against cervical cancer cells (HeLa). Anticancer Res. 2015, 35, 283–294.
- Raub, M.F.; Cardellina, J.H.; Choudhary, M.I.; Ni, C.Z.; Clardy, J.; Alley, M.C. Clavepictines A and B: Cytotoxic Quinolizidines from the Tunicate Clavelina picta. J. Am. Chem. Soc. 1991, 113, 3178–3180.
- Bracegirdle, J.; Robertson, L.P.; Hume, P.A.; Page, M.J.; Sharrock, A.V.; Ackerley, D.F.; Carroll, A.R.; Keyzers, R.A. Lamellarin Sulfates from the Pacific Tunicate Didemnum ternerratum. J. Nat. Prod. 2019, 82, 2000–2008.
- Kang, H.; Fenical, W. Polycarpine dihydrochloride: A cytotoxic dimeric disulfide alkaloid from the Indian ocean ascidian Polycarpa clavata. Tetrahedron Lett. 1996, 37, 2369–2372.
- Kobayashi, J.; Cheng, J.F.; Nakamura, H.; Ohizumi, Y.; Walchli, M.R.; Hirata, Y.; Sasaki, T. Cystodytins A, B, and C, novel tetracyclic aromatic alkaloids with potent antineoplastic activity from the Okinawan tunicate Cystodytes dellechiajei. J. Org. Chem. 1988, 53, 1800–1804.
- Zabriskie, T.M.; Mayne, C.L.; Ireland, C.M. Patellazole C: A novel cytotoxic macrolide from Lissoclinum patella. J. Am. Chem. Soc. 1988, 110, 7919–7920.
- Kobayashi, J.; Cheng, J.F.; Nakamura, H.; Ohta, T.; Nozoe, S.; Hirata, Y.; Sasaki, T. Lejimalides A and B, novel 24-membered macrolides with potent antileukemic activity from the Okinawan tunicate Eudistoma cf. rigida. J. Org. Chem. 1988, 53, 6147–6150.
- Kikuchi, Y.; Ishibashi, M.; Sasaki, T.; Kobayashi, J. Lejimalides C and D, new antineoplastic 24-membered macrolide sulfates from the okinawan marine tunicate Eudistoma cf. rigida. Tetrahedron Lett. 1991, 32, 789–797.
- Nguyen, M.H.; Imanishi, M.; Kurogi, T.; Wan, X.; Ishmael, J.E.; McPhail, K.L.; Smith, A.B. Synthetic access to the mandelalide family of macrolides: Development of an anion relay chemistry strategy. J. Org. Chem. 2018, 83, 4287–4306.
- Charyulu, G.A.; McKee, T.C.; Ireland, C.M. Diplamine, a cytotoxic polyaromatic alkaloid from the tunicate Diplosoma sp. Tetrahedron Lett. 1989, 30, 4201–4202.
- Urdiales, J.L.; Morata, P.; De Castro, I.N.; Sánchez-Jiménez, F. Antiproliferative effect of dehydrodidemnin B (DDB), a depsipeptide isolated from Mediterranean tunicates. Cancer Lett. 1996, 102, 31–37.
- Rinehart, K.L.; Holt, T.G.; Fregeau, N.L.; Stroh, J.G.; Keifer, P.A.; Sun, F.; Li, L.H.; Martin, D.G. Ecteinascidins 729, 743, 745, 759A, 759B, and 770: Potent antitumor agents from the Caribbean tunicate Ecteinascidia turbinata. J. Org. Chem. 1990, 55, 4512–4515.
- Izbicka, E.; Lawrence, R.; Raymond, E.; Eckhardt, G.; Faircloth, G.; Jimeno, J.; Clark, G.; Von Hoff, D.D. In vitro antitumor activity of the novel marine agent, Ecteinascidin-743 (ET-743, NSC-648766) against human tumors explanted from patients. Ann. Oncol. 1998, 9, 981–987.
- Berlmck, R.G.S.; Britton, R.; Piers, E.; Lim, L.; Roberge, M.; Moreira Da Rocha, R.; Andersen, R.J. Granulatimide and isogranulatimide, aromatic alkaloids with G2 checkpoint inhibition activity isolated from the Brazilian ascidian Didemnum granulatum: Structure elucidation and synthesis. J. Org. Chem. 1998, 63, 9850–9856.
- Dassonneville, L.; Wattez, N.; Baldeyrou, B.; Mahieu, C.; Lansiaux, A.; Banaigs, B.; Bonnard, I.; Bailly, C. Inhibition of topoisomerase II by the marine alkaloid ascididemin and induction of apoptosis in leukemia cells. Biochem. Pharmacol. 2000, 60, 527–537.
- Abourriche, A.; Abboud, Y.; Maoufoud, S.; Mohou, H.; Seffaj, T.; Charrouf, M.; Chaib, N.; Bennamara, A.; Bontemps, N.; Francisco, C. Cynthichlorine: A bioactive alkaloid from the tunicate Cynthia savignyi. Farmaco 2003, 58, 1351–1354.
- Torii, M.; Hitora, Y.; Kato, H.; Koyanagi, Y.; Kawahara, T.; Losung, F.; Mangindaan, R.E.P.; Tsukamoto, S. Siladenoserinols M-P, sulfonated serinol derivatives from a tunicate. Tetrahedron 2018, 74, 7516–7521.
- Holmstrom, C.; James, S.; Egan, S.; Kjelleberg, S. Inhibition of common fouling organisms by marine bacterial isolates with special reference to the role of pigmented bacteria. Biofouling 1996, 10, 251–259.
- Holmstrom, C.; Rittschof, D.; Kjelleberg, S. Inhibition of settlement by larvae of Balanus amphitrite and Ciona intestinalis by a surface-colonizing marine bacterium. Appl. Environ. Microbiol. 1992, 58, 2111–2115.
- Wang, K.L.; Xu, Y.; Lu, L.; Li, Y.; Han, Z.; Zhang, J.; Shao, C.L.; Wang, C.Y.; Qian, P.Y. Low-toxicity diindol-3-ylmethanes as potent antifouling compounds. Mar. Biotechnol. 2015, 17, 624–632.
- Stoecker, D. Resistance of a tunicate to fouling. Biol. Bull. 1978, 155, 615–626.
- Vervoort, H.C.; Pawlik, J.R.; Fenical, W. Chemical defense of the Caribbean ascidian Didemnum conchyliatum. Mar. Ecol. Prog. Ser. 1998, 164, 221–228.
- Lindquist, N.; Hay, M.E.; Fenical, W. Defense of ascidians and their conspicuous larvae: Adult vs. larval chemical defenses. Ecol. Monogr. 1992, 62, 547–568.
- Lindquist, N.; Hay, M.E. Can small rare prey be chemically defended? The case for marine larvae. Ecology 1995, 76, 1347–1358.
- Seleghim, M.H.R.; De Lira, S.P.; Campana, P.T.; Berlinck, R.G.S.; Custódio, M.R. Localization of granulatimide alkaloids in the tissues of the ascidian Didemnum granulatum. Mar. Biol. 2007, 150, 967–975.
- Núñez-Pons, L.; Carbone, M.; Vázquez, J.; Rodríguez, J.; Nieto, R.M.; Varela, M.M.; Gavagnin, M.; Avila, C. Natural products from antarctic colonial ascidians of the genera Aplidium and Synoicum: Variability and defensive role. Mar. Drugs 2012, 10, 1741–1764.
- Paul, V.J.; Lindquist, N.; Fenical, W. Chemical defenses of the tropical ascidian Atapozoa sp. and its nudibranch predators Nembrotha spp. Mar. Ecol. Prog. Ser. 1990, 59, 109–118.
- Lindquist, N.; Fenical, W. New tambjamine class alkaloids from the marine ascidian Atapozoa sp. and its nudibranch predators. Origin of the tambjamines in Atapozoa. Experientia 1991, 47, 504–506.
- Wasserman, H.H.; Friedland, D.J.; Morrison, D.A. A novel dipyrrolyldipyrromethene prodigiosin analog from Serratia marcescens. Tetrahedron Lett. 1968, 6, 641–644.
- McClintock, J.B.; Amsler, M.O.; Koplovitz, G.; Amsler, C.D.; Baker, B.J. Observations on an association between the dexaminid amphipod Polycheria antarctica f. acanthopoda and its ascidian host Distaplia cylindrica. J. Crustac. Biol. 2009, 29, 605–608.
- Cheng, M.T.; Rinehart, K.L. Polyandrocarpidines: Antimicrobial and Cytotoxic Agents from a Marine Tunicate (Polyandrocarpa sp.) from the Gulf of California. J. Am. Chem. Soc. 1978, 100, 7409–7411.
- Gorbman, A.; Whiteley, A.; Kavanaugh, S. Pheromonal stimulation of spawning release of gametes by gonadotropin releasing hormone in the chiton, Mopalia sp. Gen. Comp. Endocrinol. 2003, 131, 62–65.
- Tsukamotol, S.; Kato, H.; Hirota, H.; Fusetane, N. Lumichrome Is a putative intrinsic substance inducing larval metamorphosis in the ascidian Halocynthia roretzi. In The Biology of Ascidians; Sawada, H., Yokosawa, H., Lambert, C.C., Eds.; Springer: Tokyo, Japan, 2001; pp. 335–340.
- Watanabe, T.; Shibata, H.; Ebine, M.; Tsuchikawa, H.; Matsumori, N.; Murata, M.; Yoshida, M.; Morisawa, M.; Lin, S.; Yamauchi, K.; et al. Synthesis and complete structure determination of a sperm-activating and -attracting factor isolated from the ascidian ascidia sydneiensis. J. Nat. Prod. 2018, 81, 985–997.
- Mikami, N.; Hosokawa, M.; Miyashita, K. Effects of sea squirt (Halocynthia roretzi) lipids on white adipose tissue weight and blood glucose in diabetic/obese KK-Ay mice. Mol. Med. Rep. 2010, 3, 449–453.
- Uchimasu, H.; Matsumura, K.; Tsuda, M.; Kumagai, K.; Akakabe, M.; Fujita, M.J.; Sakai, R. Mellpaladines and dopargimine, novel neuroactive guanidine alkaloids from a Palauan Didemnidae tunicate. Tetrahedron 2016, 72, 7185–7193.
- Casertano, M.; Imperatore, C.; Luciano, P.; Aiello, A.; Putra, M.Y.; Gimmelli, R.; Ruberti, G.; Menna, M. Chemical investigation of the indonesian tunicate Polycarpa aurata and evaluation of the effects against Schistosoma mansoni of the novel alkaloids polyaurines A and B. Mar. Drugs 2019, 17, 278.
- Kubanek, J.; Williams, D.E.; de Silva, E.D.; Allen, T.; Andersen, R.J. Cytotoxic alkaloids from the flatworm Prostheceraeus villatus and its tunicate prey Clavelina lepadiformis. Tetrahedron Lett. 1995, 36, 6189–6192.
- Wright, A.D.; Goclik, E.; König, G.M.; Kaminsky, R. Lepadins D-F: Antiplasmodial and antitrypanosomal decahydroquinoline derivatives from the tropical marine tunicate Didemnum sp. J. Med. Chem. 2002, 45, 3067–3072.
- Thakur, N.L. Studies on Some Bioactive Aspects of Selected Marine Organisms; Goa University: Goa, India, 2001.
