Betel quid (BQ) chewing increased the risk of oral cancer and oral submucous fibrosis (OSMF), an oral premalignant disorder (OPMD) with malignant transformation potential. BQ components such as areca nut (AN), trauma by coarse AN fiber, catechin, copper, alkaloids, stimulated reactive oxygen species (ROS), inflammation and cytotoxicity are suggested to be the contributing factors. In this review, the expression of extracellular matrix (ECM) turnover related genes and proteins in OSMF and the relation to betel quid chewing habit is discussed. Genetic susceptibility of ECM-related genes to OSMF is also mentioned. These results can facilitate our understanding the pathogenesis of OSMF and its possible prevention/treatment in the future.
- Oral Submucous Fibrosis
- betel quid chewing
- genetic susceptibility
- extracellular matrix turnover
Introduction
Epidemiology and Risk Factor of Oral Submucous Fibrosis (OSMF)
OSMF is defined as an insidious chronic disease affecting any part of the oral cavity and sometimes pharynx. Clinically OSMF presents with burning and pain of the mouth, oral mucosal atrophy with fibrosis of submucosal tissues, mucosal rigidity and reduction in mouth opening. OSMF is common in India, Sri Lanka, Taiwan and other southeast Asian countries, and has started spreading to Europe and North America [1,2]. In Taiwan, the prevalence of OSMF increased from 8.3/100,000 person in 1996 to 16.2/100,000 person in 2013 [3]. The prevalence of betel quid (BQ) chewing, tobacco smoking and alcohol drinking habits in patients with oral premalignant lesions are about 82.9%, 95% and 22.7%, respectively [4]. OSMF may occur at any age but is frequently seen at the age of 21–30 years old. Male to female ratio is around 11:1 [5], possibly due to intrinsic differences between genders with/without the BQ chewing habit. Middle-age chewers are the more commonly involved population in India [6]. BQ components are suggested to be the major etiologic factors for OSMF and oral squamous cell carcinoma (OSCC), due to their content of inflammatory, genotoxic, carcinogenic and fibrogenic factors such as areca nut (AN), lime, arecoline, catechin, catechol, copper, and reactive oxygen species (ROS) [1,7–9].
OSMF is an oral potentially malignant disorder (OPMD) carrying risk for malignant transformation. The malignant transformation rate of OSMF is reported to be about 5.7% after 80.9 months of follow-up [4] or 7–13% [10]. In a total of 1774 cases of OSMF and OSCC in Pakistan, 765 (43.12%) cases were OSMF alone, 472 (26.60%) cases were shown to have OSCC with malignant transformation from OSMF, whereas 537 (30.27%) cases had OSCC without OSMF [11]. A 6.8-year follow-up study also elucidated that alcohol consumption is associated with the malignant transformation of patients with oral precancer [12]. Other factors including BQ chewing habit, smoking habit, environmental heavy metal exposure [13], gender, site of lesion and histological features such as epithelial dysplasia, loss of heterozygosity, aneuploidy of DNA and human papillomavirus (HPV) infection are suggested to stimulate the progression and malignant transformation of OSMF [14].
BQ and OSMF—Etiology, Clinical and Histologic Features
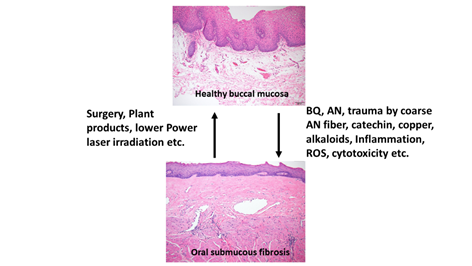
OSMF is a chronic, progressive, high-risk precancerous disease characterized by juxtaepithelial inflammatory reaction, fibrosis of lamina propria, thin parakeratinized squamous epithelium with atrophic change and loss of rete peg. Increased dense collagen fiber deposition in the lamina propria occur through time and, in the end stage, dense hyalinized fibrous tissue occupies the lamina propria and even superficial submucosa, and thus results in varied degrees of mucosal rigidity (Figure 1). OSMF is accompanied by fibroelastic hyperplasia, with/without epithelial hyperplasia/dysplasia over the oral cavity or oropharynx [15,16]. Repeated trauma causes inflammation and aggravates the fibrosis while increased collagen fiber deposition, decreased amount of blood vessels and atrophic change of the epithelium occur. Progression of OSMF, thereby, may lead to loss of tissue mobility, trismus and limited mouth opening [15,16]. However, current treatment strategies are not so effective for attenuation of OSMF. BQ chewing is shown as the major etiologic factor of OSMF [1,2,17]. There are about 200–600 million BQ chewers in the world [1]. The ingredients of BQ vary in different countries. In Taiwan and Papua New Guinea, BQ comprised of AN, and slaked lime with/without inclusion of Piper betle inflorescence or Piper betle leaf (betel leaf) [1,7]. However, in India and Sri Lanka, tobacco is popularly added as one major component of BQ [1]. Among the BQ ingredients, AN components are considered to be the main causative factors in the disease process of OSMF. This is because OSMF is widespread in Taiwan where tobacco is not added into BQ. The roles of lime, betel leaf and other ingredients in the pathogenesis of OSMF await further clarification. AN contains mainly alkaloids (such as arecoline, arecaidine, guvacoline, and guvacine), catechol, catechin, transition metals (Copper, Iron) and fibers [1,7]. The contributory role of AN components to the pathogenesis of OSMF is closely associated with the induction of ROS production [18], chronic mucositis, ulcers caused by mechanical trauma from coarse AN fibers [19,20], activation of the coagulation system [21], cytotoxicity to oral epithelial cells [8], stimulation of fibroblast proliferation/contraction [22,23], collagen synthesis/deposition [22,24], myofibroblast differentiation [10], tissue inflammation [9,25] and the inhibition of collagen degradation and phagocytosis [26,27].These AN components include AN extract (ANE), areca alkaloids (arecoline, arecaidine, guvacoline, guvacine), catechin, catechol and copper (Figures 1 and 2). However, only about 1% to 2% of BQ chewers develop OSMF, suggesting the presence of some predisposition factors toward mild or severe OSMF in these affected patients [17].
Figure 1. Proposed contributing factors (betel quid [BQ], areca nut [AN], coarse fiber of AN, arecoline, catechin, catechol, reactive oxygen species [ROS], inflammation etc.) for oral submucous fibrosis (OSMF) and the treatment methods (surgery, natural products, low power laser irradiation, enzymes, corticosteroid, vasodilator, antioxidants) for OSMF. The histologic pictures of (upper panel) normal buccal mucosa and (lower panel) late stage of OSMF. (H&E stain, A & B: 100×). Normal buccal mucosa is covered by nonkeratinized squamous epithelium with few short rete ridges and supported by lamina propria, which is composed of mainly loose fibrous connective tissue with some collagen fibrils dispersed. In contrast to normal mucosa, the mucosa in late stage OSMF patients shows thin parakeratinized squamous epithelium with atrophic change and a flat junction between epithelium and connective tissue. Dense collagen fiber deposition to hyalinized fibrous tissue involving lamina propria and superficial submucosa is evident. Some chronic inflammatory cell infiltration is frequently seen.
Genetic Susceptibility and Expression in Tissue/organ Fibrosis
A number of studies have found the association of genetic susceptibility with tissue/organ fibrosis such as pulmonary fibrosis, systemic sclerosis, liver and kidney fibrosis [28–31]. While exogenous factors such as viral hepatitis and alcohol abuse are the common causative factors of liver fibrosis, genetic predisposition may contribute to the progression of fibrosis, cirrhosis, liver failure or hepatic carcinoma [28]. Environmental factors are the key etiologic factors of lung fibrosis, but genetic factors in host defense, aging/senescence and cell-cell adhesion may also increase the risk of pulmonary fibrosis, subsequent disease progression and poor prognosis [30]. Systemic sclerosis as an autoimmune disease may involve vascular abnormalities, immune alterations and fibrosis of skin and other internal organs, where tissue inflammation and genetic susceptibility are present [31].
Several factors including nutritional deficiency, vitamin deficiencies and hypersensitivity to various dietary constituents, may also play a part in the pathogenesis of OSMF. Epidemiological studies strongly indicate that AN is the major etiologic agent that releases alkaloids to promote fibroblastic proliferation and increase collagen formation. However, only a small population of BQ chewers develop the disease, indicating that difference in genetic susceptibility plays a role in this event [32]. About 7–12% of OSMF cases progress into oral malignancy [33]. Additionally, a few individuals developed the disease after only a few contacts with BQ [34]. Therefore, the relationship between gene and OSMF is still uncertain and awaits clarification.
BQ and Collagen Turnover
3.1. Collagen-Related Genes
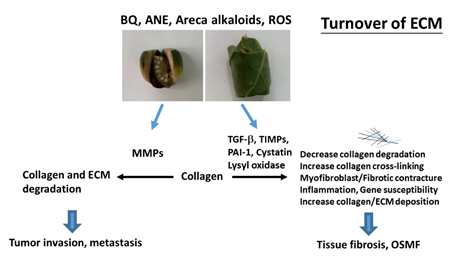
The extracellular matrix (ECM) provides a three-dimensional scaffold for cells via connection of cell surface receptors with various ECM components such as collagen, fibronectin, elastin and nonfibrillar proteins including proteoglycan, hyaluronan and glycoproteins [35]. Hypoxia and collagen-rich conditions also intensify cancer progression [36]. Impairment of ECM and ECM-cell interaction play important roles in various diseases such as osteoarthritis, fibrosis, cancer and genetic diseases [35]. OSMF is such a collagen-related disorder, with dense collagen deposition in the oral submucosa as its main characteristic feature. It has been found that fibroblasts from buccal mucosa exposed to areca alkaloid, due to BQ chewing, may result in the accumulation of collagen [22]. Therefore, it is hypothesized that collagen-related genes might play a role in OSMF pathogenesis. Transforming growth factor-β1 (TGF-β1), lysyl oxidase (LOX), cystatin (CST3), plasminogen activator inhibitor-1 (PAI-1), matrix metalloproteinases (MMPs) and tissue inhibitors of metalloproteinases (TIMPs) proved to be involved in the turnover of ECM, wound healing, tumor invasion and metastasis (Figure 2) [37,38]. However, few studies have compared protein expression and polymorphisms of the collagen-related genes situated on different chromosomes between OSMF patients and healthy controls. Some noninvasive markers (serum markers, urinary markers and image tissue stiffness markers) have been developed for evaluation of organ fibrosis, but still with some limitations [39]. Chiu et al. (2002) suggested that polymorphisms of collagen-related-genes can serve as markers of disease susceptibility in patients with OSMF [40]. Further studies on the development of noninvasive early disease markers of OSMF are crucial for disease prevention and treatment in the future.
Figure 2. Molecular mechanisms of changes in turnover of extracellular matrix (ECM) of OSMF and tumor invasion. Collagen and ECM deposition are stimulated by transforming growth factor-β1 (TGF-β1), tissue inhibitors of metalloproteinases (TIMPs), lysyl oxidase, cystatin, and plasminogen activator inhibitor-1 (PAI-1). These events may contribute to decreased ECM degradation, increased collagen cross-linking/stability and increased collagen/ECM deposition, leading to tissue fibrosis and oral submucous fibrosis (OSMF). ECM degradation is mediated mainly via metalloproteinases (MMPs) and contributes to tumor invasion/metastasis.
This entry is adapted from the peer-reviewed paper 10.3390/ijms21218104