Energy storage is one of the major barriers to achieve long-duration autonomy in robots. There are various types of rechargeable batteries such as lead-acid, nickel-cadmium, nickel-metal hydride and Lithium-ion batteries. Lead-acid batteries are the most mature rechargeable battery technology. However, Lithium-ion batteries are more suitable for mobile applications due to the high energy density properties, enabling lighter battery packs but with a higher cost.
- batteries
- robotics
- design engineering
- mobile robots
- space robots
1. Introduction
2. Current Battery Technologies
| LCO | Lithium Cobalt Oxide (LiCO2) |
| LFP | Lithium Iron Phosphate (LiFePO4) |
| LMO | Lithium Manganese Oxide (LiMn2O4) |
| LiPo | Lithium-ion Polymer |
| NCA | Lithium Nickel-Cobalt-Aluminum Oxide (LiNiCoAlO2) |
| NMC | Lithium Nickel-Manganese-Cobalt Oxide (LiNiMnCoO2) |
| LiS | Lithium Sulphur |
| LTO | Lithium Titanate (Li2TiO3) |
| Ni-MH | Nickel-Metal-Hydride |
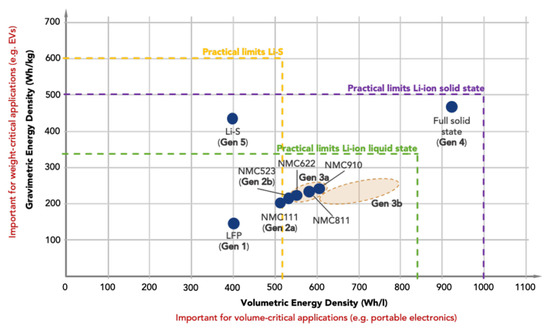
3. Capability Analysis of Battery Technologies for Robotic Applications
3.1. Battery-powered robots

- (1)
-
Space robots: the two main types of robots present in space missions are orbital and planetary robots, both having the possibility to carry robotic arms for manipulation [27].
- (2)
-
Drones: these are typically fixed-wing and multi-rotor drones.
- (3)
-
Underwater robots, more specifically Autonomous Underwater Vehicles (AUVs): These have electric motors on board to actuate their control surfaces (rudders and sterns), actuators for manipulation (robotic arms), in some cases, the thruster. Less conventional, non-commercial solutions such as snake robots, jellyfish robots or swimming (fish-like) robots are also usually actuated by electric motors [28].
- (4)
-
Wheeled and tracked mobile robots: Most unmanned ground vehicles (UGVs) fall under this category, but many other mobile robots also fit this description. The well-known humanoid robot Pepper, for example, can also be classified as a wheeled robot [29].
- (5)
- (6)
-
Wearable robots: This category includes powered prostheses and active exoskeletons, which exist for both upper and lower limbs. Exoskeletons can fulfill different functions: they can assist the wearer or augment his/her capabilities, at home or in industrial settings, or they can be used for rehabilitation purposes [33].
3.2. Suitability of battery chemistries
LFP and LTO present a good match for most robot categories. This is not a surprise, since these are also two of the most performant battery technologies. LFP emerges as the best-suited battery for all considered applications. An important disadvantage of the chemistry, however, is its self-discharge, which might trouble the balancing of the battery system [34]. LiPo presents an excellent alternative for drones, legged robots, wearable robots and wheeled robots thanks to its safety and its ability to deal with current variations. Moreover, the use of gel electrolytes instead of liquid electrolytes makes LiPo batteries safer and lighter by providing flexibility in packaging. LCO and LMO score poorly overall as these mature technologies are being overtaken by other technologies. In contrast, the emerging LiS technology still needs considerable improvements to become competitive. Finally, NMC and NCA turn out as suitable chemistries for specific applications.
It is important to note that tailor-made solutions and specificities of the chemistries can have an impact on the choice for specific chemistry. For example, the lighter packaging of LiPo batteries makes them the cell of choice for most commercial electronic devices [11]. High-rate LFP chemistry is used in only a small number of drones for shorter missions, due to its good high-current discharge abilities. NMC technology is utilized for longer missions where energy demand (specific energy) dominates the selection process.
The pressure tolerance of the battery is an additional consideration that guides the Lithium-ion chemistry choice for underwater robots [35]. Overall, there is no clear preference in the present battery selection for underwater robots. The present Lithium-ion technologies are replacing the usually used lead-acid and Ni-MH batteries providing more energy and lifespan [36]. LiPo batteries are considered a suitable choice since their packaging can be designed to resist pressure in deep water. NMC, LTO and NCA can also be good alternatives for underwater robots due to their lifespan.
In commercial wheeled and tracked robots, a variety of Lithium-ion batteries are used, including LFP and LiPo batteries. LFP technology indeed ranks first in the battery selection for this category of robots, which are very balanced in terms of requirements. LiPo batteries – the third choice after LTO – can bring light packaging and flexibility as additional advantages.
For legged robots, LTO and LiPo perform similarly as LFP. The limited commercial availability of LTO batteries makes LFP and LiPo technologies, which dominate the market share, the preferred choice. LiPo batteries are more common as the polymer electrolytes endow them with the ability to support the high current pulses typical of legged robots.
Specifics about the battery chemistry of wearable robots are rarely reported. LiPo technology is an excellent choice for these robots, as it fits most of the requirements for current variation, specific power and safety. LiPo battery packs have indeed been used in several exoskeletons, although newer exoskeletons increasingly rely on Lithium-ion chemistries [41,42]. LFP and LTO can be considered the most suitable technologies.
Due to their excellent properties in terms of current variation and lifespan, LFP and LTO technologies could be considered for space robots. However, due to high self-discharge (important for long missions) and lack of commercial availability, they are not commonly used. NCA is found to be the key technology of interest in NASA’s future consideration as they are closely studying various market available NCA batteries [44]. It is also the battery chemistry that was selected for the Mars Rover. In recent missions, robots have typically been equipped with NCO technology batteries, which are similar in terms of chemistry to NCA but lack the presence of Aluminum [43]. LCO is often used in NASA missions. The possible reasons for this may include customized battery cell, maturity of the technology, optimized anode resisting lithium-plating at low temperature, better stability etc. [45]. For satellite missions, both LiPo and Lithium-ion cells are being considered depending on the features of interest [46,47].
4. Additional Technologies for Improved Performance
4.1. Battery Management Systems
4.2. Thermal Management Systems
4.3. Recharging and Battery Swapping
4.4. Hybrid Architectures
4.5. Packaging
This entry is adapted from the peer-reviewed paper 10.3390/en16134904
