Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Oncology
纳米技术是一种新兴且有前途的治疗工具,由于具有吸光特性的纳米材料(NMs)的发展,可能会提高光疗(PT)在抗肿瘤治疗中的有效性。肿瘤靶向PTs,如光热疗法(PTT)和光动力疗法(PDT),将光能转化为热量并产生积聚在肿瘤部位的活性氧(ROS)。ROS水平的增加在致癌和疾病发展过程中诱导氧化应激(OS)。由于铜(Cu)是人体中重要的微量元素,具有局部表面等离子体共振(LSPR)特性,Cu基NMs可以表现出良好的近红外(NIR)吸收和优异的光热性能。与其他治疗方式相比,PTT/PDT可以精确地靶向肿瘤位置以杀死肿瘤细胞。此外,多种治疗方式可以与PTT / PDT结合使用,以使用基于Cu的NM治疗肿瘤。
- nanomaterials (NMs)
- photodynamic therapy (PDT)
- photothermal therapy (PTT)
- copper (Cu)
- Cu-based NMs
- reactive oxygen species (ROS)
1. 简介
癌症是一种复杂的疾病,其特征是各种遗传缺陷。恶性肿瘤发病率的增加及其转移倾向是人类健康的主要威胁,导致死亡率升高[1,2]。传统的肿瘤治疗方式(包括手术、放疗和化疗)可引起各种不良反应,包括放射性损伤、化疗药物的毒副作用或化疗引起的多药耐药,这大大限制了这些方式的治疗效果[3,4,5,6].新兴的肿瘤治疗方式,如温热疗法、免疫疗法和基因疗法,为患者带来了更大的希望[7,8]。然而,据报道,长期使用它们会严重损害免疫系统,甚至导致器官功能障碍[9,10]。发现一种对健康组织毒性低且精度更高的肿瘤治疗方式是一项势在必行且艰巨的挑战。
最近,光疗(PTs),如光热疗法(PTT)和光动力疗法(PDT),被广泛用作有效的抗肿瘤治疗策略[10]。在PTT和PDT中,光热剂(PA)和光敏剂(PSs)在受肿瘤影响的器官中充当外源性能量转换器或吸收剂[11]。这使得可见光或激光能转化为热能,在高温下诱导肿瘤细胞凋亡或坏死,同时产生或不同时产生活性氧[12,13,14,15]。PT与其他基于热的肿瘤治疗技术(包括温热疗法和微波疗法)相比具有特定的优势。其高精度使PT能够以尽可能小的损伤靶向肿瘤[16]。此外,PT是治疗肿瘤的绝佳方法,因为它具有可控性和低毒性特征。与单独作用的PTT或PDT相比,PTT和PDT联合的协同方法可以加速肿瘤细胞死亡并提高治疗效果[17]。PTT和PDT的关键成分是它们的PA和PS,目前主要包括纳米材料(NMs),它们与传统的大分子不同。NM表现出优异的特性,包括高布鲁诺尔-埃米特-泰勒(BET)、导电性、光吸收后的光谱偏移、荧光特性和降解潜力[18]。因此,它们已被稳步纳入癌症研究,推进了肿瘤研究领域[19,20,21]。关于医学治疗,已经采用了纳米级尺寸(直径1-100nm)的材料[22]。这些NM在药物运输中具有广泛的应用,可实现控释机制,增加渗透性,穿越生物屏障,并提高整体生物相容性[23,24,25]。由于其独特的大小和性状,NMs可以显着改变各种治疗过程,并显着提高治疗效果[22]。金属离子基NM,包括金(Au)、银(Ag)、铜(Cu)和其他NM,表现出独特的光学特性,特别是局部表面等离子体共振(LSPR)现象[26,27]。LSPR基于金属离子基NM强烈吸收光子能量的能力,显着增强了它们附近的电场。由于广泛的光诱导效应和产生的分子间相互作用,它们被用于肿瘤靶向PTT/PDT。然而,基于Au或Ag的NM的高成本以及一些PA/PS在体内的不可降解性限制了其在临床治疗中的应用[28]。Cu是一种容易获得的金属,具有独特的生物活性,可用于通过调节各种类型的细胞死亡来消除肿瘤细胞[29]。此外,Cu可以进行类似芬顿的反应,以催化过量的细胞内过氧化氢(H2O2),可诱导细菌和肿瘤细胞死亡[30]。铜的功能和治疗参与如图1所示。Al Kayal等人创造了覆盖有Cu纳米颗粒(NPs)的静电纺丝聚氨酯膜,并对大肠杆菌进行了抗菌测试,发现超过一半的细菌被破坏,显示出其强大的杀菌作用。此外,SARS-CoVer-2抗病毒活性近90%[31]。近年来,Cu基NM因其具有强大的近红外(NIR)吸收和光热能力[32,33]、高BET[34,35]以及用于肿瘤成像等优势而广泛用于PTT和PDT。[36]此外,基于Cu的NMs在肿瘤治疗中具有相当大的优势,因为它们的合成程序简单,并且在温和的低反应条件下具有相对较高的产量[37]。赵等人用铜合成了CCeT NMs(Ce:Chlorin e6,T:TPP-COOH)2−x以硫(S)为核心,在激光照射下光热转换效率(PCE)为10.6%。在非常低的浓度下,温度在5分钟内升高了3.10°C,并随着浓度的增加而继续升高[38],证明了PTT/PDT的治疗优势,并且使用Cu基NMs可以显着提高温度。
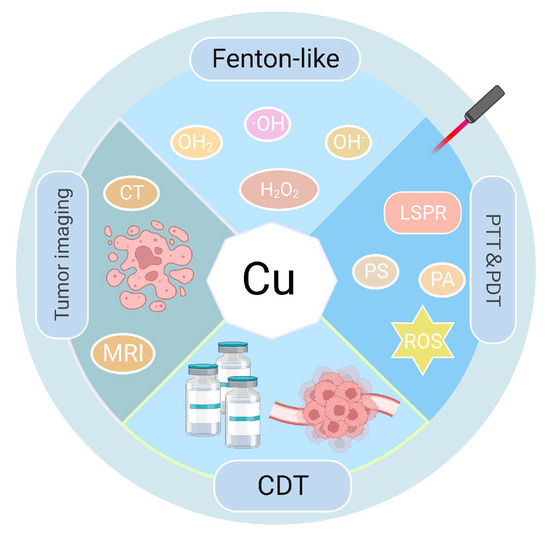
图1.铜的功能和治疗参与。CDT:化学动力学治疗。
2. 氧化铜在PTT和PDT中的应用
在过去的几十年中,各种基于Cu的NM已成为放大PTT/PDT效应的替代手段[119]。研究表明,CuO-NPs可以阻碍胰腺肿瘤的生长,特别是通过靶向肿瘤干细胞[120]。这些NPs可以诱发线粒体功能障碍,归因于Cu2+通过芬顿样反应生成ROS,并通过C-2p和Cu-3d之间的电子跃迁增强PCE[121]。Ma等人开发了CuO@CNSs-DOX(DOX:多柔比星)纳米平台,将PCE从6.7%提高到10.14%。这些平台通过DOX的药物作用和产生·羟基自由基由铜2+ [122,123]。同一组合成多功能MoS2-CuO@BSA/R837(MCBR,BSA:牛血清白蛋白,R837:咪喹莫特,一种toll样受体7(TLR7)激动剂)纳米花。在808 nm激光照射下, 这些纳米花的PCE为24.6%, ·哦生产。此外,R837的加入增加了CRT的表达,触发了ICD,有效地中和了原发性肿瘤,并抑制了转移性肿瘤[124]。利用铜的优势2O的折射率在600nm以上,Yu等人制得的核/壳结构Cu@Cu2O@polymer NP。在 660 nm 激光照射和等效铜浓度下,与 Cu@polymer NP 相比,温度升高 Cu@Cu2NPs O@polymer约为4.2 °C。 这导致相邻组织中的温度升高至少 23 °C,转化为 IC 中的 7 倍浪涌50,相对于先前的研究[125,126]。这些发现强调了Cu@Cu2O@polymer NPs具有足够的光毒性来消灭肿瘤细胞。此外,内源性H2O2LPS内毒素产生的加速Cu离子和ROS的释放,促进癌细胞的根除[33,125]。
3. 铜的应用xSy在 PTT 和 PDT 中
半导体纳米管铜xSy由于其光学和电学特性,在催化和传感领域获得了吸引力。由于其经济实惠、细胞毒性最小和高光稳定性,CuxSyNP有希望作为PAs[127,128]。田等合成了亲水、板状Cu9S5纳米晶体。使用980 nm激光器,铜的PCE9S5达到25.7%。有趣的是,在相同条件下,该PCE超过了合成的Au纳米晶体,约为23.7%。这些纳米晶体在体内迅速(不到10分钟)根除肿瘤细胞,建立Cu9S5纳米晶体作为高效的PA。当激素小鼠在注射后接受后续辐射时,观察到肿块区肿瘤细胞明显坏死和萎缩[129,130]。针对非靶向超小金属NPs作为PTT试剂在确保延长半衰期和令人满意的肿瘤部位聚集方面面临的挑战,Li等人合成了CuS NPs,并用西妥昔单抗(Ab)对其进行修饰以产生CuS-Ab NPs。在暴露于1064nm激光10分钟时,温度从23°C迅速升高至58°C,并始终保持在58°C。 此外,Ab修饰使CuS-Ab NPs在肿瘤靶向和抗血管生成能力方面具有优越性,可确保有效的肿瘤部位积累,而不会同时损害其他组织和器官[131]。PEI等阳离子聚合物可以通过加强阳离子在细胞膜上的静电吸附来实现药物递送[132]。研究表明,用PEI和PS修饰的NM有助于膀胱肿瘤的PDT[133]。基于此,Mu等人制备了HRP@CPC@HA NPs(HRP:辣根过氧化物酶,CPC:CuS-PEI-Ce6,HA:透明质酸)。这些颗粒将HRP掺入空心CuS纳米笼内[134]。在808nm激光辐射下,其PCE约为34.91%。同时,表面结合的Ce6被激活,导致大量的1O2 generation, the levels of which increased alongside temperature [133]. Additionally, HRP catalyzed the breakdown of intracellular H2O2 to oxygen, addressing the oxygen deficit in tumor cells and facilitating simultaneous multimodal tumor eradication in severely hypoxic TME [135]. Due to the increased concentration of endogenous H2S in tumor cells, Wu et al. synthesize monolocking NPs (MLNPs) [136]. When illuminated with 808 nm radiation, these MLNPs combined with H2S and produced ultra-small CuS nanodots. This interaction elevated tumor temperatures, triggering apoptosis in tumor cells. Mel is released from the NPs which causes the COX-2 enzyme to become inactive, amplifying the PTT action and inhibiting inflammation induced by PTT damage. Notably, these NPs were excreted renally [137]. The development of nanomedicine has witnessed diverse manifestations of CuxSy. Hollow structured CuxSy has particularly piqued interest in oncologic therapy, attributed to its drug-loading ability and high PCE and PAI capability [138,139], yet many extant synthesis methods for CuS-based NMs are intricate, time-consuming, and require special reaction equipment. Consequently, there is an evident need for facile, cost-effective methodologies that employ gentle reaction conditions to fabricate CuxSy NMs.
4. Application of Copper Selenides and Copper Telluride in PTT and PDT
Copper selenides, notably Cu2-XSe NMs, present promising applications in imaging and PTT for cancer treatment, attributed to their biocompatibility, excellent NIR light absorption, and increased PCE [140,141].
Du et al. synthesized CuSe/NC-DOX-DNA NPs (NC: nitrogen-doped carbon) via an environmentally friendly and simple method. Under 808 nm laser exposure, these NPs displayed a PCE of 32.9%, surpassing that of Au nanorods which had a PCE of 22.0% [142]. Moreover, the synergistic action of photocatalysis combined with the Fenton-like reaction of CuSe/NC markedly augmented ROS generation [143]. Concurrently, the encapsulated DOX functioned as a chemotherapeutic agent, realizing a threefold enhanced PTT/CDT/PCT tumor treatment method [144]. It is noteworthy that Cu2-XSe has been identified as an effective PA [141]. In another study, He et al. developed ICG@Cu2-X Se-ZIF-8 (ICG: Indocyanine Green, ZIF-8: a metal-organic framework). After 808 nm laser exposure, this compound exhibited a PCE of 15.5%. In the TME, ZIF-8 breaks down and releases Cu1+ and Cu2+, which leads to a Fenton-like reaction, controls the amount of GSH, and produces ROS [145]. Furthermore, selenium has the ability to regulate selenoprotein, allowing it to prevent the production of osteoclasts and tumor cells, resulting in the synergistic PTT/CDT prevention of malignant bone metastasis [146,147]. Moreover, CuSe is an ideal PS and has been demonstrated to biodegrade. Selenium is released during this process and has been shown to lower the risk of liver cancer, lung cancer, and prostate cancer [148,149]. Pun et al. fabricated COF-CuSe NMs (COF: covalent-organic framework); under 808 nm laser irradiation, the PCE was 26.34% after injection in mice. A large quantity of 1O2 was generated under both 650 nm and 808 nm illumination. Consequently, almost all of the HeLa cells died under laser irradiation. The PTT and PDT anti-tumor treatment strategies showed a synergistic potential [150]. At present, CuSe NMs have been poorly investigated for PTT/PDT and the development of multifunctional CuSe NM is still required to improve anti-tumor therapy for PTT/PDT.
Similar to CuSe, CuTe is considered a novel PAs candidate. Li et al. created CuTe NPs with a plasma peak at 900 nm. Under 830 nm laser irradiation, the mortality of 3T3 embryonic fibroblasts increased [151]. However, it was discovered that some cells were already dead before laser irradiation. It has been demonstrated that CuTe NPs are cytotoxic and PAs. Under 1064 nm laser irradiation, bio Cu2−XTe nanosheets synthesized by Li et al. had a PCE of 48.6%. When Cu1+ and Te are released, the nanosheets can produce ·OH and inhibit the GPx and TrxR enzymes for CDT, significantly inhibiting the proliferation of MCF-7 cells [152]. Shen et al. synthesized CM CTNPs@OVA NMs (CM: melanoma B16-OVA membrane, OVA: ovalbumin) with solid CuTe NPs. Under laser irradiation, there was a significant increase in temperature and production of ROS. In addition, B16-OVA cells produced an abundance of ATP and HMGB-1, which effectively stimulated the immune system and enhanced the anti-tumor treatment [153]. CuTe NPs anti-tumor therapy research in PTT/PDT has received less attention and requires further development and investigation.
5. Application of Cu-Based Nanocomposites in PTT and PDT
Most treatments consisting of a single therapy have a negligible impact on tumor treatment. However, a synergistic combination of multiple therapeutic modalities can improve the efficacy of treating malignant tumors [154]. The same applies to NMs. In cancer treatment, simple Cu-based NMs such as CuS may still be lacking. Therefore, it is necessary to load various materials, drugs, or fluorescents onto Cu-based NMs to create a composite material capable of fluorescence, tumor targeting, and therapy in a single NM.
As a result of the discovery of cuproptosis, Cu-based NMs may induce tumor cell death by modulating the concentration of Cu in tumor cells, offering a novel anti-tumor therapeutic modality [111]. Pan et al. produced GOx@[Cu(tz)] NPs. Under 808 nm laser irradiation, NPs entering tumor cells produced H2O2 and ·OH. In the meantime, under the influence of GSH, GOx hydrolyzed and consumed glucose, generating a large amount of H2O2 and OH that produced a ROS-adding effect [155]. Due to the depletion of glucose and GSH, the NPs bind to lipoylated mitochondrial enzymes, resulting in the aggregation of lipoylated DLAT, which induces cuproptosis and effectively inhibits tumor growth (92.4% inhibition rate) [156]. The synthesis and development of NMs that combine multiple antitumor therapeutic modalities is urgently needed. Xia et al. synthesized metal-organic skeleton nanosheets Cu-TCPP(Al)-Pt-FA (TCPP: Tetrakis (4-carboxyphenyl) porphyrin, FA: folic acid) with surface modification by platinum NPs (Pt NPs) and FA in order to solve the problem of poor oxygenation in tumor tissues. Compared to 25.2% tumor cell survival in vitro without laser irradiation, laser irradiation at 638 nm reduced tumor cell survival to 20.7%. Since Cu2+ can react with GSH via a Fenton-like reaction, it depletes intracellular GSH and increases ROS levels [157]. In the meantime, Pt NPs have catalysis activity comparable to a catalase-like reaction that can continuously convert intracellular H2O2 to O2 in order to alleviate hypoxic TME and enhance the therapeutic effect of PDT [158]. ROS concentration was increased synergistically by these two modalities, which stimulated antigen-presenting cells to activate systemic anti-tumor immune responses and increased the infiltration of cytotoxic T lymphocytes (CTLs) at the tumor site for synergistic immune anti-tumor therapy [159,160]. Xu et al. constructed d-Cu-LDH/ICG NPs (LDH: lactatedehydrogenase) in order to alleviate the tumor hypoxia problem and avoid the poor therapeutic effect caused by tumor hypoxia during PDT treatment [161]. Under 808 nm laser irradiation, the PCE was 88.7% and the production of 1O2 increased as the temperature rose. In the meantime, the rising temperature led to the dissolution of the NMs and the reduction in Cu2+ to Cu1+ by GSH, both of which can consume excessive H2O2 and generate OH in tumor cells via a Fenton-like reaction, resulting in CDT and modulation of the TME [162]. In the meantime, it was demonstrated that hormonal mice had significantly less tumor growth. Hematoxylin and eosin (H&E) staining of major organs revealed no obvious inflammation or damage and demonstrated that the effective anti-tumor agent exhibited no significant systemic toxicity. Yang et al. created NSCuCy NPs containing Cu1+ as the core [163]. Cu1+ undergoes a Fenton-like reaction with O2− to produce ·OH [164]. The fluorescence intensity of the HPF was used to detect the ROS concentration; it was discovered that the fluorescence intensity of the HPF increased rapidly in the presence of NSCuCy NPs under 660 nm irradiation. At pH 5.5, the emission intensity of HPF increased nearly 350-fold after 10 min of irradiation, demonstrating a higher ·OH production efficiency than that at pH 7.4 (180-fold) and targeting accumulation in tumor tissues to achieve complete tumor ablation [165]. Kang et al. developed Au@MSN-Cu/PEG/DSF NPs (Au@MSN: mesoporous silica-coated Au nanorods, DSF: disulfiram). The PCE under 808 nm laser irradiation is 56.32%. With an increase in temperature, the Cu-doped SiO2 framework begins to biodegrade. During the conversion of Cu2+ to Cu1+, releasing DSF can chelate with Cu2+ to produce highly cytotoxic bis (diethyldithiocarbamate) Cu (CuET) [166,167]. Cu1+ binds additionally to mitochondrial protein aggregates during the TCA cycle, inducing cuproptosis in tumor cells [111]. Photothermal therapy′s synergistic effect resulted in an 80.1% tumor inhibition rate, effectively killing tumor cells and inhibiting tumor growth [111,168].
Overall, multifunctional nanocomposite materials that integrate imaging, diagnosis, and therapy have shown significant improvements in tumor treatment compared to single-material therapy. These NMs combine PTT/PDT with drug delivery systems, immunotherapy, and chemotherapy, reducing normal medication dosage and adverse reactions while achieving effective anti-tumor treatment in the short term. However, long-term experimental data on NMs are still needed to verify their long-term toxicity, biosafety, and therapeutic effects to ensure they meet desired goals. Further investigations are required for a comprehensive understanding.
6. Application of Cu-MOF in PTT and PDT
Metal-organic frameworks (MOFs) are a novel type of NMs that combine metal ions or clusters with multisite organic ligands. These MOFs have shown great potential in anti-tumor therapy due to their inherent biodegradability, high porosity, structural diversity, and high drug-loading capacity [170,171,172,173].
For instance, Wu et al. synthesized ultrathin Cu-TCPP MOF nanosheets containing both Cu1+ and Cu2+ which enabled imaging, photothermal conversion, and anti-tumor therapy [36,174]. Under 808 nm laser irradiation, the nanosheets showed a PCE of 36.8% and a significant amount of 1O2 was generated in tumor cells. Cu2+ has unpaired 3d electrons and paramagnetic also allowed the nanosheets to be used for T1-weighted MRI. This multifunctional NMOF design holds promise for tumor PTT [59]. Tang et al. designed two metal-organic materials, MOF-1 and MOF-2, based on this premise. MOF-1 is an aluminum (AL)-MOF that does not contain Cu2+, whereas MOF-2 is an AL-Cu mixed-metal MOF[CuL-[AlOH]2n with Cu2+ at its core. Comparing the two MOFs under 650 nm laser irradiation revealed that MOF-2 has a porous physical structure, which makes the Cu2++ in MOF-2 significantly adsorb intracellular GSH and results in a decrease in GSH and an increase in ROS concentration, which enhances the efficacy of PDT [175]. Similar to the chemotherapeutic drug camptothecin (CPT), MOF-2 was able to eradicate tumor cells in an in vivo experiment. The results of a simultaneous examination of major organ tissue slices revealed little toxicity in vivo [176,177]. This MOF structure, which simultaneously decreases intracellular GSH levels and increases intracellular ROS levels, may provide not only a new strategy for intracellular GSH adsorption but also a new method for enhancing PDT.
Due to the higher cavity structure of emerging Cu-NMOFs, they can be loaded with more PSs or PAs to improve PCE and increase the diffusion range of ROS. Their porous structure also allows for chemotherapeutic drug delivery and synergistic effects with other therapies to enhance anti-tumor efficacy. However, the synthesis process of MOF PSs or PAs can be complex and might exhibit batch-to-batch variations, hindering large-scale NMOF preparation. Moreover, the presence of various metal ions necessitates further investigation into the long-term safety, biocompatibility, pharmacokinetics, and immunoreactivity of NMOFs in mammals through systematic animal studies.
This entry is adapted from the peer-reviewed paper 10.3390/pharmaceutics15092293
This entry is offline, you can click here to edit this entry!
