Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Gastroenterology & Hepatology
Non-alcoholic fatty liver disease (NAFLD) or metabolic dysfunction-associated steatotic liver disease (MASLD) and steatohepatitis (NASH) are chronic hepatic conditions leading to hepatocellular carcinoma (HCC) development. According to the recent “multiple-parallel-hits hypothesis”, NASH could be caused by abnormal metabolism, accumulation of lipids, mitochondrial dysfunction, and oxidative and endoplasmic reticulum stresses and is found in obese and non-obese patients.
- biomarker
- hepatocarcinogenesis mechanisms
- NAFLD
1. Introduction
The global epidemic of obesity and metabolic syndrome has become a significant threat to human health worldwide. Recent epidemiological evidence shows that non-alcoholic fatty liver disease (NAFLD), recently termed metabolic dysfunction-associated steatotic liver disease (MASLD), and steatohepatitis (NASH) have rapidly become leading etiologies predisposing to fibrosis, cirrhosis, and hepatocellular carcinoma (HCC) development due to the rapid increase in metabolic syndrome and obesity in the past decades and newly available therapies for hepatitis C (HCV) and B virus (HBV)-associated HCC [1,2,3,4] (Figure 1). NAFLD/NASH-HCC is known to have even lower survival rates than viral hepatitis HCC and is generally developed based on metabolic syndrome, hyperinsulinemia, insulin resistance, and dyslipidemia, and is characterized by fat accumulation in the liver, either due to the increased inflow of free fatty acids or de novo lipogenesis [3,4]. NAFLD, a hepatic manifestation of insulin resistance, is very important in the pathophysiology of type 2 diabetes (T2DM). Furthermore, T2DM confers a three-fold risk of HCC [5]. However, in recent studies, it was reported to occur in cirrhosis-free and non-obese patients (“lean NAFLD”) and to be triggered by endotoxin-related inflammation in the gut and adipocytokines, leptin, and adiponectin [4,6,7,8].
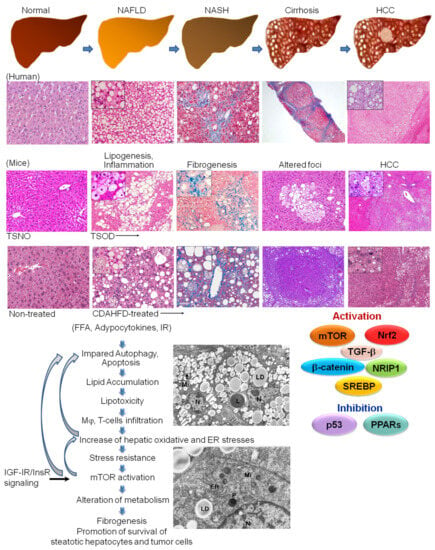
Figure 1. The developmental process of NAFLD/NASH-associated HCC in human and NASH model mice. The collagen-rich fibrotic regions are visualized via Masson’s trichrome staining. Transmission electron microscopy (TEM) is performed in TSOD mice HCC. LD: lipid droplet, Mi: mitochondria, N: nuclei, ER: endoplasmic reticulum, P: peroxisome, L: lysosome.
As for the mechanism, recently, the “multiple-parallel-hits” hypothesis was proposed, in which NASH was suggested to occur upon chronic liver damage caused by abnormal metabolism and lipotoxicity, lipid peroxidation, mitochondrial dysfunction, oxidative, endoplasmic reticulum stresses, inflammation, and cell death [9,10].
Monitoring the degree of hepatic necrotizing inflammation and fibrosis is crucial to predicting the long-term prognosis of NAFLD patients. Early intervention is necessary to slow disease progression. While liver biopsy is the gold standard for diagnosis, it is not widely used due to its invasive nature. Additionally, alpha-fetoprotein (AFP) is commonly used to diagnose HCC in NASH patients, but recent studies have found it difficult to distinguish between NASH and NASH-HCC patients [11,12]. Therefore, novel biomarkers are urgently needed for accurate and early NASH-HCC diagnosis and prognosis. The emergence of genomics, transcriptomics, proteomics, metabolomics, and lipidomics provides an opportunity to gain insight into multiple processes that contribute to the development of non-alcoholic steatohepatitis (NASH). Moreover, these technologies can aid in the identification of novel biomarkers for the diagnosis and prognosis of non-alcoholic fatty liver disease (NAFLD), NASH, and hepatocellular carcinoma (HCC). On the other hand, the discovery of NAFLD/NASH-associated physiopathological signaling cascades has benefited from large-scale omics studies describing the modulation of genes, proteins, and metabolites. In addition to well-known regulators of NAFLD/NASH hepatocarcinogenesis, such as transforming growth factor beta (TGF-β), SMAD family member 3 (SMAD3), insulin-like growth factor 1 (IGF1), peroxisome proliferation activating receptors (PPARs), sterol regulatory element-binding protein and liver X receptor α (SREBP-LXRα), CCAAT/enhancer-binding protein beta (CEBPB), nuclear factor κ-light-chain-binding protein (NFkB), c-myc, Wnt/β-catenin, signal transducer and activator of transcription 3 (STAT3), and p53, recent research has highlighted the important role of protein kinases mammalian target of rapamycin (mTOR), protein receptor sequestosome 1 (SQSTM1, p62), PI3K/Akt, PKC, MAPK, ErbB, nuclear factor (erythroid-derived 2)-like 2 (Nrf2), hepatocyte nuclear factor 1α (HNF1α) and 4α (HNF4α), and nuclear-receptor-interacting protein 1 (NRIP1; nuclear factor RIP140) in this process [12,13,14,15,16,17].
2. Signaling and Metabolic Pathways in the Regulation of NAFLD/NASH Progression, Hepatocarcinogenesis, and Associated Biomarkers
2.1. mTOR Pathway as a Multifunctional Driver of Metabolic Syndrome-Associated Hepatocarcinogenesis
Numerous clinical studies have established a causal connection between metabolic syndrome and diabetes with various types of cancer, including HCC. These conditions heighten the risk of developing cancer and facilitate the process of hepatocarcinogenesis by activating inflammatory pathways that generate proinflammatory cytokines and reactive oxygen species (ROS). This, in turn, leads to genomic instability, cellular proliferation, and the suppression of apoptosis [18] (Figure 2). The accumulation of lipids in the liver is a significant contributor to the progression of NAFLD in individuals with T2DM. The mTOR signaling pathway has a crucial role in regulating metabolic processes in various organs, including hepatic lipid metabolism, insulin resistance, chronic liver inflammation, and, ultimately, the advancement of NAFLD [18].
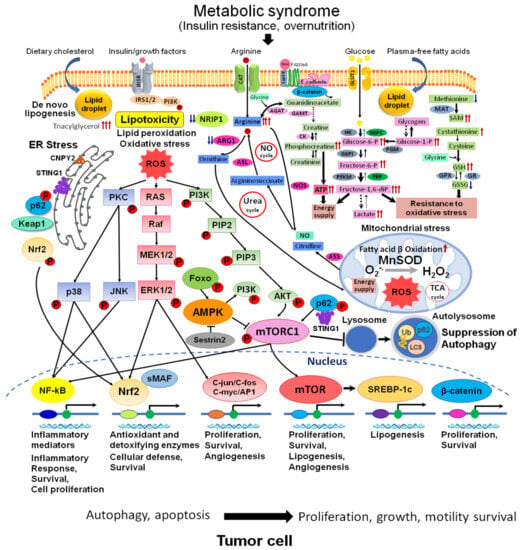
Figure 2. Metabolic and signaling pathways in NAFLD/NASH-associated HCC. Abnormal metabolism: accumulation of arginine, glucose metabolites, phosphocreatine, SAM, GSH, and alterations of urea and NO cycles are observed in NAFLD/NASH HCC. These events are associated with developed lipotoxicity, activation of de novo lipogenesis (SREBP-1), lipid peroxidation, fatty acid β oxidation, mitochondrial dysfunction, activation of oxidative (MAPK, Nrf2, NF-kB, C-jun/C-fos/C-myc/AP1) and ER stresses (p62, Keap1, Nrf2, STING, CNPY2)-related signaling, wnt/β-catenin pathway, and suppression of autophagy (mTORC1, AMPK, p62), with the central role for mTOR pathway. Abbreviations: ROS, reactive oxygen species; NO, nitric oxide; ER, endoplasmic reticulum; TCA, tricarboxylic acid cycle; INSR, insulin receptor; GLUT1, glucose trasporter 1; STING1, stimulator of interferon response cGAMP interactor 1; SAM, S-adeninemethyonine; SAH, S-adenosyl-L-homocysteine; Ub, ubiquitin; LC3, microtubule-associated protein 1A/1B−light chain 3; sMAF, small maf protein; CAT, cationic amino acid transporter; PKC, protein kinase C; MnSOD, manganese superoxide dismutase; PIP2, phosphatidylinositol 4,5-bisphosphate; PIP3, phosphatidylinositol 3,4,5-triphosphate; PI3K, phosphoinositide 3-kinase; AKT, protein kinase B (serine/threonine-specific protein kinase); p38, p38 mitogen-activated protein kinase; MEK, mitogen-activated protein kinase kinase; ERK, extracellular signal-regulated kinase; JNK, c-Jun N-terminal kinase; ARG1, arginase1; ASL, argininosuccinate lyase; ASS, argininosuccinate synthase; AGAT, arginine-glycine amidinotransferase; GAMT, guanidinoacetate N-methyltransferase; NOS, nitric oxide synthase; CK, creatine kinase; HK, hexokinase; G6PI, glucose-6-phosphate isomerase; PFKM, 6-phosphofructokinase; G6PC, glucose-6-phosphatase; FBP, fructose 1,6-bisphosphate aldolase; PGM, phosphoglycerate mutase; MAT, methionine adenosyltransferase; GPX, glutathione peroxidase, GR, glutathione reductase.
The protein kinase mTOR is the prime focus of rapamycin, an immunosuppressive drug that operates via two distinct complexes. These are mTORC1, which incorporates the regulatory-associated protein of mTOR (Raptor) and the rapamycin-associated TOR protein, and mTORC2, which consists of the rapamycin-insensitive companion of mTOR (Rictor). As there are two key regulatory components, the regulation of mTOR is very complex and is carried out by various proteins, hormones, growth factors, and nutrients [19]. mTOR controls, at least in part, four major metabolic processes responsible for the regulation and maintenance of normal levels of hepatic triglycerides (TG) amounts, which are altered in individuals with insulin resistance, obesity, and diabetes mellitus [20]. These include the delivery of plasma albumin-bound fatty acids (FAs), transferred from adipose tissue, TG synthesis from acetyl-coenzyme A (Ac-CoA) derived mainly from the metabolism of glucose in mitochondria, FAs oxidation, and TG secretion in very-low-density lipoprotein (VLDL) [19].
In recent years, protein kinases, including mTOR, MAPK, PKC, PI3K/Akt, and ErbB, were shown to govern most of the pathological pathways in NAFLD and regulate both inflammation and hepatic gluco- and liponeogenesis [21] (Figure 2). In connection with lipid metabolism alterations in NASH HCCs, proteomics research revealed overexpression of the downstream proteins of the SREBP-LXRα pathway (e.g., cytochrome P450 isoenzymes CYP51A1, CYP4F11 and CYP8B1, cortisone, etc.), and inhibition for those involved in lipid catabolism, such as molecules downstream of peroxisome proliferator-activated receptors α (PPARα) and γ (PPARγ), as well as mitochondrial and peroxisomal proteins involved in the process of oxidation of fatty acids (e.g., enoyl-CoA hydratase (ECHS1), 3-ketoacyl-CoA thiolase (ACAA2), and fatty acid binding protein 2 (FABP1)) [12,13]. In addition, the study found that NASH-HCC had increased lipid, cholesterol, and bile acid biosynthesis, elevated levels of triacylglycerol, and higher levels of AFP, AST, and cancer antigen 19-9 compared to NASH liver [12]. The pivotal role of mTOR in the regulation of metabolism, lipid biosynthesis, fibrogenesis, and NAFLD/NASH progression was confirmed with the help of omics techniques, and the close relationship with IGF1, TGF-β/SMAD, SREBP, PPARs, and other important cellular regulators was reported in human and animal studies [13,14,15]. Recent reports highlighted the crosstalk between mTOR and its upstream regulators such as Notch, Hedgehog, and Hippo [22]. It also delves into the mechanisms of the mTOR regulation of various factors, like Foxo1, Lipin1 (insulin resistance), oxidative stress (PIG3, p53, JNK), SREBPs (lipogenesis), Toll-like receptors (TLRs) (gut microbiota), autophagy, inflammation, genetic polymorphisms, and epigenetics in NAFLD [22]. Serum metabolic profiling further revealed the alteration of metabolites associated with NASH HCC and implicated in the regulation of the metabolism of lipids by PPARα, fatty acid metabolism, biogenic amine synthesis, downregulation of necroptosis, amino acid metabolism, and mTOR signaling pathway [12]. Proteome and metabolome analyses revealed the activation of mTOR in conjunction with AKT, PI3K, extracellular signal-regulated kinase 1/2 (ERK1/2) kinases, and β-catenin in the liver tumors developed in Tsumura, Suzuki, Obese Diabetic (TSOD) metabolic syndrome/T2DM NASH model mice [14,15] (Figure 2). This was also confirmed in human NASH HCCs [14]. However, mTOR activation was not prominent in CDAHFD- and DEN-treated mice and human virus-associated HCC [15]. When the proteomes of human NASH and HCV-associated HCCs were compared, altered proteins and enzymes involved in lipid, cholesterol, and bile acid biosynthesis, fatty acid oxidation and catabolism, mitochondrial dysfunction, and the downstream molecules of activated SREBP-LXRα, NRIP1, and inhibited PPARs was specific to NASH, but not the virus-associated HCC [14]. From these data, we conclude that along with IR and activation of TGF-β, the dysregulation of Wnt/β-catenin, NRIP1, Nrf2, SREBP-LXRα, PHBs, PPARs, and p53 play an important role in NASH pathogenesis. These findings caused an impulse in the discovery of novel biomarkers of NASH HCC and numerous chemopreventive and therapeutic studies with mTOR inhibitors in NAFLD/NASH/HCC [23].
Through studies investigating mTOR biomarkers and inhibitors, it has been revealed that mTOR plays a critical role in regulating adipocyte and lipogenic marker genes, including PPAR and SREBP1c. These genes are essential for lipogenesis and reducing inflammation and fibrosis. mTOR activation also influenced lipid biosynthesis in the liver preneoplastic (altered) foci and tumors of NASH model mice, inducing the tremendous accumulation of lipid droplets in the cells (ballooning) [14,15]. The discovery of hepatic G protein-coupled receptor 180 (Gpr180) has revealed its significant impact on lipid metabolism. It has been observed to increase triglyceride and cholesterol levels in the liver and plasma, leading to hepatic lipid deposition in HFD-treated Gpr180 knockout (KO) mice. Additionally, Gpr180 has been shown to decrease energy metabolism and activate adiposity [24]. Knockdown of Gpr180 in the Huh7 hepatoma cell line resulted in the downregulation of mTORC1 signaling, as detected by transcriptome analysis. Furthermore, decreases in activated SREBP1 cholesterol homeostasis and amelioration of plasma and hepatic lipid levels were observed in Gpr180 knockout mice. The study confirmed the relationship between Gpr180 and mTOR, SREBP1, and SREBP2. As a result, it was determined that Gpr180 could serve as a promising NAFLD biomarker and drug target for the treatment of NAFLD/NASH [24]. Another potential NAFLD/NASH biomarker and potential molecular target is the stimulator of interferon response cGAMP interactor 1 (STING1) [25]. Recent research indicates that STING1 plays a crucial role in the activation of MTORC1 by palmitic acid. The interaction of STING1 with various components of the MTORC1 complex and its reliance on SQSTM1 (p62), a crucial regulator of the MTORC1 pathway, enable this accomplishment. The activation of MTORC1 and STING1 were observed together in the liver tissues of NAFLD patients, providing clinical evidence that STING1 is involved in the activation of MTORC1 [25].
Concerning NASH hepatocarcinogenesis, it is also important to mention the recently discovered novel potential prognostic biomarker of human NAFLD/NASH-HCC, sphingosine 1-phosphate receptor 2 (S1PR2). The protein serves as a receptor on the cell membrane for sphingosine 1-phosphate (S1P), a lysophospholipid that is biologically active and has diverse physiological effects [26]. S1PR2 is responsible for the progression from NAFLD to NASH. The association of conjugated bile acids (CBAs) and S1PR2 alterations with the onset of NASH hepatocarcinogenesis was found in human NAFLD/NASH-HCCs in vivo and in Huh 7 and HepG2 hepatoma cell lines [26]. The cell cycle phases G1/G2 were regulated by S1PR2, which had a significant impact on the proliferation, invasion, migration, and apoptosis of both Huh 7 and HepG2 cells. Bioinformatics analysis further revealed a strong correlation between S1PR2 and several tumor-related pathways, including PI3K/AKT/mTOR, JAK/STAT, NF-kB, and ERK. Notably, high expression of S1PR2 was associated with poor prognosis in patients with NAFLD/NASH-HCC [26].
Another potential prognostic marker in metabolic syndrome/NASH-associated human liver cancer is arginase 1 (ARG1), whose expression is downregulated in NAFLD/NASH-HCC. ARG1 suppression was bound to shift from arginine to the production of guanidinoacetate and phosphocreatine (Figure 2). The study found that there is a strong correlation between low ARG1 expression and poorly differentiated tumors, higher pathological stage, and decreased patient survival, particularly in those with metabolic syndrome/T2DM. Suppression of ARG1 was associated with mTOR activation [14]. High expression of ARG1 was observed in HCV-positive HCC, while it was low in tumors associated with metabolic syndrome/NASH. Moreover, poor differentiation of NASH HCC was found to be primarily correlated with ARG1 negativity, which could explain the lower survival rates in patients.
2.2. Disturbances of Metabolism in NAFLD/NASH-HCC
Untargeted metabolomics has been recently applied to find novel and peculiar metabolite profiles in NADLD/NASH/HCC patients and NASH model mice and to give insights into discovering new biomarkers in NASH pathophysiology. The liver of TSOD NASH model mice showed an increase in lipogenesis, inflammation, peroxisome proliferation, and fibrogenesis, along with a significant rise in glucose metabolites, including glucose 1-phosphate, glucose 6-phosphate, galactose 1-phosphate, fructose 6-phosphate, and primarily fructose 1,6-diphosphate, in HCCs [14] (Figure 2). It was found that TSOD mice with liver preneoplastic lesions and tumors had significantly increased levels of polysaccharides, along with a drastic increase in amino acid L-arginine and notable increases in phosphocreatine, S-adenosylmethionine/S-adenosylhomocysteine ratio, and adenylate and guanylate energy charges [14].
Masarone et al. conducted a recent human metabolomics study that analyzed 307 subjects with steatosis, NASH, and NASH-cirrhosis. Through the study, they were able to identify specific metabolites that distinguished between the different patient classes [52]. In this investigation, an elevation of glycocholic acid, taurocholic acid, phenylalanine, and branched-chain amino-acids accompanied by a decrease in glutathione was found with the increase in the severity of the disease from steatosis to NASH and NASH-cirrhosis. Very recently, Ahmed et al. reported a depletion of amino acid leucine in NASH-HCC as compared with NASH patients [12]. In the past, branched-chain amino acids have been effective in inhibiting the growth and angiogenesis of HCC through the suppression of insulin resistance and degradation of vascular endothelial growth factor mRNA [12,53]. Significantly, they were found to trigger apoptosis in liver cancer cells by disrupting mTOR-related pathways [54]. Metabolomics profiling has proven to be an effective non-invasive diagnostic tool for distinguishing between NAFLD/NASH-HCC and identifying the different stages of the disease [14,52].
The emerging area of NAFLD/NASH-associated cancer development and progression was devoted to research on lipid metabolism [55]. The risk of NAFLD was reported to be increased with an imbalance between fatty acid uptake, oxidation, and secretion and a high level of plasma phospholipids (e.g., phosphatidylethanolamine), which can act as signal transductors with oncogenic and tumor-suppressive roles [12]. In addition, suppression of mitochondrial β-oxidation with increased peroxisomal and microsomal ω-oxidation accompanied by accumulation of hepatic fatty acids in NASH resulted in the activation of lipotoxicity and progression of the disease.
The HCCs in TSOD mice showed alterations in intracellular pathways, such as suppression of the urea cycle, methionine and putrescine degradation pathways, and an increase in cellular antioxidants like glutathione (GSH) levels. This indicates a boost in antioxidant activity in the HCCs [13]. Proteome analysis of HCCs revealed that increased levels of L-arginine significantly inhibited urea cycle enzymes, such as ARG1, argininosuccinate lyase (ASL), argininosuccinate synthase 1 (ASS1), and ornithine carbamoyltransferase (OTC). The study proposes that liver tumor-initiating cells contribute to the development of T2DM/NASH hepatocarcinogenesis by increasing glucose metabolites and L-arginine levels. This triggers glucose uptake, creatine metabolism, oxidative stress resistance, and tumor cell proliferation [14]. These findings highlighted the association between elevated L-arginine and inhibition of these enzymes.
2.3. Formation of Oxidative Stress and Resistance Mechanisms
Various sources contribute to the formation of oxidative stress, including the oxidation of free fatty acids, iron overload, inflammatory cytokines, lipid peroxidation, and mitochondrial dysfunction. Oxidative stress plays a crucial role in the progression of simple steatosis to NASH and hepatocarcinogenesis [56]. When the balance between the production of ROS (such as hydroxyl (OH•), superoxide (O2•−) radicals, and nitric oxide (•NO) and non-radical species (H2O2), as well as the protection from oxidative damage through antioxidant enzyme systems and DNA repair, is disrupted in the liver as in NAFLD/NASH, it can lead to the onset and progression of hepatocarcinogenesis [57]. Excessive ROS production can harm DNA and proteins, disrupt mitochondrial conditions, trigger apoptosis, and alter cellular signaling pathways like MAPK. This can impact regenerative cell proliferation, growth, and angiogenesis [57].
The development of oxidative stress and its influence on hepatocarcinogenesis was studied in NASH animal models. Formation of oxidative stress and damage to liver cell DNA in terms of 8-hydroxy-2-deoxyguanosine (8-OHdG) in TSOD and fatty liver Shionogi (FLS) NASH model mice was considered a key event in the initiation of T2DM/NASH-associated hepatocarcinogenesis [14,58]. From proteomics studies, NASH-associated hepatocarcinogenesis was found to be associated with the activation of nuclear factor (erythroid-derived 2)-like 2 (Nrf2)-mediated signaling, which is known to mediate resistance to oxidative stress injury [13]. In addition, alteration of protein expression of numerous enzymes involved in the control of mitochondrial function was also detected in NASH-associated human HCCs. Human and STAM NASH model mice HCCs exhibited strong overexpression of transcriptional factors prohibitin 1 (PHB1) and prohibitin 2 (PHB2). These factors play crucial roles in controlling mitochondrial respiration, function, cell proliferation, and apoptosis [13]. In addition, the study revealed an increase in the activity of superoxide dismutase manganese [Mn], mitochondrial (SOD2), and thioredoxin (TXN), while catalase (CAT) was found to be decreased in tumors [13]. In another NASH model containing mice fed with a methionine and choline-deficient diet (MCD), proteomic analysis of livers revealed significant elevation of peroxiredoxins 1 (Prdx1) and 6 (Prdx6) in correlation with TGFβ1, TNFα, and TLR4, CYP2E1, cytokeratin 8 (CK8), cytokeratin 18 (CK18), fructose-1,6-bisphosphatase 1 and vimentin, and downregulation of proteins involved in methionine (Met) metabolism and oxidative stress in NAFLD/NASH [27]. From those studies, all observed metabolome and proteome alterations pointed to the formation of oxidative stress resistance in liver tumor cells as a consequence of lipotoxicity and insulin resistance.
Among proteins involved in the development of liver tissue cellular defense against oxidative stress, oxidative DNA damage, inflammation, and steatohepatitis, peroxiredoxins 1 (Prx1) and 6 (Prdx6) and sestrin 2 (SESN2) were considered promising potential diagnostic biomarkers for the early stage of NAFLD/NASH, which may also influence the mTOR [27,28]. Prdx1 and Prdx6 are involved in Akt/mTOR, AMPK/mTOR, PI3K/Akt, FoxO and p53 signaling pathways, and ubiquitin-mediated proteolysis [59,60]. It was also demonstrated that protein levels of SESN2 were positively correlated with AMPK/mTOR pathway components and induced expression of lipogenesis-related genes in NAFLD model HFD-fed C57BL/6 mice liver and palmitic acid (PA)-stimulated HepG2 cells [28].
2.4. Glycoproteins-Related Pathways in NASH HCC and Potential Biomarkers
Ramachandran et al. have revealed through their study of human NASH-HCC serum samples that cholesterol and fatty acid metabolism regulators, LXR/RXR and FXR/RXR, along with acute phase response signaling, complement system, and clathrin-mediated endocytosis signaling are involved in pathways related to glycoproteins in the development of NASH and HCC [16]. Alpha-2-macroglobulin (A2MG), haptoglobin (HP), apolipoprotein C3 (APOC3), complement factor H (CFAH), serotransferrin (TRFE), vitronectin precursor (VTNC), ceruloplasmin (CERU), and alpha-1 antitrypsin (A1AT) exhibited unidirectional differences in abundance across the three phenotypes. Among the upstream regulators participating in NASH-associated hepatocarcinogenesis, HNF1α, HNF4α, and sterol regulatory element binding factor (SREBF1) were identified as associated with NASH HCC transcription factors.
Zhang et al. found that alpha-1-acid glycoprotein (AGP1) glycopeptides had significantly higher glycan branching, sialylation, and fucosylation in samples from NASH and cirrhosis patients compared to controls. This suggests that AGP-1 glycan could potentially serve as a diagnostic biomarker for NASH and associated HCC [29]. In addition, Kamada et al. discovered that fucosylated and hyper-sialylated variants of HP are valuable indicators for distinguishing NASH from NAFLD and HCC from control groups [30]. HP glycoforms could be therefore applied as markers for the diagnosis of NASH and associated HCC.
In addition to various glycosylated protein markers, the APOC3 protein exhibits notably decreased levels in NASH HCC, and can be applied for NASH-HCC diagnosis [31]. Moreover, specific glycopeptide moieties located at amino acid position 1424 of A2MG are detectable in the plasma of HCC patients [31].
2.5. Immune Dysregulation and Activation of Liver Fibrogenesis with the Central Role for TGF-β/SMAD Pathway
In the pathogenesis of NAFLD and NASH, an increase in plasma insulin levels, de novo lipogenesis, TG, and hepatic gluconeogenesis finally resulted in lipotoxicity, damage to hepatocytes, and an influx of immune cells, activation of transforming growth factor β (TGF-β)/SMAD signaling in hepatic stellate cells (HSCs), their differentiation into myofibroblasts, and finally proceeding to progressive fibrosis in the chronic state of NASH [61,62]. It has been shown that stress-related signaling pathways, like c-Jun N-terminal kinase (JNK) and NF-κB, are activated by pro-inflammatory cytokines, such as TNF-α, IL-1, IL-6, STAT3, and ERK. This activation leads to increased hepatocyte proliferation and ultimately promotes the development of hepatocarcinogenesis [61]. HSCs that are not strongly activated generate growth factors and cytokines, including hepatocyte growth factor (HGF), which can help prevent hepatocyte death and the development of HCC [62]. However, in the case of chronic liver injury, a shift to tumor-promoting HSC occurs, giving rise to a highly active HSC with myofibroblastic phenotype with increased deposition of type I collagen and other matrix proteins, which promotes cell proliferation and secreting TGF-β and platelet-derived growth factor (PDGF) [63]. In addition, TGF-β activation boosts the production of TGF-β within the cell and extends the lifespan of activated HSCs by decreasing apoptosis [64]. In addition, integrins, which act as cell surface mechanoreceptors, can interfere with TGF-β1 and PDGF and modulate the proliferation and survival of hepatocytes and HSC [65]. The regenerative repair pathways, such as the Hedgehog signaling pathway, are induced during the process of fibrogenesis in NASH and predict the induction of hepatocarcinogenesis [61].
Besides the hepatocytes, tumor microenvironment stromal cells, endothelial cells, immune cells, cytokines, and extracellular matrix (ECM) have been suggested to play a key role in the initiation and progression of HCC [66,67]. In proteomic studies with human NASH-associated biopsies, in addition to alteration proteins associated with inflammation, lipid deposition, insulin resistance, and oxidative stress, significant elevation of numerous ECM and cytoskeleton proteins, with the majority being downstream of TGF-β, including different collagens, fibronectin, vimentin, beta actin-like protein 2, tropomyosin α-4 chain, myosin 9, tubulin α-1C chain, moesin, and others, have been demonstrated [13,68]. On the contrary, the intermediate filament members CK8 and CK18 were strongly downregulated in human NASH biopsies and HCCs but not in virus-associated HCCs [13,69]. The CK18 fragments in the blood can indicate NASH and the severity of NAFLD in patients with uncontrolled hepatocyte apoptosis, necrosis, and caspase cleavage. A previous study on a diverse group of patients with biopsy-proven NAFLD highlights the potential clinical value of this test [32].
In addition, a large ECM glycoprotein thrombospondin-I (TSP-1) was reported to be a potential target of interest in NAFLD/NASH as it can help to develop anti-fibrotic therapeutics [33]. TSP-1 regulates inflammation, cell adhesion, angiogenesis, and several signaling pathways, such as TGF-β1. A comparison of the transcriptomic profiles based on genotype revealed significant differences in the expression of the PPAR pathway and amino acid metabolism pathways in TSP-1 null mice. These findings were further supported by a noticeable decrease in serum lipid levels, indicating activation of the PPAR pathway [33].
The influence of gut bacterial dysbiosis and microbial metabolites in the promotion of NASH hepatocarcinogenesis, worsening liver disease through enterohepatic circulation, has been recently reported [70,71]. It is known that the liver carries out immunological functions and participates in the physiological connection between gut-derived molecules and the systemic circulation, in which liver macrophages, Kupffer cells (KCs) producing pro-inflammatory and pro-fibrogenic cytokines and chemokines, liver resident cells, and monocytes play a very important role [61]. M1 macrophages promote inflammation, whereas M2 macrophages exert inhibitory effects and induce tissue repair. Moreover, pro-inflammatory cytokines, including TNF-α, IL-1β, IL-6, IL-12, C-C chemokine ligand 2 (CCL2) and 5 (CCL5) are secreted by M1 macrophages and exhibit increased amounts of nitric oxide species (NOS) and ROS [61]. Obesity, whether caused by diet or genetics, can affect the gut microbiota and increase the levels of deoxycholic acid (DCA), a metabolite produced by gut bacteria. This can lead to the activation of senescence-associated secretory phenotype (SASP) in hepatic stellate cells (HSCs), resulting in the secretion of inflammatory and tumor-promoting factors (such as IL-1, 6, and 8) in the liver. This process can contribute to the development of hepatocellular carcinoma (HCC) in both human and mouse models [8]. Recent metabolome research has revealed a reduction in serum levels of indole-3-propionic acid, a potential biomarker and a deamination metabolite of tryptophan maintaining intestinal homeostasis, in NASH-HCC patients [12,34].
The use of choline-deficient, L-amino acid-defined, high-fat diet (CDAHFD)-fed NASH model mice in animal experiments revealed severe inflammation, fibrosis, impaired mitochondrial respiration, and severe oxidative stress [72]. However, de novo lipogenesis (DLG), gluconeogenesis, and antioxidant enzymes in the liver were found to decrease. These changes were associated with a reduction in mitochondrial DNA copy number and complex proteins [73]. When exploring NASH biomarkers with gene expression analysis of the whole blood, a marked increase in the inflammation markers such as TNF-α, monocyte chemoattractant protein-1 (MCP1), IL1-β and lipocalin 2 (LCN2) (inflammation), collagen 1a1 (COL1A1) and 3a1 (COL3A1) (fibrosis), and Nrf2 targets NAD(P)H quinone dehydrogenase 1 (NQO1) and heme oxygenase (HO1) was found in the CDAHFD group. On the other hand, CDAHFD induced suppression of stearoyl-coenzyme A desaturase 1 (SCD1), fatty acid synthase (FASN), SREBF-1 (lipogenesis), G6PC and PEPCK (gluconeogenesis), and GPX2, CAT, and SOD3 (antioxidant enzymes) [73]. Recent findings in CDAHFD-treated mice suggested that proteins vitronectin (VN), monoacylglycerol acyltransferase 2 (MGAT2), retinoic acid-inducible gene-I (RIG-I)-like receptor (RLR), and formyl peptide receptor 2 (FPR2) may become potential diagnostic biomarkers and/or promising therapeutic opportunities for NAFLD/NASH by modulating the inflammatory reaction and contributing to the development of fibrosis via the activation of HSCs [35,36,37,38]. VN, being an EMT component protein, was shown to increase the COL1a2 mRNA expression levels, decrease CD11b and F4/80, macrophage markers, as well as TNF-α and IL-1β inflammatory cytokines and α-SMA, but did not affect the fatty acid synthases expression [35]. The regulation of TG absorption and homeostasis is crucially dependent on the highly expressed T2 enzyme in both the human small intestine and liver [36]. RLR and FPR2 could be also involved in inflammatory reactions in NASH development [37].
2.6. The Role of NRIP1 in NAFLD/NASH and Associated HCC
Recent proteome and bioinformatic analyses of NASH human liver biopsies have revealed a fascinating connection between the development of NAFLD and NASH and the activation of the transcription cofactor NRIP1 (nuclear-receptor-interacting protein 1), also known as RIP140 (receptor-interacting protein, 140 kDa) [13]. NRIP1 is a multifunctional transcriptional co-regulator, which plays important physiological roles in the control of a large number of metabolic nuclear receptors and transcription factors, such as the estrogen-related receptor (ESR1), nuclear receptor subfamily 3 group C member 1 (NR3C1) and member 2 (NR3C2), PPARα, PPARβ/δ, PPARγ, liver-X-receptor (LXRα), retinoid-X-receptor alpha (RXRα), apoAI enhancer, Wnt/β-catenin, RAR related orphan receptor A (RORA), and RORC, and is involved in the regulation of the ARNTL/BMAL1, CLOCK, and CRY1 circadian clock genes expression [74,75,76,77,78]. The NRIP1 gene plays a crucial role in regulating several bodily functions, including energy expenditure, lipid and glucose metabolism, inflammatory response, intestinal homeostasis, ovulation, mammary gland development, behavior, and cognition [74,79,80]. Compared to their wild-type counterparts on an HFD, NRIP1-null mice exhibit remarkable leanness, lack of obesity and hepatic steatosis, improved glucose tolerance, and heightened insulin responsiveness [81]. NRIP1 function as a coactivator for LXR appears to have an impact on its ability to regulate FAS expression in the liver [74,81].
NRIP1 also participates in the regulation of key steps and various oncogenic signaling pathways during cancer initiation and progression, especially in breast, ovary, liver, and colon tumors, and the development of atherosclerosis [79,82,83]. The impact of NRIP1 on HCC has been studied, and findings indicate that decreased levels of NRIP1 (both mRNA and protein) can lead to increased growth and migration of cancer cells in HCC. This is believed to be due to the direct interaction between NRIP1 and β-catenin, leading to repression of β-catenin/T-cell factor/lymphoid enhancer factor (TCF) signaling [78]. Furthermore, in Huh7 and HepG2 human liver cancer cells, overexpression of NRIP1 suppressed the malignant potential of liver cancer cells by inhibiting NF-kappaB (NF-kB)-mediated alternative polarization of macrophages [84], while the downregulation of miR-140-3-p miRNA, which targets the NRIP1 mRNA, was suggested to influence the hepatocarcinogenesis by stimulating anti-apoptotic signaling [85]. In an adrenoleukodystrophy mouse model, the genetic inactivation of NRIP1 prevented mitochondrial depletion and dysfunction, bioenergetic failure, and inflammatory dysregulation. This was achieved through a redox-dependent mechanism driven by very-long-chain fatty acids [86]. From these studies, NRIP1 is implicated in the development of NAFLD/NASH due to metabolic and inflammatory dysregulation and is downregulated in HCC, stimulating anti-apoptotic signaling and acting as an enhancer of proliferation, growth, and malignant potential of liver cancer cells. Further deciphering the link between NRIP1, hormone nuclear receptors, LXR/Fas, PPARs, Wnt/β-catenin/T-cell factor (TCF), and its epigenetic regulation could lead to novel strategies against the development of NASH and associated HCC. Furthermore, the NRIP1 in the form of circulating RNA (circRNA) could become a potential biomarker of NAFLD/NASH-HCC, which needs further investigation.
This entry is adapted from the peer-reviewed paper 10.3390/cancers15184566
This entry is offline, you can click here to edit this entry!
