Bio-oils are produced from biomass using three main processing techniques, namely (i) organic solvent liquefaction, which involves the utilization of organic solvents at relatively low temperatures to liquefy biomass; (ii) fast pyrolysis, which involves the liquefaction of biomass at elevated temperatures in the absence of oxygen and a solvent; and (iii) hydrothermal liquefaction, which breaks down biomass in water at elevated pressures and temperatures. It is noteworthy that even though the products from the abovementioned three liquefaction processes are all called bio-oil, the properties of these bio-oils, such as hydroxyl number, chemical composition, and molecular weight, differ greatly. Therefore, the final physical properties of bio-oil-based epoxy resins depend on the processing conditions used for the corresponding bio-oils.
- bio-oil
- epoxy resin
- thermochemical processing
- sustainable material
1. Introduction
2. Bio-Based Epoxy Resins
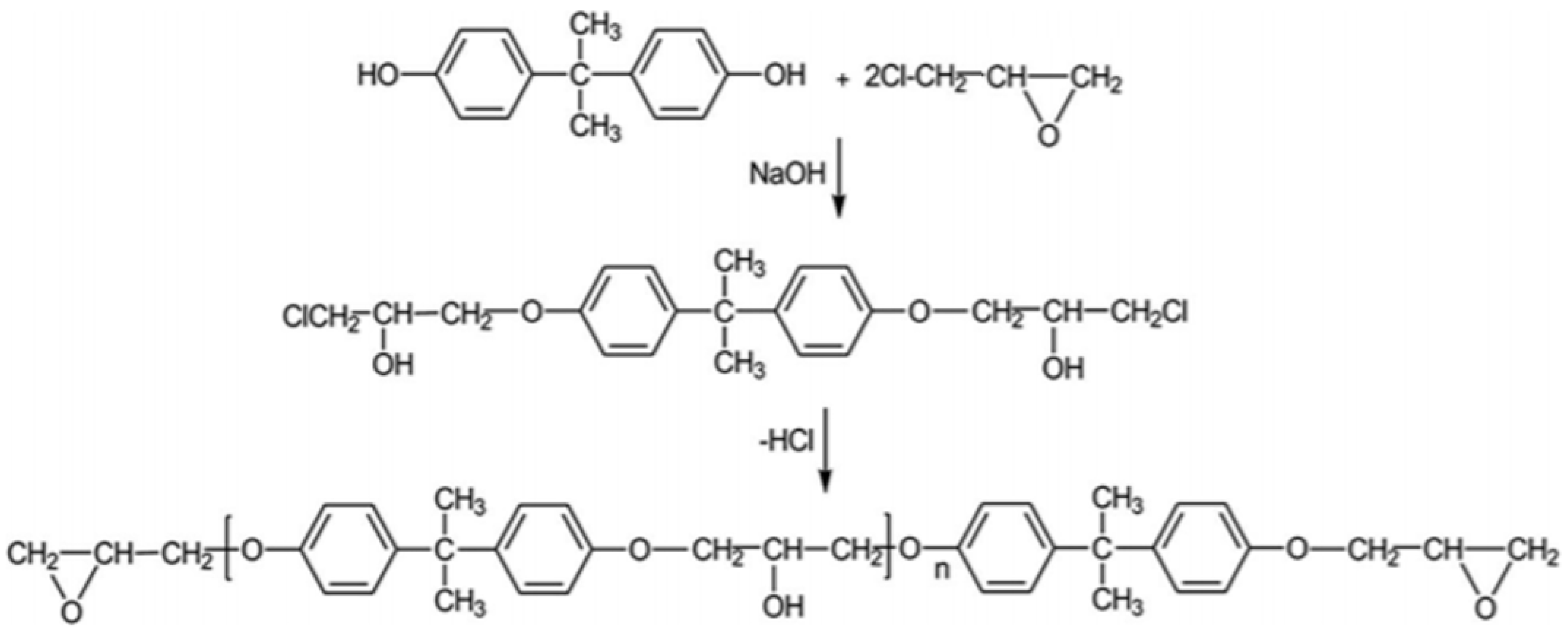
3. Organic Solvent Liquefaction
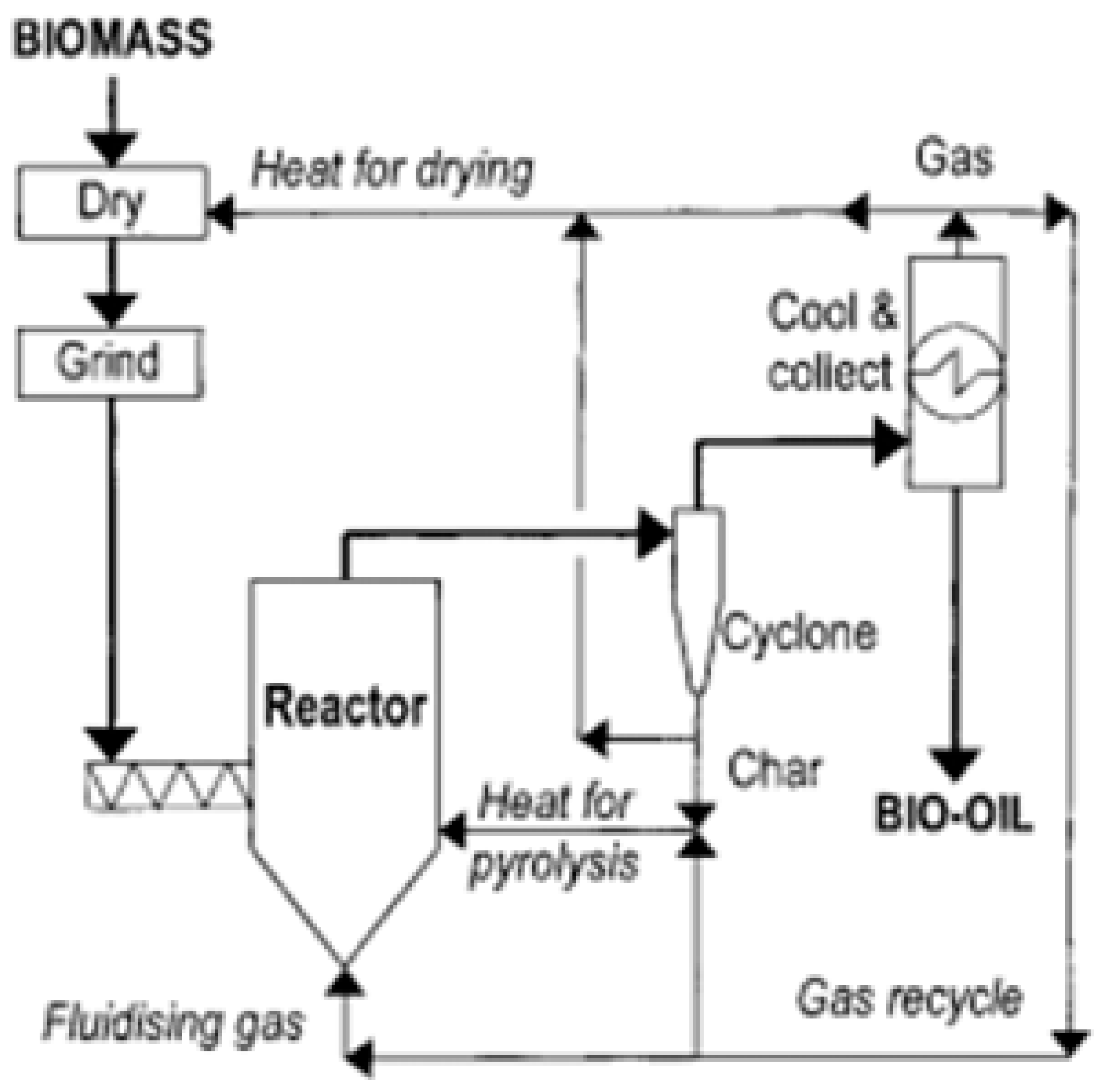
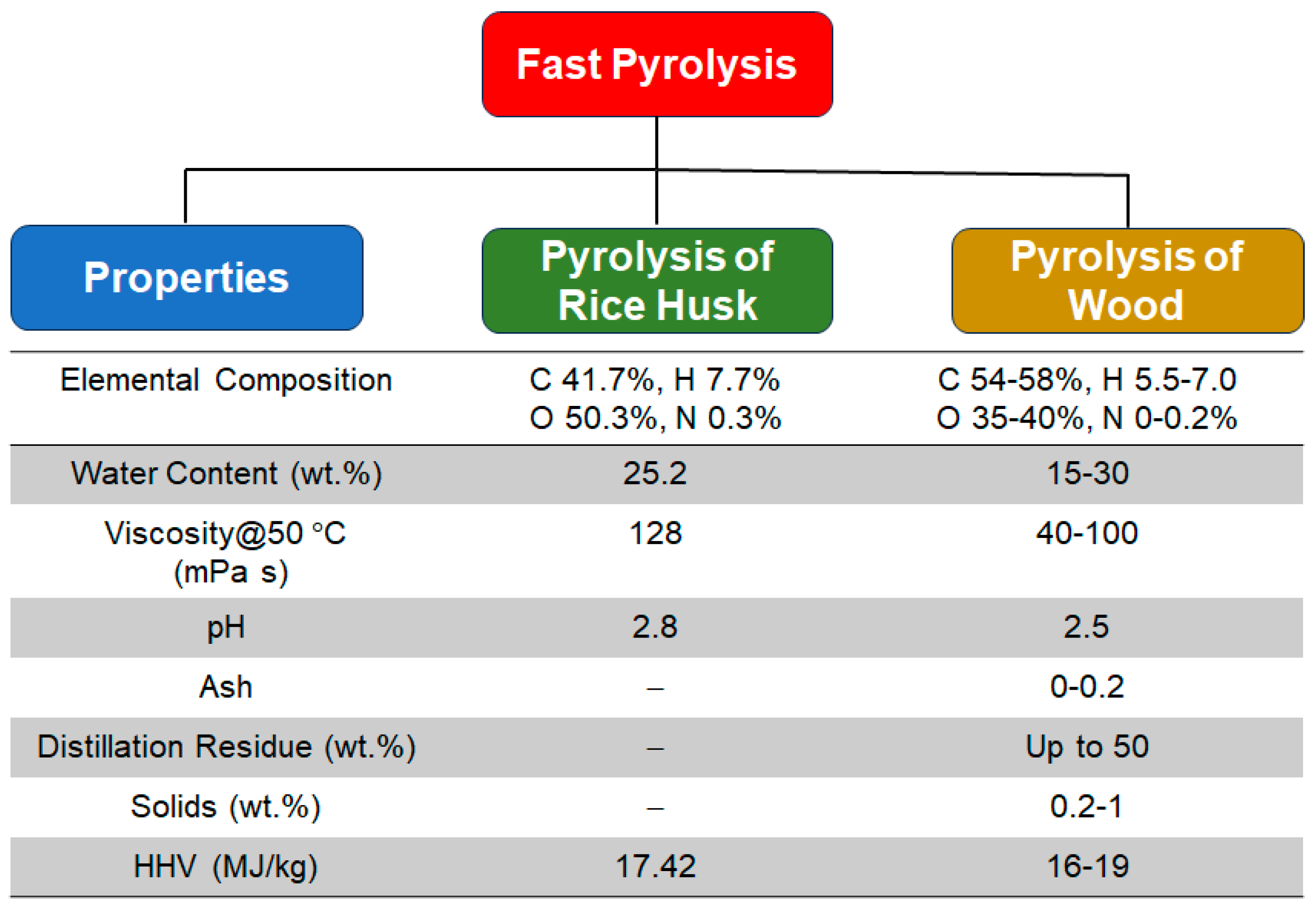
4. Fast Pyrolysis
5. Hydrothermal Liquefaction
| Feedstock | Temperature (°C) | Pressure (MPa) | Time (s) | Yield of Oil (%) | Calorific Value (MJ/Kg) |
Reference |
|---|---|---|---|---|---|---|
| Dairy Manure | 250–380 | 10–34 | – | 50 | – | [29] |
| Sewage Sludge | 300 | 10 | 30–1200 | 48 | 37–39 | [30] |
| Rubbish | 250–340 | 6–8 | 360–7200 | 27.6 | 36 | [31] |
| Sewage Sludge | 150–300 | 8–20 | 0–10,800 | 44.5 | 35.7 | [32] |
| Sewage Sludge | 250–350 | 8–20 | – | 30.7 | 36.4 | [25] |
| Municipal Solid Waste | 260–340 | 13–24 | – | 32 | 46 | [33] |
| Municipal Solid Waste | 295–450 | – | 1200–5400 | 35–63.3 | – | [34] |
| Sewage Sludge | 300–360 | 10–18 | 5–20 | – | 30–35 | [35] |
| Swine Manure | 305 | 10.3 | 80 | 70 | 25–33 | [36] |
This entry is adapted from the peer-reviewed paper 10.3390/jcs7090374
References
- May, C.A. Epoxy Resins: Chemistry and Technology, 2nd ed.; Marcel Dekker: New York, NY, USA, 1988; p. 1.
- Bilyeu, B.; Brostow, W.; Menard, K.P. Epoxy thermosets and their applications I: Chemical structures and applications. J. Mater. Educ. 1999, 21, 281–286.
- Ueki, T.; Nishijima, S.; Izumi, Y. Designing of epoxy resin systems for cryogenic use. Cryogenics 2005, 45, 141–148.
- Karnati, S.R.; Agbo, P.; Zhang, L. Applications of silica nanoparticles in glass/carbon fiber-reinforced epoxy nanocomposite. Compos. Commun. 2020, 17, 32–41.
- Celikbag, Y.; Meadows, S.; Barde, M.; Adhikari, S.; Buschle-Diller, G.; Auad, M.L.; Via, B.K. Synthesis and characterization of bio-oil-based self-curing epoxy resin. Ind. Eng. Chem. Res. 2017, 56, 9389–9400.
- Ellis, B. Introduction to the chemistry, synthesis, manufacture and characterization of epoxy resins. In Chemistry and Technology of Epoxy Resins; Springer Science + Business Media: Dordrecht, The Netherlands, 1993; pp. 1–36.
- Liu, J.-Q.; Bai, C.; Jia, D.-D.; Liu, W.-L.; He, F.-Y.; Liu, Q.-Z.; Yao, J.-S.; Wang, X.-Q.; Wu, Y.-Z. Design and fabrication of a novel superhydrophobic surface based on a copolymer of styrene and bisphenol A diglycidyl ether monoacrylate. RSC Adv. 2014, 4, 18025–18032.
- Petrie, E.M. Epoxy Adhesive Formulations; McGraw-Hill: New York, NY, USA, 2006; pp. 1–26.
- Gandini, A.; Belgacem, M.N.. The State of the Art. In Monomers, Polymers and Composites from Renewable Resources; Belgacem, M.N.; Gandini, A., Eds.; Elsevier: Amsterdam, 2008; pp. 1-16.
- Zhang, Q.; Philips, H.R.; Purchel, A.; Hexum, J.K.; Reineke, T.M.; Sustainable and degradable epoxy resins from trehalose, cyclodextrin, and soybean oil yield tunable mechanical performance and cell adhesion. ACS Sustain. Chem. Eng. 2018, 6, 14967-14978, .
- Ortiz, P.; Vendamme, R.; Eevers, W.; Fully biobased epoxy resins from fatty acids and lignin.. Molecules 2020, 25, 1158, .
- Naik, N.; Shivamurthy, B.; Thimmappa, B.H.S.; Guo, Z.; Bhat, R.; Bio-based epoxies: Mechanical characterization and their applicability in the development of eco-friendly composites.. J. Compos. Sci. 2022, 6, 294, .
- Pan, H. Synthesis of polymers from organic solvent liquefied biomass: A review. Renew. Sustain. Energy Rev. 2011, 15, 3454–3463.
- Demirbaş, A. Mechanisms of liquefaction and pyrolysis reactions of biomass. Energy Convers. Manag. 2000, 41, 633–646.
- Demirbaş, A. Biomass resource facilities and biomass conversion processing for fuels and chemicals. Energy Convers. Manag. 2001, 42, 1357–1378.
- Zhang, L.; Xu, C.C.; Champagne, P. Overview of recent advances in thermo-chemical conversion of biomass. Energy Convers. Manag. 2010, 51, 969–982.
- Lange, J.-P. Lignocellulose liquefaction to biocrude: A tutorial review. ChemSusChem 2018, 11, 997–1014.
- Liang, L.; Mao, Z.; Li, Y.; Wan, C.; Wang, T.; Zhang, L.; Zhang, L. Liquefaction of crop residues for polyol production. BioResources 2006, 1, 248–256.
- Czernik, S.; Bridgwater, A. Overview of applications of biomass fast pyrolysis oil. Energy Fuels 2004, 18, 590–598.
- Bridgwater, A.; Peacocke, G. Fast pyrolysis processes for biomass. Renew. Sustain. Energy Rev. 2000, 4, 1–73.
- Celikbag, Y.; Robinson, T.J.; Via, B.K.; Adhikari, S.; Auad, M.L. Pyrolysis oil substituted epoxy resin: Improved ratio optimization and crosslinking efficiency. J. Appl. Polym. Sci. 2015, 132, 42239.
- Lu, Q.; Li, W.-Z.; Zhu, X.-F. Overview of fuel properties of biomass fast pyrolysis oils. Energy Convers. Manag. 2009, 50, 1376–1383.
- Zhang, L.; Liu, R.; Yin, R.; Mei, Y. Upgrading of bio-oil from biomass fast pyrolysis in China: A review. Renew. Sustain. Energy Rev. 2013, 24, 66–72.
- Elliott, D.C.; Biller, P.; Ross, A.B.; Schmidt, A.J.; Jones, S.B. Hydrothermal liquefaction of biomass: Developments from batch to continuous process. Bioresour. Technol. 2015, 178, 147–156.
- Xiu, S.; Shahbazi, A.; Shirley, V.; Cheng, D. Hydrothermal pyrolysis of swine manure to bio-oil: Effects of operating parameters on products yield and characterization of bio-oil. J. Anal. Appl. Pyrolysis 2010, 88, 73–79.
- Akhtar, J.; Amin, N.A.S. A review on process conditions for optimum bio-oil yield in hydrothermal liquefaction of biomass. Renew. Sustain. Energy Rev. 2011, 15, 1615–1624.
- Cheng, S.; D’cruz, I.; Wang, M.; Leitch, M.; Xu, C. Highly efficient liquefaction of woody biomass in hot-compressed alcohol- water co-solvents. Energy Fuels 2010, 24, 4659–4667.
- Celikbag, Y.; Via, B.K.; Adhikari, S.; Buschle-Diller, G.; Auad, M.L. The effect of ethanol on hydroxyl and carbonyl groups in biopolyol produced by hydrothermal liquefaction of loblolly pine: 31P-NMR and 19F-NMR analysis. Bioresour. Technol. 2016, 214, 37–44.
- Ogi, T.; Yokoyama, S.-Y.; Koguchi, K. Direct liquefaction of wood by alkali and alkaline earth salt in an aqueous phase. Chem. Lett. 1985, 14, 1199–1202.
- Itoh, S.; Suzuki, A.; Nakamura, T.; Yokoyama, S.-Y. Production of heavy oil from sewage sludge by direct thermochemical liquefaction. Desalination 1994, 98, 127–133.
- Minowa, T.; Murakami, M.; Dote, Y.; Ogi, T.; Yokoyama, S.-Y. Oil production from garbage by thermochemical liquefaction. Biomass Bioenergy 1995, 8, 117–120.
- Suzuki, A.; Nakamura, T.; Yokoyama, S.-Y. Effect of operating parameters on thermochemical liquefaction of sewage sludge. J. Chem. Eng. Jpn. 1990, 23, 6–11.
- Gharieb, H.K.; Faramawy, S.; Zaki, N. Liquefaction of cellulosic waste V. Water formation and evaluation of pyrolytic char as a by-product of pyrolysis reaction. Fuel Sci. Technol. Int. 1995, 13, 895–909.
- He, B.J.; Zhang, Y.; Yin, Y.; Funk, T.L.; Riskowski, G.L. Operating temperature and retention time effects on the thermochemical conversion process of swine manure. Trans. ASAE 2000, 43, 1821.
- Balat, M. Mechanisms of thermochemical biomass conversion processes. Part 3: Reactions of liquefaction. Energy Sources Part A 2008, 30, 649–659.
- He, B.J.; Zhang, Y.; Funk, T.L.; Riskowski, G.L.; Yin, Y. Thermochemical conversion of swine manure: An alternative process for waste treatment and renewable energy production. Trans. ASAE 2000, 43, 1827.