Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Hyaluronic acid (HA) is as naturally occurring glycosaminoglycan composed of repeating disaccharide units consisting of glucuronic acids and N-acetylglucosamine, resulting in different molecular weights. HA plays a crucial pathophysiological role in rheumatic diseases, especially concerning joint health and function.
- hyaluronic acid
- rheumatology
- osteoarthritis
- rheumatoid arthritis
- joint lubrication
1. Introduction
Hyaluronic acid (HA) plays an important role in a wide range of medical physiological and pathological conditions: Notably, it finds application in dermatology, ophthalmology, cosmetic medicine, and rheumatology [1][2][3]. Its significance extends to wound healing, granulation, and cell migration [4]. However, the efficacy of HA in rheumatology remains a subject of controversy at times [5][6].
HA is as naturally occurring glycosaminoglycan composed of repeating disaccharide units consisting of glucuronic acids and N-acetylglucosamine (Figure 1), resulting in different molecular weights [7][8][9]. This structural variability imparts diverse functional implications in both physiological and pathological contexts [10]. HA is commercially produced through extraction from animal tissues, such as chicken combs, and from Streptococci bacteria [11].
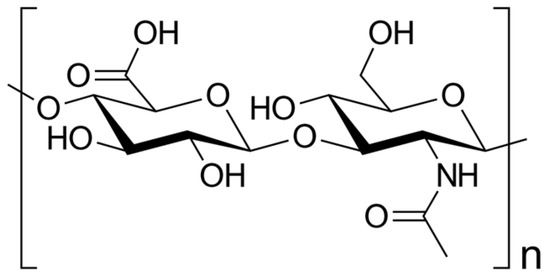
Figure 1. Skeletal formula of hyaluronan—a polymer consisting of D-glucuronic acid and N-acetyl-D-glucosamine linked via alternating β-(1→4) and β-(1→3) glycosidic bonds [7].
Functionally, HA demonstrates remarkable water-binding capacity, rendering it an essential constituent of the extracellular matrix (ECM) [12]. Its elongated, unbranched chains create a gel-like network, imparting hydration and lubrication to crucial tissues like the skin, cartilage, and synovial fluid [13][14][15].
In the field of rheumatology, HA has garnered substantial attention, owing to its pivotal involvement in joint health and its relevance to diseases like osteoarthritis (OA) and rheumatoid arthritis (RA) [16][17].
OA is a degenerative joint disorder characterized by the gradual deterioration of articular cartilage, resulting in pain, stiffness, and diminished joint function [18]. There is no cure for OA, so doctors usually treat OA symptoms with a combination of therapies [18]. HA serves as a lubricant and shock absorber within the synovial fluid, facilitating smooth joint movements [19]. However, in OA, the concentration and quality of HA decrease, compromising its protective and viscoelastic properties due to heightened degradation and decreased synthesis. Consequently, this leads to impaired cartilage function and joint degeneration [20]. As a therapeutic approach, supplementation with exogenous HA has emerged to alleviate symptoms and enhance joint function in OA patients [21]. By restoring synovial fluid viscosity and promoting cartilage repair, HA aids in improving joint mobility and reducing pain [16].
On the other hand, RA is an autoimmune and inflammatory disease characterized by the immune system mistakenly attacking healthy cells, resulting in inflammation, particularly in the joints, leading to painful swelling [22]. RA can be effectively treated and managed with medication(s) and self-management strategies [22]. In RA, the level of HA in the synovial fluid is significantly diminished, causing reduced lubrication and increased inflammation and pain [23].
Numerous studies have explored the potential therapeutic benefits of exogenous HA supplementation in rheumatic diseases. Through intra-articular injections, HA has shown promise in improving joint mobility, reducing pain, and promoting cartilage repair by restoring synovial fluid viscosity [24].
Furthermore, HA has demonstrated immunomodulatory effects, including the suppression of pro-inflammatory cytokines and the promotion of anti-inflammatory cytokine production [25]. This suggests that HA may also hold promise in the treatment of immune-mediated rheumatic diseases [26].
Moreover, researchers have explored HA’s potential applications in drug delivery systems and tissue engineering due to its biocompatibility and biodegradability [27].
However, the use of HA therapy in rheumatology remains a topic of controversy, with conflicting evidence regarding its efficacy and safety [5][28].
In summary, HA represents a promising therapeutic option in the field of rheumatology due to its potential to enhance joint function and alleviate inflammation and pain [29]. Nevertheless, further investigation is required to fully elucidate its therapeutic potential in rheumatic diseases.
2. Hyaluronic Acid: Structure, Function, and Biochemistry
HA plays a crucial role in diverse cellular and tissue processes, encompassing hydration, lubrication, tissue repair, regulation of inflammation, and cell signaling [14]. It is naturally synthesized by various cell types, predominantly fibroblasts, chondrocytes, and synoviocytes [30]. The biosynthesis of HA takes place in the plasma membrane through the coordinated activity of specific enzymes, including hyaluronan synthases [30].
2.1. Molecular Structure of Hyaluronic Acid
Hyaluronan synthases catalyze the addition of glucuronic acid and N-acetylglucosamine, leading to the formation of the repeating disaccharide units that constitute HA [31]. The molecular weight of HA displays highly variability, ranging from several hundred kilo Da to millions of kilo Da, exerting a direct impact on its functional properties [32]: Notably, higher-molecular-weight HA exhibits increased viscosity [33], thereby affecting its flow and lubrication capability, i.e., in joints [34]. As a result, high-molecular-weight HA provides superior lubrication and cushioning effects [35].
2.2. Biosynthesis and Degradation of Hyaluronic Acid
HA turnover in tissues is intricately regulated by a delicate balance between biosynthesis and degradation processes [36]. The degradation of HA primarily involves the action of enzymes known as hyaluronidases, which cleave HA into smaller fragments [37]. Hyaluronidase enzymes are categorized into several families, including HYAL1, HYAL2, and PH-20 [38], and they play a pivotal role in maintaining the appropriate concentration and size distribution of HA within tissues [39]. Furthermore, the degradation of HA can be modulated by reactive oxygen species, matrix metalloproteinases (MMPs), and other factors present in the extracellular environment [40].
2.3. Physiological Functions and Distribution in Tissues of Hyaluronic Acid
HA plays a critical role in tissue repair and remodeling processes within the human body [41]. It participates in various stages of wound healing, encompassing inflammation, cell migration, proliferation, and ECM remodeling [42]. As a scaffolding molecule, HA provides essential structural support and aids in cell migration during tissue repair [43] (Table 1).
Table 1. Physiological functions of hyaluronic acid (HA) in rheumatology.
| Function | Mechanisms | References |
|---|---|---|
| Lubrication of joints | HA provides lubrication and viscoelastic properties to synovial fluid, reducing friction between joint surfaces and enhancing joint mobility. | [44] |
| Chondroprotection | HA exhibits chondroprotective effects by promoting cartilage matrix synthesis, reducing matrix degradation, and inhibiting the activity of proteolytic enzymes | [45][46] |
| Anti-inflammatory activity | HA can modulate inflammation by reducing the expression of pro-inflammatory cytokines and enzymes, inhibiting leukocyte migration, and suppressing immune responses. | [47][48] |
| Tissue repair and remodeling | HA plays a role in tissue repair and remodeling processes by promoting cell migration, angiogenesis, and extracellular matrix remodeling | [44][49] |
| Viscoelasticity | HA contributes to the viscoelastic properties of connective tissues, maintaining tissue integrity, elasticity, and shock-absorbing capabilities. | [48][50] |
Selected studies on the physiological role of HA in rheumatology. This is not an exhaustive list, and further research and clinical trials have been conducted in this field. For more detailed information, it is recommended to refer to the referenced papers.
During the inflammatory phase of wound healing, HA is involved in the recruitment and activation of immune cells, such as macrophages and neutrophils [51]. HA fragments generated during tissue injury can function as damage-associated molecular patterns and trigger immune responses [51]. Moreover, HA promotes the infiltration of immune cells into the wound site, facilitating the removal of debris and pathogens [52]. In the subsequent proliferative phase, HA contributes to cell migration and proliferation [53]. It forms a provisional matrix that guides cell movement and stimulates cell proliferation [53]. HA receptors, such as CD44 and RHAMM, mediate cell adhesion, migration, and signal transduction, enabling cells to migrate into the wound area and contribute to tissue repair [54]. Furthermore, HA plays a regulatory role in ECM remodeling during tissue repair [55]. It interacts with other components, such as fibronectin and collagen, promoting their assembly and organization [56]. HA also influences the activity of enzymes involved in ECM remodeling, such as MMPs and tissue inhibitors of MMPs, which are essential for matrix turnover and remodeling [57].
Beyond its involvement in tissue repair, HA also plays a role in tissue remodeling processes, such as embryonic development, organ morphogenesis, and angiogenesis [58]. HA provides a structural framework for cell migration and tissue organization during these processes [59]. It regulates cell behavior, including cell differentiation, proliferation, and survival, through interactions with specific receptors and signaling pathways [60].
Overall, HA exhibits a multifaceted role in tissue repair and remodeling, contributing to inflammation resolution, cell migration, proliferation, and ECM (re)organization [61]. Its involvement in these processes highlights its significance in wound healing, tissue regeneration, and developmental biology [62].
Regarding tissue distribution, HA is widely distributed throughout the body, with particularly high concentrations found in connective tissues, such as the skin, synovial fluid, and cartilage [63]. In the skin, HA resides in the ECM and contributes to tissue hydration, elasticity, and wound healing [64]. In the synovial fluid of joints, HA forms a viscous, gel-like substance that provides lubrication, shock absorption, and nutrient supply to the articular cartilage [65]. Additionally, HA is present in the vitreous humor of the eye, where it helps maintain the transparency and shape of the eyeball [66]. Moreover, HA is found in other tissues, including the umbilical cord, umbilical vessels, and embryonic tissues, where it plays crucial roles in development and tissue morphogenesis [67].
3. Pathophysiological Role of Hyaluronic Acid in Rheumatic Diseases
HA plays a crucial pathophysiological role in rheumatic diseases, especially concerning joint health and function [68]. In a healthy joint, HA’s viscoelastic properties facilitate smooth movement of the joint surfaces and shield the cartilage from excessive mechanical stress [69]. HA serves as a shock absorber, mitigating the impact on the joint and thereby reducing the risk of damage. However, in rheumatic conditions such as OA and RA, significant alterations occur in the HA metabolism and distribution within the joint [70].
3.1. Osteoarthritis
3.1.1. Role of Hyaluronic Acid in Joint Lubrication
In OA, there is a notable reduction in the concentration of HA within the synovial fluid, leading to diminished joint lubrication and compromised cartilage protection [69]. This decline in HA levels can be attributed to an imbalance between HA synthesis and degradation, with increased activity of hyaluronidases, the enzymes responsible for HA breakdown [71]. Consequently, the cartilage becomes vulnerable to wear and tear, resulting in pain, stiffness, and functional impairment [72]. Dysregulated HA metabolism, including increased synthesis and degradation, contributes to cartilage degradation, synovitis, and pain in OA [73]. The altered biomechanical properties of HA affect joint lubrication, chondrocyte activity, and inflammation, further exacerbating disease progression [74].
3.1.2. Protective Effects on Chondrocytes and Cartilage Matrix
HA serves as a fundamental component of the cartilage matrix, playing a crucial role in maintaining joint integrity. However, in OA, changes occur in the quantity, molecular weight, and distribution of HA, which can impact its functional properties [75]. The molecular weight of HA influences its retention time within the tissues [76]. Generally, higher-molecular-weight HA exhibits a longer half-life, meaning it remains in the joint or tissue for a more extended period before being broken down and cleared by the body [8]. This prolonged presence may result in longer-lasting effects [8]. On the other hand, lower-molecular-weight HA can more easily penetrate the ECM and reach target cells, while higher-molecular-weight HA may have more limited diffusion [8].
3.1.3. Modulation of Inflammation and Synovial Fluid Changes
Inflammation is a hallmark of rheumatic diseases [77], and HA plays a multifaceted role in this process [78]. Pathological conditions often lead to changes in the quantity and molecular weight of HA in synovial fluid, with implications for disease severity and progression [79]. During the inflammatory phase, HA actively participates in the recruitment and activation of immune cells, such as macrophages and neutrophils [79]. HA engages with specific cell surface receptors, such as CD44 [80], and the binding affinity of HA to these receptors can vary depending on its molecular weight [81]. Distinct fractions of HA, characterized by different molecular weights, can have specific effects on cell signaling, inflammation, and tissue repair processes [82].
3.2. Rheumatoid Arthritis
In RA, the inflammatory process significantly disrupts HA homeostasis [83]. Synovial inflammation induces the release of pro-inflammatory cytokines and enzymes that promote the degradation of HA, leading to a decrease in its concentration and alterations in its molecular weight distribution [84]. These changes in HA metabolism have profound effects on joint lubrication, exacerbate cartilage damage, and perpetuate the inflammatory cycle [85].
3.2.1. Altered Hyaluronic Acid Synthesis and Breakdown
In RA, alterations in HA synthesis and breakdown contribute to the pathogenesis of the disease [70]. Synovial fibroblasts, key players in RA pathophysiology, exhibit dysregulated HA synthesis, leading to increased production and accumulation of HA in the synovial fluid and tissues [86]. This abnormal HA synthesis is influenced by various factors, including pro-inflammatory cytokines and growth factors, which stimulate the expression of HA synthases [87]. Concurrently, increased HA degradation occurs due to upregulated expression and activity of hyaluronidases [88]. The imbalance between HA synthesis and degradation results in the accumulation of fragmented HA in the synovial fluid, exacerbating inflammation and joint damage [89]. Moreover, the presence of HA fragments in the synovium further amplifies the inflammatory response by activating immune cells and promoting the production of pro-inflammatory mediators [90]. The presence of HA fragments and their interaction with CD44 receptors contribute to the perpetuation of chronic inflammation and joint damage in RA [91].
3.2.2. Inflammatory Mediator Modulation
Moreover, the anti-inflammatory properties of HA, which typically involve the modulation of the inflammatory responses within the joints, are disturbed in RA [83]. The inhibitory action of HA on the production of pro-inflammatory cytokines, such as tumor necrosis factor-alpha and interleukin-1 beta, and the suppression of inflammatory enzymes, such as cyclooxygenase-2 and MMPs, become dysregulated [92]. During the subsequent proliferative phase, HA continues to play a role in cell migration and proliferation, forming a provisional matrix that guides cell movement and stimulates cell proliferation [93].
3.2.3. Effects on Synovial Hyperplasia and Pannus Formation
HA exerts significant effects on synovial hyperplasia and pannus formation in RA [93]. It plays a pivotal role in promoting synovial hyperplasia by stimulating the proliferation and migration of synovial fibroblasts [94]. Furthermore, it interacts with CD44 receptors on the surface of these fibroblasts, triggering intracellular signaling cascades that promote cell survival, proliferation, and ECM production [95]. Additionally, HA enhances the expression of various pro-inflammatory mediators, such as cytokines and chemokines, further driving synovial hyperplasia and inflammation [96]. Additionally, HA fragments, generated due to the increased breakdown of HA in RA, can stimulate the production of matrix-degrading enzymes, leading to cartilage and bone destruction [97].
This entry is adapted from the peer-reviewed paper 10.3390/pharmaceutics15092247
References
- Monfort, J.; Lisbona, M.P.; Gimenez-Arnau, A.; Iglesias, M.; Llorente-Onaindia, J.; Benito, P. Immunoallergic reaction following intraarticular injection of high molecular weight hyaluronic acid. Jt. Bone Spine 2013, 80, 551–552.
- Roszkowska, A.M.; Inferrera, L.; Spinella, R.; Postorino, E.; Gargano, R.; Oliverio, G.W.; Aragona, P. Clinical Efficacy, Tolerability and Safety of a New Multiple-Action Eyedrop in Subjects with Moderate to Severe Dry Eye. J. Clin. Med. 2022, 11, 6975.
- Borzabadi-Farahani, A.; Mosahebi, A.; Zargaran, D. A Scoping Review of Hyaluronidase Use in Managing the Complications of Aesthetic Interventions. Aesthetic Plast. Surg. 2022.
- Wollina, U. Medical use of hyaluronic acid—A 2023 perspective. CosmoDerma 2023, 3, 86.
- Chen, L.H.; Xue, J.F.; Zheng, Z.Y.; Shuhaidi, M.; Thu, H.E.; Hussain, Z. Hyaluronic acid, an efficient biomacromolecule for treatment of inflammatory skin and joint diseases: A review of recent developments and critical appraisal of preclinical and clinical investigations. Int. J. Biol. Macromol. 2018, 116, 572–584.
- Miller, L.E. Towards reaching consensus on hyaluronic acid efficacy in knee osteoarthritis. Clin. Rheumatol. 2019, 38, 2881–2883.
- Hyaluronic Acid. Available online: https://en.wikipedia.org/wiki/Hyaluronic_acid (accessed on 3 June 2023).
- Fraser, J.R.; Laurent, T.C.; Laurent, U.B. Hyaluronan: Its nature, distribution, functions and turnover. J. Intern. Med. 1997, 242, 27–33.
- Saari, H.; Konttinen, Y.T.; Friman, C.; Sorsa, T. Differential effects of reactive oxygen species on native synovial fluid and purified human umbilical cord hyaluronate. Inflammation 1993, 17, 403–415.
- Bruyère, O.; Reginster, J.Y.; Honvo, G. Health economic evaluation of a high and low molecular weight hyaluronic acid formulation for the treatment of knee osteoarthritis. Post hoc analyses from a randomized clinical trial. Rheumatol. Ther. 2022, 9, 1119–1128.
- Sze, J.; Brownlie, J.C.; Love, C.A. Biotechnological production of hyaluronic acid: A mini review. 3 Biotech 2016, 6, 67.
- Ohnishi, T.; Novais, E.J.; Risbud, M. Alterations in ECM signature underscore multiple sub-phenotypes of intervertebral disc degeneration. Matrix Biol. Plus 2020, 21, 100036.
- Galvez-Martin, P.; Soto-Fernandez, C.; Romero-Rueda, J.; Cabañas, J.; Torrent, A.; Castells, G.; Martinez-Puig, D. A Novel Hyaluronic Acid Matrix Ingredient with Regenerative, Anti-Aging and Antioxidant Capacity. Int. J. Mol. Sci. 2023, 24, 4774.
- Matheson, A.R.; Sheehy, E.J.; Jay, G.D.; Scott, W.M.; O’Brien, F.J.; Schmidt, T.A. The role of synovial fluid constituents in the lubrication of collagen-glycosaminoglycan scaffolds for cartilage repair. J. Mech. Behav. Biomed. Mater. 2021, 118, 104445.
- Mustonen, A.; Capra, J.; Rilla, K.; Lehenkari, P.; Oikari, S.; Kääriäinen, T.; Joukainen, A.; Kröger, H.; Paakkonen, T.; Matilainen, J.; et al. Characterization of hyaluronan-coated extracellular vesicles in synovial fluid of patients with osteoarthritis and rheumatoid arthritis. BMC Musculoskelet. Disord. 2021, 22, 247.
- Householder, N.A.; Raghuram, A.; Agyare, K.; Thipaphay, S.; Zumwalt, M. A Review of Recent Innovations in Cartilage Regeneration Strategies for the Treatment of Primary Osteoarthritis of the Knee: Intra-articular Injections. Orthop. J. Sports Med. 2023, 11, 23259671231155950.
- Marcolina, A.; Vu, K.; Chang Chien, G. Peripheral Joint Injections. Phys. Med. Rehabil. Clin. N. Am. 2022, 33, 267–306.
- Osteoarthritis (OA) Arthritis CDC. Available online: https://www.cdc.gov/arthritis/basics/osteoarthritis.htm (accessed on 4 August 2023).
- Ferreira, N.D.R.; Sanz, C.K.; Raybolt, A.; Pereira, C.M.; DosSantos, M.F. Action of Hyaluronic Acid as a Damage-Associated Molecular Pattern Molecule and Its Function on the Treatment of Temporomandibular Disorders. Front. Pain Res. 2022, 3, 852249.
- Li, Y.; Yuan, Z.; Yang, H.; Zhong, H.; Peng, W.; Xie, R. Recent Advances in Understanding the Role of Cartilage Lubrication in Osteoarthritis. Molecules 2021, 26, 6122.
- Costa, F.R.; Costa Marques, M.R.; Costa, V.C.; Santos, G.S.; Martins, R.A.; Santos, M.D.S.; Santana, M.H.A.; Nallakumarasamy, A.; Jeyaraman, M.; Lana, J.V.B.; et al. Intra-Articular Hyaluronic Acid in Osteoarthritis and Tendinopathies: Molecular and Clinical Approaches. Biomedicines 2023, 11, 1061.
- Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Division of Population Health. Available online: https://www.cdc.gov/arthritis/basics/rheumatoid-arthritis.html (accessed on 4 August 2023).
- Ragan, C.; Meyer, K. The hyaluronic acid of synovial fluid in rheumatoid arthritis. J. Clin. Investig. 1949, 28, 56–59.
- De Lucia, O.; Murgo, A.; Pregnolato, F.; Pontikaki, I.; De Souza, M.; Sinelli, A.; Cimaz, R.; Caporali, R. Hyaluronic Acid injections in the treatment of osteoarthritis secondary to primary inflammatory rheumatic diseases: A systematic review and qualitative synthesis. Adv. Ther. 2020, 37, 1347–1359.
- Knopf-Marques, H.; Pravda, M.; Wolfova, L.; Velebny, V.; Schaaf, P.; Vrana, N.E.; Lavalle, P. Hyaluronic Acid and Its Derivatives in Coating and Delivery Systems: Applications in Tissue Engineering, Regenerative Medicine and Immunomodulation. Adv. Healthc. Mater. 2016, 5, 2841–2855.
- Altman, R.M.; Bedi, A.; Manjoo, A.; Niazi, F.; Shaw, P.; Mease, P. Anti-Inflammatory Effects of Intra-Articular Hyaluronic Acid: A Systematic Review. Cartilage 2019, 10, 43–52.
- Salathia, S.; Gigliobianco, M.; Casadidio, C.; Di Martino, P.; Censi, R. Hyaluronic Acid-Based Nanosystems for CD44 Mediated Anti-Inflammatory and Antinociceptive Activity. Int. J. Mol. Sci. 2023, 24, 7286.
- Richardson, C.; Plaas, A.; Block, J.A. Intra-articular Hyaluronan Therapy for Symptomatic Knee Osteoarthritis. Rheum. Dis. Clin. N. Am. 2019, 45, 439–451.
- Harrer, D.; Sanchez Armengol, E.; Friedl, J.D.; Jalil, A.; Jelkmann, M.; Leichner, C.; Laffleur, F. Is hyaluronic acid the perfect excipient for the pharmaceutical need? Int. J. Pharm. 2021, 15, 120589.
- Pendyala, M.; Woods, P.S.; Brubaker, D.K.; Blaber, E.A.; Schmidt, T.A.; Chan, D.D. Endogenous production of hyaluronan, PRG4, and cytokines is sensitive to cyclic loading in synoviocytes. PLoS ONE 2022, 17, e0267921.
- Atkins, E.D.; Sheehan, J.K. The molecular structure of hyaluronic acid. Biochem. J. 1971, 125, 92P.
- Jabbari, F.; Babaeipour, V.; Saharkhiz, S. Comprehensive review on biosynthesis of hyaluronic acid with different molecular weights and its biomedical applications. Int. J. Biol. Macromol. 2023, 240, 124484.
- Kobayashi, Y.; Okamoto, A.; Nishinari, K. Viscoelasticity of hyaluronic acid with different molecular weights. Biorheology 1994, 31, 235–244.
- Zhang, L.; Weifeng, L.; Yaxun, F.; Nir, K.; Yilin, W.; Klein, J. Effects of Hyaluronan Molecular Weight on the Lubrication of Cartilage-Emulating Boundary Layers. Biomacromolecules 2020, 21, 4345–4354.
- Gupta, R.C.; Lall, R.; Srivastava, A.; Sinha, A. Hyaluronic Acid: Molecular Mechanisms and Therapeutic Trajectory. Front. Vet. Sci. 2019, 6, 192.
- Tobisawa, Y.; Fujita, N.; Yamamoto, H.; Ohyama, C.; Irie, F.; Yamaguchi, Y.J. The cell surface hyaluronidase TMEM2 is essential for systemic hyaluronan catabolism and turnover. J. Biol. Chem. 2021, 297, 101281.
- Wu, G.T.; Kam, J.; Bloom, J.D. Hyaluronic Acid. Basics and Rheology. Clin. Plast. Surg. 2023, 50, 391–398.
- Csoka, A.B.; Frost, G.I.; Stern, R. The six hyaluronidase-like genes in the human and mouse genomes. Matrix Biol. 2001, 20, 499–508.
- Valcarcel, J.; García, M.; Varela, U.R.; Vázquez, J.A. Hyaluronic acid of tailored molecular weight by enzymatic and acid depolymerization. Int. J. Biol. Macromol. 2020, 145, 788–794.
- Guan, Y.; You, H.; Cai, J.; Zhang, Q.; Yan, S.; You, R. Physically crosslinked silk fibroin/hyaluronic acid scaffolds. Carbohydr. Polym. 2020, 239, 116232.
- Casale, J.; Crane, J.S. Biochemistry, Glycosaminoglycans. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023.
- De Francesco, F.; Saparov, A.; Riccio, M. Hyaluronic acid accelerates re-epithelialization and healing of acute cutaneous wounds. Eur. Rev. Med. Pharmacol. Sci. 2023, 7, 37–45.
- Menezes, R.; Vincent, R.; Osorno, L.; Hu, P.; Arinzeh, T.L. Biomaterials and tissue engineering approaches using glycosaminoglycans for tissue repair: Lessons learned from the native extracellular matrix. Acta Biomater. 2023, 163, 210–227.
- Henrotin, Y.; Raman, R.; Richette, P.; Bard, H.; Jerosch, J.; Conrozier, T.; Chevalier, X.; Migliore, A. Consensus statement on viscosupplementation with hyaluronic acid for the management of osteoarthritis. Semin. Arthritis Rheum. 2015, 45, 140–149.
- Filardo, G.; Di Matteo, B.; Di Martino, A.; Merli, M.L.; Cenacchi, A.; Fornasari, P.; Marcacci, M.; Kon, E. Platelet-Rich Plasma Intra-articular Knee Injections Show No Superiority Versus Viscosupplementation: A Randomized Controlled Trial. Am. J. Sports Med. 2015, 43, 1575–1582.
- Altman, R.D.; Manjoo, A.; Fierlinger, A.; Niazi, F.; Nicholls, M. The mechanism of action for hyaluronic acid treatment in the osteoarthritic knee: A systematic review. BMC Musculoskelet. Disord. 2015, 16, 321.
- Daheshia, M.; Yao, J.Q. The interleukin 1beta pathway in the pathogenesis of osteoarthritis. J. Rheumatol. 2008, 35, 2306–2312.
- Genasetti, A.; Vigetti, D.; Viola, M.; Karousou, E.; Moretto, P.; Rizzi, M.; Bartolini, B.; Clerici, M.; Pallotti, F.; De Luca, G.; et al. Hyaluronan and human endothelial cell behavior. Connect. Tissue Res. 2008, 49, 120–123.
- Papakonstantinou, E.; Roth, M.; Karakiulakis, G. Hyaluronic acid: A key molecule in skin aging. Dermatoendocrinol 2012, 4, 253–258.
- McAlindon, T.E.; Bannuru, R.R. Osteoarthritis: Is viscosupplementation really so unsafe for knee OA? Nat. Rev. Rheumatol. 2012, 8, 635–636.
- Ghosh, S.; Hoselton, S.A.; Dorsam, G.P.; Schuh, J.M. Hyaluronan fragments as mediators of inflammation in allergic pulmonary disease. Immunobiology 2015, 220, 575–588.
- Johnson, P.; Arif, A.A.; Lee-Sayer, S.S.M.; Dong, Y. Hyaluronan and Its Interactions With Immune Cells in the Healthy and Inflamed Lung. Front. Immunol. 2018, 9, 2787.
- Parnigoni, A.; Viola, M.; Karousou, E.; Rovera, S.; Giaroni, C.; Passi, A.; Vigetti, D. Hyaluronan in pathophysiology of vascular diseases: Specific roles in smooth muscle cells, endothelial cells, and macrophages. Am. J. Physiol. Cell Physiol. 2022, 323, C505–C519.
- Naor, D. Interaction Between Hyaluronic Acid and Its Receptors (CD44, RHAMM) Regulates the Activity of Inflammation and Cancer. Front. Immunol. 2016, 8, 39.
- Leng, Y.; Abdullah, A.; Wendt, M.; Calve, S. Hyaluronic acid, CD44 and RHAMM regulate myoblast behavior during embryogenesis. Matrix Biol. 2019, 78–79, 236–254.
- Hackett, T.L.; Osei, E.T. Modeling Extracellular Matrix-Cell Interactions in Lung Repair and Chronic Disease. Cells 2021, 10, 2145.
- Ambili, M.; Sudhakaran, P.R. Modulation of neutral matrix metalloproteinases of involuting rat mammary gland by different cations and glycosaminoglycans. J. Cell Biochem. 1999, 73, 218–226.
- Yung, S.; Chan, T.M. Hyaluronan-regulator and initiator of peritoneal inflammation and remodeling. Int. J. Artif. Organs 2007, 30, 477–483.
- Lierova, A.; Kasparova, J.; Filipova, A.; Cizkova, J.; Pekarova, L.; Korecka, L.; Mannova, N.; Bilkova, Z.; Sinkorova, Z. Hyaluronic Acid: Known for Almost a Century, but Still in Vogue. Z. Pharm. 2022, 14, 838.
- Amorim, S.; Reis, C.A.; Reis, R.L.; Pires, R.A. Extracellular Matrix Mimics Using Hyaluronan-Based Biomaterials. Trends Biotechnol. 2021, 39, 90–104.
- Reitinger, S.; Lepperdinger, G. Hyaluronan, a ready choice to fuel regeneration: A mini-review. Gerontology 2013, 59, 71–76.
- Knudson, C.B.; Toole, B.P. Hyaluronate-cell interactions during differentiation of chick embryo limb mesoderm. Dev. Biol. 1987, 124, 82–90.
- Bastow, E.R.; Byers, S.; Golub, S.B.; Clarkin, C.E.; Pitsillides, A.A.; Fosang, A.J. Hyaluronan synthesis and degradation in cartilage and bone. Cell Mol. Life Sci. 2008, 65, 395–413.
- Legouffe, R.; Jeanneton, O.; Gaudin, M.; Tomezyk, A.; Gerstenberg, A.; Dumas, M.; Heusèle, C.; Bonnel, D.; Stauber, J.; Schnebert, S. Hyaluronic acid detection and relative quantification by mass spectrometry imaging in human skin tissues. Anal. Bioanal. Chem. 2022, 414, 5781–5791.
- Laurent, T.C.; Laurent, U.B.; Fraser, J.R. The structure and function of hyaluronan: An overview. Immunol. Cell Biol. 1996, 74, A1–A7.
- Deguine, V.; Labat-Robert, J.; Ferrari, P.; Pouliquen, Y.; Menasche, M.; Robert, L. Aging of the vitreous body. Role of glycation and free radicals. Pathol. Biol. 1997, 45, 321–330. (In French)
- Klein, J.; Meyer, F.A. Tissue structure and macromolecular diffusion in umbilical cord. Immobilization of endogenous hyaluronic acid. Biochim. Biophys. Acta 1983, 755, 400–411.
- Schmidt, T.; Gastelum, N.S.; Nguyen, Q.T.; Schumacher, B.L.; Sah, R.L. Boundary lubrication of articular cartilage: Role of synovial fluid constituents. Arthritis Rheum. 2007, 56, 882–891.
- Mederake, M.; Trappe, D.; Jacob, C.; Hofmann, U.K.; Schüll, D.; Dalheimer, P.; Exner, L.; Walter, C. Influence of hyaluronic acid on intra-articular friction—A biomechanical study in whole animal joints. BMC Musculoskelet. Disord. 2022, 23, 927.
- Kosinska, M.K.; Ludwig, T.E.; Liebisch, G.; Zhang, R.; Siebert, H.C.; Wilhelm, J.; Kaesser, U.; Dettmeyer, R.B.; Klein, H.; Ishaque, B.; et al. Articular Joint Lubricants during Osteoarthritis and Rheumatoid Arthritis Display Altered Levels and Molecular Species. PLoS ONE 2015, 10, e0125192.
- Yoshida, M.; Sai, S.; Marumo, K.; Tanaka, T.; Itano, N.; Kimata, K.; Fujii, K. Expression analysis of three isoforms of hyaluronan synthase and hyaluronidase in the synovium of knees in osteoarthritis and rheumatoid arthritis by quantitative real-time reverse transcriptase polymerase chain reaction. Arthritis Res. Ther. 2004, 6, R514–R520.
- Zhang, J.; Nishida, Y.; Koike, H.; Ito, K.; Zhuo, L.; Nishida, K.; Kimata, K.; Ikuta, K.; Sakai, T.; Urakawa, H.; et al. Hyaluronan in articular cartilage: Analysis of hip osteoarthritis and osteonecrosis of femoral head. J. Orthop. Res. 2023, 41, 307–315.
- Papaneophytou, C.; Alabajos-Cea, A.; Viosca-Herrero, E.; Calvis, C.; Costa, M.; Christodoulides, A.E.; Kroushovski, A.; Lapithis, A.; Lapithi, V.; Papayiannis, I.; et al. Associations between serum biomarkers of cartilage metabolism and serum hyaluronic acid, with risk factors, pain categories, and disease severity in knee osteoarthritis: A pilot study. BMC Musculoskelet. Disord. 2022, 23, 195.
- Sun, Z.P.; Wu, S.P.; Liang, C.D.; Zhao, C.X.; Sun, B.Y. The synovial fluid neuropeptide PACAP may act as a protective factor during disease progression of primary knee osteoarthritis and is increased following hyaluronic acid injection. Innate Immun. 2019, 25, 255–264.
- Sasaki, E.; Tsuda, E.; Yamamoto, Y.; Maeda, S.; Inoue, R.; Chiba, D.; Fujita, H.; Takahashi, I.; Umeda, T.; Nakaji, S.; et al. Serum hyaluronic acid concentration predicts the progression of joint space narrowing in normal knees and established knee osteoarthritis—A five-year prospective cohort study. Arthritis Res. Ther. 2015, 17, 283.
- Ye, T.; Zhang, W.; Sun, M.; Yang, R.; Song, S.; Mao, Y.; Yao, J.; Yang, X.; Wang, S.; Pan, W. Study on intralymphatic-targeted hyaluronic acid-modified nanoliposome: Influence of formulation factors on the lymphatic targeting. Int. J. Pharm. 2014, 471, 245–257.
- Berenbaum, F. Osteoarthritis as an inflammatory disease (osteoarthritis is not osteoarthrosis!). Osteoarthr. Cartil. 2013, 21, 16–21.
- Jin, L.; Xu, K.; Liang, Y.; Du, P.; Wan, S.; Jiang, C. Effect of hyaluronic acid on cytokines and immune cells change in patients of knee osteoarthritis. BMC Musculoskelet. Disord. 2022, 23, 812.
- Lu, K.H.; Lu, P.W.; Lin, C.W.; Lu, E.W.; Yang, S.F. Different molecular weights of hyaluronan research in knee osteoarthritis: A state-of-the-art review. Matrix Biol. 2023, 117, 46–71.
- Bayer, I.S. Hyaluronic Acid and Controlled Release: A Review. Molecules 2020, 25, 2649.
- Kinoshita, M.; Kakehi, K. Analysis of the interaction between hyaluronan and hyaluronan-binding proteins by capillary affinity electrophoresis: Significance of hyaluronan molecular size on binding reaction. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2005, 816, 289–295.
- Mizrahy, S.; Raz, S.R.; Hasgaard, M.; Liu, H.; Soffer-Tsur, N.; Cohen, K.; Dvash, R.; Landsman-Milo, D.; Bremer, M.G.; Moghimi, S.M.; et al. Hyaluronan-coated nanoparticles: The influence of the molecular weight on CD44-hyaluronan interactions and on the immune response. J. Control. Release 2011, 156, 231–238.
- Guo, Y.; Wei, T.; Hu, N.; Zhou, X. Disrupted homeostasis of synovial hyaluronic acid and its associations with synovial mast cell proteases of rheumatoid arthritis patients and collagen-induced arthritis rats. Immunol. Res. 2021, 69, 584–593.
- Goto, M.; Hosako, Y.; Katayama, M.; Yamada, T. Biochemical analysis of rheumatoid synovial fluid after serial intra-articular injection of high molecular weight sodium hyaluronate. Int. J. Clin. Pharmacol. Res. 1993, 13, 161–166.
- Poole, A.R.; Dieppe, P. Biological markers in rheumatoid arthritis. Semin. Arthritis Rheum. 1994, 23 (Suppl. S2), 17–31.
- Chang, X.; Yamada, R.; Yamamoto, K. Inhibition of antithrombin by hyaluronic acid may be involved in the pathogenesis of rheumatoid arthritis. Arthritis Res. Ther. 2005, 7, R268–R273.
- Butler, D.M.; Vitti, G.F.; Leizer, T.; Hamilton, J.A. Stimulation of the hyaluronic acid levels of human synovial fibroblasts by recombinant human tumor necrosis factor alpha, tumor necrosis factor beta (lymphotoxin), interleukin-1 alpha, and interleukin-1 beta. Arthritis Rheum. 1988, 31, 1281–1289.
- Pásztói, M.; Nagy, G.; Géher, P.; Lakatos, T.; Tóth, K.; Wellinger, K.; Pócza, P.; György, B.; Holub, M.C.; Kittel, A.; et al. Gene expression and activity of cartilage degrading glycosidases in human rheumatoid arthritis and osteoarthritis synovial fibroblasts. Arthritis Res. Ther. 2009, 11, R68.
- Wollheim, F.A. Predictors of joint damage in rheumatoid arthritis. APMIS 1996, 104, 81–93.
- Olsson, M.; Bremer, L.; Aulin, C.; Harris, H.E. Fragmented hyaluronan has no alarmin function assessed in arthritis synovial fibroblast and chondrocyte cultures. Innate Immun. 2018, 24, 131–141.
- Fujii, K.; Tanaka, Y.; Hübscher, S.; Saito, K.; Ota, T.; Eto, S. Crosslinking of CD44 on rheumatoid synovial cells augment interleukin 6 production. Lab. Investig. 1999, 79, 1439–1446.
- Hashizume, M.; Mihara, M. High molecular weight hyaluronic acid inhibits IL-6-induced MMP production from human chondrocytes by up-regulating the ERK inhibitor, MKP-1. Biochem. Biophys. Res. Commun. 2010, 403, 184–189.
- Brondello, J.M.; Djouad, F.; Jorgensen, C. Where to Stand with Stromal Cells and Chronic Synovitis in Rheumatoid Arthritis? Cells 2019, 8, 1257.
- Shiozawa, S.; Yoshihara, R.; Kuroki, Y.; Fujita, T.; Shiozawa, K.; Imura, S. Pathogenic importance of fibronectin in the superficial region of articular cartilage as a local factor for the induction of pannus extension on rheumatoid articular cartilage. Ann. Rheum. Dis. 1992, 51, 869–873.
- Al-Sharif, A.; Jamal, M.; Zhang, L.X.; Larson, K.; Schmidt, T.A.; Jay, G.D.; Elsaid, K.A. Lubricin/Proteoglycan 4 Binding to CD44 Receptor: A Mechanism of the Suppression of Proinflammatory Cytokine-Induced Synoviocyte Proliferation by Lubricin. Arthritis Rheumatol. 2015, 67, 1503–1513.
- Campo, G.M.; Avenoso, A.; D’Ascola, A.; Prestipino, V.; Scuruchi, M.; Nastasi, G.; Calatroni, A.; Campo, S. The stimulation of adenosine 2A receptor reduces inflammatory response in mouse articular chondrocytes treated with hyaluronan oligosaccharides. Matrix Biol. 2012, 31, 338–351.
- Saxne, T.; Heinegård, D. Synovial fluid analysis of two groups of proteoglycan epitopes distinguishes early and late cartilage lesions. Arthritis Rheum. 1992, 35, 385–390.
This entry is offline, you can click here to edit this entry!
