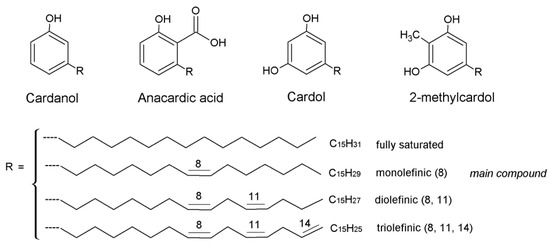
Cashew nut shell liquid (CNSL), obtained as a byproduct of the cashew industry, represents an important natural source of phenolic compounds, with important environmental benefits due to the large availability and low cost of the unique renewable starting material, that can be used as an alternative to synthetic substances in many industrial applications. The peculiarity of the functional groups of CNSL components, such as phenolic hydroxyl, the aromatic ring, acid functionality, and unsaturation(s) in the C15 alkyl side chain, permitted the design of interesting nanostructures. Cardanol (CA), anacardic acid (AA), and cardol (CD), opportunely isolated from CNSL, served as building blocks for generating an amazing class of nanomaterials with chemical, physical, and morphological properties that can be tuned in view of their applications, particularly focused on their bioactive properties.
- CNSL
- cardanol
- anacardic acid
- nanostructures
- green chemistry
- renewable materials
1. Introduction
2. CNSL-Based Nanomaterials
2.1. Cardanol-Based Nanomaterials
Some examples of CA-based metal nanomaterials have been produced via coordination polymerization of the phenolic compound with metal ions or molecules derived from chemical modifications of CA that have shown very versatile properties (optical, photophysical, electrochemical, etc.) and are used to prepare composite nanomaterials with metal oxides. A promising approach, applied to water purification, is heterogeneous photocatalysis using nanostructured semiconductors. In this context, nanocomposite materials based on ZnO nanostructures, impregnated with CA-derived lipophilic porphyrins (H2Pp-metal-free and CuPp-copper porphyrin), provide an alternative technology to efficiently remove toxic substances from water under environmental conditions [16]. In this way, nanomaterials with a diameter of 55 nm were obtained. Moreover, FTIR studies confirm the noncovalent nature of the interactions between CA-porphyrins and ZnO. The photocatalytic activity was investigated via the degradation of rhodamine B (RhB) in an aqueous solution under visible-light irradiation and natural sunlight. Porphyrins are photosensitizing agents for semiconductors; thus, the composite nanomaterials showed better absorption in the visible region than bare ZnO did.
An advanced functional material, like a metal–organic framework (MOF), was produced via the microwave-assisted synthesis of a renewable organic ligand CA and nontoxic endogenous cation Mn(II) bivalent salt [17]. The synthesis was carried out by a “solvent-free in situ” approach, and FTIR spectroscopy was used to confirm the structure and verify the curing of the material (MnIIMicCol). The morphological characterization of the nanomaterial investigated through XRD, optical microscopy, SEM, and TEM showed an amorphous and layered morphology and mesoporous (pore diameter of 8.0286 nm) behavior. The thermal behavior measured by the TGA/DTG/DSC techniques confirmed a high inherent thermal stability. Antibacterial activity was tested against Gram-negative (E. coli and K. pneumoniae) and Gram-positive (B. subtilis and S. aureus) bacterial strains, revealing two bactericidal mechanisms: (i) damage to the bacterial cell membrane and (ii) the production of reactive oxygen species (ROS), which may cause oxidative stress on bacteria cells and damage to both DNA and RNA. The excellent chemical–physical properties and the moderate antibacterial activity make this nanostructured MOF usable in thermally stable (up to 230–250 °C) antimicrobial coating materials.
In newer research, sheet-like nano-biocomposites, based on CA thermosetting resin [20], were obtained. The inclusion of cellulose nanofibrils and nanoplatelets of expanded graphite improved in synergy the flammability, thermal, mechanical, and water absorption properties of nano-biocomposites, required for applicability in coating systems and automotive applications, where weight reduction and a reduction in VOCs in the environment are of great importance. The assembly of this composite material allowed for improving the dispersion of nanofiller cellulose nanofibrils with a high specific surface area and a high percentage of exposed atoms on their surfaces. Furthermore, the CA resin showed a stabilizing effect from the expanded graphite in the nanosheets.
The versatile behavior of CA and its derivative small molecules were found to be attractive in functional soft nanomaterials research to generate self-assembled morphologies down to 100 nm dimensions, such as nanotubes, nanofibers, gels, surfactants, and liquid crystals [21,22]. Gels are systems delicately balanced between molecules’ precipitation and solubilization in a solvent that, self-assembling through noncovalent interactions, form a fibrous network that traps the solvent through capillary forces and resists the flow of the medium. Pyrene-coupled coumarin derivatives with varying hydrophobic chains have been synthesized via aldol condensation, starting from CA-aldehyde, obtained through electrophilic aromatic substitution reactions [23]. The formation of transparent fluorescent organogels occurred via supramolecular self-assembly through the π–π stacking of pyrene units and hydrogen bonding. In particular, absorbance and emission and 1HNMR studies showed that hydrogen bonding between carbonyl groups of coumarin coupled pyrene with the hydroxyl group of a solvent, and π–π stacking interactions have driven the self-aggregation and gel formation processes. The presence of saturated and unsaturated hydrophobic tails affects the gelation efficiency tested in different solvents, strongly influencing the optical properties of π-conjugated derivatives. From these results, self-assembly nanoflakes were derived, and in vitro fluorescence imaging reveals that these compounds inhibit the proliferation of PC3 prostate cancer cells, making them potentially applicable in the cell imaging field. Another study reported the synthesis of coumarin-tris-based amphiphiles, which in turn have been derived from CA [24]. This small amphiphilic system showed the ability to form a stable supramolecular hydrogel sensitive to external stimuli such as pH or the presence of the biologically important Fe3+ ion. Optical microscopy and high-resolution transmission electron microscopy (HRTEM) investigations revealed a reversible morphological transition from self-assembled gels at neutral and basic pH levels to vesicles and nanotubes when pH is acidic. 1HNMR and XRD studies suggested that the π–π stacking interactions and hydrogen bonding were the driving forces for the gelation process.
2.2. Anacardic Acid-Based Nanomaterials
This entry is adapted from the peer-reviewed paper 10.3390/nano13172486
