Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Crystalline carbon-based nanomaterials, encompassing graphene, graphene oxide, reduced graphene oxide, carbon nanotubes, and graphene quantum dots, have emerged as promising materials for the development of medical devices in various biomedical applications. These materials possess inorganic semiconducting attributes combined with organic π-π stacking features, allowing them to efficiently interact with biomolecules and present enhanced light responses.
- crystalline carbon-based nanomaterials
- biomedical applications
- cancer treatment
- biosensors
1. Introduction
Graphite, a natural material with a long history, holds the distinction of being one of the oldest and most widely utilized substances. While traditionally recognized as the primary component of pencil lead, giving rise to its name, graphite has found expanded application in various large-scale industrial sectors. These include its use in steelmaking for carbon raising, battery electrodes, and industrial-grade lubricants [1]. Notably, the demand for synthetic graphite has experienced a significant upsurge in recent years. Scientific investigations have extensively explored graphite, revealing that its distinctive combination of physical properties arise from its intricate macromolecular arrangement, which comprises successive hexagonal arrays of sp2 carbon stacked together. Advancements and growing recognition in nanofabrication methods and nanomaterials over the past two decades have spurred the active utilization of graphite as a foundational material for engineering diverse types of carbon-based nanomaterials (CBNs) [2].
CBNs demonstrate remarkable mechanical strength, electrical and thermal conductivity, and optical characteristics [3]. Consequently, extensive research has been dedicated to capitalizing on these favorable properties for diverse applications, including the development of high-strength composite materials and advancements in electronics. Notably, carbon materials encompass a wide range of forms within the carbon family, spanning from zero-dimensional (0D) to three-dimensional (3D) structures [4][5][6][7][8] (Figure 1). Carbon-based quantum dots and carbon nanotubes (CNTs) such as single or multi-walled nanotubes, nanoribbons, graphene, and substances derived from compounds like graphene oxide, biomass or polymer-derived carbon, fullerenes, and nanodiamonds are among the commonly employed crystalline carbon materials [9][10][11][12].
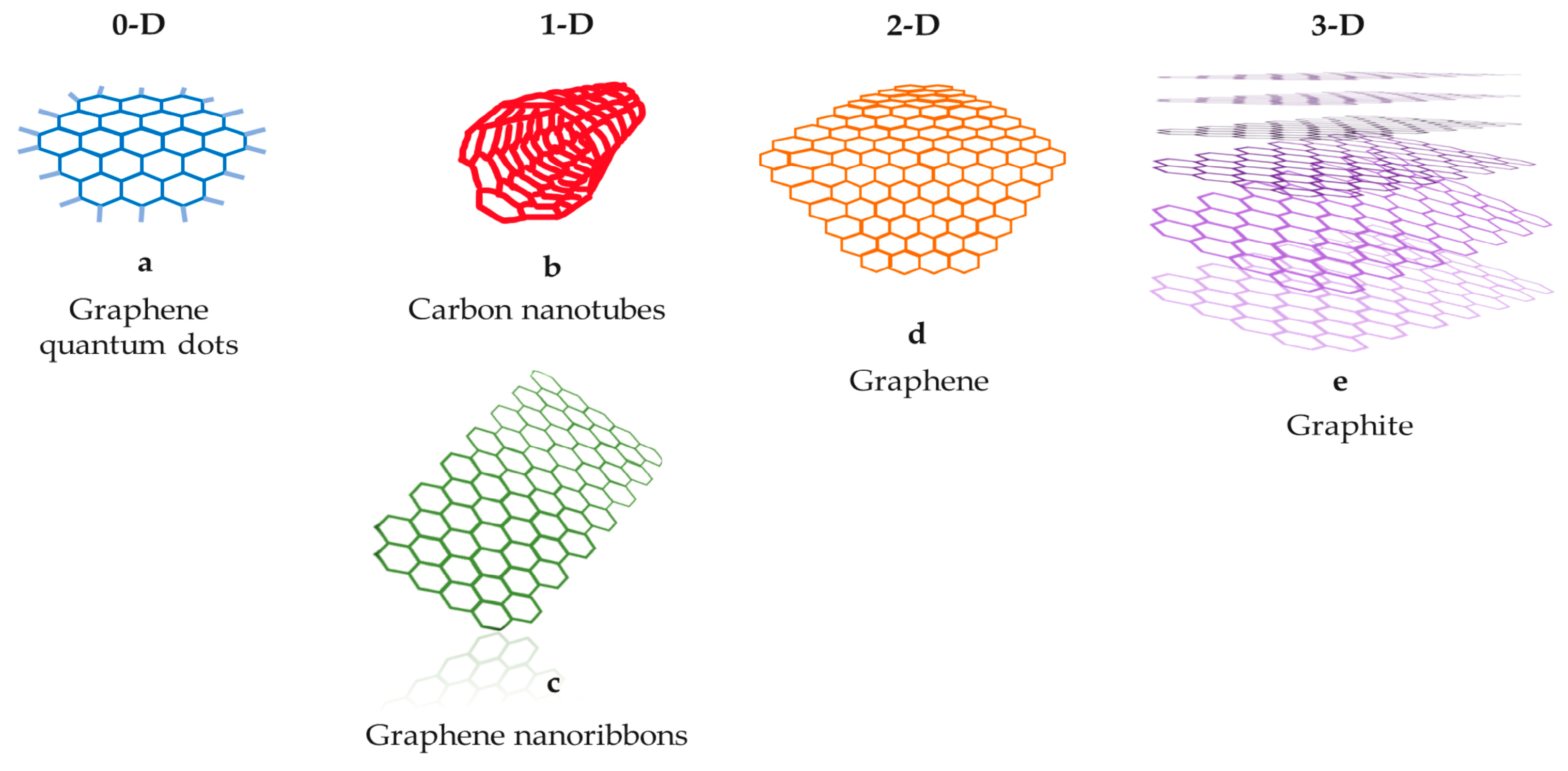
Figure 1. The main carbon-based nanomaterials. (a) Graphene quantum dots, (b) carbon nanotubes, (c) graphene nanoribbons, (d) graphene, (e) graphite.
In recent times, the field of biomedical engineering has warmly welcomed the growing popularity and influence of CBNs due to their crucial role in numerous applications, which heavily rely on biomaterial performance. Nanoscale biomaterials, in particular, are highly sought after due to their small size [13][14][15][16][17][18]. Among these materials, CBNs have gained considerable recognition as appealing biomaterials [19] because of their compelling characteristics, such as exceptional mechanical properties, enhanced specific surface area, environmentally friendly nature, and ease on surficial alterations [20][21][22][23]. Furthermore, they possess the highest distribution rate within the body [24]. Incorporating CBNs into existing biomaterials can enhance their functionalities, leading to their extensive utilization in various biomedical research areas [25], including drug delivery systems, the reinforcement of tissue scaffolds, and the development of cellular sensors [26].
2. Carbon Nanotubes (CNTs)
Subsequent to their initial identification, carbon nanotubes (CNTs) have emerged as the prevalently employed CBNs [27][28]. CNTs are typically synthesized through processes like arc discharge or chemical vapor deposition of graphite. Characterized by their cylindrical structure, CNTs exhibit a broad array of both electrical and optical features. These characteristics do not solely stem from their sp2 carbon structure but also from their customizable physical features, including their diameter, length, whether they are single-walled or multi-walled, their surface functionalization capacity, and even their chirality [28]. The CNTs’ versatile and beneficial properties have the motivated exploration of numerous industrialized purposes [29]. For instance, the exceptional mechanical strength constitutes one of the features comprising known CNTs, surpassing that of various widely employed robust materials, such as high-tensile steel and Kevlar. As a result, CNTs are employed as reinforcing constituents in composite materials, including plastics, as well as metal alloys, leading to the commercialization of several products [30]. Nevertheless, the potential for CNT composites to serve as ultra-high-strength load-bearing substances has not yielded adequate outcomes thus far. This is primarily attributed to the inadequate load transmission from the surrounding matrices to CNTs, stemming from the poor interaction between them [30].
In recent times, numerous research endeavors have focused on integrating CNTs into several substances to leverage their versatile properties. Instead of solely emphasizing composites’ mechanical strength, attention has been directed towards utilizing CNTs’ diverse attributes. For instance, the remarkable electrical features of CNTs, combined with their nano-dimensions, have garnered significant attention in electronics for constructing nano-electronic circuits [31][32]. Moreover, CNTs exhibit a decreased threshold for electric fields in field emission, surpassing that of the commonly utilized field emitters [33][34]. Consequently, there is ongoing dynamic research on CNTs for their exceptional performance in electron emission devices like electron microscopes, flat display panels, and gas-discharge tubes. Moreover, their ability to exhibit robust luminescence through field emission opens up promising avenues for their use in lighting components [32].
The utilization of CNTs in diverse biomedical applications has garnered significant attention, due to their aforementioned attributes, making them highly favorable contenders for the advancement of biomaterials [35]. Moreover, CNTs are able to be chemically modified to introduce distinct entities, such as functional groups, molecules, as well as polymers, to enhance their suitability for biomedical purposes. These modifications can enhance solubility, biocompatibility, and cellular responsiveness [36]. For this reason, CNTs are the most applied CBNs in biomedicine, particularly in sensoring, drug delivery, cancer treatment, and tissue engineering. Their toxicity can be regulated through functionalization. Thus, they are considered ideal for biomedical applications compared to the other available CBNs.
The main biomedical applications of CNTs are presented in Figure 2 and are discussed in the following subsections.
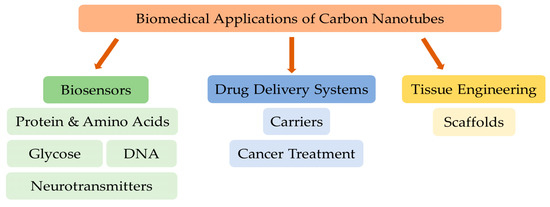
Figure 2. The main biomedical applications of carbon nanotubes.
2.1. CNTs as Biosensors
Given their remarkable attributes, CNTs have been recognized as innovative nanoprobes [37]. Their high aspect ratio, conductivity, chemical robustness, as well as sensitivity [38], along with their rapid electron-transfer rate [39], render them highly suitable for biosensing purposes. The fundamental aspect of CNTs-based biosensors involves the surficial immobilization of biomolecules, thereby enhancing recognition, as well as facilitating the signal transduction process. The biosensors are commonly classified as electrochemical and electronic CNTs-based biosensors, as well as optical ones, resting on their target sensing and communication mechanisms. CNTs have gained significant acclaim as advantageous materials for enhancing electron transfer, making them well-suited for integrating electrochemical and electronic biosensors [40].
2.1.1. Sensing of Glucose
Several glucose biosensors utilizing CNTs in conjunction with glucose oxidase have been developed [26][41].
Compared to the first-generation sensor where the electrode and glucose oxidase were used [42], the second generation of glucose sensor incorporates an electron carrier within the enzyme electrode [43]. In this phase, the electron medium actively participates within the reaction, replacing the role of O2. The enhanced exclusivity and acuteness of the second-generation sensor have resulted in its commercial exploitation in glucose sensing. Enzyme-only sensors tend to be unsteady, as enzymes are susceptible to deactivation and detachment. In order to address this, enzyme immobilization has been identified as an effective approach to assure enzyme robustness and recyclability [44]. Additionally, leveraging the unique properties of the material can further enhance enzyme efficiency. For example, glucose oxidase can be anchored using polymers, as well as a vertical array of CNTs (VACNTs) [45]. Chemical vapor deposition (CVD) is utilized to modify the CNT wall with polyaniline (PANI) for fixing glucose oxidase (GOx), resulting in improved electrode stability and accelerated electron transfer. The sensor’s range of linearity spans between 2 and 426 mM, having a detectable limit equal to 1.1 mM. Importantly, it is not affected by interference from uric acid and ascorbic acid. To overcome enzyme activity reduction or denaturation and to facilitate enzyme adsorption, metal or metal oxide nanoparticles are employed for enzyme immobilization. A ZnFe2O4-CNTs-glucose oxidase composite has been fabricated for use in glucose sensing [46]. The as-mentioned sensor demonstrated stable catalytic efficiency for 20 days, as well as recyclability >5 times, benefiting from the ZnFe2O4′s-increased catalytic effectiveness, the enhanced surface area ratio of CNTs, and the elevated loading capacity of GOx.
An electrode consisting of Au nanoparticles/MnOx-VOx/CNT/GCE was synthesized using electrochemical pulsed deposition by combining metal oxides [47]. MnOx-VOx was applied as a surficial modification of a glassy carbon electrode (CE) with CNT (CNT/GCE), while Au nanoparticles were deposited onto the oxide film. Glucose detection exhibited a linear range of 0.1–1.0 mM, and the detection limit was measured at 0.02 mM. Another approach involved the utilization of CNTs altered with platinum nanoparticles as electrodes for glucose detection [48]. The researchers initially formulated composite solutions comprising carboxyl functionalized MWCNTs and chemically derived graphene (CDG). These solutions were then deposited as thin films onto the gold electrode, followed by the deposition of Pt nanoparticles through an electrochemical process. The linear range for glucose detection was 0.5–13.5 mM, with a low detection limit (1.3 mM).
Ensuring long-lasting durability and optimal current density for continuous monitoring are crucial considerations for glucose sensors. The rate of alteration also impacts the efficiency of CNT materials. The presence of lignosulfonate can influence the composites’ electrical effectiveness containing multi-walled CNTs (MWCNTs), subsequently affecting their electrocatalytic functionality [49]. The chemical alteration of glucose oxidase can enhance its efficiency and selectivity simultaneously. By combining glucose oxidase and MWCNTs modified with β-cyclodextrin, the sensor exhibited 95.8% stability in current retention over a span of 14 days [50].
In contrast to the conventional one-factor-at-a-time (OFAT) approach, several researchers have suggested the use of a design of experiments (DOE) approach to simultaneously maximize the current density and robustness [51].
The enzymes’ chemical features are composed of proteinic molecules that are prone to denaturation when exposed to pH value modifications, temperature, and various environmental factors. This leads to decreased sensor stability and affects the effectiveness of detection. While physical immobilization helps preserve the enzymes, it does not guarantee a reduction in activity. Enzyme leakage occurs when enzymes detach and access the solution, rendering them ineligible for involvement in the reaction. To address the issue of poor stability, a nGOx/N-CNTs-Chi/GCE (Chitosan/Glassy CE) sensor was developed [52]. A polymerization approach was suggested to enclose glucose oxidase in single-molecule enzyme nanocapsules (SMENs) in situ. The synthesized SMENs of GOx-substituted traditional glucose oxidase, improving enzyme activity preservation. In comparison to traditional CNTs, n-doped CNTs (NCNTs) were employed to enhance the catalytic conductivity, specific surface area, and biocompatibility. Another approach utilized glucose oxidase micro-particles instead of independent enzymes [53]. The relevant sensitivity persisted >86% for 9 days. Additionally, the issue of enzyme leakage was effectively addressed through the implementation of a hollow fiber membrane with a gradient structure. The range of linear detection spanned from 0 to 24 mM. The enzymes’ catalytic efficiency was intimately linked to temperature. Microencapsulated phase change materials (MEPCMs) were employed to inhibit enzyme leakage and attain temperature control, thereby enhancing the enzyme-sensing performance at increased temperatures [54].
Several crucial aspects are considered during the development of enzymatic sensors to enhance robustness and sensitivity. Firstly, effective screening is carried out to identify enzymes with high activity. Secondly, efforts are made to maintain the activity and stability of the enzymes. Lastly, reliable fixation of the enzymes to the semiconductor is ensured. To address the aforementioned challenges, a third generation of glucose sensors resting on direct electron transfer (DET) has emerged. These sensors operate without the need for media or chemical reactions, enabling a direct transfer of electrons from glucose to the electrode. In one study, a GOx-Chit-CNT/ITO electrode was created by modifying the composite chitosan-CNTs onto an indium tin oxide (ITO) electrode [55]. Various solutions with diverse mass ratios of CNTs were prepared, and it was found that chitosan-CNT85 exhibited the optimal performance. In the long run, glucose detection biosensors are expected to be transformed into wearable devices for convenient in vitro sensing [56]. Another approach involved utilizing a dual enzyme system consisting of glucose oxidase and horseradish peroxidase in a CNTs-EVA composite membrane [57]. The glucose oxidase/horseradish peroxidase/CNT-EVA electrode was instantly employed for glucose detection in sweat. A non-intrusive glucose sensor utilizing two electrodes demonstrated superior performance compared to existing invasive glucose meters, presenting sensitivity equal to 14.45 ± 2.97 mA mM−1 cm−2 [58].
Zhu and his team [59] employed non-woven fabrics made of CNTs (CNTFs) to detect glucose by incorporating glucose oxidase into a polyvinyl alcohol solution. In addition, Gaitán and co-researchers focused on the impact of surficial chemistry, as well as glucose oxidase-coated MWCNTs’ structure in electrochemical glucose sensing [60].
Non-enzymatic sensors possess several advantageous features, such as long-lasting durability, low-cost, enhanced sensitivity, effective electron transfer, and strong electro-catalytic efficiency [61][62]. These benefits overcome the constraints of enzyme-based sensors, which are prone to instability and storage difficulties. Consequently, numerous research studies have been conducted [63][64][65], encouraging advancements in glucose sensor technology. Ideal glucose sensors should exhibit an extensive linear span, decreased limit of detection, and increased sensitivity. While enzyme-based sensors offer a broad linear range and sensitivity, their fixation and preparation present challenges. Moreover, enzyme stability issues can lead to discrepancies. Consequently, extensive studies are dedicated to the fabrication of non-enzymatic sensors to enhance their sensing capabilities. Non-enzymatic sensors typically involve the modification of electrodes with metal nanoparticles. For instance, an electrode composed of stacked cup CNTs (SCCNT) modified with nickel oxide (NiO) was utilized [66]. Another approach involved a bimetallic nanoparticle-altered electrode (Fe, Ni/CNTs/GCE) that harnessed the advantageous electrochemical features of Fe and Ni [67]. The impact of an NiO layer’s thickness on sensing has been also investigated. Pt nanoparticles have been employed to modify MWCNTs, with the functionalization of 3-mercaptophenylboronic acid (3MPBA) attached to the platinum particle surface [68]. The linear range of sensing was 0–10 mM, presenting a detection limit equal to 4.5 mM. Moreover, PdNi@f-MWCNT/GCE was produced by modifying functionalized MWCNTs with Pd and Ni nanoparticles [69]. This particular sensor presented a linear range of 0.01–1.4 mM, and an exceptionally decreased LOD of 0.026 mM.
Transition metal oxide/carbon hybrid electrodes offer enhanced sensitivity and improved electron transfer efficiency. A fluorescent nanosensor utilizing a hybrid nanocomplex of cobalt and CNTs (Co-CNT) was documented [70]. This sensor employs unlabeled and enzyme-free fluorescence ratio approaches for the simultaneous sensing of H2O2 and glucose. The detection sensitivity for H2O2 reached 150 nM, while for glucose, it was also 150 nM. A straightforward approach was proposed for the preparation of a cobalt-containing electrode using melamine foam [71]. The glucose sensor employing the CoeCo3O4/CNT/CF/GCE electrode exhibited a detection range of 1.2 mM to 2.29 mM. The catalytic properties of binary metal composites outperformed those of metal oxides, making Ni and Co-based alloys with functionalized MWCNTs (f-MWCNTs) highly effective in glucose detection [72]. The combination of cobalt phthalocyanines (CoPc) with single-walled CNTs (SWCNTs) and reduced graphene oxide (rGO) allowed for a rapid response, responding within 1.2 s to changes in glucose concentrations in saliva [73].
With a focus on nanomaterials, researchers have become increasingly dedicated to nano-structured design and novel approaches [74]. An electrode composed of Co3O4/NCNTs, based on a metal–organic skeleton, was created for detecting glucose and H2O2 [75]. A unique dodecahedral CNT array was achieved through the carbonization of a specific zeolitic imidazolate framework material (ZIF-67), with Co3O4 nanoparticles encapsulated within the CNTs ends. Furthermore, to enhance the robustness and catalytic effectiveness of binary metal composites, porous carbon materials were coupled with a CoCu organic framework catalyst, resulting in the development of nitrogen-doped CNTs@MOF@CoCu biosensors [76].
Cu2O presents itself as a favorable option for a metal oxide electrode. Along with material selection, the utilization of various nanostructures can enhance the performance of detection. For instance, a non-enzymatic sensor employed a copper nanowire-bilayer (CuNW-CNT-BL) configuration [77]. The sensor presented a linear range of 10–2000 mM, characterized by a limit of quantification equal to 1.1 nM and a low detection limit equal to 0.33 nM. Moreover, this sensor demonstrated selectivity towards uric acid, dopamine, and various molecules. In the Cu2O-MSs/S-MWCNTs/GCE composite electrode, the Cu2O material exhibited a spherical design [78]. By incorporating sulfur atoms, the electrocatalytic performance of CNTs was enhanced. The linear range spanned from 4.95 mM to 7 mM, with a comparatively decreased detection limit (1.46 mM). In order to enhance manufacturing accessibility, researchers have explored novel approaches, like microfluidic technology, which enables the regulation and sensing of complex fluids at the micro-scale. A decreased-fluid-consuming sensor built upon Pt-Ni NPs-MWCNTs/screen-printed CE was also introduced [79]. Additionally, microfluidic cloth-based analytical devices (mCADs) with multiple empty channels have been synthesized for enzyme-free glucose sensing [80]. The utilization of a method named photoelectrochemical (PEC) monitoring involved CdS quantum dots and poly (dimethyl diadly ammonium chloride)-functionalized MWCNTs, enabling the efficient and straightforward detection of glucose in saliva possessing a decreased detection limit (15.99 nM).
2.1.2. Sensing of DNA
Deoxyribonucleic acid (DNA) plays a crucial role as a genetic information carrier in organisms, and its analysis can aid in the timely identification and management of diseases [81]. Cancer, a major cause of mortality, benefits greatly from early detection, increasing the chances of successful treatment. For instance, urokinase plasminogen activator (uPA) serves as a biomarker for distinct categories of prostate cancer [82], while alpha-fetoprotein (AFP) is associated with liver cancer [83]. Colorectal cancer diagnosis could be facilitated through detecting the glycoprotein CEACAM5 [84]. However, conventional methods, like polymerase chain reaction (PCR), for the clinical detection of diseases have limitations, including the risk of contamination [85]. Consequently, there is a requirement for economical and exceptionally sensitive methods for biomolecule sensing in medical applications. CNT-based biosensors have emerged as promising candidates [86]. Over time, notable strides have been taken in the evolution of DNA sensors based on CNTs, employing both optical [87] and electrochemical techniques [88] in the medical field.
For instance, HIV is a contagious illness that devastates the immune system and carries a high fatality rate. Detecting the infection at an early stage can impede its progression. A wearable device utilizing a paper electrode MOF-based sensor exhibited remarkable tensile attributes and demonstrated promising prospects. Furthermore, it showcased excellent detection capabilities [89]. The detection range spanned from 10 nM to 1 mM, with an impressively low limit of detection (0.13 nM). The unexpected emergence of COVID-19 [90] and the escalating infection incidents have presented significant difficulties in swiftly identifying nucleic acids. By utilizing ss-DNA-modified SWCNTs, it binding with the RNA of the virus target becomes possible. Moreover, the ss-DNA can be substituted to capture other viruses, making it a versatile and adaptable solution [91].
CNTs were modified with extremely spinning Fe atoms to perform label-free cancer gene testing repeatedly [85][92]. A Pt-interdigitated electrode surface was coated with Au/Fe2O3 magnetic nanoparticles and CNTs prepared from two different types of nanoparticles for influenza virus detection using an altered DNA probe approach [93]. A label-free DNA sensor with a 3D spatial framework, consisting of four DNA types, was developed [94]. Furthermore, CNTs selectively integrate with DNA that was immobilized on an ITO electrode, and the detection of DNA was achieved using the electrochemical luminescence approach [95]. In this procedure, CNTs had a vital role in suppressing electrochemical reactions and augmenting electro-chemiluminescence signals. In order to surpass the complexity of fabricating CNTS electrodes on supporting matrixes, like polymer-based and glassy CEs, self-standing three-dimensional CNTs structures have been introduced. Prakash and his team recently developed a standalone CNTs aerogel electrode for exclusive DNA sensing, achieving a detection limit equal to 1 pM [89]. The as-mentioned electrode, imposed with a multi-directional CNT network, exhibited favorable electrical features, enabling increased sensitivity in DNA sensing. A sensor using AuNPs/Chit-MWCNTs/GCE was prepared with methylene blue as a procedure indicator [96]. CNTs/chitosan, Au nanoparticle thin films, and ss-DNA probes were successively altered on the surface of a glassy CE. Additionally, polymer- and MWCNT-modified electrodes were used in DNA, drugs, and interaction sensing by modifying double-stranded DNA (ds-DNA) [97].
Nano-biosensors utilizing a field-effect transistor (FET) offer remarkable advantages, including high label-free detection specificity. In this approach, the probe-ss-DNA is immobilized within the interval between the nanotube and the Au-gate, forming a double-stranded DNA structure when coupled with the target ss-DNA [98]. However, the substrate used in traditional FETs can hinder the sensing performance. To overcome this limitation, a suspended CNT FET was proposed, which separates the CNTs from the substrate and exhibits a decreased detection limit [99]. Liang and his team further enhanced the sensitivity by optimizing the FET structure, introducing a Y2O3 thin film between the Au nanoparticles and the CNT layers [100]. Additionally, nitrogen-containing CNTs with a chiral structure can detect DNA damage, which is associated with aging, gene mutation, and carcinogenesis, providing insights into pathogenesis [101]. Nanobridges developed by wrapping MWCNTs with elongated DNA strands can detect DNA damage resulting from various free radicals [102]. Another innovative approach is the CNT thin-film-transistor (CNT-TFT) sensor, which incorporates peptide electrodes at the ends of CNTs’ channels and functionalizes CNTs. This enables the quantitative detection of cancer cell genes, expanding the range of sensing and reducing the detection limit [103].
2.1.3. Sensing of Proteins and Amino Acids
Proteins, which are involved in various physiological activities, play a crucial role in body metabolism [104]. Despite the use of multiple techniques for protein detection, there is still a need for rapid and simple sensing methods [105][106]. Recently, CNTs have been utilized in point-of-care protein sensing, with the aim of increasing sensitivity and reducing the response time. Janssen and co-researchers designed a label-free biosensor based on CNTs to detect bovine serum albumin, which demonstrated a detection limit of 2.89 ng/mL [107]. This biosensor incorporates cellulose paper, SWCNTs, and BSA-specific antibodies, utilizing the principle of electrical percolation for the simplified and rapid quantification of BSA. Additionally, Huang et al. introduced an Fe3O4 nanoparticle–CNT-based biosensor for the visual detection of Rabbit IgG without complicated sample processing [108]. The biosensor employs Fe3O4 nanoparticles decorated on the surface of magnetized CNTs, which are altered with specific antibodies. This approach favored the immediate sensing of target proteins in whole blood possessing a low detection limit (10 ng/mL). Such low-cost and visual protein detection methods hold promise for clinical diagnosis. Furthermore, a redox probe-free electrochemical biosensor was developed by incorporating copper nanoparticle-doped CNTs as the electroactive film and a plastic antibody used as the bio-recognition constituent [109]. The biosensor eliminated the need for a redox probe-reading stage in conventional electrochemical detection. It exhibited a detection limit equal to 1.0 ng/mL for protein detection, and its application in serum samples suggests its potential for direct sample readings during in situ biomedical analysis.
CNT-FET biosensors employing the chemo-dosimetric detection principle have been fabricated for cysteine’s detection by [110]. The molecular interaction among the selective chemo-dosimeter and cysteine induces a chemical alteration, affecting the surficial distribution of charge of CNTs and triggering electrical responses in the CNT-FET. The acquired data demonstrate that the as-mentioned biosensor enabled label-free, selective, and highly sensitive cysteine sensing (LOD = 0.45 fM). Additionally, the interaction between serine amino acid and CNTs was investigated [111]. The chirality of serine amino acid significantly influences CNTs’ dielectric features, proposing the CNT-based sensors’ potential to discriminate chiral molecules. Furthermore, a dual-signal electrochemical biosensor was constructed using chiral CNTs coupled with platinum nanoparticles and cyclodextrin [112]. This biosensor exhibited the ability to differentiate among amino acids, specifically between tyrosine and tyrosine, presenting the limits of detection equal to 0.107 nM and 0.133 nM, respectively.
2.1.4. Sensing of Neurotransmitters
Neurotransmitters like dopamine, acetylcholine, and norepinephrine are crucial chemical components involved in cell-to-cell communication [113]. Acetylcholine, for instance, is related to the nervous system and is linked to the increased threat of Alzheimer’s disease [114]. Epinephrine contributes to regulating blood pressure and heart function [115]. Dopamine serves as a hormone and neurotransmitter, and insufficient levels of it can cause conditions like Parkinson’s disease and mental dysfunctions. Typically, neurotransmitters are absent in isolation within the body but exist alongside other biomolecules like ascorbic acid, uric acid, and tyrosine [116], which are crucial for preserving normal physiological function. To enable the simultaneous sensing of dopamine, ascorbic acid, and uric acid without interference, a green reduction process was employed using Pelargonium graveolens extract to reduce hydroxylated MWCNTs [117], followed by hybridization with Pb nanotubes. The detection platform involved modifying a graphite electrode through the hybridization of reduced carboxylated MWCNTs and Pd nanotubes.
Guan and his team conducted a hydroxylation process on both SWCNTs and MWCNTs in order to acquire the MWCNT-COOH/SWCNT-OH-modified glassy CE sensor [118]. The arrangement of the two types of nanotubes in a staggered distribution created a simple grid structure, which facilitated electron mobility. Metal-based electrodes incorporating CNTs are commonly used in electrochemical sensors. For instance, GC/Ni-CNT electrodes have been employed to sense dopamine and adrenaline [119]. PPy has been extensively utilized in biosensor fabrication, and in this case, sodium dodecyl sulfate was used to modify the sensor, resulting in the overoxidized PPy/sodium dodecyl sulfate-modified MWCNT electrode [120]. This sensor demonstrated high sensitivity in detecting DA while minimizing interference from glucose and other substances. Another innovative approach involved the development of a molecularly imprinted polymer (MIP)/MWCNT/graphene aerogel composite electrode (MIP/MWCNT/GAs/GCE), where the MIP was created through electro-polymerization of pyrrole [121]. Initially, MWCNT/graphene aerogels were synthesized, while the resulting mixture was applied onto the glassy CE surface using drop casting. Furthermore, a novel type of graphene quantum dots was fabricated from glucose [122], and these dots were integrated with modified MWCNTs to enable the sensing of dopamine. The linear detection range was 250 nM–250 mM, with a detection limit equal to 95 nM. Gupta et al. suggested a CNT array-based method for dopamine detection, utilizing increasingly densified CNTs fibers [123]. The range of detection of this approach was 1 nM–100 mM, with an LOD of 32 pM.
Uric acid and dopamine are present alongside ascorbic acid in living organisms, making their detection crucial. Electrochemical techniques are extensively employed in neurotransmitter detection because of their benefits, including heightened sensitivity, rapid response, and cost effectiveness [124]. To detect dopamine and uric acid, a sensitive and selective biosensor was developed using Au nanoparticle-loaded MWCNTs altered with PCA [125]. Serotonin, comprising an essential gastrointestinal neurotransmitter, has also garnered significant attention in medical and life sciences. A remarkably sensitive electrochemical sensor for serotonin sensing was developed by using Cu2O encapsulated in CNTs with platinum nanoparticles [126]. The as-described sensor exhibited exceptional electro-catalytic performance towards serotonin oxidation, achieving a low detection limit (3 nM). Furthermore, MWCNTs modified with capsaicin were employed to alternate glassy CEs towards the quantitative sensing of xanthurenic acid, dopamine, epinephrine, ascorbic acid, and uric acid at a micromolar level [127]. A ternary nanocomposite of N-doped CQDs@Fe2O3/MWCNTs enabled the sensing of uric acid and xanthine, characterized by detection limits equal to 0.106 mM and 0.092 mM, respectively [128]. The practical applicability of this nanocomposite was demonstrated in a human urine sample.
2.2. CNTs for Drug Delivery
CNTs have gained tremendous attention as an efficient means of transferring several drug molecules into vital cells due to their unique structure, which facilitates non-intrusive permeation across biological membranes [129]. Typically, drug molecules are anchored to CNT sidewalls either through covalent or non-covalent bonds with functionalized CNTs [130]. Nevertheless, each of these methods comes with its own set of benefits and limitations. Covalent interactions provide stability to the drug-loaded CNTs both outside and inside cells, but they lack controlled drug release within the cancer cells’ cellular micro-environment that comprises a limitation of the drug delivery system. On the other hand, non-covalent interactions allow for gradual drug release in the acidic tumor sites’ conditions but suffer from robustness issues at extracellular pH values [131]. Therefore, utilizing the inner hollow cavity of CNTs for drug loading offers an optimal solution by providing drug isolation from the physiological environment. To overcome these challenges related to drug release, researchers have investigated various external stimuli, either individually or in combination, including temperature, electric field, and light. For instance, a chitosan-modified CNT with a thermosensitive polymer (NIPAAm) and NIPAAm-co-Vim was developed and encapsulated in bovine serum albumin at body temperature (37 °C) to evaluate the temperature-responsive discharge of biomolecules. Bovine serum albumin was released just above the lower critical solution temperature of polyVIm (38–40 °C) [18][132].
Shi and co-researchers [133] employed an electric field to release ibuprofen from a hybrid hydrogel that consisted of sodium alginate, bacterial cellulose, and MWCNTs. Estrada and his team [134] investigated the temperature and NIR light-responsive discharge of methylene blue from a hydrogel composed of MWCNTs and k-carrageenan. CNTs have been utilized as carriers for a range of drugs, such as doxorubicin, paclitaxel, docetaxel, and oxaliplatin, in both in vitro and in vivo cancer therapy. Modified CNTs for drug delivery purposes have been thoroughly researched, developing strategies to enhance water solubility and entrap drug molecules [135]. Jain and Mehra, in 2015, compared the cancer-targeting abilities of doxorubicin-loaded folic acid and estrone-anchored PEG-modified MWCNTs in tumor-bearing mice, showing improved survival with the use of DOX/ES-PEG-MWCNTs [136]. Calcium phosphate-crowned MWCNTs as nanocapsules for intracellular drug transfer have been also explored, demonstrating pH-stimulated dissolution and controlled discharge within lysosomes [137]. Xu and co-researchers [138] developed an amine-PEG-modified polydopamine-CNT nanosystem for drug transfer to enhance the biocompatibility of CNTs. Finally, Picaud et al. [139] carried out theoretical investigations on the loading and discharge of cisplatin from CNTs.
2.3. CNTs for Cancer Treatment
Since cancer remains a major health issue, several studies have focused on increasing the understanding of the molecular mechanisms that mediate tumorigenesis [140], oxidative-stress [141], and cellular senescence and aging [142], and others have tried to provide alternative therapeutic approaches [15][16][17][18].
CNTs possess versatile properties that make them highly valuable in biomedical applications. They are extensively utilized as carriers for anticancer drugs, genes, and proteins in chemotherapy, making them a promising option [143]. Additionally, their enhanced absorption of NIR light enables effective photothermal therapy. Su and his team [144] synthesized iRGD-PEI modified MWCNTs, which were then conjugated with candesartan. This functionalized nanocomposite targeted αvβ3-integrin and AT1R receptors on tumor endothelium and lung cancer cells, respectively. The combination of candesartan with [pAT (2)] resulted in the downregulation of VEGF and the effective inhibition of angiogenesis [144].
Another study by Zhang and his team [19] involved the design of a nanocomposite consisting of DOX-loaded MWCNTs and magneto-fluorescent CQDs for combined chemo- and photothermal therapy [19]. The negatively charged GdN@CQDs-MWCNTs enabled the bonding with positively charged DOX molecules. This nanocomposite exhibited strong NIR irradiation absorption. In in vivo photothermal therapy experiments, laser irradiation of tumor sites led to a temperature increase of 51.8 °C in the mice provided with GdN@CQDs-MWCNTs/DOX-EGFR, while the control group showed no significant change in temperature. The localized heating effect facilitated the release of DOX and led to successful photothermal therapy, as demonstrated by a reduction in the tumor volume.
In a recent study, a nanosystem consisting of TAT-chitosan functionalized MWCNTs loaded with DOX was utilized by Dong et al. [145] to combine chemo and photothermal therapy. To promote apoptosis in cancer cells, a PEG-coated CNT-ABT737 nanodrug was employed, which targeted the mitochondria [146]. The discharge of the nanodrug in the cytosol induced apoptosis in lung cancer cells by disrupting the mitochondrial membrane. In vivo, this approach showed impressive therapeutic effectiveness, further enhanced by the localized heating effect triggered by NIR light irradiation. Another development involved an Au nanoparticle-coated CNT ring that exhibited exceptional Raman and optical signal characteristics, leading to enhanced photoacoustic signal and photothermal conversion behavior [147]. The as-mentioned composite indicated promising results in image-guided cancer therapy. Additionally, the photothermal cancer-killing efficacy was improved by the surface plasmon resonance absorption of Au in SWNT-Au-PEG-FA nanomaterials [148].
2.4. CNTs for Reinforcing Tissue Engineering Scaffolds
The utilization of CNTs in composite reinforcements of tissue engineering scaffolds has predominantly emphasized increasing their mechanical attributes [149]. Traditional scaffold materials, like hydrogels and fibrous scaffolds, are commonly pliable to emulate the inherent rigidity of tissues, resulting in decreased structural robustness and support. However, by introducing CNTs into the as-mentioned materials, their mechanical properties can be significantly improved. For instance, Shin et al. conducted a study where they incorporated CNTs into gelatin hydrogels, resulting in notable enhancement of tensile strength [150]. Similarly, Sen and his team illustrated the enhanced tensile strength of CNTs-reinforced polystyrene and polyurethane fibrous membranes [151]. Lately, researchers have explored the versatile capabilities of CNTs in tissue scaffold engineering, particularly their ability to confer electrical conductivity. Most biomaterials utilized in tissue engineering are electrically insulating due to their composition of non-conductive polymers [152][153][154]. Nevertheless, specific applications, like neural and cardiac tissues, require conductive scaffolds to efficiently transfer electrical signals and support optimal electro-physiological operations. For instance, Kam and co-researchers conducted experiments where they employed electrical stimulation to neural stem cells cultured on CNT–laminin composite thin films, resulting in increased activity potential and the functional neural networks’ differentiation [153]. Similarly, Shin and his team cultured cardiomyocytes on CNT-reinforced gelatin hydrogels, which exhibited increased electro-physiological efficiency and eventually produced functional cardiac tissue [155]. The aforementioned studies demonstrate the capability of CNTs to successfully supply electrical conductivity to previously non-conductive biomaterials.
2.5. Cytotoxicity of CNTs
Although CNTs have been successfully applied in biomedical engineering, there is a growing apprehension regarding their harmlessness. Several contemporary in vitro studies have documented the elevated cytotoxicity of CNTs, which can be attributed to factors such as cellular uptake, aggregation, and induced oxidative stress [156][157][158]. The contradictory results regarding the CNTs’ biocompatibility primarily arise from variations in their properties (such as size, surficial characteristics, and functionalization), as well as the different testing protocols employed (including in vitro and in vivo studies, cell type, tissues, and animals tested). Furthermore, the insufficient elimination of metal catalysts utilized during CNT preparation has been associated with increased cytotoxicity [157]. The majority of in vivo studies have indicated the minimal toxicity of CNTs and their clearance through renal pathways, although some accumulation in organs like lungs, liver, and spleen has been observed, potentially leading to inflammation [156][157][158]. Nevertheless, cytotoxicity appears to demonstrate increased fluctuation and significance at the cellular level, as indicated by numerous in vitro cell culture studies [159][160]. Given the expanding use of CNTs in diverse biomedical fields, it is crucial to conduct comprehensive biological evaluations considering the various chemical and physical properties of CNTs to ascertain their pharmaco-kinetics, cytotoxicity, and optimized dosages. Nonetheless, a plethora of research studies have demonstrated that functionalizing CNTs utilizing biocompatible polymers or surfactants can effectively minimize toxicity and prevent aggregation [161][162].
3. Graphene and Graphene Oxide
Graphene, a novel nanomaterial, has gained significant attention in recent years. Geim and Novoselov’s groundbreaking research introduced a facile technique for isolating graphene from graphite through exfoliation and uncovered its remarkable electrical attributes [163][164]. While graphene and CNTs share comparable electrical, optical, and thermal characteristics, the unique 2D graphene’s atomic structure allows for more wide-ranging electronic properties. Notably, the presence of the quantum Hall effect and massless Dirac fermions accounts for its low-energy charge excitation in ambient conditions, as well as the optical transparency across the NIR and visible light spectrum. Furthermore, graphene exhibits structural strength combined with exceptional flexibility, making it an appealing choice for producing thin and flexible materials [165][166].
It was expected that those characteristics would be exploited in biomedical applications, and thus graphene and graphene oxide are used in the sensoring of nucleic acids, as a basic component of drug delivery systems, focusing on cancer treatment. Their use in biomedicine is not as wide as that of CNTs, and concerns about their toxicity still limit their biomedical applications.
3.1. Graphene and Graphene Oxide as Biosensors
Graphene oxide exhibits dynamic interactions with the probe and can transduce specific responses towards target molecules. This transmission procedure utilizes fluorescence, Raman scattering, as well as electrochemical reactions. As a result, graphene oxide is widely employed as a biosensor [146]. Graphene-based nanomaterials have been intensively utilized towards the electrochemical detection of single- and double-stranded DNA with high selectivity [167]. The superior graphene’s electrochemical features, coupled with the robust ionic interaction among the negatively charged -COOH groups and positively charged nucleobases, as well as the sturdy π-π stacking among nucleobases and the carbon structure, contribute to its exceptional sensitivity. Reduced graphene nanowires have been introduced as biosensors for detecting DNA bases through the oxidation signals of individual nucleotide bases [168]. The as-mentioned nanowires exhibited remarkable robustness, presenting only a 15% variation in oxidation signals during the increment of the differential pulse voltammetry up to 100 scans. Lately, Cheng and colleagues developed -COOH-modified graphene oxide and polyaniline-altered graphene oxide to effectively detect DNA through differential pulse voltammetry within the range of 1 × 10−6–1 × 10−14 [169]. Moreover, Ping and colleagues developed a label-free DNA biosensor by employing graphene field-effect transistors that were modified with single-stranded probe DNA. The as-described ultra-sensitive biosensor demonstrated an extensive analytical range possessing a limit of detection equal to1 fM for 60-mer DNA oligonucleotides [170]. In another study, researchers fabricated a device using physical vapor deposition, incorporating Au nanoparticle-decorated graphene field-effect transistors. Thiol-modified Au NP-Gr-FETs successfully detected DNA (LOD = 1 nM) and presented increased specificity of non-complementary DNA [171]. Furthermore, a single-layer graphene-based FET biosensor achieved the detection of extremely low DNA concentrations (10 fM) [172]. Kim and his team [173] synthesized a graphene surface-modified vertically aligned Si nanowire for the sensitive and selective detection of oligonucleotides.
They initiated the surficial attachment of oligonucleotides onto the silicon nanowire arrays, which then underwent hybridization with the probe, leading to an amplification of the biosensor’s response. The biosensor current was observed to increase from 19% to 120% as the DNA concentration ranged from 0.1 nM to 500 nM [173]. The process of single-and double- stranded DNA being absorbed and desorbed on graphene oxide was investigated by Park and co-researchers [174]. It was found that single-stranded DNA exhibited a higher affinity for graphene oxide, while double-stranded DNA showed lower affinity. Additionally, recent studies have indicated that the adsorption of DNA on graphene oxide depends on the length of the DNA molecules [175]. Prabowoa and his team [176] proposed a novel approach for the sensing of Mycobacterium tuberculosis DNA hybridization utilizing graphene integrated onto a surface plasmon resonance-sensing chip. The utilization of graphene oxide-based nanomaterials towards glucose detection has been rapidly advancing [177]. A device incorporating graphene electrodes with glucose oxidase demonstrated excellent selectivity and increased glucose sensitivity, characterized by a low limit of detection (0.5 mM) [178]. Reduced graphene oxide with C6H5(CH2)3COOH as a glucose-binding linker has been also developed. The as-proposed sensor device exhibited the capability to detect glucose levels ranging from 1 to 4 mM [179]. In addition, researchers have successfully developed a more durable and reusable graphene-bismuth composite device that demonstrated impressive glucose detection capabilities. It covered a broad linear range (1–12 mM) with an enhanced sensitivity and a reduced limit of detection (0.35 mM) [180]. A carbon-functionalized graphene/fullerene C60 composite was successfully fabricated for glucose detection (0.1–12.5 mM), with an LOD = 35 µM and enhanced sensitivity [181]. Hydroxyapatite 1D nanorods on a graphene nanosheet-altered glassy CE were fabricated, demonstrating excellent sensing capabilities across a broad concentration range (0.1–11.5 mM), with an LOD = 0.03 mM and increased sensitivity [182].
3.2. Graphene and Graphene Oxide for Drug Delivery
Graphene, with its enhanced specific surface area and abundant π electrons, has proven to be a suitable carrier for drug delivery. Wang and his team [183] grafted a substantial amount of doxorubicin onto graphene coated with a phospholipid monolayer and observed sustained release of DOX, particularly at extremely low pH values as compared to a basic pH [184]. Another approach involved loading DOX onto a graphene sheet through physisorption, accompanied by surficial alteration with PEG-NH2 to increase robustness and compatibility within a biological environment [185]. Nandi and co-researchers successfully loaded both DOX and indomethacin onto PNIPAM (PolyN-isopropylacrylamide)-grafted graphene oxide using π-π interactions, H-bonding, and hydrophobic interactions [186]. PNIPAM was covalently grafted onto graphene oxide through free radical polymerization to achieve the controlled discharge of DOX, particularly in an acidic pH environment, where increased hydrophilicity, higher DOX solubility , and minimized hydrogen bonding interactions were observed between the DOX and the grafted graphene oxide surface. Xu and his team [187] loaded paclitaxel onto graphene oxide-PEG through π-π stacking and hydrophobic interactions, achieving a loading efficiency of 11.2 wt%.
Zhao and co-researchers [188] developed well-defined PMMA-coated polyethylene glycol-functionalized graphene oxide nanoparticles, which displayed outstanding dispersion in PBS solution and acted as a proficient drug delivery system. The PMMA coating enabled the controlled release of DOX, with reduced release in healthy tissues and accelerated discharge in tumor tissues upon the presence of glutathione as a reducing agent. In another study, DOX-loaded graphene oxide was synthesized, followed by alteration with hyaluronic acid to enhance stability and serve as a targeting agent for the HAGO-DOX nanohybrid [189]. Recently, Mahdavi et al. [190] conducted simulation research on the loading and release of DOX in graphene oxide at several pH values. A DOX-loaded PEI, biotin, β-Cyclodextrin-conjugated, reduced-graphene oxide nanosystem was also developed, where the PEI and biotin contributed to increased robustness and targeting effectiveness, respectively. The β-Cyclodextrin served as a host molecule to accommodate guest molecules, including water-insoluble anticancer drugs [191].
3.3. Graphene and Graphene Oxide for Cancer Treatment
The unique features of graphene and graphene oxide also render them ideal candidates for various cancer therapy applications [177]. Yu and his team developed GO functionalized with αvβ6-targeting peptide and coated with the photosensitizer HPPH. This functionalized graphene oxide triggered dendritic cells and efficiently hindered tumor growth and lung metastasis via promoting the infiltration of cytotoxic CD8+ T lymphocytes into tumors [192]. A combination of reduced graphene oxide and gold nanorods loaded with doxorubicin (DOX) was engineered as a vehicle for simultaneous photothermal therapy and chemotherapy. The discharge of DOX was significantly enhanced by the photothermal heating effect induced by NIR light and the acidic conditions within the micro-environment of the tumor [193]. The compact arrangement of Au nanoparticles on graphene oxide resulted in an increased absorption peak. Under laser light, the Au nanoparticle–graphene oxide system exhibited a maximum temperature enhancement equal to 23.2 °C [132]. Cheon and his team [194] proposed a novel approach for combined chemo- and photothermal therapy for brain tumors using a DOX-loaded bovine serum albumin-modified graphene sheet.
Su and co-researchers [195] devised a novel material comprising sponge-like carbon grafted with dual chemotherapeutics and supported lipid bilayers on graphene nanosheets altered with a tumor-targeting protein. The as-described composite exhibited remarkable accumulation at the tumor site, resulting in the effective suppression of xenograft tumors within 16 days.
Shao and his team [196] fabricated a polydopamine-functionalized reduced graphene oxide coated with mesoporous silica and subsequently modified with hyaluronic acid to facilitate DOX loading. This system demonstrated pH-dependent and NIR-triggered release of DOX, making it an efficient chemo-photothermal agent. In a study by Dai and co-researchers [197], a smart material composed of TiO2-MnOx-conjugated graphene composite was synthesized for tumor elimination purposes.
3.4. Cytotoxicity of Graphene and Graphene Oxide
Graphene and graphene oxide, similar to CNTs, have limitations in their biomedical application given their potential toxicity. Ou and his team [198] extensively discussed graphene’s toxicity in various organs. Numerous studies have examined graphene’s toxicity in both animals and cells [199]. Several factors, including the concentration, size, surface properties, and functional groups, have been identified as crucial in influencing its toxicity in biological systems [200]. Li and co-researchers [201] reported that when GLC-82 cells were incubated with graphene oxide (100 mg/L) for 24 h, it induced the development of reactive oxygen species (ROS) and resulted in toxicity. To mitigate the noxious effects of graphene oxide in distinct biomedical applications, researchers have explored the modification of graphene sheets by coating them with biological molecules, as demonstrated in a study on reducing the toxic effects of graphene through a blood protein coating [202].
4. Graphene Quantum Dots (GQDs)
Quantum dots (QDs) are semiconducting nanoparticles with unique properties, and there is increasing interest for their various applications [203]. Graphene quantum dots (GQDs) are a very promising material in nanomedicine since they combine high biocompatibility and low cytotoxicity in parallel with fluorescent potential. Thus, they are ideal in diagnostics and therapeutics, and recently, in theragnostics.
4.1. GQDs as Biosensors
In recent times, considerable advancements have been made in the field of biosensors, particularly in the utilization of graphene quantum dots (GQDs), which can detect biomacromolecules with high selectivity and sensitivity. GQDs exhibit outstanding chemical and optical properties, such as photoluminescence and electro chemiluminescence, which are key properties for biosensors used in clinical analysis and diagnosis [203]. Researchers [204] have developed a DNA detection approach relying on the fluorescence sensing technique called Fourier resonance energy transfer (FRET), using reduced graphene quantum dots (GQDs) functionalized with DNA probes. They created a conjugate of phosphorylated peptide-GQD coordinated with Zr4+, which was capable of detecting casein kinase II (CK2) at concentration as low as 0.03 mL−1 [205]. Another group [19] developed a glucose sensor by fabricating graphene quantum dots (GQDs) with pyrene-1-boronic acid. They observed a shift in Dirac voltage as they increased the concentration of pyrene-1-boronic acid, which indicated there was a strong relation between the PBA concentration and glucose sensitivity. Another reason that this functionalization proved to be successful was the increase in the relative capacitance with higher glucose concentrations. In a separate study, an electro-chemi-fluorescent nanofiber composed of polyvinyl alcohol (PVA) and GQDs was created for the highly sensitive and selective detection of both H2O2 and glucose [178]. The mechanism relies on the incorporation of glucose oxidase into the composite, leading to the generation, which can be detected by the fluorescent GQD. Xi and colleagues designed a hybrid material based on N-doped GQDs and palladium nanoparticles for the detection of many different cancer cells [206]. These types of sensors can become instrumental in the early detection of cancer.
4.2. GQDs for Drug Delivery
Very often, drug developers face challenges when designing new drug molecules due to their bad stability in the body and also the need for more targeted delivery. Graphene quantum dots are being investigated as potential drug delivery systems thanks to their unique properties, such as extremely small size and an oxygen-rich surface. Also, GQDs are fluorescent, which allows for monitoring the delivery of the drug to the targeted tissue [207]. As a result, there are numerous studies using GQDs for drug delivery. For instance, Tian and his team created a zeolite imidazolate framework embedded with graphene quantum dots as a delivery system for doxorubicin. The system released the drug only in acidic conditions, which enabled the targeted release of this anticancer drug to the low pH environment of tumor sites [208]. In another method, intracellular drug delivery and the real-time monitoring of drug release were achieved through DOX-loaded aptamer/GQD-capped fluorescent mesoporous silica nanoparticles. The system can be triggered by the adenosine triphosphate molecule, which is present in high doses in cancer cells and releases the loaded anticancer drug, providing targeted release and monitoring [178]. Leveraging the notable physicochemical properties of GQDs, Wei and coworkers developed a graphene quantum dot system conjugated with Cy5.5 dye using a cathepsin D-responsive peptide for the delivery of doxorubicin [209]. They reported improved therapeutic performance both in laboratory tests (in vitro) and live subjects. The result was attributed to better tissue penetration and high cellular uptake. In their study, they used confocal laser scanning microscopy (CLSM). They detected blue fluorescence from the cells that were treated with the conjugate, while the absence of visible fluorescence from Cy5.5 was promising for the biocompatibility of the conjugate. The release of the therapeutic molecule was detected thanks to its green fluorescence. This signal can be used in vivo to monitor the release of the anticancer drug into the cancer tissue. Nigam and his team (2014) designed a targeted drug delivery nanocarrier, which included GQD-conjugated gemcitabine-loaded human serum albumin (HSA). Albumin facilitated the transportation of gemcitabine to tumor cells using the gp60 pathway [210]. In another study, Iannazzo and his team [211] designed a different nano-system for targeted anticancer drug delivery using biotin-conjugated graphene quantum dots and loaded them with doxorubicin. Sui and coworkers created a nanoconjugate of cisplatin and GQD for cancer treatment. GQDs that facilitated cellular uptake and cisplatin that interacted with DNA, improving nuclear uptake, have also been used [212]. Wang and his team designed a GQD-folic acid nanocomposite loaded with doxorubicin. They reported successful targeting of the cancer tissue, high cellular uptake, and easy monitoring of the process [213].
4.3. GQDs for Cancer Treatment
Graphene quantum dots exhibit many properties that make them ideal for use in nanomedicine. They are stable in the environment of the body and non-toxic. They can help improve the hydrophilicity of other molecules and have surface functional groups that facilitate interactions with other molecules. Their strong fluorescence is another important factor that makes them very promising for many therapeutic applications, especially cancer treatment [210]. Lee and his team loaded the chemotherapy drug curcumin into graphene quantum dots in order to improve its hydrophilicity [214]. Ge and coworkers focused on the potential of singlet oxygen generation by graphene quantum dots, aiming to create an effective sensitizer for photodynamic therapy [215]. They successfully created graphene quantum dots smaller than 6 nm. They added norpempidine and studied the generation of (1O2) under irradiation. They also used a scavenger for singlet oxygen to prove that no other reactive oxygen species were generated. Furthermore, no significant diffusion of GQDs was observed at the injection site. In vivo studies in mice using this method resulted in regression of the tumors after a few days.
It is also possible to use magnetic nanoparticles to generate heat under an alternating magnetic field in order to kill cancer cells. Yao and his team used a composite of graphene quantum dots and magnetic silica nanoparticles to study this effect [216]. Near-infrared radiation can be used to amplify this effect. A different group used folic acid to functionalize graphene quantum dots and then loaded them with IR780. They used a NIR laser to target tumors in mice, which had been given the drug. After a few minutes, the temperature of the target was increased, which had a significant effect on the cancer cells. The tumors almost disappeared after two weeks [217].
In their thorough review, Ailuno and her colleagues indicated the potential of quantum dots and boron carbide nanoparticles in cancer treatment. Boron carbide(B4C) was investigated as a potential material for boron neutron capture therapy (BNCT) agents due to its high neutron absorptivity. Several studies have explored different approaches to functionalize B4C nanoparticles (NPs) for targeted BNCT applications. These studies collectively demonstrate the promising features of modified B4C NPs and boron-doped quantum dots in targeted BNCT applications, providing promising avenues for cancer therapy [218].
4.4. GQDs in Theragnostic Systems
GQDs can be utilized for bioimaging and sensoring for early disease diagnosis as well as in drug delivery systems targeting particularly cancer cells. Since GQDs can be quite easily functionalized, many studies have investigated their potential use in theragnostic systems [219].
The functionalization of GQDs can be maintained during the synthesis process or the molecular binding step, and it generally decreases their toxicity, thus improving their biocompatibility and increasing their internalization and endocytosis [219]. Polymers (e.g., PEG), lipid-like materials, and peptides can be used to increase biocompatibility and internalization [220]. In other studies, therapeutic monoclonal antibodies have been attached to GQDs for advanced immunotherapy through transmembrane receptors [221] and drugs such as DOX for efficient chemotherapy [222]. Their fluorescent properties make them desirable for bioimaging and simultaneous targeting.
4.5. Cytotoxicity of GQDs
Graphene quantum dots (GQDs) have gained significant attention for biological applications due to their unique properties in relation to other carbon nanomaterials. However, understanding the toxicity of graphene quantum dots is crucial, since they can defer from other carbon nanomaterials that have been studied, and thus, new research is required. The research suggests that there are many factors that influence the toxicity of carbon nanodots. A key factor is their size, and, in this case, it seems that their incredibly small diameter is an advantage over other carbon nanomaterials. The research on cell viability by Wang and coworkers proved that toxicity is very low when the nanodot size is under 10 nm [223]. As for any substance, the concentration is extremely important when accessing the toxicity of nanomaterials. In this area, there have been some conflicting results, indicating that more research is needed. Simulations provided some theoretical results based on size and concentration [224]. The simulations showed that graphene quantum dots with smaller sizes had the ability to penetrate the POPC membrane [225]. The presence of GQDs affected the thickness of the POPC lipid membrane by their permeation. Larger graphene quantum dots were only absorbed by the membrane. Something else to consider when assessing toxicity are the functional groups at the surface of graphene quantum dots. The reports again vary. Studies on hydroxylated graphene quantum dots, by one group, showed low cell viability for A549 and H1299 cells [226]. At the same time, a different group did not observe any significant toxic effects on various cancer cells (KB, MDA-MB231, A549) or normal cell lines (MDCK). They followed these results with in vivo studies that focused on any long-term effects, and again, did not find any significant damage [227]. Unfortunately, clear information concerning the influence of various functional groups on GQD toxicity is still lacking in the available literature.
This entry is adapted from the peer-reviewed paper 10.3390/cryst13081236
References
- Pierson, H.O. Handbook of Carbon, Graphite, Diamond, and Fullerenes: Properties, Processing, and Applications; Noyes Publications: Park Ridge, NJ, USA, 1993.
- Krüger, A. Carbon Materials and Nanotechnology; Wiley-CH: Weinheim, Germany, 2010.
- Maiti, D.; Tong, X.; Mou, X.; Yang, K. Carbon-Based Nanomaterials for Biomedical Applications: A Recent Study. Front. Pharmacol. 2019, 9, 1401.
- Patel, K.D.; Singh, R.K.; Kim, H.-W. Carbon-based nanomaterials as an emerging platform for theranostics. Mater. Horiz. 2019, 6, 434–469.
- Shi, X.; Meng, H.; Sun, Y.; Qu, L.; Lin, Y.; Li, Z.; Du, D. Far-red to near-infrared carbon dots: Preparation and applications in biotechnology. Small 2019, 15, e1901507.
- Zhang, J.; Zhang, H. Emerging carbons. Adv. Mater. 2019, 31, e1808208.
- Wang, H.T.; Mi, X.Y.; Li, Y.; Zhan, S.Z. 3D graphene-based macrostructures for water treatment. Adv. Mater. 2020, 32, e1806843.
- Lin, H.; Sturmberg, B.C.P.; Lin, K.T.; Yang, Y.; Zheng, X.; Chong, T.K.; de Sterke, C.M.; Jia, B. A 90-nm-thick graphene metamaterial for strong and extremely broadband absorption of unpolarized light. Nat. Photonics 2019, 13, 270–276.
- Liu, H.; Qiu, H. Recent advances of 3D graphene-based adsorbents for sample preparation of water pollutants: A review. Chem. Eng. J. 2020, 393, 124691.
- Yang, X.; Wan, Y.; Zheng, Y.; He, F.; Yu, Z.; Huang, J.; Wang, H.; Ok, H.S.; Jiang, Y.; Gao, B. Surface functional groups of carbon-based adsorbents and their roles in the removal of heavy metals from aqueous solutions: A critical review. Chem. Eng. J. 2019, 366, 608–621.
- Song, P.; Liu, B.; Liang, C.; Ruan, K.; Qiu, H.; Ma, Z.; Guo, Y.; Gu, J. Lightweight, flexible cellulose-derived carbon Aerogel@Reduced graphene oxide/PDMS composites with outstanding EMI shielding performances and excellent thermal conductivities. Nanomicro Lett. 2021, 13, 91.
- Tang, T.; Liu, D.; Wang, Y.J.; Cui, L.; Ignaszak, A.; Yu, Y.; Zhang, J. Research advances in biomass-derived nanostructured carbons and their composite materials for electrochemical energy technologies. Prog. Mater. Sci. 2021, 118, 100770.
- Auffan, M.; Rose, J.; Bottero, J.-Y.; Lowry, G.V.; Jolivet, J.-P.; Wiesner, M.R. Towards a definition of inorganic nanoparticles from an environmental, health and safety perspective. Nat. Nanotechnol. 2009, 4, 634–641.
- Tremi, I.; Havaki, S.; Georgitsopoulou, S.; Lagopati, N.; Georgakilas, V.; Gorgoulis, V.G.; Georgakilas, A.G. A Guide for Using Transmission Electron Microscopy for Studying the Radiosensitizing Effects of Gold Nanoparticles In Vitro. Nanomaterials 2021, 11, 859.
- Katifelis, H.; Nikou, M.-P.; Mukha, I.; Vityuk, N.; Lagopati, N.; Piperi, C.; Farooqi, A.A.; Pippa, N.; Efstathopoulos, E.P.; Gazouli, M. Ag/Au Bimetallic Nanoparticles Trigger Different Cell Death Pathways and Affect Damage Associated Molecular Pattern Release in Human Cell Lines. Cancers 2022, 14, 1546.
- Gatou, M.-A.; Lagopati, N.; Vagena, I.-A.; Gazouli, M.; Pavlatou, E.A. ZnO Nanoparticles from Different Precursors and Their Photocatalytic Potential for Biomedical Use. Nanomaterials 2023, 13, 122.
- Lagopati, N.; Kotsinas, A.; Veroutis, D.; Evangelou, K.; Papaspyropoulos, A.; Arfanis, M.; Falaras, P.; Kitsiou, P.V.; Pateras, I.; Bergonzini, A.; et al. Biological Effect of Silver-modified Nanostructured Titanium Dioxide in Cancer. Cancer Genom. Proteom. 2021, 18 (Suppl. S3), 425–439.
- Papadopoulou-Fermeli, N.; Lagopati, N.; Pippa, N.; Sakellis, E.; Boukos, N.; Gorgoulis, V.G.; Gazouli, M.; Pavlatou, E.A. Composite Nanoarchitectonics of Photoactivated Titania-Based Materials with Anticancer Properties. Pharmaceutics 2023, 15, 135.
- Zhang, D.-Y.; Zheng, Y.; Tan, C.-P.; Sun, J.-H.; Zhang, W.; Ji, L.-N.; Mao, Z.-W. Graphene oxide decorated with Ru(II)–polyethylene glycol complex for lysosome-targeted imaging and photodynamic/photothermal therapy. ACS Appl. Mater. Interfaces 2017, 9, 6761–6771.
- Huang, C.H.; Zhao, Y.L.; Li, Y.L. Graphdiyne: The fundamentals and application of an emerging carbon material. Adv. Mater. 2019, 31, e1904885.
- Lopez Barreiro, D.; Martin-Moldes, Z.; Yeo, J.; Shen, S.; Hawker, M.J.; Martin-Martinez, F.J.; Kaplan, D.L.; Buehler, M.J. Conductive silk-based composites using biobased carbon materials. Adv. Mater. 2019, 31, e1904720.
- Panwar, N.; Soehartono, A.M.; Chan, K.K.; Zeng, S.; Xu, G.; Qu, J.; Coquet, P.; Yong, K.T.; Chen, X. Nanocarbons for biology and medicine: Sensing, imaging, and drug delivery. Chem. Rev. 2019, 119, 9559–9656.
- Wang, H.; Li, S.; Wang, Y.; Wang, H.; Shen, X.; Zhang, M.; Lu, H.; He, M.; Zhang, Y. Bioinspired fluffy fabric with in situ grown carbon nanotubes for ultrasensitive wearable airflow sensor. Adv. Mater. 2020, 32, e1908214.
- Juliette, S.; Emmanuel, F.; Muriel, G. Overview of carbon nanotubes for biomedical applications. Materials 2019, 12, 624.
- Lagopati, N.; Efstathopoulos, E.P.; Veroutis, D.; Katifelis, H.; Theocharous, G.; Pantelis, P.; Evangelou, K.; Gorgoulis, V.G.; Gazouli, M. Hybrid Multifunctional Nanomaterials for Diagnostic and Therapeutic Applications. In Pharmaceutical Nanobiotechnology for Targeted Therapy, 1st ed.; Barabadi, H., Mostafavi, E., Saravanan, M., Eds.; Nanotechnology in the Life Sciences; Springer: Cham, Switzerland, 2022; pp. 489–519.
- Lagopati, N.; Valamvanos, T.-F.; Proutsou, V.; Karachalios, K.; Pippa, N.; Gatou, M.-A.; Vagena, I.-A.; Cela, S.; Pavlatou, E.A.; Gazouli, M.; et al. The Role of Nano-Sensors in Breath Analysis for Early and Non-Invasive Disease Diagnosis. Chemosensors 2023, 11, 317.
- Iijima, S.; Ichihashi, T. Single-shell carbon nanotubes of 1-nm diameter. Nat. Nanotechnol. 1993, 363, 603–605.
- Saito, R.; Dresselhaus, G.; Dresselhaus, M.S. Physical Properties of Carbon Nanotubes; Imperial College Press: London, UK, 1998.
- Baughman, R.H.; Zakhidov, A.A.; de Heer, W.A. Carbon nanotubes: The route toward applications. Science 2002, 297, 787–792.
- Yamamoto, T.; Watanabe, K.; Hernandez, E.R. Mechanical properties, thermal stability and heat transport in carbon nanotubes. In Carbon Nanotubes: Advanced Topics in the Synthesis, Structure, Properties and Applications; Jorio, A., Dresselhaus, G., Dresselhaus, M.S., Eds.; Springer: Berlin/Heidelberg, Germany, 2008.
- Choi, W.B.; Bae, E.; Kang, D.; Chae, S.; Cheong, B.-h.; Ko, J.-h.; Lee, E.; Park, W. Aligned Carbon Nanotubes for Nanoelectronics. Nanotechnology 2007, 15, 512–516.
- Endo, M.; Strano, M.S.; Ajayan, P.M. Potential applications of carbon nanotubes. In Carbon Nanotubes: Advanced Topics in the Synthesis, Structure, Properties and Applications; Jorio, A., Dresselhaus, G., Dresselhaus, M.S., Eds.; Springer: Berlin, Germany, 2008; pp. 13–61.
- Bonard, J.M.; Salvetat, J.P.; Stöckli, T.; Forró, L.; Chatelain, A. Field emission from carbon nanotubes: Perspectives for applications and clues to the emission mechanism. Appl. Phys. A Mater. Sci. Process. 1999, 69, 245–254.
- Ajayan, P.M.; Zhou, O.Z. Applications of carbon nanotubes. In Carbon Nanotubes: Synthesis, Structure, Properties, and Applications; Dresselhaus, M.S., Dresselhaus, G., Avouris, P., Eds.; Springer-Verlag: New York, NY, USA, 2001; pp. 391–425.
- Harrison, B.S.; Atala, A. Carbon nanotube applications for tissue engineering. Biomaterials 2007, 28, 344–353.
- Bianco, A.; Kostarelos, K.; Partidos, C.D.; Prato, M. Biomedical applications of functionalized carbon nanotubes. Chem. Commun. 2005, 5, 571–577.
- Tîlmaciu, C.M.; Morris, M.C. Carbon nanotube biosensor. Front. Chem. 2015, 3, 59.
- Zhao, Q.; Gan, Z.; Zhuang, Q. Electrochemical sensors based on carbon nanotubes. Electroanalysis 2002, 14, 1609–1613.
- Lin, Y.; Lu, F.; Tu, Y.; Ren, Z. Glucose biosensors based on carbon nanotube nanoelectrode ensembles. Nano Lett. 2004, 4, 191–195.
- Zribi, B.; Roy, E.; Pallandre, A.; Chebil, S.; Koubaa, M.; Mejri, N.; Magdinier Gomez, H.; Sola, C.; Korri-Youssoufi, H.; Haghiri-Gosnet, A.-M. A microfluidic electrochemical biosensor based on multiwall carbon nanotube/ferrocene for genomic DNA detection of Mycobacterium tuberculosis in clinical isolates. Biomicrofluidics 2016, 2, 014115.
- Kaloumenou, M.; Skotadis, E.; Lagopati, N.; Efstathopoulos, E.; Tsoukalas, D. Breath Analysis: A Promising Tool for Disease Diagnosis—The Role of Sensors. Sensors 2022, 22, 1238.
- Otero, F.; Magner, E. Biosensors-recent advances and future challenges in electrode materials. Sensors 2020, 20, 3561.
- Wang, J. Electrochemical glucose biosensors. Chem. Rev. 2008, 108, 814–825.
- Meena, J.; Gupta, A.; Ahuja, R.; Singh, M.; Kumar Panda, A. Recent advances in nano-engineered approaches used for enzyme immobilization with enhanced activity. J. Mol. Liq. 2021, 338, 116602.
- Azimi, S.; Farahani, A.; Sereshti, H. Plasma-functionalized highly aligned CNT-based biosensor for point of care determination of glucose in human blood plasma. Electroanalysis 2019, 32, 394–403.
- Wang, C.; Li, J.; Tan, R.; Wang, Q.; Zhang, Z. Colorimetric method for, glucose detection with enhanced signal intensity using ZnFe2O4-carbon nanotube glucose oxidase composite material. Analyst 2019, 144, 1831–1839.
- Akogulları, S.; Çinar, S.; Ozdokur, K.V.; Aydemir, T.; Ertas, F.N.; Koçak, S. Pulsed deposited manganese and vanadium oxide film modified with carbon nanotube and gold nanoparticle: Chitosan and ionic liquid-based biosensor. Electroanalysis 2019, 32, 445–453.
- Hossain, M.F.; Slaughter, G. Pt NPs decorated chemically derived graphene and carbon nanotubes for sensitive and selective glucose biosensing. J. Electroanal. Chem. 2020, 861, 113990.
- Plocienniczak, P.; Rebis, T.; Leda, A.; Milczarek, G. Lignosulfonate-assisted synthesis of platinum nanoparticles deposited on multi-walled carbon nanotubes for biosensing of glucose. Colloids Surf. B Biointerfaces 2021, 210, 112222.
- Zou, B.; Wang, P.; Xia, J.; Qian, J.; Yan, Y.; Mutombo, J. Enzyme biosensors systems based on Co-modification of carbon nanotubes and enzyme for detection of glucose in food. J. Electrochem. Soc. 2021, 168, 065501.
- Jayakumar, K.; Bennett, R.; Leech, D. Electrochemical glucose biosensor based on an osmium redox polymer and glucose oxidase grafted to carbon nanotubes: A design-of-experiments optimization of current density and stability. Electrochim. Acta 2021, 371, 137845.
- Dhanjai, X.L.; Wu, L.; Chen, J.; Lu, Y. Robust single-molecule enzyme nanocapsules for biosensing with significantly improved biosensor stability. Anal. Chem. 2020, 92, 5830–5837.
- Huang, H.; Li, T.; Jiang, M.; Wei, C.; Ma, S.; Chen, D.; Tong, W.; Huang, X. Construction of flexible enzymatic electrode based on gradient hollow fiber membrane and multi-wall carbon tubes meshes. Biosens. Bioelectron. 2020, 152, 112001.
- Sun, Z.; Liu, H.; Wang, X. Thermal self-regulatory intelligent biosensor based on carbon-nanotubes-decorated phase-change microcapsules for enhancement of glucose detection. Biosens. Bioelectron. 2021, 195, 113586.
- Choi, Y.-B.; Kim, H.-S.; Jeon, W.-Y.; Lee, B.-H.; Shin, U.S.; Kim, H.-H. The electrochemical glucose sensing based on the chitosan-carbon nanotube hybrid. Biochem. Eng. J. 2019, 144, 227–234.
- Zheng, Y.; Tang, N.; Omar, R.; Hu, Z.; Duong, T.; Wang, J.; Wu, W.; Haick, H. Smart materials enabled with artificial intelligence for healthcare wearables. Adv. Funct. Mater. 2021, 31, 2105482.
- Xia, H.-q.; Tang, H.; Zhou, B.; Li, Y.; Zhang, X.; Shi, Z.; Deng, L.; Song, R.; Li, L.; Zhang, Z.; et al. Mediator-free electron-transfer on patternable hierarchical meso/macro porous bienzyme interface for highly-sensitive sweat glucose and surface electromyography monitoring. Sens. Actuators B Chem. 2020, 312, 127962.
- Yao, Y.; Chen, J.; Guo, Y.; Lv, T.; Chen, Z.; Li, N.; Cao, S.; Chen, B.; Chen, T. Integration of interstitial fluid extraction and glucose detection in one device for wearable non-invasive blood glucose sensors. Biosens. Bioelectron. 2021, 179, 113078.
- Zhu, L.; Deng, C.; Chen, P.; You, X.-D.; Su, H.-B.; Yuan, Y.-H.; Zhu, M.-F. Glucose oxidase biosensors based on carbon nanotube non-woven fabrics. Carbon 2014, 67, 795–796.
- Gaitán, C.G.; Rosas, R.R.; Morallón, E.; Amorós, D.C. Effects of the surface chemistry and structure of carbon nanotubes on the coating of glucose oxidase and electrochemical biosensors performance. RSC Adv. 2017, 7, 26867–26878.
- Liu, Q.; Zhong, H.; Chen, M.; Zhao, C.; Liu, Y.; Xi, F.; Luo, T. Functional nanostructure-loaded three-dimensional graphene foam as a non-enzymatic electrochemical sensor for reagentless glucose detection. RSC Adv. 2020, 10, 33739–33746.
- Aun, T.T.; Salleh, N.M.; Ali, U.F.M.; Manan, N.S.A. Non-enzymatic glucose sensors involving copper: An electrochemical perspective. Crit. Rev. Anal. Chem. 2021, 53, 1967720.
- Ahmad, R.; Khan, M.; Tripathy, N.; Khan, M.I.R.; Khosla, A. Hydrothermally synthesized nickel oxide nanosheets for non-enzymatic electrochemical glucose detection. J. Electrochem. Soc. 2020, 167, 107504.
- Abrori, S.A.; Septiani, N.L.W.; Nugraha; Anshori, I.; Suyatman; Suendo, V.; Yuliarto, B. Metal-organic-framework FeBDC-derived Fe3O4 for nonenzymatic electrochemical detection of glucose. Sensors 2020, 20, 4891.
- Viswanathan, P.; Wang, K.; Li, J.; Hong, J.-D. Multicore-shell Ag-CuO networked with CuO nanorods for enhanced non-enzymatic glucose detection. Colloids Surf. Physicochem. Eng. Asp. 2020, 598, 124816.
- Movlaee, K.; Raza, H.; Pinna, N.; Leonardi, S.G.; Neri, G. Ultrasensitive nonenzymatic electrochemical glucose sensor based on NiO/CNT composite. Sensors 2019, 539, 135–140.
- Naqvi, S.T.R.; Shirinfar, B.; Hussain, D.; Majeed, S.; Ashiq, M.N.; Aslam, Y.; Ahmed, N. Electrochemical sensing of ascorbic acid, hydrogen peroxide and glucose by bimetallic (Fe, Ni)-CNTs composite modified electrode. Electroanalysis 2019, 31, 851–857.
- Silva-Carrillo, C.; Felix-Navarro, R.M.; Perez Sicairos, S.; Trujillo-Navarrete, B.; Paraguay-Delgado, F.; Lin-Ho, S.W.; Reynoso-Soto, E.A. Electrokinetic carbohydrates sensor by streaming potential with 3MPBA-PtNPs-CNTs composite material. J. Electrochem. Soc. 2020, 167, 046516.
- Karimi-Maleh, H.; Cellat, K.; Arikan, K.; Savk, A.; Karimi, F.; Sen, F. Palladium nickel nanoparticles decorated on functionalized-MWCNT for high precision non-enzymatic glucose sensing. Mater. Chem. Phys. 2020, 250, 123042.
- Qian, P.; Qin, Y.; Lyu, Y.; Li, Y.; Wang, L.; Wang, S.; Liu, Y. A hierarchical cobalt/carbon nanotube hybrid nanocomplex-based ratiometric fluorescent nanosensor for ultrasensitive detection of hydrogen peroxide and glucose in human serum. Anal. Bioanal. Chem. 2019, 411, 1517–1524.
- Han, J.; Miao, L.; Song, Y. Preparation of co-Co3O4/carbon nanotube/carbon foam for glucose sensor. J. Mol. Recogn. 2019, 33, e2820.
- Arikan, K.; Burhan, H.; Bayat, R.; Sen, F. Glucose nano biosensor with nonenzymatic excellent sensitivity prepared with nickel-cobalt nanocomposites on f-MWCNT. Chemosphere 2021, 291, 132720.
- Adeniyi, O.; Nwahara, N.; Mwanza, D.; Nyokong, T.; Mashazi, P. Nanohybrid electrocatalyst based on cobalt phthalocyanine-carbon nanotube-reduced graphene oxide for ultrasensitive detection of glucose in human saliva. Sens. Actuators B Chem. 2021, 348, 130723.
- Scognamiglio, V.; Arduini, F. The technology tree in the design of glucose biosensors. Trends Anal. Chem. 2019, 120, 115642.
- Qin, Y.; Sun, Y.; Li, Y.; Li, C.; Wang, L.; Guo, S. MOF derived Co3O4/N-doped carbon nanotubes hybrids as efficient catalysts for sensitive detection of H2O2 and glucose. Chin. Chem. Lett. 2020, 31, 774–778.
- Kim, S.e.; Muthurasu, A. Highly oriented nitrogen-doped carbon nanotube integrated bimetallic cobalt copper organic framework for non-enzymatic electrochemical glucose and hydrogen peroxide sensor. Electroanalysis 2021, 33, 1333–1345.
- Palve, Y.P.; Jha, N. A novel bilayer of copper nanowire and carbon nanotube electrode for highly sensitive enzyme free glucose detection. Mater. Chem. Phys. 2020, 240, 122086.
- Waqas, M.; Wu, L.; Tang, H.; Liu, C.; Fan, Y.; Jiang, Z.; Wang, X.; Zhong, J.; Chen, W. Cu2O microspheres supported on sulfur-doped carbon nanotubes for glucose sensing. ACS Appl. Nano Mater. 2020, 3, 4788–4798.
- Najmi, A.; Saidi, M.S.; Shahrokhian, S.; Hosseini, H.; Kazemzadeh Hannani, S. Fabrication of a microdialysis-based nonenzymatic microfluidic sensor for regular glucose measurement. Sens. Actuators B Chem. 2021, 333, 129569.
- Mao, X.; Zhang, C. A microfluidic cloth-based photoelectrochemical analytical device for the detection of glucose in saliva. Talanta 2022, 238, 123052.
- Kaiser, J. DNA test to predict odds of severe COVID-19 draws scrutiny. Science 2021, 372, 1139.
- Williams, R.M.; Lee, C.; Heller, D.A. A fluorescent carbon nanotube sensor detects the metastatic prostate cancer biomarker uPA. ACS Sens. 2018, 3, 1838–1845.
- Ganbat, K.; Pan, D.; Chen, K.; Ning, Z.; Xing, L.; Zhang, Y.; Shen, Y. One-pot electrografting preparation of bifunctionalized carbon nanotubes for sensitive electrochemical immunosensing. J. Electroanal. Chem. 2020, 860, 113906.
- Gulati, P.; Mishra, P.; Khanuja, M.; Narang, J.; Islam, S.S. Nano-moles detection of tumor specific biomarker DNA for colorectal cancer detection using vertically aligned multi-wall carbon nanotubes based flexible electrodes. Process. Biochem. 2020, 90, 184–192.
- Grushevskaya, H.V.; Krylova, N.G.; Lipnevich, I.V.; Egorova, V.P.; Babenka, A.S. Single nucleotide polymorphism genotyping using DNA sequencing on multiwalled carbon nanotubes monolayer by CNT-plasmon resonance. Int. J. Mod. Phys. B 2018, 32, 1840033.
- Ye, Q.; Xu, X.; Paghi, A.; Bamford, T.; Horrocks, B.R.; Houlton, A.; Barillaro, G.; Dimitrov, S.; Palma, M. Solution-processable carbon nanotube nanohybrids for multiplexed photoresponsive devices. Adv. Funct. Mater. 2021, 31, 2105719.
- Lambert, B.P.; Gillen, A.J.; Boghossian, A.A. Synthetic biology: A solution for tackling nanomaterial challenges. J. Phys. Chem. Lett. 2020, 11, 4791–4802.
- Prakash, J.; Dey, A.; Uppal, S.; Alexander, R.; Kaushal, A.; Misra, H.S.; Dasgupta, K. Label-free rapid electrochemical detection of DNA hybridization using ultrasensitive standalone CNT aerogel biosensor. Biosens. Bioelectron. 2021, 191, 113480.
- Lu, Q.; Su, T.; Shang, Z.; Jin, D.; Shu, Y.; Xu, Q.; Hu, X. Flexible paper-based Ni-MOF composite/AuNPs/CNTs film electrode for HIV DNA detection. Biosens. Bioelectron. 2021, 184, 113229.
- Belogiannis, K.; Florou, V.A.; Fragkou, P.C.; Ferous, S.; Chatzis, L.; Polyzou, A.; Lagopati, N.; Vassilakos, D.; Kittas, C.; Tzioufas, A.G.; et al. SARS-CoV-2 Antigenemia as a Confounding Factor in Immunodiagnostic Assays: A Case Study. Viruses 2021, 13, 1143.
- Jeong, S.; Gonzalez-Grandio, E.; Navarro, N.; Pinals, R.L.; Ledesma, F.; Yang, D.; Landry, M.P. Extraction of viral nucleic acids with carbon nanotubes increases SARS-CoV-2 quantitative reverse transcription polymerase chain reaction detection sensitivity. ACS Nano 2021, 15, 10309–10317.
- Grushevskaya, H.V.; Krylova, N.G.; Lipnevich, I.V.; Babenka, A.S.; Egorova, V.P.; Chakukov, R.F. CNT-based label-free electrochemical sensing of native DNA with allele single nucleotide polymorphism. Semiconductors 2018, 52, 1836–1838.
- Lee, J.; Morita, M.; Takemura, K.; Park, E.Y. A multi-functional gold/iron-oxide nanoparticle-CNT hybrid nanomaterial as virus DNA sensing platform. Biosens. Bioelectron. 2018, 102, 425–431.
- Han, S.; Liu, W.; Zheng, M.; Wang, R. Label-free and ultrasensitive electrochemical DNA biosensor based on urchinlike carbon nanotube-gold nanoparticle nanoclusters. Anal. Chem. 2020, 92, 4780–4787.
- Huang, R.; Wang, L.; Gai, Q.; Wang, D.; Qian, L. DNA-mediated assembly of carbon nanotubes for enhancing electrochemiluminescence and its application. Sens. Actuators B Chem. 2018, 256, 953–961.
- Heydarzadeh, S.; Roshanfekr, H.; Peyman, H.; Kashanian, S. Modeling of ultrasensitive DNA hybridization detection based on gold nanoparticles/carbon-nanotubes/chitosan-modified electrodes. Colloids Surf. Physicochem. Eng. Asp. 2020, 587, 124219.
- Kuralay, F.; Dükar, N. Polypyrrole-based nanohybrid electrodes: Their preparation and potential use for DNA recognition and paclitaxel quantification. ChemistrySelect 2020, 5, 4708–4714.
- Tamersit, K.; Djeffal, F. Carbon nanotube field-effect transistor with vacuum gate dielectric for label-free detection of DNA molecules: A computational investigation. IEEE Sens. J. 2019, 19, 9263–9270.
- Sun, Y.; Peng, Z.; Li, H.; Wang, Z.; Mu, Y.; Zhang, G.; Chen, S.; Liu, S.; Wang, G.; Liu, C.; et al. Suspended CNT-based FET sensor for ultrasensitive and label-free detection of DNA hybridization. Biosens. Bioelectron. 2019, 137, 255–262.
- Liang, Y.; Xiao, M.; Wu, D.; Lin, Y.; Liu, L.; He, J.; Zhang, G.; Peng, L.M.; Zhang, Z. Wafer-scale uniform carbon nanotube transistors for ultrasensitive and label-free detection of disease biomarkers. ACS Nano 2020, 8, 8866–8874.
- Cui, M.; Zhao, Q.; Zhang, Q.; Fu, M.; Liu, Y.; Fan, X.; Wang, H.; Zhang, Y.; Wang, H. Nitrogen doped chiral carbonaceous nanotube for ultrasensitive DNA direct electrochemistry, DNA hybridization and damage study. Anal. Chim. Acta 2018, 1038, 41–51.
- Zhao, H.; Liu, M.; Jiang, T.; Xu, J.; Zhang, H.; Yu, C.; Liu, Z.; Wang, Y.; Tang, L. Ultrasensitive monitoring of DNA damage associated with free radicals’ exposure using dynamic carbon nanotubes bridged interdigitated electrode array. Environ. Int. 2020, 139, 105672.
- Li, W.; Gao, Y.; Zhang, J.; Wang, X.; Yin, F.; Li, Z.; Zhang, M. Universal DNA detection realized by peptide-based carbon nanotube biosensors. Nanoscale Adv. 2020, 2, 717–723.
- Balchin, D.; Hayer-Hartl, M.; Hartl, F.U. In vivo aspects of protein folding and quality control. Science 2016, 353, 43–54.
- Rafiq, H.S.; Fatima, B.; Hussain, D.; Mohyuddin, A.; Majeed, S.; Manzoor, S.; Imran, M.; Nawaz, R.; Shabbir, S.; Mukhtar, S.; et al. Selective electrochemical sensing of hemoglobin from blood of 8-thalassemia major patients by tellurium nanowires-graphene oxide modified electrode. Chem. Eng. J. 2021, 419, 129706.
- Khan, N.I.; Maddaus, A.G.; Song, E. A low-cost inkjet-printed aptamer-based electrochemical biosensor for the selective detection of lysozyme. Biosensors 2018, 8, 7.
- Janssen, J.; Lambeta, M.; White, P.; Byagowi, A. Carbon nanotube-based electrochemical biosensor for label-free protein detection. Biosensors 2019, 9, 144.
- Huang, Y.; Wen, Y.Q.; Baryeh, K.; Takalkar, S.; Lund, M.; Zhang, X.J.; Liu, G.D. Magnetized carbon nanotubes for visual detection of proteins directly in whole blood. Anal. Chim. Acta 2017, 993, 79–86.
- Moreira, F.T.C.; Rodriguez, B.A.G.; Dutra, R.A.F.; Sales, M.G.F. Redox probe-free readings of a beta-amyloid-42 plastic antibody sensory material assembled on copper@carbon nanotubes. Sens. Actuators B Chem. 2018, 264, 1–9.
- Lee, C.S.; Kim, J.S.; Kim, T.H. A chemodosimeter-modified carbon nanotube field effect transistor: Toward a highly selective and sensitive electrical sensing platform. RSC Adv. 2019, 9, 28414–28420.
- Naveh, Z.M.H.; Khajehdad, M.M.; Sarmazdeh, M.M. A theoretical study on the chirality detection of serine amino acid based on carbon nanotubes with and without stone-wales defects. Struct. Chem. 2020, 31, 455–464.
- Ning, G.; Wang, H.; Fu, M.; Liu, J.; Sun, Y.; Lu, H.; Fan, X.; Zhang, Y.; Wang, H. Dual signals electrochemical biosensor for point-of-care testing of amino acids enantiomers. Electroanalysis 2021, 34, 316–325.
- Xiong, M.; Tao, Y.Z.; Gao, Q.Q.; Feng, B.; Yan, W.; Zhou, Y.Y.; Kotsonis, T.A.; Yuan, T.L.; You, Z.W.; Wu, Z.Y.; et al. Human stem cell-derived neurons repair circuits and restore neural function. Cell Stem Cell 2021, 28, 112–126.
- Vu, T.T.; Dau, T.N.N.; Ly, C.T.; Pham, D.C.; Nguyen, T.T.N.; Pham, V.T. Aqueous electrodeposition of (AuNPs/MWCNT-PEDOT) composite for high-affinity acetylcholinesterase electrochemical sensors. J. Mater. Sci. 2020, 55, 9070–9081.
- Gopal, P.; Narasimha, G.; Reddy, T.M. Development, validation and enzyme kinetic evaluation of multi walled carbon nano tubes mediated tyrosinase based electrochemical biosensing platform for the voltammetric monitoring of epinephrine. Process. Biochem. 2020, 92, 476–485.
- Li, Y.; Ye, W.; Cui, Y.; Li, B.; Yang, Y.; Qian, G. A metal-organic frameworks@carbon nanotubes based electrochemical sensor for highly sensitive and selective determination of ascorbic acid. J. Mol. Struct. 2020, 1209, 127986.
- Shruthi, C.D.; Venkataramanappa, Y.; Suresh, G.S. Reduced MWCNTs/palladium nanotubes hybrid fabricated on graphite electrode for simultaneous detection of ascorbic acid, dopamine and uric acid. J. Electrochem. Soc. 2018, 165, 458–465.
- Guan, J.-F.; Zou, J.; Liu, Y.-P.; Jiang, X.-Y.; Yu, J.-G. Hybrid carbon nanotubes modified glassy carbon electrode for selective, sensitive and simultaneous detection of dopamine and uric acid. Ecotoxicol. Environ. Saf. 2020, 201, 110872.
- Manivel, P.; Thamilselvan, A.; Rajagopal, V.; Nesakumar, N.; Suryanarayanan, V. Enhanced electrocatalytic activity of Ni-CNT nanocomposites for simultaneous determination of epinephrine and dopamine. Electroanalysis 2019, 31, 2387–2396.
- Eom, G.; Oh, C.; Moon, J.; Kim, H.; Kim, M.K.; Kim, K.; Seo, J.-W.; Kang, T.; Lee, H.J. Highly sensitive and selective detection of dopamine using overoxidized polypyrrole/sodium dodecyl sulfate-modified carbon nanotube electrodes. J. Electroanal. Chem. 2019, 848, 113295.
- Ma, X.; Gao, F.; Dai, R.; Liu, G.; Zhang, Y.; Lu, L.; Yu, Y. Novel electrochemical sensing platform based on a molecularly imprinted polymer-decorated 3Dmulti-walled carbon nanotube intercalated graphene aerogel for selective and sensitive detection of dopamine. Anal. Methods 2020, 12, 1845–1851.
- Arumugasamy, S.K.; Govindaraju, S.; Yun, K. Electrochemical sensor for detecting dopamine using graphene quantum dots incorporated with multiwall carbon nanotubes. Appl. Surf. Sci. 2020, 508, 145294.
- Gupta, P.; Tsai, K.; Ruhunage, C.K.; Gupta, V.K.; Rahm, C.E.; Jiang, D.; Alvarez, N.T. True picomolar neurotransmitter sensor based on open-ended carbon nanotubes. Anal. Chem. 2020, 92, 8536–8545.
- Shukla, R.P.; Aroosh, M.; Matzafi, A.; Ben-Yoav, H. Partially functional electrode modifications for rapid detection of dopamine in urine. Adv. Funct. Mater. 2021, 31, 2004146.
- Posha, B.; Kuttoth, H.; Sandhyarani, N. 1-Pyrene carboxylic acid functionalized carbon nanotube-gold nanoparticle nanocomposite for electrochemical sensing of dopamine and uric acid. Microchim. Acta 2019, 186, 672.
- Ashraf, G.; Asif, M.; Aziz, A.; Iftikhar, T.; Liu, H. Rice-spikelet-like copper oxide decorated with platinum stranded in the CNT network for electrochemical in vitro detection of serotonin. ACS Appl. Mater. Interfaces 2021, 13, 6023–6033.
- Da Silva, L.V.; Dos Santos, N.D.; De Almeida, A.K.A.; Dos Santos, D.D.E.R.; Ferreira Santos, A.C.; Franca, M.C.; Lima, D.J.P.; Lima, P.R.; Goulart, M.O.F. A new electrochemical sensor based on oxidized capsaicin/multi-walled carbon nanotubes/glassy carbon electrode for the quantification of dopamine, epinephrine, and xanthurenic, ascorbic and uric acids. J. Electroanal. Chem. 2021, 881, 114919.
- Ganesan, M.; Ramadhass, K.D.; Chuang, H.-C.; Gopalakrishnan, G. Synthesis of nitrogen-doped carbon quantum dots@Fe2O3/multiwall carbon nanotubes ternary nanocomposite for the simultaneous electrochemical detection of 5-fluorouracil, uric acid, and xanthine. J. Mol. Liq. 2021, 331, 115768.
- Panczyk, T.; Wolski, P.; Lajtar, L. Coadsorption of doxorubicin and selected dyes on carbon nanotubes. Theoretical investigation of potential application as a pH-controlled drug delivery system. Langmuir 2016, 32, 4719–4728.
- Singh, B.; Lohan, S.; Sandhu, P.S.; Jain, A.; Mehta, S.K. Functionalized carbon nanotubes and their promising applications in therapeutics and diagnostics. Nanobiomaterials Med. Imaging 2016, 2016, 455–478.
- Zare, H.; Ahmadi, S.; Ghasemi, A.; Ghanbari, M.; Rabiee, N.; Bagherzadeh, M.; Karimi, M.; Webster, T.J.; Hamblin, M.R.; Mostafavi, E. Carbon Nanotubes: Smart Drug/Gene Delivery Carriers Int. J. Nanomed. 2021, 16, 1681–1706.
- Kang, J.H.; Kima, H.S.; Shin, U.S. Thermo conductive carbon nanotube-framed membranes for skin heat signal-responsive transdermal drug delivery. Polym. Chem. 2017, 8, 3154–3163.
- Shi, X.; Zheng, Y.; Wang, C.; Yue, L.; Qiao, K.; Wang, G.; Wang, L.; Quan, H. Dual stimulus responsive drug release under the interaction of pH value and pulsatile electric field for a bacterial cellulose/sodium alginate/multi-walled carbon nanotube hybrid hydrogel. RSC Adv. 2015, 5, 41820–41829.
- Estrada, A.C.; Silva, A.L.D.; Trindade, T. Photothermally enhanced drug release by κ-carrageenan hydrogels reinforced with multi-walled carbon nanotubes. RSC Adv. 2013, 3, 10828–10836.
- Dhar, S.; Liu, Z.; Thomale, J.; Dai, H.; Lippard, S.J. Targeted single-wall carbon nanotube-mediated Pt (IV) prodrug delivery using folate as a homing device. J. Am. Chem. Soc. 2008, 130, 11467–111476.
- Mehra, N.K.; Jain, N.K. Optimization of a pretargeted strategy for the PET imaging of colorectal carcinoma via the modulation of radioligand pharmacokinetics. Mol. Pharm. 2015, 12, 630–643.
- Banerjee, S.S.; Todkar, K.J.; Khutale, G.V.; Chate, G.P.; Biradar, A.V.; Gawande, M.B.; Zboril, R.; Khandare, J.J. Calcium phosphate nanocapsule crowned multiwalled carbon nanotubes for pH triggered intracellular anticancer drug release. J. Mater. Chem. B 2015, 3, 3931–3939.
- Xu, H.; Liu, M.; Lan, M.; Yuan, H.; Yu, W.; Tian, J.; Wan, Q.; Zhang, X.; Wei, Y. Mussel-inspired PEGylated carbon nanotubes: Biocompatibility evaluation and drug delivery applications. Toxicol. Res. 2016, 5, 1371–1379.
- Mejri, A.; Vardanega, D.; Tangour, B.; Gharbi, T.; Picaud, F. Substrate temperature to control moduli and water uptake in thin films of vapor deposited N,N’-Di(1-naphthyl)-N,N’-diphenyl-(1,1′-biphenyl)-4,4′-diamine (n.d.). J. Phys. Chem. B 2015, 119, 604–611.
- Lagopati, N.; Belogiannis, K.; Angelopoulou, A.; Papaspyropoulos, A.; Gorgoulis, V. Non-Canonical Functions of the ARF Tumor Suppressor in Development and Tumorigenesis. Biomolecules 2021, 11, 86.
- Pantelis, P.; Theocharous, G.; Lagopati, N.; Veroutis, D.; Thanos, D.-F.; Lampoglou, G.-P.; Pippa, N.; Gatou, M.-A.; Tremi, I.; Papaspyropoulos, A.; et al. The Dual Role of Oxidative-Stress-Induced Autophagy in Cellular Senescence: Comprehension and Therapeutic Approaches. Antioxidants 2023, 12, 169.
- Barbouti, A.; Lagopati, N.; Veroutis, D.; Goulas, V.; Evangelou, K.; Kanavaros, P.; Gorgoulis, V.G.; Galaris, D. Implication of Dietary Iron-Chelating Bioactive Compounds in Molecular Mechanisms of Oxidative Stress-Induced Cell Ageing. Antioxidants 2021, 10, 491.
- Hwang, Y.S.; Park, S.H.; Lee, J.W. Applications of functionalized carbon nanotubes for the therapy and diagnosis of cancer. Polymers 2017, 9, 13.
- Su, Y.; Hu, Y.; Li, M.; Zhou, J.; Liu, Y.; Wang, W.; Wang, Y.; Xu, X.; Yuan, Y.; Li, Y.; et al. A precision-guided MWNT mediated reawakening the sunk synergy in RAS for anti-angiogenesis lung cancer therapy. Biomaterials 2017, 139, 75–90.
- Dong, X.; Sun, Z.; Wang, X.; Leng, X. An innovative MWCNTs/DOX/TC nanosystem for chemo-photothermal combination therapy of cancer. Nanomed. Nanotech. Biol. Med. 2017, 13, 2271–2280.
- Kim, S.W.; Lee, Y.K.; Lee, J.Y.; Hong, J.H.; Khang, D. PEGylated anticancer-carbon nanotubes complex targeting mitochondria of lung cancer cells. Nanotechnology 2017, 28, 465102.
- Song, J.; Wang, F.; Yang, X.; Ning, B.; Harp, M.G.; Culp, S.H.; Hu, S.; Huang, P.; Nie, L.; Chen, J.; et al. Gold nanoparticle coated carbon nanotube ring with enhanced Raman scattering and photothermal conversion Property for theranostic applications. J. Am. Chem. Soc. 2016, 138, 7005–7015.
- Bao, Z.; Liu, X.; Liu, Y.; Liu, H.; Zhao, K. Near-infrared light-responsive inorganic nanomaterials for photothermal therapy. AJPS 2016, 11, 349–364.
- Sahithi, K.; Swetha, M.; Ramasamy, K.; Srinivasan, N.; Selvamurugan, N. Polymeric composites containing carbon nanotubes for bone tissue engineering. Int. J. Biol. Macromol. 2010, 46, 281–283.
- Shin, S.R.; Bae, H.; Cha, J.M.; Mun, J.Y.; Chen, Y.-C.; Tekin, H.; Shin, H.; Farshchi, S.; Dokmeci, M.R.; Tang, S.; et al. Carbon nanotube reinforced hybrid microgels as scaffold materials for cell encapsulation. ACS Nano 2011, 6, 362–372.
- Sen, R.; Zhao, B.; Perea, D.; Itkis, M.E.; Hu, H.; Love, J.; Bekyarova, E.; Haddon, R.C. Preparation of single-walled carbon nanotube reinforced polystyrene and polyurethane nanofibers and membranes by electrospinning. Nano Lett. 2004, 4, 459–464.
- Lau, C.; Cooney, M.J.; Atanassov, P. Conductive macroporous composite chitosan carbon nanotube scaffolds. Langmuir 2008, 24, 7004–7010.
- Kam, N.W.S.; Jan, E.; Kotov, N.A. Electrical stimulation of neural stem cells mediated by humanized carbon nanotube composite made with extracellular matrix protein. Nano Lett. 2009, 9, 273–278.
- Worsley, M.A.; Kucheyev, S.O.; Kuntz, J.D.; Hamza, A.V.; Satcher, J.J.H.; Baumann, T.F. Stiff and electrically conductive composites of carbon nanotube aerogels and polymers. J. Mater. Chem. 2009, 19, 3370–3372.
- Shin, S.R.; Jung, S.M.; Zalabany, M.; Kim, K.; Zorlutuna, P.; Kim, S.; Nikkhah, M.; Khabiry, M.; Azize, M.; Kong, J.; et al. Carbon-nanotube-embedded hydrogel sheets for engineering cardiac constructs and bioactuators. ACS Nano 2013, 7, 2369–2380.
- Yang, S.-T.; Luo, J.; Zhou, Q.; Wang, H. Pharmacokinetics, Metabolism and Toxicity of Carbon Nanotubes for Biomedical Purposes. Theranostics 2012, 2, 271–282.
- Lam, C.-W.; James, J.T.; McCluskey, R.; Arepalli, S.; Hunter, R.L. A Review of carbon nanotube toxicity and assessment of potential occupational and environmental health risks. Crit. Rev. Toxicol. 2006, 36, 189–217.
- Firme, C.P.; Bandaru, P.R. Toxicity issues in the application of carbon nanotubes to biological systems. Nanomed. Nanotechnol. Biol. Med. 2010, 6, 245–256.
- Sato, Y.; Yokoyama, A.; Shibata, K.-I.; Akimoto, Y.; Ogino, S.-I.; Nodasaka, Y.; Kohgo, T.; Tamura, K.; Akasaka, T.; Uo, M.; et al. Influence of length on cytotoxicity of multi-walled carbon nanotubes against human acute monocytic leukemia cell line THP-1 in vitro and subcutaneous tissue of rats in vivo. Mol. BioSyst. 2005, 1, 176–182.
- Wick, P.; Manser, P.; Limbach, L.K.; Dettlaff-Weglikowska, U.; Krumeich, F.; Roth, S.; Stark, W.J.; Bruinink, A. The degree and kind of agglomeration affect carbon nanotube cytotoxicity. Toxicol. Lett. 2007, 168, 121–131.
- Duch, M.C.; Budinger, G.R.S.; Liang, Y.T.; Soberanes, S.; Urich, D.; Chiarella, S.E.; Campochiaro, L.A.; Gonzalez, A.; Chandel, N.S.; Hersam, M.C.; et al. Minimizing oxidation and stable nanoscale dispersion improves the biocompatibility of graphene in the lung. Nano Lett. 2011, 11, 5201–5207.
- Yang, K.; Wan, J.; Zhang, S.; Zhang, Y.; Lee, S.-T.; Liu, Z. In vivo pharmacokinetics, long-term biodistribution, and toxicology of pegylated graphene in mice. ACS Nano 2011, 5, 516–522.
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric field effect in atomically thin carbon films. Science 2004, 306, 666–669.
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nat. Mater. 2007, 6, 183–191.
- Kim, K.S.; Zhao, Y.; Jang, H.; Lee, S.Y.; Kim, J.M.; Kim, K.S.; Ahn, J.-H.; Kim, P.; Choi, J.-Y.; Hong, B.H. Large-scale pattern growth of graphene films for stretchable transparent electrodes. Nature 2009, 457, 706–710.
- Eda, G.; Fanchini, G.; Chhowalla, M. Large-area ultrathin films of reduced graphene oxide as a transparent and flexible electronic material. Nat. Nanotechnol. 2008, 3, 270–274.
- Tang, L.; Wang, Y.; Li, J. The graphene/nucleic acid nanobiointerface. Chem. Soc. Rev. 2015, 44, 6954–6980.
- Akhavan, O.; Ghaderi, E.; Rahighi, R. Toward single-DNA electrochemical biosensing by graphene nanowalls. ACS Nano 2012, 6, 2904–2916.
- Cheng, D.; Yang, L.; Li, X.; Zhou, J.; Chen, Q.; Yan, S.; Li, N.; Chu, M.; Dong, Y.; Xie, Z.; et al. An electrochemical DNA sensing platform using carboxyl functionalized graphene as the electrode modified material. J. Electrochem. Soc. 2017, 164, 345–351.
- Ping, J.; Vishnubhotla, J.R.; Vrudhula, A.; Johnson, A.T.C. Scalable production of high-sensitivity, label-free DNA biosensors based on backgated graphene field Effect transistors. ACS Nano 2016, 10, 8700–8704.
- Gao, Z.; Kang, H.; Naylor, C.H.; Streller, F.; Ducos, P.; Serrano, M.D.; Ping, J.; Zauberman, J.; Rajesh; Carpick, R.W.; et al. Scalable production of sensor arrays based on high-mobility hybrid graphene field effect transistors. ACS Appl. Mater. Interfaces 2016, 8, 27546–27552.
- Zheng, X.T.; Ananthanarayanan, A.; Luo, K.Q.; Chen, P. Glowing graphene quantum dots and carbon dots: Properties, syntheses, and biological applications. Small 2015, 11, 1620–1636.
- Kim, J.; Park, S.-Y.; Kim, S.; Lee, D.H.; Kim, J.H.; Kim, J.M.; Kang, H.; Han, J.-S.; Park, J.W.; Lee, H.; et al. Precise and selective sensing of DNA-DNA hybridization by graphene/Si-nanowires diode-type biosensors. Sci. Rep. 2016, 6, 31984.
- Park, J.S.; Goo, N.I.; Kim, D.E. Mechanism of DNA adsorption and desorption on graphene oxide. Langmuir 2014, 30, 12587–12595.
- Huang, Z.; Liu, J. Length-dependent diblock DNA with poly-cytosine (Poly-C) as high-affinity anchors on graphene oxide. Langmuir 2018, 34, 1171–1177.
- Prabowo, B.A.; Alom, A.; Secario, M.K.; Masim, F.C.P.; Lai, H.-C.; Hatanaka, K.; Liu, K.-C. Graphene-based portable SPR Sensor for the detection of Mycobacterium tuberculosis DNA strain. Procedia Eng. 2016, 168, 541–545.
- Kumar, S.; Amala, G.; Gowtham, S.M. Graphene based sensors in the detection of glucose in saliva—A promising emerging modality to diagnose diabetes mellitus. RSC Adv. 2017, 7, 36949–36976.
- Zhang, M.; Liao, C.; Mak, C.H.; You, P.; Mak, C.L.; Yan, F. Highly sensitive glucose sensors based on enzyme-modified whole-graphene solution gated transistors. Sci. Rep. 2015, 5, 8311.
- Park, B.; Park, H.G.; Ji, J.; Cho, J.; Jun, S.C. A Reduced graphene oxide based radio frequency glucose sensing device using multi-dimensional parameters. Micromachines 2016, 7, 136.
- Mani, V.; Devasenathipathy, R.; Chen, S.M.; Subramani, B.; Govindasamy, M. A novel glucose biosensor at glucose oxidase immobilized graphene and bismuth nanocomposite film modified electrode. Int. J. Electrochem. Sci. 2015, 10, 691–700.
- Thirumalraj, B.; Palanisamy, S.; Chen, S.M.; Yang, C.Y.; Periakaruppan, P.; Lou, B.S. Direct electrochemistry of glucose oxidase and sensing of glucose at a glassy carbon electrode modified with a reduced graphene oxide/fullerene-C60 composite. RSC Adv. 2015, 5, 77651–77657.
- Bharath, G.; Madhu, R.; Chen, S.M.; Veeramani, V.; Balamurugan, A.; Mangalaraj, D. Enzymatic electrochemical glucose biosensors by mesoporous 1D hydroxyapatite-on-2D reduced graphene oxide. J. Mater. Chem. B 2015, 3, 1360–1370.
- Wang, X.; Wang, C.; Cheng, L.; Lee, S.T.; Liu, Z. Noble metal coated single-walled carbon nanotubes for applications in surface enhanced Raman scattering imaging and photothermal therapy. J. Am. Chem. Soc. 2012, 134, 7414–7422.
- Liu, Y.; Dong, X.; Chen, P. Biological and chemical sensors based on graphene materials. Chem. Soc. Rev. 2012, 41, 2283–2307.
- Zhang, H.; Grüner, G.; Zhao, Y. Recent advancements of graphene in biomedicine. J. Mater. Chem. B 2013, 1, 2542–2567.
- Kundu, A.; Nandi, S.; Das, P.; Nandi, A.K. Fluorescent graphene oxide via polymer grafting: An efficient nanocarrier for both hydrophilic and hydrophobic drugs. ACS Appl. Mater. Interfaces 2015, 7, 3512–3523.
- Xu, Z.; Wang, S.; Li, Y.; Wang, M.; Shi, P.; Huang, X. Covalent functionalization of graphene oxide with biocompatible poly (ethylene glycol) for delivery of paclitaxel. ACS Appl. Mater. Interfaces 2014, 6, 17268–17276.
- Zhao, X.; Yang, L.; Li, X.; Jia, X.; Liu, L.; Zeng, J.; Guo, J.; Liu, P. Functionalized graphene oxide nanoparticles for cancer cell specific delivery of antitumor drug. Bioconjugate Chem. 2015, 26, 128–136.
- Lerra, L.; Farfalla, A.; Sanz, B.; Cirillo, G.; Vittorio, O.; Voli, F.; Le Grand, M.; Curcio, M.; Nicoletta, F.P.; Dubrovska, A.; et al. Graphene Oxide Functional Nanohybrids with Magnetic Nanoparticles for Improved Vectorization of Doxorubicin to Neuroblastoma Cells. Pharmaceutics 2018, 11, 3.
- Mahdavi, M.; Rahmani, F.; Nouranian, S. Molecular simulation of pH-dependent diffusion, loading, and release of doxorubicin in graphene and graphene oxide drug delivery systems. J. Mater. Chem. B 2016, 4, 7441–7451.
- Wei, G.; Dong, R.; Wang, D.; Feng, L.; Dong, S.; Song, A.; Hao, J. Functional materials from the covalent modification of reduced graphene oxide and βcyclodextrin as a drug delivery carrier. New J. Chem. 2014, 38, 140–145.
- Yu, X.; Gao, D.; Gao, L.; Lai, J.; Zhang, C.; Zhao, Y.; Zhong, L.; Jia, B.; Wang, F.; Chen, X.; et al. Inhibiting metastasis and preventing tumor relapse by triggering host immunity with tumor-targeted photodynamic therapy using photosensitizer-loaded functional nanographenes. ACS Nano 2017, 11, 10147–10158.
- Song, L.; Shi, J.; Lu, J.; Lu, C. Structure observation of graphene quantum dots by single-layered formation in layered confinement space. Chem. Sci. 2015, 6, 4846–4850.
- Cheon, Y.A.; Bae, J.H.; Chung, B.G. Reduced graphene oxide nanosheet for chemo-photothermal therapy. Langmuir 2016, 32, 2731–2736.
- Su, S.; Wang, J.; Vargas, E.; Wei, J.; Martínez-Zaguilán, R.; Sennoune, S.R.; Pantoya, M.L.; Wang, S.; Chaudhuri, J.; Qiu, J. Porphyrin immobilized nanographene oxide for enhanced and targeted photothermal therapy of brain cancer. ACS Biomater. Sci. Eng. 2016, 2, 1357–1366.
- Shao, L.; Zhang, R.; Lu, J.; Zhao, C.; Deng, X.; Wu, Y. Mesoporous silica coated polydopamine functionalized reduced graphene oxide for synergistic targeted chemo-photothermal therapy. ACS Appl. Mater. Interfaces 2017, 9, 1226–1236.
- Dai, C.; Zhang, S.; Liu, Z.; Wu, R.; Chen, Y. Two-dimensional graphene augments nanosonosensitized sonocatalytic tumor eradication. ACS Nano 2017, 11, 9467–9480.
- Ou, L.; Song, B.; Liang, H.; Liu, J.; Feng, X.; Deng, B.; Sun, T.; Shao, L. Toxicity of graphene-family nanoparticles: A general review of the origins and mechanisms. Part. Fibre Toxicol. 2016, 13, 57.
- Shareena, T.P.; McShan, D.; Dasmahapatra, A.K.; Tchounwou, P.B. A review on graphene-based nanomaterials in biomedical applications and risks in environment and health. Nanomicro Lett. 2018, 10, 53.
- Alshehri, R.; Ilyas, A.M.; Hasan, A.; Arnaout, A.; Ahmed, F.; Memic, A. Carbon nanotubes in biomedical applications: Factors, mechanisms, and remedies of toxicity. J. Med. Chem. 2016, 59, 8149–8167.
- Li, Y.; Wu, Q.; Zhao, Y.; Bai, Y.; Chen, P.; Xia, T.; Wang, D. Response of MicroRNAs to in vitro treatment with graphene oxide. ACS Nano 2014, 8, 2100–2110.
- Chong, Y.; Ge, C.; Yang, Z.; Gárate, J.A.; Gu, Z.; Weber, J.K.; Liu, J.; Zhou, R. Reduced cytotoxicity of graphene nanosheets mediated by blood-protein coating. ACS Nano 2015, 9, 5713–5724.
- Perini, G.; Palmieri, V.; Ciasca, G.; De Spirito, M.; Papi, M. Unravelling the Potential of Graphene Quantum Dots in Biomedicine and Neuroscience. Int. J. Mol. Sci. 2020, 21, 3712.
- Kumawat, M.K.; Thakur, M.; Gurung, R.B.; Srivastava, R. Graphene quantum dots for cell proliferation, nucleus imaging, and photoluminescent sensing applications. Sci. Rep. 2017, 7, 15858.
- Qian, Z.S.; Shan, X.Y.; Chai, L.J.; Ma, J.J.; Chen, J.R.; Feng, H. A universal fluorescence sensing strategy based on biocompatible graphene quantum dots and graphene oxide for the detection of DNA. Nanoscale 2014, 6, 5671–5674.
- Wang, Y.; Zhang, L.; Liang, R.P.; Bai, J.M.; Qiu, J.D. Using graphene quantum dots as photoluminescent probes for protein kinase sensing. Anal. Chem. 2013, 85, 9148–9155.
- Xi, J.; Xie, C.; Zhang, Y.; Wang, L.; Xiao, J.; Duan, X.; Ren, J.; Xiao, F.; Wang, S. Ultrafine Pd nanoparticles encapsulated in microporous Co3O4 hollow nanospheres for in situ molecular detection of living cells. ACS Appl. Mater. Interfaces 2016, 8, 22563–22573.
- Srivastava, A.; Yadav, T.; Sharma, S.; Nayak, A.; Kumari, A.; Mishra, N. Polymers in drug delivery. J. Biosci. Med. 2016, 4, 69–84.
- Tian, Z.; Yao, X.; Ma, K.; Niu, X.; Grothe, J.; Xu, Q.; Liu, L.; Kaskel, S.; Zhu, Y. Metal-organic framework/graphene quantum dot nanoparticles used for synergistic chemoand photothermal therapy. ACS Omega 2017, 2, 1249–1258.
- Ding, H.; Zhang, F.; Zhao, C.; Lv, Y.; Ma, G.; Wei, W.; Tian, Z. Beyond a Carrier: Graphene quantum dots as a probe for programmatically monitoring anti-cancer drug delivery, release, and response. ACS Appl. Mater. Interfaces 2017, 9, 27396–27401.
- Nigam, P.; Waghmode, S.; Louis, M.; Wangnoo, S.; Chavan, P.; Sarkar, D. Graphene quantum dots conjugated albumin nanoparticles for targeted drug delivery and imaging of pancreatic cancer. J. Mater. Chem. B 2014, 2, 3190–3195.
- Iannazzo, D.; Pistone, A.; Salamò, M.; Galvagno, S.; Romeo, R.; Giofré, S.V.; Branca, C.; Visalli, G.; Di Pietro, A. Graphene quantum dots for cancer targeted drug delivery. Int. J. Pharm. 2017, 518, 185–192.
- Sui, X.; Luo, C.; Wang, C.; Zhang, F.; Zhang, J.; Guo, S. Graphene quantum dots enhance anticancer activity of cisplatin via increasing its cellular and nuclear uptake. Nanomedicine 2016, 12, 1997–2006.
- Wang, L.; Wang, X.; Bhirde, A.; Cao, J.; Zeng, Y.; Huang, X.; Sun, Y.; Liu, G.; Chen, X. Carbondot-based two-photon visible nanocarriers for safe and highly efficient delivery of siRNA and DNA. Adv. Healthc. Mater. 2014, 3, 1203–1209.
- Some, S.; Gwon, A.-R.; Hwang, E.; Bahn, G.-H.; Yoon, Y.; Kim, Y.; Kim, S.-H.; Bak, S.; Yang, J.; Jo, D.-G.; et al. Cancer therapy using ultrahigh hydrophobic drug-loaded graphene derivatives. Sci. Rep. 2014, 4, 6314.
- Ge, J.; Lan, M.; Zhou, B.; Liu, W.; Guo, L.; Wang, H.; Jia, Q.; Niu, G.; Huang, X.; Zhou, H.; et al. A graphene quantum dot photodynamic therapy agent with high singlet oxygen generation. Nat. Commun. 2014, 5, 4596.
- Yao, X.; Niu, X.; Ma, K.; Huang, P.; Grothe, J.; Kaskel, S.; Zhu, Y. Graphene quantum dots-capped magnetic mesoporous silica nanoparticles as a multifunctional platform for controlled drug delivery, magnetic hyperthermia, and photothermal therapy. Small 2017, 13, 1602225.
- Li, K.; Liu, W.; Ni, Y.; Li, D.; Lin, D.; Su, Z.; Wei, G. Technical synthesis and biomedical applications of graphene quantum dots. J. Mater. Chem. B 2017, 5, 4811–4826.
- Ailuno, G.; Balboni, A.; Caviglioli, G.; Lai, F.; Barbieri, F.; Dellacasagrande, I.; Florio, T.; Baldassari, S. Boron Vehiculating Nanosystems for Neutron Capture Therapy in Cancer Treatment. Cells 2022, 11, 4029.
- Schroeder, K.L.; Goreham, R.V.; Nann, T. Graphene Quantum Dots for Theranostics and Bioimaging. Pharm. Res. 2016, 33, 2337–2357.
- Su, Z.; Shen, H.; Wang, H.; Wang, J.; Li, J.; Nienhaus, G.U.; Shang, L.; Wei, G. Motif-designed peptide nanofibers decorated with graphene quantum dots for simultaneous targeting and imaging of tumor cells. Adv. Funct. Mater. 2015, 25, 5472–5478.
- Zheng, X.T.; He, H.L.; Li, C.M. Multifunctional graphene quantum dots-conjugated titanate nanoflowers for fluorescence-trackable targeted drug delivery. RSC Adv. 2013, 3, 24853–24857.
- Huang, C.-L.; Huang, C.-C.; Mai, F.-D.; Yen, C.-L.; Tzing, S.-H.; Hsieh, H.-T.; Ling, Y.-C.; Chang, J.-Y. Application of paramagnetic graphene quantum dots as a platform for simultaneous dual-modality bioimaging and tumor-targeted drug delivery. J. Mater. Chem. B. 2015, 3, 651–664.
- Wang, J.; Cao, S.; Ding, Y.; Ma, F.; Lu, W.; Sun, M. Theoretical investigations of optical origins of fluorescent graphene quantum dots. Sci. Rep. 2016, 6, 24850.
- Liang, L.; Kong, Z.; Kang, Z.; Wang, H.; Zhang, L.; Shen, J.W. Theoretical evaluation on potential cytotoxicity of graphene quantum dots. ACS Biomater. Sci. Eng. 2016, 2, 1983–1991.
- Tian, X.; Xiao, B.-B.; Wu, A.; Yu, L.; Zhou, J.; Wang, Y.; Wang, N.; Guan, H.; Shang, Z.-F. Hydroxylated graphene quantum dots induce cells senescence in both p53-dependent and -independent manner. Toxicol. Res. 2016, 5, 1639–1648.
- Nurunnabi, M.; Khatun, Z.; Huh, K.M.; Park, S.Y.; Lee, D.Y.; Cho, K.J.; Lee, Y.-K. In Vivo biodistribution and toxicology of carboxylated graphene quantum dots. ACS Nano 2013, 7, 6858–6867.
This entry is offline, you can click here to edit this entry!
