A good pairing is based on the complementary role of wine on the four critical factors of food: components, textures, flavors, and colors. Everyday wine and food examples of food preparation with wine or vine products, such as grapes or vine leaves, exemplify numerous similarities and contrasting characteristics in those four parameters, which we can find in an enjoyable meal. The knowledge of the internal anatomy and composition of both grape berries, which includes the skin, the mesocarp, and the endocarp, and vine leaf, which consists of the vascular system, parenchymatous mesophyll, and epidermis, is essential to know how to develop new, tastier and healthier dishes.
- alcoholic drinks
- grapes
- vine leaves
- cooking
1. Grape Berries’ Composition
|
Compound |
Grape Variety |
|||||||
|---|---|---|---|---|---|---|---|---|
|
Red Grape Varieties |
White Grape Varieties |
|||||||
|
Berry |
Skin |
Seed |
Reference |
Berry |
Skin |
Seed |
Reference |
|
|
Sucrose (g/L) |
1.7–4.4 |
[3] |
1.6–3.7 |
[3] |
||||
|
Glucose (g/L) |
81.6–93.7 |
[3] |
77.3–93.9 |
[3] |
||||
|
93.6–107.0 |
[4] |
86.4–104.9 |
[4] |
|||||
|
Fructose (g/L) |
79.1–95.9 |
[3] |
80.9–102.1 |
[3] |
||||
|
80.4–94.1 |
[4] |
73.1–88.5 |
[4] |
|||||
|
Total sugar (g/L) |
181.2–198.9 |
[4] |
163.5–197.8 |
[4] |
||||
|
Tartaric acid (g/L) |
4.3–4.9 |
[3] |
5.0–6.2 |
[3] |
||||
|
3.8–5.2 |
[4] |
4.8–5.0 |
[4] |
|||||
|
Malic acid (g/L) |
2.2–2.9 |
[3] |
1.5–1.9 |
[3] |
||||
|
3.0–3.1 |
[4] |
2.8–3.6 |
[4] |
|||||
|
Citric acid (g/L) |
0.3–0.4 |
[4] |
0.2–0.3 |
[4] |
||||
|
(+)-Catechin (mg/kg FW) |
197–515 |
[5] |
70.3–659.1 |
[5] |
||||
|
39.2–533 |
[6] |
61.0–481 |
[6] |
|||||
|
(−)-Epicatechin (mg/kg FW) |
32.4–231.2 |
[5] |
67.1–172.7 |
[5] |
||||
|
63.5–467.3 |
[6] |
84.3–144 |
[6] |
|||||
|
Procyanidin B1 |
13.39–15.95 a |
8.4–22 c |
74–170 e |
2.4–3.3 b |
12–48 c |
200–620 d |
||
|
4.2–7.2 b |
0.09–0.26 d |
5.3–5.92 d |
1.1–1.9 d |
0.2–2.0 d |
[11] |
|||
|
1.38–3.58 d |
[9] |
|||||||
|
Procyanidin B2 |
5.74–10.39 a |
0.75–2.2 c |
21–41 e |
0.3–2.0 b |
0.06–0.2 d |
15–33 e |
||
|
1.9–4.3 b |
0.011–0.020 d |
24–30 d |
3.8–5.6 d |
[11] |
||||
|
1.75–5.23 d |
[9] |
|||||||
|
Procyanidin B3 |
16–39 c |
43–64 e |
[8] |
21–37 c |
39–59 e |
[8] |
||
|
0.013 d |
5–7.3 d |
0.2–0.3 d |
0.9–1.8 d |
[11] |
||||
|
0.45–0.96 d |
[9] |
|||||||
|
Procyanidin B4 |
33–80 e |
[8] |
0.04 d |
53–95 e |
||||
|
8–12.24 d |
[9] |
2.5–2.9 d |
[11] |
|||||
|
0.65–2.57 d |
[8] |
|||||||
|
Delphinidin-3-glucoside (mg/kg FW) |
9.2–13.9 |
[4] |
||||||
|
Cyanidin-3-glucoside (mg/kg FW) |
3.3–9.6 |
[4] |
||||||
|
Petunidin-3-glucoside (mg/kg FW) |
7.4–13.6 |
[4] |
||||||
|
Peonidin-3-glucoside (mg/kg FW) |
28.0–38.7 |
[4] |
||||||
|
Malvidin-3-glucoside (mg/kg FW) |
35.0–76.6 |
[4] |
||||||
|
Hexanal (µg/kg equiv of 2-octanol) |
2104–2317 |
[4] |
2193–2642 |
[4] |
||||
|
Linalool(µg/kg equiv of 2-octanol) |
21–418 |
[4] |
27–324 |
[4] |
||||
|
Geraniol (µg/kg equiv of 2-octano) |
48 |
[4] |
26 |
[4] |
||||
|
2-Phenylethanol (µg/kg equiv of 2-octane) |
19 |
[4] |
10 |
[4] |
||||
|
Isobutyl-methoxypyrazine (ng/L) |
6.5–9.9 |
[12] |
||||||
|
Ethylmethoxypyrazine (ng/L) |
2.4–47.1 |
[12] |
||||||
2. Grape Berries’ Anatomy and Histology
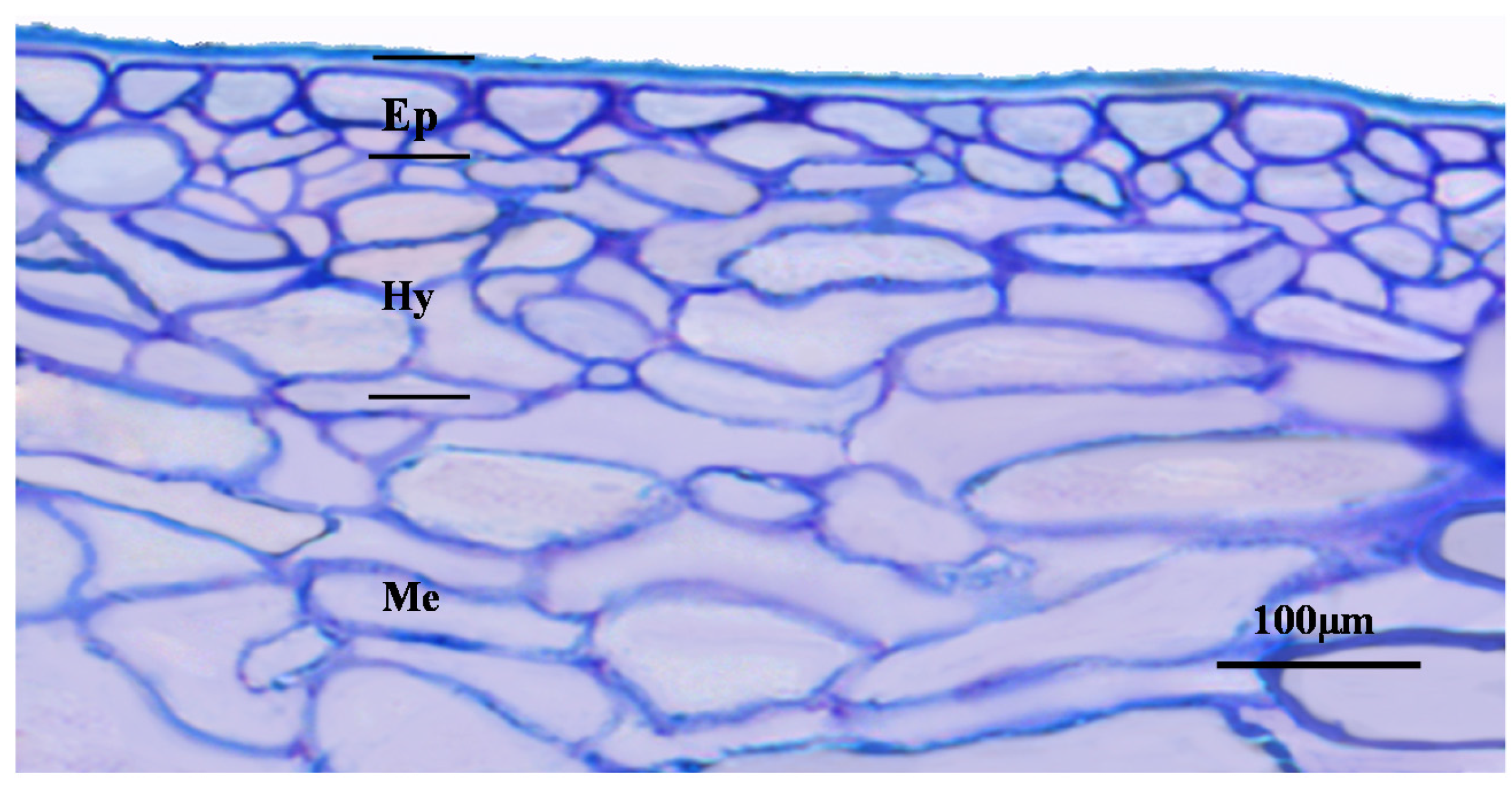
3. Cooking with Grapes, Sensory Aspects
This entry is adapted from the peer-reviewed paper 10.3390/beverages3030042
References
- Liu, H.F.; Wu, B.H.; Fan, P.G.; Li, S.H.; Li, L.S. Sugar and acid concentrations in 98 grape cultivars analyzed by principal component analysis. J. Sci. Food Agric. 2006, 86, 1526–1536.
- Robinson, S.P.; Davies, C. Molecular biology of grape berry ripening. Aust. J. Grape Wine Res. 2000, 6, 175–188.
- Sabir, A.; Kafkas, E.; Tangolar, S. Distribution of major sugars, acids and total phenols in juice of five grapevine (Vitis spp.) cultivars at different stages of berry development. Span. J. Agric. Res. 2010, 8, 425–433.
- Aubert, C.; Chalot, G. Chemical composition, bioactive compounds, and volatiles of six table grape varieties (Vitis vinifera L.). Food Chem. 2017.
- Balìk, J.; Kyseláková, M.; Vrchotová, N.; Triska, J.; Kumsta, M.; Veverka, J.; HÍc, P.; Totusek, J.; Lefnerová, D. Relations between polyphenols content and antioxidant activity in vine grapes and leaves. Czech J. Food Sci. 2008, 26, S25–S32.
- Mikes, O.; Vrchotová, N.; Triska, J.; Kyseláková, M.; Smidrkal, J. Distribution of major polyphenolic compounds in vine grapes of different cultivars growing in South Moravian vineyards. Czech J. Food Sci. 2008, 26, 182–189.
- Pérez-Trujillo, J.P.; Hernández, Z.; López-Bellido, F.J.; Hermosín-Guiteérrez, L. Characteristic phenolic composition of single-cultivar red wines of the Canary Islands (Spain). J. Agric. Food Chem. 2011, 59, 6150–6154.
- Montalegre, R.R.; Peces, R.R.; Vozmediano, J.L.S.; Gascueña, J.M.; Romero, E.G. Phenolic compunds in skins and seeds of ten grape Vitis vinifera varieties grown in a warm climate. J. Food Compos. Anal. 2006, 19, 687–693.
- Mateus, N.; Marques, S.; Gonçalves, A.C.; Machado, J.M.; De Freitas, V.A.P. Proanthocyanidin composition of red Vitis vinifera varieties from the Douro Valley during ripening: Influence of cultivation altitude. Am. J. Enol. Vitic. 2001, 52, 115–121.
- De Freitas, V.A.P.; Glories, Y.; Monique, A. Developmental changes of procyanidins in grapes of red Vitis vinifera varieties and their composition in respective wine. Am. J. Enol. Vitic. 2000, 51, 397–403.
- De Freitas, V.A.P.; Glories, Y. Concentration and compositional changes of procyanidines in grape seeds and skin of white Vitis vinifera varieties. J. Sci. Food Agric. 1999, 79, 1601–1606.
- Allen, M.S.; Lacey, M.J. Methoxypyrazine grape flavor components: Influence of grape cultivarescultivars. In Proceedings of the Eighth Australian Wine Industry Technical Conference, Melbourne, Australia, 25–29 October 1992; Stockley, C.S., Johnstone, R.S., Leske, P.A., Lee, T.H., Eds.; Winetitles: Adelaide, Australia, 1992; p. 1995.
- Kanellis, A.K.; Roubelakis-Angelakis, K.A. Grape. In Biochemistry of Fruit Ripening; Seymour, G., Taylor, J., Tucker, G., Eds.; Chapman &Hall: London, UK, 1993; pp. 189–234.
- Lamikanra, O.; Inyang, I.D.; Leong, S. Distribution and Effect of Grape Maturity on Organic Acid Content of Red Muscadine Grapes. J. Agric. Food Chem. 1995, 43, 3026–3028.
- Kliewer, W.M. Sugars and Organic Acids of Vitis vinifera. Plant Physiol. 1966, 41, 923–931.
- Wermelinger, B. Nitrogen Dynamics in Grapevine. Physiology and Modeling. In Proceedings of the International Symposium on Nitrogen in Grapes and Wine, Seatle, WA, USA, 18–19 June 1991; Rantz, J.M., Ed.; American Society for Enology and Viticulture: Davis, CA, USA, 1991; pp. 23–31.
- Huang, Z.; Ough, C.S. Amino acid profiles of commercial grape juices and wines. Am. J. Enol. Vitic. 1991, 42, 261–267.
- Hernandez-Orte, P.; Ibraz, M.J.; Cacho, J.; Ferriera, V. Amino acid determination in grape juices and wines by HPLC using a modification of the 6-aminoquinolyl-nhydroxysuccinimidyl carbamate (AQC) method. Chromatographia 2003, 58, 29–35.
- Lorrain, B.; Chira, K.; Teissedre, P.-L. Phenolic composition of Merlot and Cabernet–Sauvignon grapes from Bordeaux vineyard for the 2009-vintage: Comparison to 2006, 2007 and 2008 vintages. Food Chem. 2011, 126, 1991–1999.
- Jordão, A.M.; Ricardo-da-Silva, J.M.; Laureano, O. Evolution of catechins and oligomeric procyanidins during grape maturation of Castelão Francês and Touriga Francesa. Am. J. Enol. Vitic. 2001, 53, 231–234.
- Pozo-Bayon, M.A.; Hernandez, M.T.; Martin-Alvarez, P.J.; Polo, M.C. Study of low molecular weight phenolic compounds during the aging of sparkling wines manufactured with red and white grape varieties. J. Agric. Food Chem. 2003, 51, 2089–2095.
- Vanhoenacker, G.; De Villiers, A.; Lazou, K.; Keukeleire, D.; Sandra, P. Comparison of high performance liquid chromatography—Mass spectroscopy and capillary electrophoresis—Mass spectroscopy for the analysis of phenolic compounds in diethyl ether extracts of red wines. Chromatographia 2001, 54, 309–315.
- Hernandez-Jimenez, A.; Gomez-Plaza, E.; Martinez-Cutillas, A.; Kennedy, J.A. Grape skin and seed proanthocyanidins from Monastrell x Syrah grapes. J. Agric. Food Chem. 2009, 57, 10798–10803.
- Cheynier, V.; Rigaud, J. HPLC separation and characterization of flavonols in the skins of Vitis Vinifera var. Cinsault. Am. J. Enol. Vitic. 1986, 37, 248–252.
- Pastrana-Bonilla, E.; Akoh, C.C.; Sellappan, S.; Krewer, G. Phenolic content and antioxidant capacity of Muscadine grapes. J. Agric. Food Chem. 2003, 51, 5497–5503.
- Alcalde-Eon, C.; Escribano-Bailon, M.T.; Santos-Buelga, C.; Rivas Gonzalo, J.C. Changes in the detailed pigment composition of red wine maturity and ageing—A comprehensive study. Anal. Chem. Acta 2006, 563, 238–254.
- Vidal, S.; Hayasaka, Y.; Meudec, E.; Cheynier, V.; Skouroumounis, G. Fractionation of grape anthocyanin classes using multilayer coil countercurrent chromatography with step gradient elution. J. Agric. Food Chem. 2004, 52, 713–719.
- De Pascual-Teresea, S.; Rivas-Gonzalo, J.C.; Santos-Buelga, C. Prodelphinidins and related flavanols in wine. Int. J. Food Sci. Technol. 2000, 35, 33–40.
- Cosme, F.; Ricardo-da-Silva, J.M.; Laureano, O. Tannic profiles of Vitis vinifera L. cv. red grapes growing in Lisbon and from their monovarietal wines. Food Chem. 2009, 112, 197–204.
- Mane, C.; Souquet, J.M.; Olle, D.; Verries, C.; Veran, F.; Mazerolles, G.; Cheynier, V.; Fulcrand, H. Optimization of simultaneous flavanol, phenolic acid, and anthocyanin extraction from grapes using an experimental design: Application to the characterization of Champagne grape varieties. J. Agric. Food Chem. 2007, 55, 7224–7233.
- Williams, P.J.; Strauss, C.R.; Wilson, B. Hydroxylated linalool derivatives of volatile monoterpenes of Muscat grapes. J. Agric. Food Chem. 1980, 28, 766–771.
- Torregrosa, L.; Vialet, S.; Adivèze, A.; Iocco-Corena, P.; Thomas, M.R. Grapevine (Vitis vinifera L.). Methods Mol. Biol. 2015, 1224, 177–194.
- Keller, M. The Science of Grapevines: Anatomy and Physiology, 2nd ed.; Academic Press: Tokyo, Japan, 2015; pp. 1–57.
- Fontes, N.; Gerós, H.; Delrot, S. Grape Berry Vacuole: A Complex and Heterogeneous Membrane System Specialized in the Accumulation of Solutes. Am. J. Enol. Vitic. 2011, 62, 270–278.
- Conde, C.; Silva, P.; Fontes, N.; Dias, A.C.P.; Tavares, R.M.; Sousa, M.J.; Agasse, A.; Delrot, S.; Gerós, H. Biochemical changes throughout grape berry development and fruit and wine quality. Food 2007, 1, 1–22.
- Hardie, W.J.; O’Brien, T.P.; Jaudzems, V.G. Morphology, anatomy and development of the pericarp after anthesis in grape, Vitis Vinifera L. Aust. J. Grape Wine Res. 1996, 2, 97–142.
- Ribéreau-Gayon, P.; Dubourdieu, D.; Donèche, B.; Lonvaud, A. The Microbiology of Wine and Vinifications, 1st ed.; Handbook of Enology; Wiley: Chichester, UK, 2000; Volume 1.
- Wilson, B.; Strauss, C.R.; Williams, P.J. The distribution of free and glycosidically-bound monoterpenes among skin, juice and pulp fractions of some white grape varieties. Am. J. Enol. Vitic. 1986, 37, 107–111.
- Schlosser, J.N.; Olsson, M.; Weis, K.; Reid, F.; Peng, S.; Lund, P.B. Cellular expansion and gene expression in the developing grape (Vitis vinifera L.). Protoplasma 2008, 232, 255–265.
- Roth, I. Fruits of the Angiosperms; Gebrüder Bornträger: Berlin, Germany, 1977.
- Cho, S.-M.; Kim, J.-H.; Park, H.-J.; Chun, H.-K. Manufacturing of Korean traditional rice wine by using Gardenia jasminoides. Korean J. Microbiol. Biotechnol. 2009, 37, 413–415.
- Vilela, A.; Cosme, F. Drink Red: Phenolic Composition of Red Fruit Juices and Their Sensorial Acceptance. Beverages 2016, 2, 29.
- Falcão, A.P.; Chaves, E.S.; Falcão, L.D.; Gauche, C.; Barreto, P.L.M.; Bordignon-Luiz, M.T. Rheological behavior and color stability of anthocyanins from Merlot (Vitis vinifera L.) and Bordô (Vitis labrusca L.) grapes in a jam model system. Ciênc. Tecnol. Aliment. 2009, 29, 857–862.
- Pop, I.M.; Pascariu, S.M.; Simeanu, D. The grape pomace influence on the broiler chickens growing rate. Lucrari Stiintifice Seria Zootehnie 2015, 64, 34–39.
