Food samples such as milk, beverages, meat and chicken products, fish, etc. are complex and demanding matrices. Various novel materials such as molecular imprinted polymers (MIPs), carbon-based nanomaterials carbon nanotubes, graphene oxide and metal-organic frameworks (MOFs) have been recently introduced in sample preparation to improve clean up as well as to achieve better recoveries, all complying with green analytical chemistry demands. Metal-organic frameworks are hybrid organic inorganic materials, which have been used for gas storage, separation, catalysis and drug delivery.
- metal-organic frameworks
- MOF
- sample preparation
- HPLC
- GC
- food samples
1. Introduction
2. Food Matrices
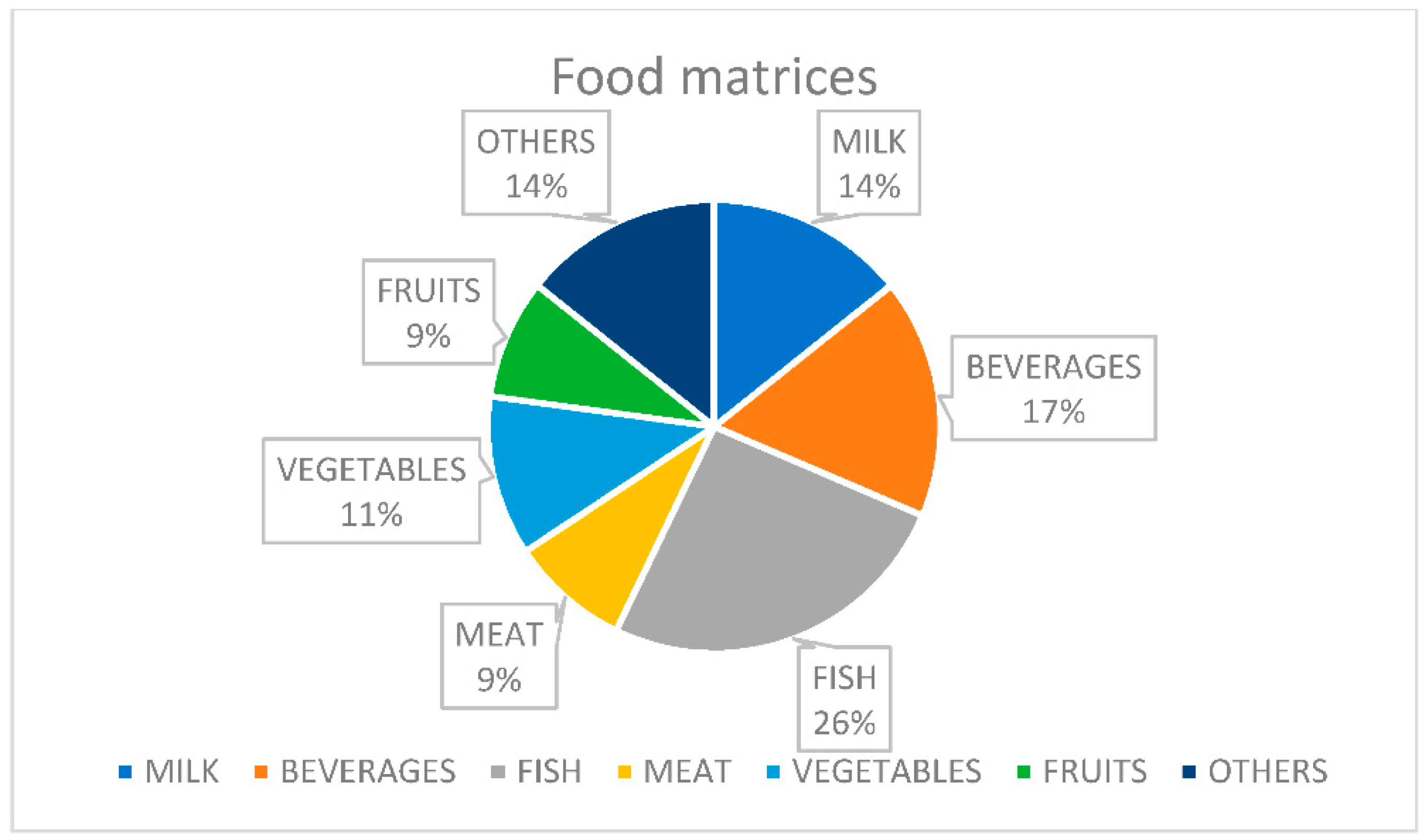
|
Matrix |
Analytes |
Analytical Technique |
MOF Material |
Sample Preparation Technique |
Recovery |
LODs |
Reference |
|---|---|---|---|---|---|---|---|
|
Milk |
Sulfonamides |
UHPLC-MS/MS |
MIL-101(Cr)@GO |
d-μSPE |
79.83–103.8% |
0.012–0.145 μg/L |
[29] |
|
Milk |
Penicillins |
UHPLC-TUV |
MIL-53 |
In tube SPME |
80.8–90.9% |
0.06–0.26 μg/L |
[30] |
|
Milk |
Tetracyclines |
HPLC-PDA |
ZIF-8 |
on-line SPE |
70.3–107.4% |
1.5–8.0 μg/L |
[31] |
|
Milk |
Estrogens |
HPLC-UV |
MOF-5 |
SPME |
73.1–96.7% |
0.17–0.56 ng/mL |
[32] |
|
Fruit tea |
Polycyclic aromatic hydrocarbons |
UHPLC-FLD |
Fe3O4@HKUST-1 |
D-μSPE |
On average 75% |
0.8 ng/L |
[33] |
|
Tea samples |
Pyrethroids |
GC-ECD |
MIL-101(Cr) |
MSPE-DLLME-SFO |
>0.015 ng/mL |
78.3–103.6% |
[34] |
|
Chrysanthemum tea |
Luteolin |
Square wave anodic stripping voltammetry |
Cu3(BTC)2/GO |
SPE |
7.9 × 10−10 mol/L |
99.4–101.0% |
[35] |
|
In tea and mushroom |
Hg(II) |
AFS |
JUC-62 |
SPE |
>0.58 mg/kg |
On average 93.3% |
[36] |
|
Fish |
Polychlorinated biphenyls |
GC-MS |
Fe3O4-MOF-5(Fe) |
SBSE |
0.061–0.096 ng/g |
>80% |
[37] |
|
Fish |
Polychlorinated biphenyls |
GC-MS |
MOF-5 |
SBSE |
0.003–0.004 ng/mL |
>80% |
[38] |
|
Fish |
Aromatic hydrocarbons and gibberellic acids |
GC-MS LC-MS/MS |
MOF-5 |
MSPE |
0.91–1.96 ng/L for PAHs and 0.006–0.08 μg/L for GAs |
66.4–120.0% for PAHs and 90.5–127.4% for GAs |
[39] |
|
Fish |
Triphenylmethane dyes |
HPLC-MS/MS |
MOF-5 |
MSPE |
0.30–0.80 ng/mL |
83.15–96.53 |
[40] |
|
Fish |
Cd(II) and Pb(II) |
FAAS |
MOF-199 |
MSPE |
0.2–1.1 μg/L |
92.8–117% |
[41] |
|
Fish |
Cd(II), Zn(II), Ni(II), and Pb(II) |
FAAS |
MOF-199 |
MSPE |
0.12–1.2 ng/mL |
>90% |
[42] |
|
Fish |
Hg(II) |
Cold Vapor AAS |
MOF-199 |
MSPE |
10 ng/L |
95–102% |
[43] |
|
Fish and shrimps |
Cd(II), Pb(II), and Ni(II) |
FAAS |
Fe3O4@TAR |
MSPE |
0.15–0.8 ng/mL |
NA |
[44] |
|
Shrimp samples, chicken and pork meat |
Sulfonamides |
HPLC-DAD |
Fe3O4@JUC-48 |
MSPE |
1.73–5.23 ng/g, |
76.1–102.6% |
[45] |
|
Chicken breast |
Drug traces |
LC-MS/MS |
MIL-101(Cr)@GO |
d-μSPE |
0.08 and 1.02 ng/kg |
88.9–102.3% |
[46] |
|
Lettuce |
Pesticides |
GC-MS |
∞[(La0.9Eu0.1)2(DPA)3(H2O)3] |
MSPD |
0.02–0.05 mg/kg |
78–107% |
[47] |
|
Fruits and vegetables |
Phytohormones |
HPLC-FLD |
UiO-66 |
Pipette Tip SPE |
0.01–0.02ng/mL |
88.3–105.2% |
[48] |
|
Fruits |
Plant growth regulator |
HPLC-FLD |
UIO-67 |
d-SPE |
89.3–102.3% |
0.21–0.57 ng/mL |
[49] |
|
Fruits |
Phytohormones |
HPLC-UV |
Zeolitic imidazolate framework-8 |
SBSE |
82.7–111% |
0.11–0.51μg/L |
[50] |
|
Fruits and vegetables |
of insecticides |
HPLC-UV |
Fe3O4@SiO2-GO MOF |
MSPE |
81.2–105.8% |
0.30–1.58 μg/L |
[51] |
|
Shellfish |
Shellfish poisoning toxin |
LC-MS/MS |
Fe3O4@SiO2@UiO-66 |
MSPE |
93.1% and 107.3% |
1.45 pg/mL |
[52] |
|
Rice |
Herbicides |
HPLC-UV |
MIL-101(Cr) |
MSPE |
83.9–103.5% |
0.010–0.080 μg/kg |
[53] |
|
Tomato sauce |
Sudan dyes |
HPLC-DAD |
Fe3O4-NH2@MIL-101 |
MSPE |
69.6–92.9% |
0.5–2.5 μg/kg |
[54] |
|
Peanuts |
Herbicides |
HPLC-DAD |
MIL-101(Cr) |
d-SPE |
89.5–102.7% |
0.98–1.9 μg/kg |
[55] |
|
In cereal, beverages and water samples |
Lead |
FAAS |
MOF-545 |
Vortex Assisted SPE |
91–96% |
1.78 μg/L |
[56] |
3. MOF-Derived Carbon Materials
|
Matrix |
Analytes |
Analytical Technique |
Precursor MOF Material |
Sample Preparation Technique |
Recovery |
LODs |
Reference |
|---|---|---|---|---|---|---|---|
|
Apples |
Carbamates |
HPLC-UV |
MOF-5 |
MSPE |
89.3–109.7% |
0.1–0.2 ng/g |
[59] |
|
Grapes and bitter gourd |
Herbicides |
HPLC-UV |
ZIF-67 |
MSPE |
88.9–105.1% for grapes, 89.6–105.0% for bitter gourd |
0.17–0.4 ng/g for grapes, 0.23–0.46 ng/g for bitter gourd |
[60] |
|
Fatmelon |
Neonicotinoid insecticides |
HPLC-UV |
ZIF-67 |
MSPE |
93.0–99.3% |
0.2–0.5 ng/g |
[61] |
|
Honey tea |
Chlorophenols |
HPLC-UV |
ZIF-8 |
MSPE |
83.0–114.0% |
0.1–0.2 ng/mL |
[62] |
|
Fruit juice and milk |
Endocrine disrupting compounds |
UHPLC-FLD |
MIL-53 |
MSPE |
92.2–108.3% |
0.05–0.10 ng/mL |
[63] |
|
Mushrooms |
Chlorophenols |
HPLC-UV |
MOF-5 |
MSPE |
0.25–0.30 ng/g |
85.4–97.5% |
[64] |
|
Chicken |
Fluoroquinolones |
HPLC-UV |
Cu based MOF |
DSPE |
0.18–0.58 ng/g |
81.3–104.3% |
[65] |
This entry is adapted from the peer-reviewed paper 10.3390/molecules23112896
References
- Manousi, N.; Raber, G. Recent advances in microextraction techniques of antipsychotics in biological fluids prior to liquid chromatography analysis. Separations 2017, 4, 18.
- Green Chemistry. Available online: http://www.epa.gov/greenchemistry (accessed on 30 September 2018).
- Arthur, L.C.; Pawliszyn, J. Solid phase microextraction with thermal desorption using fused silica optical fibers. Anal. Chem. 1990, 62, 2145–2148.
- Anthemidis, A.N.; Ioannou, K.I. On-line sequential injection dispersive liquid-liquid microextraction system for flame atomic absorption spectrometric determination of copper and lead in water samples. Talanta 2009, 30, 86–91.
- Karageorgou, E.; Manousi, N.; Samanidou, V.; Kabir, A.; Furton, K.G. Fabric phase sorptive extraction for the fast isolation of sulfonamides residues from raw milk followed by high performance liquid chromatography with ultraviolet detection. Food Chem. 2016, 196, 428–436.
- Samanidou, F.V.; Karageorgou, E. Carbon nanotubes in sample preparation. Curr. Org. Chem. 2012, 16, 1645–16699.
- Kyzas, G.Z.; Deliyanni, E.A.; Matis, K.A. Graphene oxide and its application as an adsorbent for wastewater treatment. J. Chem. Technol. Biotechnol. 2014, 89, 196–205.
- Maya, F.; Cabello, C.P.; Frizzarin, R.M.; Estela, J.M.; Palomino, G.T.; Cerda, V. Magnetic solid-phase extraction using metal-organic frameworks (MOFs) and their derived carbons. Trends Anal. Chem. 2017, 90, 142–152.
- Rostami, S.; Pour, A.N.; Salimi, A.; Abolghasempour, A. Hydrogen adsorption in metal-organic frameworks (MOFs): Effects of adsorbent architecture. Int. J. Hydrog. Energy 2018, 43, 7072–7080.
- Meek, S.T.; Greathouse, J.A.; Allendorf, M.D. Metal-organic frameworks: A rapidly growing class of versatile nanoporous materials. Adv. Mater. 2011, 23, 249–267.
- Yaghi, O.M.; Li, H. Hydrothermal synthesis of a metal-organic framework containing large rectangular channels. J. Am. Chem. Soc. 1995, 117, 10401–10402.
- Zhou, H.; Long, J.R.; Yaghi, O.M. Introduction to metal-organic frameworks. Chem. Rev. 2012, 112, 673–674.
- Hashemi, B.; Zohrabi, P.; Raza, N.; Kim, K. Metal-organic frameworks as advanced sorbents for the extraction and determination of pollutants from environmental, biological, and food media. Trends Anal. Chem. 2017, 97, 65–82.
- Furukawa, H.; Cordova, K.E.; O’Keeffe, M.; Yagh, O.M. The chemistry and applications of metal-organic frameworks. Science 2013, 341.
- Chen, Y.; Zhang, W.; Zhang, Y.; Deng, Z.; Zhao, W.; Du, H.; Ma, X.; Yin, D.; Xie, F.; Chen, W.; et al. In situ preparation of core-shell magnetic porous aromatic framework nanoparticles for mixed-mode solid-phase extraction of trace multitarget analytes. J. Chromatogr. A 2018, 1556, 1–9.
- Wu, H.B.; Lou, X.W. Metal-organic frameworks and their derived materials for electrochemical energy storage and conversion: Promises and challenges. Sci. Adv. 2017, 13.
- Chen, W.; Wu, C. Synthesis, functionalization, and applications of metal-organic frameworks in biomedicine. Dalton Trans. 2018, 47, 2114–2133.
- Chen, D.; Yang, D.; Dougherty, C.A.; Lu, W.; Wu, H.; He, X.; Cai, T.; Van Dort, M.E.; Ross, B.D.; Hong, H. In vivo targeting and positron emission tomography imaging of tumor with intrinsically radioactive metal-organic frameworks nanomaterials. ACS Nano 2017, 25, 4315–4327.
- Peller, M.; Böll, K.; Zimpel, A.; Wuttke, S. Metal-organic framework nanoparticles for magnetic resonance imaging. Inorg. Chem. Front. 2018, 5, 1760–1779.
- Chen, B.; Liang, C.; Yang, J.; Contreras, D.S.; Clancy, Y.L.; Lobkovsky, E.B.; Yaghi, O.M.; Dai, S. A microporous metal-organic framework for gas-chromatographic separation of alkanes. Angew. Chem. 2006, 45, 1390–1393.
- Gu, Z.-Y.; Yan, X.-P. Metal-organic framework MIL-101 for high-resolution gas chromatographic separation of xylene isomers and ethylbenzene. Angew. Chem. 2010, 49, 1477–1480.
- Gu, Z.-Y.; Jiang, J.-Q.; Yan, X.-P. Fabrication of isoreticular metal-organic framework coated capillary columns for high-resolution gas chromatographic separation of persistent organic pollutants. Anal. Chem. 2011, 83, 5093–5100.
- Chang, N.; Gu, Z.-Y.; Wang, H.-F.; Yan, X.-P. Metal-organic frameworks-based tandem molecular sieves as a dual platform for selective microextraction and high resolution gas chromatographic separation of n-alkanes in complex matrixes. Anal. Chem. 2011, 83, 7094–7101.
- Yang, C.-X.; Yan, X.-P. Metal-organic framework MIL-101(Cr) for high-performance liquid chromatographic separation of substituted aromatics. Anal. Chem. 2011, 83, 7144–7150.
- Liu, S.-S.; Yang, C.-X.; Wang, S.-W.; Yan, X.-P. Metal-organic frameworks for reverse-phase high-performance liquid chromatography. Analyst 2012, 137, 816–818.
- Yang, C.-X.; Chen, Y.-J.; Wang, H.-F.; Yan, X.-P. High-performance separation of fullerenes on metal-organic framework MIL-101(Cr). Chem. Eur. J. 2011, 17, 11734–11737.
- Gu, Z.-Y.; Yang, C.X. Metal-organic frameworks for analytical chemistry: From sample collection to chromatographic separation. Acc. Chem. Res. 2012, 45, 734–745.
- Chaikittisilp, W.; Ariga, K.; Yamauchi, Y. A new family of carbon materials: Synthesis of MOF-derived nanoporous carbons and their promising applications. J. Mater. Chem. A 2013, 1, 14–19.
- Jia, X.; Zhao, P.; Ye, X.; Zhang, L.; Wang, T.; Chen, Q.; Hou, X. A novel metal-organic framework composite MIL-101(Cr)@GO as an efficient sorbent in dispersive micro-solid phase extraction coupling with UHPLC-MS/MS for the determination of sulfonamides in milk samples. Talanta 2017, 169, 227–238.
- Lirio, S.; Liu, W.-L.; Lin, C.-L.; Lin, C.-H.; Huang, H.Y. Aluminum based metal-organic framework-polymer monolith in solid-phase microextraction of penicillins in river water and milk samples. J. Chromatogr. A 2016, 1428, 236–245.
- Yang, X.-Q.; Yang, C.-X.; Yan, X.-P. Zeolite imidazolate framework-8 as sorbent for on-line solid-phase extraction coupled with high-performance liquid chromatography for the determination of tetracyclines in water and milk samples. J. Chromatogr. A 2013, 1304, 28–33.
- Lan, H.; Pan, D.; Sun, Y.; Guo, Y.; Wu, Z. Thin metal-organic frameworks coatings by cathodic electrodeposition for solid-phase microextraction and analysis of trace exogenous estrogens in milk. Anal. Chim. Acta 2016, 937, 53–60.
- Rocío-Bautista, P.; Pino, V.; Ayala, J.H.; Pasán, J.; Ruiz-Pérez, C.; Afonso, A.M. A magnetic-based dispersive micro-solid-phase extraction method using the metal-organic framework HKUST-1 and ultra-high-performance liquid chromatography with fluorescence detection for determining polycyclic aromatic hydrocarbons in waters and fruit tea infusions. J. Chromatogr. A 2016, 1436, 42–50.
- Lu, N.; He, X.; Wang, T.; Liu, S.; Hou, X. Magnetic solid-phase extraction using MIL-101(Cr)-based composite combined with dispersive liquid-liquid microextraction based on solidification of a floating organic droplet for the determination of pyrethroids in environmental water and tea samples. Microchem. J. 2018, 137, 449–455.
- Wang, Y.; Wu, Y.; Ge, H.; Chen, H.; Ye, G.; Hu, X. Fabrication of metal-organic frameworks and graphite oxide hybrid composites for solid-phase extraction and preconcentration of luteolin. Talanta 2014, 122, 91–96.
- Wu, Y.; Xu, G.; Wei, F.; Song, Q.; Tang, T.; Wang, X.; Hu, Q. Determination of Hg (II) in tea and mushroom samples based on metal-organic frameworks as solid phase extraction sorbents. Microporous Mesoporous Mater. 2016, 235, 204–210.
- Lin, S.; Gan, N.; Qiao, L.; Zhang, J.; Cao, Y.; Chen, Y. Magnetic metal-organic frameworks coated stir bar sorptive extraction coupled with GC-MS for determination of polychlorinated biphenyls in fish samples. Talanta 2015, 144, 1139–1145.
- Lin, S.; Gan, N.; Zhang, J.; Qiao, L.; Chen, Y.; Cao, Y. Aptamer-functionalized stir bar sorptive extraction coupled with gas chromatography–mass spectrometry for selective enrichment and determination of polychlorinated biphenyls in fish samples. Talanta 2016, 149, 266–274.
- Hu, Y.; Huang, Z.; Liao, J.; Li, G. Chemical bonding approach for fabrication of hybrid magnetic metal-organic framework-5: High efficient adsorbents for magnetic enrichment of trace analytes. Anal. Chem. 2013, 85, 6885–6893.
- Zhou, Z.; Fu, Y.; Qin, Q.; Lu, X.; Shia, X.; Zhao, C.; Xu, G. Synthesis of magnetic mesoporous metal-organic framework-5 for the effective enrichment of malachite green and crystal violet in fish samples. J. Chromatogr. A 2018, 1560, 19–25.
- Sohrabi, M.R.; Matbouie, Z.; Asgharinezhad, A.A.; Dehghani, A. Solid phase extraction of Cd(II) and Pb(II) using a magnetic metal-organic framework, and their determination by FAAS. Microchim. Acta 2013, 180, 589–597.
- Taghizadeh, M.; Asgharinezhad, A.A.; Pooladi, M.; Barzin, M.; Abbaszadeh, A.; Tadjarodi, A. A novel magnetic metal-organic framework nanocomposite for extraction and preconcentration of heavy metal ions, and its optimization via experimental design methodology. Microchim. Acta 2013, 180, 1073–1084.
- Tadjarodi, A.; Abbaszadeh, A. A magnetic nanocomposite prepared from chelator-modified magnetite (Fe3O4) and HKUST-1 (MOF-199) for separation and preconcentration of mercury(II). Microchim. Acta 2016, 183, 1391–1399.
- Ghorbani-Kalhor, E. A metal-organic framework nanocomposite made from functionalized magnetite nanoparticles and HKUST-1 (MOF-199) for preconcentration of Cd(II), Pb(II), and Ni(II). Microchim. Acta 2016, 183, 2639–2647.
- Xia, L.; Liu, L.; Lv, X.; Qu, F.; Li, G.; You, J. Towards the determination of sulfonamides in meat samples: A magnetic and mesoporous metal-organic framework as an efficient sorbent for magnetic solid phase extraction combined with high-performance liquid chromatography. J. Chromatogr. A 2017, 1500, 24–31.
- Wang, Y.; Dai, X.; He, X.; Chen, L.; Hou, X. MIL-101(Cr)@GO for dispersive micro-solid-phase extraction of pharmaceutical residue in chicken breast used in microwave-assisted coupling with HPLC-MS/MS detection. J. Pharm. Biomed. Anal. 2017, 145, 440–446.
- Barreto, A.S.; da Silva, R.L.; Dos Santos Silva, S.C.; Rodrigues, M.O.; de Simone, C.A.; de Sá, G.F.; Júnior, S.A.; Navickiene, S.; de Mesquita, M.E. Potential of a metal-organic framework as a new material for solid-phase extraction of pesticides from lettuce (Lactuca sativa), with analysis by gas chromatography-mass spectrometry. J. Sep. Sci. 2010, 33, 3811–3816.
- Yan, Z.; Wu, M.; Hu, B.; Yao, M.; Zhang, L.; Lu, Q.; Pang, J. Electrospun UiO-66/polyacrylonitrile nanofibers as efficient sorbent for pipette tip solid phase extraction of phytohormones in vegetable samples. J. Chromatogr. A 2018, 1542, 19–27.
- Liu, L.; Xia, L.; Wu, C.; Qu, F.; Li, G.; Sun, Z.; You, J. Zirconium(IV)-based metal-organic framework(UIO-67) as efficient sorbent in dispersive solid phase extraction of plant growth regulator from fruits coupled with HPLC fluorescence detection. Talanta 2016, 154, 23–30.
- You, L.; He, M.; Chen, B.; Hu, B. One-pot synthesis of zeolitic imidazolate framework-8/poly (methylmethacrylate-ethyleneglycol dimethacrylate) monolith coating for stir bar sorptive extraction of phytohormones from fruit samples followed by high performance liquid chromatography-ultraviolet detection. J. Chromatogr. A 2017, 1524, 57–65.
- Wang, X.; Ma, X.; Huang, P.; Wang, J.; Du, T.; Dua, X.; Lu, X. Magnetic Cu-MOFs embedded within graphene oxide nanocomposites for enhanced preconcentration of benzenoid-containing insecticides. Talanta 2018, 181, 112–117.
- Zhang, W.; Yan, Z.; Gao, J.; Tong, P.; Liu, W.; Zhang, L. Metal-organic framework UiO-66 modified core-shell magnetic microspheres for magnetic solid-phase extraction of domoic acid from shellfish sample. J. Chromatogr. A 2015, 1400, 10–18.
- Liang, L.; Wang, X.; Sun, Y.; Ma, P.; Li, X.; Piao, H.; Jiang, Y.; Song, D. Magnetic solid-phase extraction of triazine herbicides from rice using metal-organic framework MIL-101(Cr) functionalized magnetic particles. Talanta 2019, 179, 512–519.
- Shi, X.-R.; Chen, X.-L.; Hao, Y.-L.; Li, L.; Xu, H.-J.; Wang, M.-M. Magnetic metal-organic frameworks for fast and efficient solid-phase extraction of six Sudan dyes in tomato sauce. J. Chromatogr. B 2018, 1086, 146–152.
- Li, N.; Wang, Z.; Zhang, L.; Nian, L.; Lei, L.; Yang, X.; Zhang, H.; Yu, A. Liquid-phase extraction coupled with metal-organic frameworks-based dispersive solid phase extraction of herbicides in peanuts. Talanta 2014, 128, 345–353.
- Tokalıoglu, S.; Yavuz, E.; Demir, S.; Patat, S. Zirconium-based highly porous metal-organic framework (MOF-545) as an efficient adsorbent for vortex assisted-solid phase extraction of lead from cereal, beverage and water samples. Food Chem. 2017, 237, 707–715.
- Introduction to Inorganic Chemistry/Coordination Chemistry and Crystal Field Theory. Available online: https://en.wikibooks.org/wiki/Introduction_to_Inorganic_Chemistry/Coordination_Chemistry_and_Crystal_Field_Theory (accessed on 27 September 2018).
- Liu, B.; Shioyama, H.; Akita, T.; Xu, Q. Metal-organic framework as a template for porous carbon synthesis. J. Am. Chem. Soc. 2008, 30, 5390–5391.
- Liu, X.; Wang, C.; Wu, Q.; Wang, Z. Magnetic porous carbon-based solid-phase extraction of carbamates prior to HPLC analysis. Microchim. Acta 2015, 183, 415–421.
- Liu, X.; Wang, C.; Wu, Q.; Wang, Z. Metal-organic framework-templated synthesis of magnetic nanoporous carbon as an efficient absorbent for enrichment of phenylurea herbicides. Anal. Chim. Acta 2015, 870, 67–70.
- Hao, L.; Wang, C.; Wu, Q.; Li, Z.; Zang, X.; Wang, Z. Metal-organic framework derived magnetic nanoporous carbon: Novel adsorbent for magnetic solid-phase extraction. Anal. Chem. 2014, 86, 12199–12205.
- Li, M.; Wang, J.; Jiao, C.; Wang, C.; Wu, Q.; Wang, Z. Magnetic porous carbon derived from a Zn/Co bimetallic metal-organic framework as an adsorbent for the extraction of chlorophenols from water and honey tea samples. J. Sep. Sci. 2016, 39, 1884–1891.
- Liu, X.; Feng, T.; Wang, C.; Hao, L.; Wang, C.; Wu, Q.; Wang, C. A metal-organic framework-derived nanoporous carbon/iron composite for enrichment of endocrine disrupting compounds from fruit juices and milk samples. Anal. Methods 2016, 8, 3528.
- Hao, L.; Liu, X.-L.; Wang, J.-T.; Wang, C.; Wu, Q.-H.; Wang, Z. Metal-organic framework derived magnetic nanoporous carbon as an adsorbent for the magnetic solid-phase extraction of chlorophenols from mushroom sample. Chin. Chem. Lett. 2016, 27, 783–788.
- Wang, Y.; Tong, Y.; Xu, X.; Zhang, L. Metal-organic framework-derived three-dimensional porous graphitic octahedron carbon cages-encapsulated copper nanoparticles hybrids as highly efficient enrichment material for simultaneous determination of four fluoroquinolones. J. Chromatogr. A 2018, 1533, 1–9.
- An Underused Framework for Simpler Sample Prep? Available online: https://theanalyticalscientist.com/issues/0618/an-underused-framework-for-simpler-sample-prep/ (accessed on 1 October 2018).
