With rapidly increasing environmental pollution, there is an urgent need for the development of fast, low-cost, and effective sensing devices for the detection of various organic and inorganic substances. Silver nanoparticles (AgNPs) are well known for their superior optoelectronic and physicochemical properties, and have, therefore, attracted a great deal of interest in the sensor arena. The introduction of AgNPs onto the surface of two-dimensional (2D) structures, incorporation into conductive polymers, or within three-dimensional (3D) nanohybrid architectures is a common strategy to fabricate novel platforms with improved chemical and physical properties for analyte sensing.
- silver nanoparticles
- chemical reduction
- voltammetric sensors
- amperometric sensors
- environmental analysis
1. Introduction
2. Application of Silver Nanoparticles in Voltammetric and Amperometric Sensors
2.1. Working Principles of Voltammetric and Amperometric Sensing Techniques
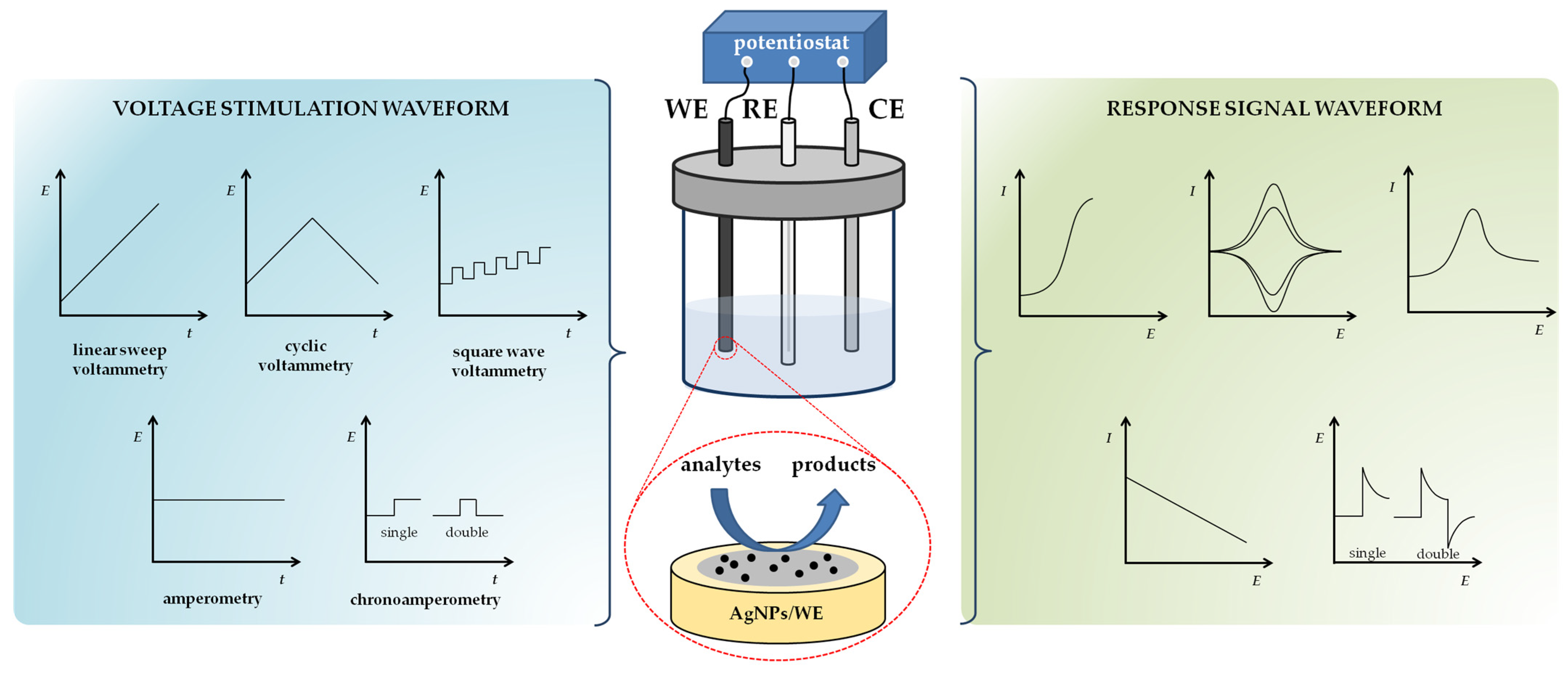
2.2. Electrochemical Sensors for Detection of Heavy Metal Ions
2.2.1. Electrochemical Sensors for Divalent HMs
2.2.2. Electrochemical Sensors for Trivalent and Hexavalent HMs
2.3. Electrochemical Sensors for Nitrogen-Containing Inorganic Species
2.3.1. Electrochemical Sensors for Nitrite (NO2−) and Ammonium (NH4+) Detection
2.3.2. Electrochemical Sensors for Nitrate (NO3−) and Ammonia (NH3) Detection
2.4. Electrochemical Sensors for Phenolic Compounds
2.5. Electrochemical Sensors for Pharmaceuticals
2.6. Electrochemical Sensors for Nitroaromatics
2.7. Electrochemical Sensors for Natural and Synthetic Estrogens
3. Discussion
3.1. Electrochemical Sensor Technology
Electrochemical sensor technology has become an important aspect of modern analytical chemistry, and great efforts are being made to develop novel and cost-effective sensing platforms that provide a fast, accurate, and repeatable response to the analyte of interest. Compared to ubiquitous colorimetric sensors, the main advantage of electrochemical detection is the ability to measure in turbid samples [71]. Moreover, working electrodes can also be made in a planar and/or flexible mode [72][73][74][75], i.e., they can be easily miniaturized and integrated into more hierarchical devices [76]. Electrochemical sensors can be used in various fields, including pharmaceuticals, medicine, chemistry, synthesis, materials engineering, and biotechnology [77][78][79]. In addition, electrochemical sensors based on disc (3D) or planar (2D) electrodes decorated with AgNPs have proven to be simple but reliable analytical tools for the rapid and selective detection of emerging water pollutants (i.e., heavy metal ions, nitrogen-containing inorganic species, phenolic compounds, nitroaromatics, pharmaceuticals, and natural and synthetic hormones), which is highlighted.
3.2. Wet Chemical Synthesis of AgNPs for Sensing Applications
For the fabrication of highly selective and sensitive voltammetric and amperometric electrochemical sensors, the usage of silver nanomaterials with precise particle size, shape, crystalline facets, and morphology is essential. Hence, controlled synthesis is a paramount for achieving excellent electrocatalytic activity for practical sensing application. AgNPs are usually synthesized using a chemical reduction from soluble silver(I) precursors (mostly AgNO3), and many of these synthetic processes are carried out under harsh reaction conditions applying toxic hydrazine [80] or borohydride [81][82][83] reducing agents and/or (volatile) organic solvents [84]. Although there has been more recent focus towards green synthesis [85][86][87], in 64 of the 86 electrochemical sensors presented the reduction of silver(I) to zero-valent form was assisted by electric current or inorganic reducing agents. It is important to emphasize that if the chemical reduction process using strong inorganic agents is precisely controlled, i.e., if no hazardous chemicals are released into the environment during the synthesis [88], such an approach does not necessarily contradict to the principles of green synthesis. Moreover, AgNPs of smaller size and cleaner particle shapes can be produced with strong reducing agents: (i) nanoball-shaped AgNPs (d = 5–6 nm) via the polyol process [89], (ii) spherical crystals (mean diameter of 10.6 nm) obtained via borohydride reduction [90], or (iii) globular particles with a diameter of ~20 nm via ethanolamine reduction [91]. On the other hand, biosynthesized AgNPs generally exhibit irregular morphologies, are polydisperse, and often form larger particle agglomerates [92][93][94]. Nevertheless, even strong reducing agents can render AgNPs of various morphologies (quasi-spherical, cubic, twinned structure, and triangles) and larger size distributions when deposited on highly wrinkled and folded graphene nanosheets [95], or when the Ag(I) precursor is previously encapsulated with a massive chelating ligand [96], while a precisely controlled green approach can produce remarkably small particles (d = 5 nm) with high catalytic efficiency [97]. AgNPs of precise size and shape can also be formed and fine-tuned using electric current [98][99][100][101]. In electrochemical sensors based on electrodeposited silver nanomaterial [102][75], the addition of silver material was found to play a key role in the trace-level quantification of both trivalent and hexavalent chromium through formation and stabilization of functional bimetallic metal oxides [102], and in the formation of dual-region WE for simultaneous detection of inorganic nitrogen-containing species (nitrate/ammonia) through a signal current channel [75].
3.3. Electroanalytical Techniques for Characterization of AgNPs-Modified Electrodes and Analyte Quantification
In general, cyclic voltymmetry and impedance measurements are used to study the changes in the interfacial phenomena between the sensor body and solution when different electrode coating materials are used. In other words, CV and the EIS technique are indispensable tools to observe in detail the stepwise evolution of the electrochemical sensor fabrication pathway. To implement both techniques the usage of a ferri/ferrocyanide redox probe is needed, and measurements are usually performed in aqueous 0.1 M KCl [103][104][105][106][107] or in a phosphate buffer solution [108][109]. With the introduction of AgNPs as electrode modifier material, both the increase of current peak and the reduction in peak separation are noticeable with cyclic voltammetry [89]. The AgNPs modification of the bare electrode is also evident in electrochemical impedance spectroscopy, where the value of electron transfer resistance, which corresponds to the semicircle diameter of the Nyquist diagram, is significantly lower compared to the bare electrode (Figure 2). Furthermore, the diffusion-limited process is represented by the linear portion of the Nyquist plot at lower frequencies, while the process is related to the part of the diagram at higher frequencies. In summary, the specific role of the nanosilver material in the exchange of the electron transfer rate between the electroactive species (redox couple) and the modified sensing surface is attributed to the enhancement of the maximum peak current (CV analysis) and the diminution of the impedance (EIS analysis) .
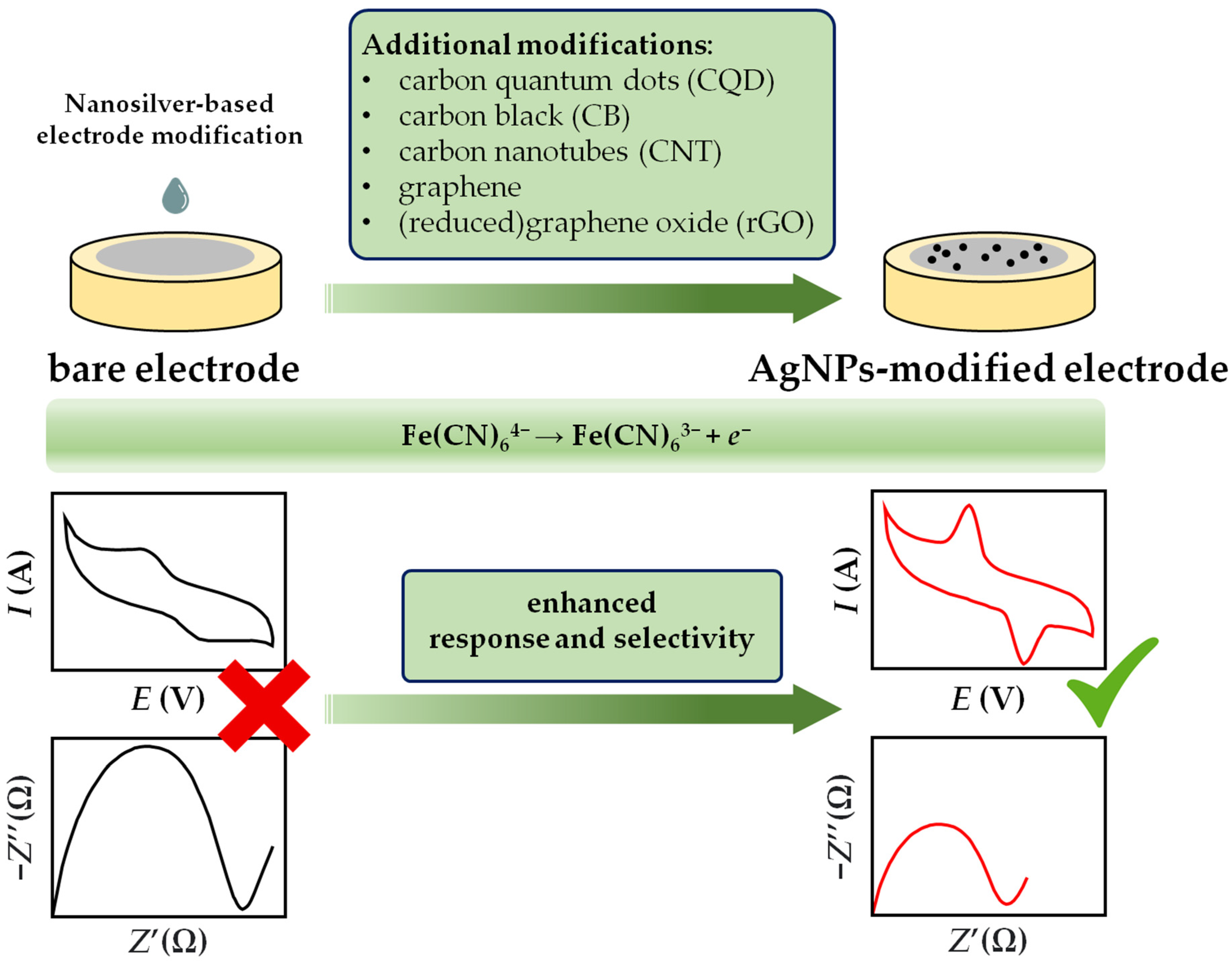
Figure 2. Schematic illustration of AgNPs electrochemical signal boost through the changes in current (CV) and impedance (EIS) responses on bare and nanosilver-modified electrodes.
Voltammetry and amperometry are particularly attractive techniques for the analysis of complex sample matrices such as various environmental waters (groundwater, lakes, rivers, streams, seawater, etc.) The choice of voltammetric technique depends primarily on the location of the sample and the expected concentration of the analyte. Although CV can be used to determine analyte concentration [110][111][112][75], DPV or SWV methods have been shown to be better for quantitative analysis. SWV is known for its superior sensitivity and measurement speed compared to DPV technique [113]. For greater sensitivity, analyte preconcentration can be performed by adsorptive square wave stripping voltammetry [114][115].
3.4. Mechanisms of AgNPs-based Voltammetric and Amperometric Sensors
The detection mechanism of nanosilver-based voltammetric and amperometric sensors for water pollutant monitoring summarized is based on several different mechanisms: (i) AgNPs aggregation; (ii) AgNPs displacement; (iii) AgNPs electronic conductive channels enhancement/inhibition via selective site recognition; and (iv) OR reaction of inorganic/organic water pollutants.
(i) Aggregation of AgNPs triggered by the addition of the analyte is a common optical sensing approach that can be converted to voltammetric [116] and amperometric sensors [117]. This strategy is feasible usually in the form of dual- [116][118] or multisensing [119][120][121][122] platforms, with optical, fluorescent, and electrochemical response towards the detection of HMs. In contrast to the colorimetric assay, electrochemical aggregation of functional silver material results in voltammetric/amperometric signal amplification or inhibition, depending on the chemistry of the nanoparticle stabilizer and the target analyte. For the aggregation-induced sensing mechanism, the role of biogenic synthesized AgNPs is highlighted because biogenic molecules are rich in electron-donor groups that successfully act as analyte-chelating ligands. AgNPs synthesized using fungus Agaricus bisporus [116] and the Mimosa diplotricha leaf extract [118] were found to be potent sensors towards hazardous mercury, boosting the DPV responses through strong stabilizer complexation. Green AgNPs also play a prominent role as multisensor probes for mercury [119], cadmium [120], arsenic [123], and chromium [122].
(ii) Electrochemical sensors based on the displacement of functional nanomaterials are considered a novel tool for highly selective and sensitive detection of analytes. In two examples of the reviewed scientific papers, the mechanism of mercury detection was associated with the removal of the surface-bound stabilizer (calixarene moiety), leaving the AgNPs uncapped [117], or with galvanic displacement of silver by mercury [124]. As thermodynamically unstable species, the bare nanoparticles tend to aggregate into larger clusters, or form a amalgam [117]. This leads to signal inhibition, which is visible in the optical mode by a reduction in the SPR band in the UV-Vis spectrum, and in the amperometric mode by a reduction in current output. In the galvanic displacement method, the AgNPs interact with divalent mercury ions in the solution and convert the zero-valent silver into ionic silver species, resulting in loss of stripping signal. Since only small amounts of the analyte are required to displace measurable amounts of silver, this sensor can quantify inorganic mercury in the picomolar range.
(iii) Aggravation or enhancement of the electron transfer pathway of AgNPs-modified WE leads to the amplification or decrement of voltammetric performance. Two voltammetric platforms based on MIPs supported by AgNPs-decorated GCE have been developed for highly selective and sensitive detection of natural estrogen in environmental and drinking water with picomolar and femtomolar detection limits [101][125]. The detection of the analyte is catalyzed by the AgNPs (signal amplification or inhibition), while the analyte capture and selective recognition were performed by the MIPs. In the presence of E2, more imprinted cavities of the poly(p-aminophenol) (MIP-pAPh) were occupied, resulting in direct amplification of the current response by AgNPs signal amplification [101]. The reverse sensing mechanism was proposed for the AgNP/GO/MIP (poly-imidazole; PImi) functional platform [125]. In addition to the imidazole moiety (p-type-electron acceptor), additional functional sites of GO (n-type electron donor) were available for analyte binding, which blocked the diffusion of the ferri/ferrocyanide redox probe at the electrode/solution interface, reducing the SWV current responses.
(iv) Voltammetric and amperometric sensors, which detect inorganic/organic pollutants based on their OR response, are the most represented electrochemical sensor class. AgNPs increase the active surface area of the bare and/or modified electrode, facilitate electron transfer, and serve as active catalytic sites for oxidation/reduction of the aquatic pollutant. It has been reported that the reaction rate constant (k) is proportional to the total effective surface area of the nanosilver material [126]. Therefore, the homogeneity of particle size and shape of AgNPs anchored on sensing platforms is of utmost importance for catalytic efficiency. The adsorption of the analyte (the first step in the sensing mechanism) is more promoted on small particles with larger effective surface area, which facilitates the OR process of electroactive species on the modified electrodes [127]. The interactions between the AgNPs, electrode support and/or other electrode modifiers, and the target analyte, depends on the nature of the surface-active species. On the one hand, stabilizers can be interpreted as a barrier between the AgNPs and the electrode material, controlling the electron transfer kinetics and, thus, affecting the analytical performance of the sensor, especially in terms of achieving lower detection limits [103]. Therefore, clean and uncovered active surface sites of nanoclusters are preferred for catalytic and sensing applications [128]. However, since unprotected AgNPs tend to aggregate, the presence of a stabilizer is extremely important. The catalytic activity of green-capped AgNPs can be altered by the size of the stabilizer (bulky ligands or small molecules) [129][130], and by the adsorption of intermediary molecules (formed during the sensing mechanism) onto the biostabilizers, as was the case in the amperometric quantification of nitrobenzene [131]. The determination of nitrites based on their irreversible electrochemical oxidation via two-electron transfer was reported for various AgNPs-functionalized electrodes, i.e., nanospheres decorated GCE [93], core-shell Au@Ag structure anchored over carboxylated graphene/GCE [132], AgNPs-(r)GO platform [133][134], AgNPs/rGO nanohybrid in conjunction with PPy [135] and PANI [136] conducting polymers, AgNPs/ZnO nanocomposite [137], and AgNPs/HNT/MoS2 platform [138]. In contrast, one-electron transfer was demonstrated in nitrite sensing with a nanosilver-polymer composite (chitosan, PEDOT:PSS) [139], and also with an AgNPs-MWCNTs hybrid platform [140]. AgNP/SWCNT/CPE was used to detect DNL by the irreversible oxidation of phenolic groups to (semi)quinone species [141]. The overall chemical–electrochemical mechanism involves a transfer of two protons and two electrons, but the final product depends on the applied potential and the electrode modifier used. Oxidation products of E3, obtained using GCE coated with AgNP/CNB [89] and AgNPs-rGO [142], correspond to ketone derivatives. For ultrasensitive detection of AM and AT drugs, an oxidation mechanism involving rapid electron transfer from the analytes through the COOH-CNTs/Ag/NH2-CNTs ternary sandwich architecture to the GCE was proposed [115]. The overall electrochemical oxidation (single SWASV profile) of the electroactive center 1,4-dihydropyridine (AM) occurs through a two-electron/two-proton process, whereas the pyrrole center of AT exhibits two-electron/one-proton transfer. The reversible oxidation of CC and HQ phenolic compounds to o- and p-benzokinone species via a two-electron/two-proton transfer was mediated by the cobalt active site of the AgNPs/TACoPc/PANI ternary composite using the DPV technique [143] and with AgNPs/Fe3O4-rGO hybrid material [144]. Quantitative determination of BPA via electrooxidation to 2,2-bis(4-phenylquinone) was successfully performed with an AgNPs-rGO composite on a ITO substrate [72], and with an AgNPs-rGO/PLL-coated GCE [80]. Conversely, several phenolic compounds were detected using the reduction mechanism. The nitro groups of 4-nitrophenol [108][128][145], 2,6-dinitrophenol [146], 4-nitrotoluene [147], nitrofurantoin [107], and nitrobenzene [148][94][131] were irreversibly converted into hydroxylamine or aminophenol groups by an electrochemical mechanism involving four protons and four electrons. Moreover, nitrobenzene was detected on a silicate sol–gel matrix GCE functionalized with AgAu alloy, depending on the electrochemical reduction of the nitro group involving six protons and six electrons [149]. Gold electrode coated with AgNPs [150][84], in conjunction with the SWV technique, proved to be a highly efficient combination for the detection of nitrates in seawater due to their two-electron reduction to nitrites. In addition, nitrate was detected on a Cu(II)-terephthalate decorated SPCE via a Cu2+/Cu+ redox couple [151]. The adsorption of nitrates is promoted on the Cu(I) active site by copper–oxygen coordination interactions, which is further supported by the charge transferability of MOFs and the enhanced transfer of two electrons to the catalytic AgNP sites.
3.5. Design of AgNPs-based Voltammetric and Amperometric Sensors for Detection of Aquatic Pollutants
The choice of bare electrode substrate (supporting material) is of paramount importance in manufacturing process of electrochemical sensors. The common denominator in the use of pure metal electrodes (Au or Pt), or bare carbon-based electrodes (GCE, GPE, PGE, CPE, SP(C)E, etc.), is the difficult electron transfer between the electroactive analyte molecule and the sensing electrode materials. This leads to low selectivity towards the analyte (the redox reaction is possible at potentials substantially higher than thermodynamic potentials), and inability to distinguish the target molecule from interfering species in real matrices. Therefore, modification of bare electrodes with (hybrid) nanomaterials, especially noble metallic nanoparticles, offers a unique combination of excellent catalytic and sensory properties [110][152][153]. Silver nanoparticles outperform gold and platinum nanomaterials as a low-cost and efficient electrocatalysts for practical sensing applications. Moreover, the synergistic effects of bimetallic [154][132][155], AgNPs/metal oxide composites [156][137] can be also used to promote and/or enhance catalytic sensing.
Due to its diverse structural and morphological forms, carbon is remarkably important and one of the mostly widely used electrode materials [157]. In 54% of the voltammperometric sensors presented , GCE was used as the core material. The main advantages of GCE are its resistance to acidic and alkaline environments and the improved adhesion of the AgNPs to the glassy carbon material [158]. Only a few examples of GCE modified exclusively with AgNPs for the detection of nitrites [110][159][93], nitrobenzene [106][131], and 4-nitrophenol [128] have been found; the vast majority of GCE-based electrochemical sensors are modified with composite silver (nano)materials. One of the frequently used strategies in the development of novel sensor surfaces is the anchoring of AgNPs on the surface of 2D nanomaterials, mostly graphene-derived, due to their large surface area, high chemical stability, and mechanical strength [160][161]. The GO platform is rich in oxygen-containing functional groups, which greatly improves its hydrophilicity and functionality. Therefore, GO is easily dispersed in both water and organic solvents to obtain a homogeneous dispersion, and can be easily applied to electrode substrates by simple immersion or drop casting technique [133][91]. In addition, GO has an enhanced ability to capture target molecules due to its rich edge oxygen chemistry, a property that has been used to develop a nitrite sensing platform [133]. To increase the sensitivity of the sensor, the strategy of combining AgNPs with a more conductive reduced GO form (restoration of sp2 hybrid carbon networks) was developed [80][144][95][142]. Thus, (r)GO serves as an outstanding platform for the immobilization of various electroactive species through covalent or noncovalent bonds. In addition to (r)GO sheets, other 2D layered structures with semiconducting properties such as activated C3N4 [105][148][162], MOFs [151][163], and transition metal dichalogenides [164] have also emerged as prospective candidates for improved catalyst supports for simultaneous voltammetric detection of HMs, selective oxidation of nitrite, amperometric nitrate detection, and selective dopamine quantification, respectively. 3D MWCNTs, especially functionalized by acid treatment, possess excellent thermal and electrical conductivity, which, combined with their large surface area, significantly improves the catalytic properties for the oxidation of ammonium and nitrite species [140][112][165], and the detection of phenolic compounds [99][166] in real water matrices. In only one scientific paper, SWCNTs were used in conjunction with AgNPs as a sensing surface for the quantification of the synthetic estrogen dienestrol in river water [141]. Another approach to increase the sensitivity is to use the CPs in the nanosilver-based sensing hierarchy in the form of an AgNPs@p-1,8-DAN layer [167] or an AgNPs/CB/PEDOT:PSS film [90] built over disc GCE, or ternary hybrid structures of nanosilver on the (r)GO platform with PPy [135] or PANI [168]. Due to their (high) conductivity, redox reversibility, long-term environmental stability, high solution process ability, and simple synthetic procedures with controllable thickness on the sensing electrode, CPs are often employed in voltammperometric sensor design. Synthetic MIPs based on GCE sensing platforms are the best alternative to circumvent the stability and cost issues associated with biological receptors traditionally used for the detection of (bio)molecules [169]. Since the size, shape, and orientation of the recognition sites of the cavities imprinted in MIP directly reflect the properties of the analyte, the molecular imprint technology is more than efficient for the development of highly selective sensors. MIPs in combination with AgNPs and AgNPs/GO composites have proven to be exquisite sensing platforms—picomolar and femtomolar LOD values have been achieved for the detection of the endocrine-disrupting 17-β-estradiol in real water matrices [101][125].
In addition to GCE, another robust carbon-based 3D electrode—CPE—can be quickly fabricated, modified with various (nano)materials, and its surface is easy to clean. Although their sensitivity is (usually) lower than that of GCEs, CPEs decorated with AgNPs have proven to be a reliable tool for amperometric detection of synthetic estrogen (nM LOD) [141], amperometric quantification of nitrites in aquatic solutions (μM LOD) [138], selective voltammetric reduction of p-nitroaniline in wastewater (nM LOD) [170], and electrochemical sensing of lead ions in tap water and wastewater specimens (μM LOD) [171]. Another simple and inexpensive form of a carbon-based electrode, with advantages such as high mechanical strength, good quality, stability, and reproducibility, is the PGE [172]. This user-friendly electrode can be used as disposable electrode, eliminating the time-consuming cleaning of solid electrodes. A PGE sensing probe modified with an AgNP-ZnO nanocomposite has been successfully used for the determination of nitrites in lake water and pickled water samples [137], and one decorated with biosynthesized AgNPs was selective towards divalent mercury [111].
Noble metal electrodes, such as gold and platinum material, find application in a variety of electrochemical processes due to their high electrical conductivity, mechanical stability, and chemical resistance. Nevertheless, only a few electrochemical sensors based on nanosilver-decorated noble metal electrodes have been presented . Gold (Au) disc electrodes predominate in the development of voltammetric sensors for the detection of arsenic in river water and lake water [173][100][123], but are also suitable as substrates for the selective and sensitive detection of nitrates in seawater matrices [150][84]. Platinum (Pt) electrodes, decorated with AgNPs biosynthesized utilizing Agaricus bisporus mushroom extract [116] and Mimosa diplotricha plant leaf extract [118], were found to be an excellent solution for the selective and sensitive detection of toxic Hg(II) in lake water, tap water, and river water specimens, respectively. In addition, a Pt disc electrode decorated with bark green AgNPs (Moringa oleifera bark extract) was utilized to detect hazardous Cu(II) from electroplating plant effluents [121], while AgNPs produced with garlic extract were used for the quantification of Cd(II) in lakes [120].
Planar screen-printed (carbon) electrodes are suitable for on-site analyses because they are built on the same substrate with a three-electrode setup (WE, RE, and CE). Planar SPE surfaces can be modified in the same way as disc electrodes by using a: (i) hybrid of AgNPs and Cu(II)–terephthalate MOFs for the quantification of nitrate in drinking water [151], (ii) silver nanoseed/carbon nanofiber modifier for simultaneous detection of Cu(II) and Pb(II) in groundwater matrices [81], (iii) a curcumin-stabilised AgNPs-FeCo2O4 nanosheets-rGO complex architecture for the detection of 4-NP in industrial wastewater [174], (iv) a ternary Ag/Co3O4/chitosan composite for the removal of 4-nitrophenol in sewage [175], (v) bimetallic silver-gold-oxide nanoparticles for simultaneous determination of trivalent and hexavalent chromium in tap water [176]. In addition to carbon-based planar surfaces, AgNPs-decorated glass [177], ITO substrates [72][178], and FTO glass substrates [179] were successfully employed for the selective detection of nitrite, nitrate, BPA, and hazardous copper, respectively.
3.6. Summary and Future Perspective
In summary, the use of silver nanomaterial-decorated electrodes as voltammperometric sensors offers several advantages based on the electroanalysis output: (i) efficient catalysis; (ii) rapid mass transfer of the target analyte from the bulk solution to the sensing surface and vice versa; (iii) large sensor surface area; and (iv) the ability to precisely tune the electrode microenvironment. Although electrochemical sensing technologies may achieve quantification of analytes even at femtomolar levels, they still lack quality assurance and reliability for long-term measurements in complex water matrices.
Therefore, to ensure an advanced and self-sufficient next generation of nanosilver-based voltammetric/amperometric probes, future research must focus on the: (i) the development of low-cost and submersible sensor platforms for real-time detection of pollutants in water matrices; (ii) fabrication of lighter weight and small-sized devices—miniaturization and planar (all-printed all-solid state) electrode design; (iii) construction of wireless and portable sensors for measurements at remote locations with minor facilities; (iv) sensitivity and selectivity augmentation overcoming interference and fouling challenges; and (v) development of self-sustained devices.
4. Conclusions and Outlooks
Electrochemical sensing in combination with nanosilver-based materials and various electroanalytical techniques offers attractive opportunities to meet the requirements for accurate detection of emerging water pollutants. The high conductivity of AgNPs facilitates rapid charge transfer during detection of the target species, while superior catalytic efficiency can be easily tuned by changing the particle size, shape, and temperature of the reaction mixture. Compared to other metal-based nanoparticles, AgNPs have relatively lower cost and toxicity, ensuring their great potential for practical applications. Modification of electrode substrates with silver hybrid architectures by introducing 2D carbon (graphene and its derivatives, carbon black, carbon cloth), 3D carbon nanomaterials (quantum dots, carbon nanotubes), and MOFs or CPs as support materials forms adventurous sensing surfaces for detection of various analytes. Molecular imprinting is by far the most sensitive targeting technology, enabling rapid and ultrasensitive quantification of analytes by fine-tuning the recognition sites in the MIP cavities.
So far, different synthesis strategies have been developed to prepare Ag-based nanocomposite materials decorated WE, from simple immersion and drop casting to in situ reduction method. By combining the advantages of the different synthesis methods and the various modification techniques, high-performance detection is possible for practical in-field applications. Although numerous studies on the synthesis and environmental applications of AgNPs-based electrochemical sensors have been conducted recently, the development of highly selective and sensitive platforms with the possibility of miniaturization and integration into portable devices for real-time measurements and sustainable use of water resources remains a challenge.
This entry is adapted from the peer-reviewed paper 10.3390/s23073692
References
- Khalil, M.; Kadja, G.T.; Ilmi, M.M. Advanced nanomaterials for catalysis: Current progress in fine chemical synthesis, hydrocarbon processing, and renewable energy. J. Ind. Eng. Chem. 2021, 93, 78–100.
- Khalil, M.; Jan, B.M.; Tong, C.W.; Berawi, M.A. Advanced nanomaterials in oil and gas industry: Design, application and challenges. Appl. Energy 2017, 191, 287–310.
- Pareek, V.; Gupta, R.; Panwar, J. Do physico-chemical properties of silver nanoparticles decide their interaction with biological media and bactericidal action? A review. Mater. Sci. Eng. C 2018, 90, 739–749.
- Torrinha, A.; Oliveira, T.; Ribeiro, F.; Correia, A.; Lima-Neto, P.; Morais, S. Application of Nanostructured Carbon-Based Electrochemical (Bio)Sensors for Screening of Emerging Pharmaceutical Pollutants in Waters and Aquatic Species: A Review. Nanomaterials 2020, 10, 1268.
- Karthik, V.; Selvakumar, P.; Kumar, P.S.; Satheeskumar, V.; Vijaysunder, M.G.; Hariharan, S.; Antony, K. Recent advances in electrochemical sensor developments for detecting emerging pollutant in water environment. Chemosphere 2022, 304, 135331.
- Tajik, S.; Beitollahi, H.; Nejad, F.G.; Dourandish, Z.; Khalilzadeh, M.A.; Jang, H.W.; Venditti, R.A.; Varma, R.S.; Shokouhimehr, M. Recent Developments in Polymer Nanocomposite-Based Electrochemical Sensors for Detecting Environmental Pollutants. Ind. Eng. Chem. Res. 2021, 60, 1112–1136.
- Azzouz, A.; Kailasa, S.K.; Kumar, P.; Ballesteros, E.; Kim, K.-H. Advances in functional nanomaterial-based electrochemical techniques for screening of endocrine disrupting chemicals in various sample matrices. TrAC Trends Anal. Chem. 2019, 113, 256–279.
- Li, D.; Wang, T.; Li, Z.; Xu, X.; Wang, C.; Duan, Y. Application of Graphene-Based Materials for Detection of Nitrate and Nitrite in Water—A Review. Sensors 2020, 20, 54.
- Su, S.; Chen, S.; Fan, C. Recent advances in two-dimensional nanomaterials-based electrochemical sensors for environmental analysis. Green Energy Environ. 2018, 3, 97–106.
- Zhang, Z.; Shen, W.; Xue, J.; Liu, Y.; Liu, Y.; Yan, P.; Liu, J.; Tang, J. Recent advances in synthetic methods and applications of silver nanostructures. Nanoscale Res. Lett. 2018, 13, 18.
- Hanko, M.; Švorc, L.; Planková, A.; Mikuš, P. Overview and recent advances in electrochemical sensing of glutathione—A review. Anal. Chim. Acta 2019, 1062, 1–27.
- Manivannan, S.; Jeong, J.; Kang, D.-K.; Kim, K. One-step Synthesis of AuAg Alloy Nanodots and its Electrochemical Studies towards Nitrobenzene Reduction and Sensing. Electroanalysis 2018, 30, 57–66.
- Bezerra, A.P.; Santos, A.O.; Abrantes-Coutinho, V.E.; Filho, E.D.S.; Soares, J.M.; Castro, S.S.; Oliveira, T.M. Theoretical and experimental findings regarding the electroanalysis of dienestrol in natural waters using a silver nanoparticles/single-walled carbon nanotubes-based amperometric sensor. J. Electroanal. Chem. 2021, 880, 114821.
- Wan, Y.; Zheng, Y.F.; Wan, H.T.; Yin, H.Y.; Song, X.C. A novel electrochemical sensor based on Ag nanoparticles decorated multi-walled carbon nanotubes for applied determination of nitrite. Food Control. 2017, 73, 1507–1513.
- Cheng, Y.; Li, H.; Fang, C.; Ai, L.; Chen, J.; Su, J.; Zhang, Q.; Fu, Q. Facile synthesis of reduced graphene oxide/silver nanoparticles composites and their application for detecting heavy metal ions. J. Alloys Compd. 2019, 787, 683–693.
- Bao, Z.-L.; Zhong, H.; Li, X.-R.; Zhang, A.-R.; Liu, Y.-X.; Chen, P.; Cheng, Z.-P.; Qian, H.-Y. Core-shell nanoparticles on carboxylated graphene for simultaneous electrochemical sensing of iodide and nitrite. Sens. Actuators B Chem. 2021, 345, 130319.
- Abraham, T.; Rejil, K.R.; George, J.M.; Antony, A.; Pillai, S.C.; Hinder, S.J.; Mathew, B. Magnetic Fe3O4–reduced graphene oxide composite decorated with Ag nanoparticles as electrochemical sensor and self-cleaning material for organic pollutants. J. Porous Mater. 2020, 27, 303–318.
- Meng, L.; Bian, R.; Guo, C.; Xu, B.; Liu, H.; Jiang, L. Aligning Ag Nanowires by a Facile Bioinspired Directional Liquid Transfer: Toward Anisotropic Flexible Conductive Electrodes. Adv. Mater. 2018, 30, 9.
- Terán-Alcocer, A.; Bravo-Plascencia, F.; Cevallos-Morillo, C.; Palma-Cando, A. Electrochemical Sensors Based on Conducting Polymers for the Aqueous Detection of Biologically Relevant Molecules. Nanomaterials 2021, 11, 252.
- Paixao, T. Measuring Electrochemical Surface Area of Nanomaterials versus the Randles−Ševčík Equation. Chemelectrochem 2020, 7, 3414–3415.
- Ryu, H.; Thompson, D.; Huang, Y.; Li, B.; Lei, Y. Electrochemical sensors for nitrogen species: A review. Sens. Actuators Rep. 2020, 2, 100022.
- Mohammadi, S.Z.; Beitollahi, H.; Dehghan, Z.; Hosseinzadeh, R. Electrochemical determination of ascorbic acid, uric acid and folic acid using carbon paste electrode modified with novel synthesized ferrocene derivative and core-shell magnetic nanoparticles in aqueous media. Appl. Organomet. Chem. 2018, 32, e4551.
- Liu, X.; Yao, Y.; Ying, Y.; Ping, J. Recent advances in nanomaterial-enabled screen-printed electrochemical sensors for heavy metal detection. Trends Anal. Chem. 2019, 115, 187–202.
- Ashrafi, A.M.; Koudelkova, Z.; Sedlackova, E.; Richtera, L.; Adam, V. Review—Electrochemical Sensors and Biosensors for Determination of Mercury Ions. J. Electrochem. Soc. 2018, 165, B824–B834.
- Eksin, E.; Erdem, A.; Fafal, T.; Kıvçak, B. Eco-friendly Sensors Developed by Herbal Based Silver Nanoparticles for Electrochemical Detection of Mercury (II) Ion. Electroanalysis 2019, 31, 1075–1082.
- Sebastian, M.; Aravind, A.; Mathew, B. Green silver-nanoparticle-based dual sensor for toxic Hg(II) ions. Nanotechnology 2018, 29, 355502.
- Punnoose, M.S.; Bijimol, D.; Abraham, T.; Plathanam, N.J.; Mathew, B. Green Synthesized Unmodified Silver Nanoparticles as Reproducible Dual Sensor for Mercuric Ions and Catalyst to Abate Environmental Pollutants. Bionanoscience 2021, 11, 739–754.
- Suhani, I.; Sahab, S.; Srivastava, V.; Singh, R.P. Impact of cadmium pollution on food safety and human health. Curr. Opin. Toxicol. 2021, 27, 1–7.
- Aravind, A.; Sebastian, M.; Mathew, B. Green silver nanoparticles as a multifunctional sensor for toxic Cd(ii) ions. New J. Chem. 2018, 42, 15022–15031.
- Wani, A.L.; Ara, A.; Usmani, J.A. Lead toxicity: A review. Interdiscip. Toxicol. 2015, 8, 55–64.
- Ganash, A.A.; Alghamdi, R.A. Fabrication of a novel polyaniline/green-synthesized, silver-nanoparticle-modified carbon paste electrode for electrochemical sensing of lead ions. JCCS 2021, 68, 2312–2325.
- Dash, S.R.; Bag, S.S.; Golder, A.K. Synthesis of highly structured spherical core-shell NPs using bio-analytes for electrocatalytic Pb(II) sensing. Sens. Actuators B Chem. 2020, 314, 128062.
- Mahmoudian, M.R.; Basirun, W.J.; Zalnezhad, E.; Ladan, M.; Alias, Y. l-Glutamine-assisted synthesis of flower-like NiO and ball-flower-like NiO/Ag as an electrochemical sensor for lead(ii) detection. RSC Adv. 2017, 7, 30870–30878.
- Thakkar, S.; Dumée, L.F.; Gupta, M.; Singh, B.R.; Yang, W. Nano–Enabled sensors for detection of arsenic in water. Water Res. 2021, 188, 116538.
- Kempahanumakkagari, S.; Deep, A.; Kim, K.-H.; Kailasa, S.K.; Yoon, H.-O. Nanomaterial-based electrochemical sensors for arsenic—A review. Biosens. Bioelectron. 2017, 95, 106–116.
- Sonkoue, B.M.; Tchekwagep, P.M.S.; Nanseu-Njiki, C.P.; Ngameni, E. Electrochemical Determination of Arsenic Using Silver Nanoparticles. Electroanalysis 2018, 30, 2738–2743.
- Salunke, R.S.; Chavan, P.G.; Shirale, D.J. Anodic stripping voltammetry studies of electrochemically engineered silver nanoparticles over single polypyrrole nanowire device for tracing of arsenic(III): An environmental perspective. Nanotechnol. Environ. Eng. 2018, 3, 12.
- Wen, S.-H.; Liang, R.-P.; Zhang, L.; Qiu, J.-D. Multimodal Assay of Arsenite Contamination in Environmental Samples with Improved Sensitivity through Stimuli-Response of Multiligands Modified Silver Nanoparticles. ACS Sustain. Chem. Eng. 2018, 6, 6223–6232.
- Hilali, N.; Mohammadi, H.; Amine, A.; Zine, N.; Errachid, A. Recent Advances in Electrochemical Monitoring of Chromium. Sensors 2020, 20, 5153.
- Ikhsan, N.I.; Rameshkumar, P.; Pandikumar, A.; Shahid, M.M.; Huang, N.M.; Kumar, S.V.; Lim, H.N. Facile synthesis of graphene oxide–silver nanocomposite and its modified electrode for enhanced electrochemical detection of nitrite ions. Talanta 2015, 144, 908–914.
- Li, Y.; Wang, H.; Liu, X.; Guo, L.; Ji, X.; Wang, L.; Tian, D.; Yang, X. Nonenzymatic nitrite sensor based on a titanium dioxide nanoparticles/ionic liquid composite electrode. J. Electroanal. Chem. 2014, 719, 35–40.
- Wang, K.; Wu, C.; Wang, F.; Liu, C.; Yu, C.; Jiang, G. In-situ insertion of carbon nanotubes into metal-organic frameworks-derived α-Fe2O3 polyhedrons for highly sensitive electrochemical detection of nitrite. Electrochim. Acta 2018, 285, 128–138.
- Li, X.; Ping, J.; Ying, Y. Recent developments in carbon nanomaterial-enabled electrochemical sensors for nitrite detection. Trends Anal. Chem. 2019, 113, 1–12.
- Yang, Z.; Zhou, X.; Yin, Y.; Fang, W. Determination of Nitrite by Noble Metal Nanomaterial-Based Electrochemical Sensors: A Minireview. Anal. Lett. 2021, 54, 2826–2850.
- Jiang, C.; He, Y.; Liu, Y. Recent advances in sensors for electrochemical analysis of nitrate in food and environmental matrices. Anal. 2020, 145, 5400–5413.
- Legrand, D.C.; Barus, C.; Garçon, V. Square Wave Voltammetry Measurements of Low Concentrations of Nitrate Using Au/AgNPs Electrode in Chloride Solutions. Electroanalysis 2017, 29, 2882–2887.
- Amali, R.; Lim, H.; Ibrahim, I.; Huang, N.; Zainal, Z.; Ahmad, S. Significance of nanomaterials in electrochemical sensors for nitrate detection: A review. Trends Environ. Anal. Chem. 2021, 31, e00135.
- Aliabadi, M.H.; Esmaeili, N.; Jahromi, H.S. An electrochemical composite sensor for phenol detection in waste water. Appl. Nanosci. 2020, 10, 597–609.
- Jebril, S.; Fdhila, A.; Dridi, C. Nanoengineering of eco-friendly silver nanoparticles using five different plant extracts and development of cost-effective phenol nanosensor. Sci. Rep. 2021, 11, 22060.
- Zhu, Y. Synthesis of Ag Nanoparticles Decorated Carbon Nanotubes as an Electrochemical Sensor for Determination of Phenolic Compounds in Shale Gas Wastewater. Int. J. Electrochem. Sci. 2021, 16, 21074.
- Goulart, L.A.; Gonçalves, R.; Correa, A.A.; Pereira, E.C.; Mascaro, L.H. Synergic effect of silver nanoparticles and carbon nanotubes on the simultaneous voltammetric determination of hydroquinone, catechol, bisphenol A and phenol. Microchim. Acta 2018, 185, 9.
- Sudhakara, S.M.; Devendrachari, M.C.; Kotresh, H.M.N.; Khan, F. Silver nanoparticles decorated phthalocyanine doped polyaniline for the simultaneous electrochemical detection of hydroquinone and catechol. J. Electroanal. Chem. 2021, 884, 115071.
- Kalaiyarasi, G.M.; Elakkiya, R.; Kundu, M.; Jin, W.; Sasidharan, M.; Maduraiveeran, G. Uncapped Silver Nanoclusters as Potential Catalyst for Enhanced Direct-Electrochemical Oxidation of 4-Nitrophenol. J. Clust. Sci. 2019, 30, 393–402.
- Ahmad, N.; Al-Fatesh, A.S.; Wahab, R.; Alam, M.; Fakeeha, A.H. Synthesis of silver nanoparticles decorated on reduced graphene oxide nanosheets and their electrochemical sensing towards hazardous 4-nitrophenol. J. Mater. Sci. Mater. Electron. 2020, 31, 11927–11937.
- Boumya, W.; Taoufik, N.; Achak, M.; Bessbousse, H.; Elhalil, A.; Barka, N. Electrochemical sensors and biosensors for the determination of diclofenac in pharmaceutical, biological and water samples. Talanta Open 2020, 3, 100026.
- Dao, K.C.; Yang, C.-C.; Chen, K.-F.; Tsai, Y.-P. Recent Trends in Removal Pharmaceuticals and Personal Care Products by Electrochemical Oxidation and Combined Systems. Water 2020, 12, 1043.
- Zamarchi, F.; Vieira, I.C. Determination of paracetamol using a sensor based on green synthesis of silver nanoparticles in plant extract. J. Pharm. Biomed. Anal. 2021, 196, 113912.
- Wong, A.; Santos, A.M.; Fatibello-Filho, O. Simultaneous determination of paracetamol and levofloxacin using a glassy carbon electrode modified with carbon black, silver nanoparticles and PEDOT:PSS film. Sens. Actuators B Chem. 2018, 255, 2264–2273.
- Dou, N.; Zhang, S.; Qu, J. Simultaneous detection of acetaminophen and 4-aminophenol with an electrochemical sensor based on silver–palladium bimetal nanoparticles and reduced graphene oxide. RSC Adv. 2019, 9, 31440–31446.
- Tang, J.; Liu, Y.; Hu, J.; Zheng, S.; Wang, X.; Zhou, H.; Jin, B. Co-based metal-organic framework nanopinnas composite doped with Ag nanoparticles: A sensitive electrochemical sensing platform for simultaneous determination of dopamine and acetaminophen. Microchem. J. 2020, 155, 104759.
- Pandiyarajan, C.; Rameshkumar, P.; Murugesan, S.; Selvaraj, M. Silver nanoparticles-supported graphitic-like carbon nitride for the electrochemical sensing of nitrobenzene and its derivatives. J. Mater. Sci. Mater. Electron. 2021, 32, 19912–19924.
- Shivakumar, M.; Dharmaprakash, M.S.; Manjappa, S.; Nagashree, K.L. Green synthesis of silver nanoparticles (SNPs)-modified electrode for electrochemical detection of nitrobenzene. J. Iran. Chem. Soc. 2020, 17, 893–900.
- Karthik, R.; Govindasamy, M.; Chen, S.-M.; Cheng, Y.-H.; Muthukrishnan, P.; Padmavathy, S.; Elangovan, A. Biosynthesis of silver nanoparticles by using Camellia japonica leaf extract for the electrocatalytic reduction of nitrobenzene and photocatalytic degradation of Eosin-Y. J. Photochem. Photobiol. B Biol. 2017, 170, 164–172.
- Kappen, J.; Aravind, M.K.; Varalakshmi, P.; Ashokkumar, B.; John, S.A. Quantitative removal of Hg(II) as Hg(0) using carbon cloths coated graphene quantum dots and their silver nanoparticles composite and application of Hg(0) for the sensitive determination of nitrobenzene. Colloids Surfaces A Physicochem. Eng. Asp. 2022, 641, 128542.
- Kaya, S.I.; Yıldırım, S.; Cetinkaya, A.; Erkmen, C.; Uslu, B.; Ozkan, S.A. Nanomaterial-based electroanalytical sensors for the selected prohibited anabolic agents, hormones and metabolic modulators and their sensitive assays. Trends Anal. Chem. 2021, 145, 116457.
- Musa, A.M.; Kiely, J.; Luxton, R.; Honeychurch, K.C. Recent progress in screen-printed electrochemical sensors and biosensors for the detection of estrogens. Trends Anal. Chem. 2021, 139, 116254.
- Manjunatha, J. Electroanalysis of estriol hormone using electrochemical sensor. Sens. Bio-Sens. Res. 2017, 16, 79–84.
- Singh, A.K.; Agrahari, S.; Gautam, R.K.; Tiwari, I. Fabrication of an innovative electrochemical sensor based on graphene-coated silver nanoparticles decorated over graphitic carbon nitride for efficient determination of estradiol. Environ. Sci. Pollut. Res. 2022.
- Regasa, M.B.; Nyokong, T. Design and fabrication of electrochemical sensor based on molecularly imprinted polymer loaded onto silver nanoparticles for the detection of 17-β-estradiol. J. Mol. Recognit. 2022, 35, e2978.
- Regasa, M.B.; Nyokong, T. Synergistic recognition and electrochemical sensing of 17β-Estradiol using ordered molecularly imprinted polymer-graphene oxide-silver nanoparticles composite films. J. Electroanal. Chem. 2022, 922, 116713.
- Ivanišević, I.; Milardović, S.; Kassal, P. Recent Advances in (Bio)Chemical Sensors for Food Safety and Quality Based on Silver Nanomaterials. Food Technol. Biotechnol. 2021, 59, 216–237.
- Verma, D.; Dhiman, T.K.; Das Mukherjee, M.; Solanki, P.R. Electrophoretically Deposited Green Synthesized Silver Nanoparticles Anchored in Reduced Graphene Oxide Composite Based Electrochemical Sensor for Detection of Bisphenol A. J. Electrochem. Soc. 2021, 168, 097504.
- Pérez-Ràfols, C.; Bastos-Arrieta, J.; Serrano, N.; Díaz-Cruz, J.M.; Ariño, C.; De Pablo, J.; Esteban, M. Ag Nanoparticles Drop-Casting Modification of Screen-Printed Electrodes for the Simultaneous Voltammetric Determination of Cu(II) and Pb(II). Sensors 2017, 17, 1458.
- Ivanišević, I.; Milardović, S.; Ressler, A.; Kassal, P. Fabrication of an All-Solid-State Ammonium Paper Electrode Using a Graphite-Polyvinyl Butyral Transducer Layer. Chemosensors 2021, 9, 333.
- Wang, J.; Diao, P. Simultaneous detection of ammonia and nitrate using a modified electrode with two regions. Microchem. J. 2020, 154, 104649.
- Teodoro, K.B.; Shimizu, F.M.; Scagion, V.P.; Correa, D.S. Ternary nanocomposites based on cellulose nanowhiskers, silver nanoparticles and electrospun nanofibers: Use in an electronic tongue for heavy metal detection. Sens. Actuators B Chem. 2019, 290, 387–395.
- Boumya, W.; Taoufik, N.; Achak, M.; Bessbousse, H.; Elhalil, A.; Barka, N. Electrochemical sensors and biosensors for the determination of diclofenac in pharmaceutical, biological and water samples. Talanta Open 2020, 3, 100026.
- Li, P.; Li, X.; Chen, W. Recent advances in electrochemical sensors for the detection of 2,4 6-trinitrotoluene. Curr. Opin. Electrochem. 2019, 17, 16–22.
- Alahi, E.E.; Mukhopadhyay, S. Detection methods of nitrate in water: A review. Sens. Actuators A Phys. 2018, 280, 210–221.
- Li, Y.; Wang, H.; Yan, B.; Zhang, H. An electrochemical sensor for the determination of bisphenol A using glassy carbon electrode modified with reduced graphene oxide-silver/poly-l-lysine nanocomposites. J. Electroanal. Chem. 2017, 805, 39–46.
- Pérez-Ràfols, C.; Bastos-Arrieta, J.; Serrano, N.; Díaz-Cruz, J.M.; Ariño, C.; De Pablo, J.; Esteban, M. Ag Nanoparticles Drop-Casting Modification of Screen-Printed Electrodes for the Simultaneous Voltammetric Determination of Cu(II) and Pb(II). Sensors 2017, 17, 1458.
- Wong, A.; Santos, A.M.; Fatibello-Filho, O. Simultaneous determination of paracetamol and levofloxacin using a glassy carbon electrode modified with carbon black, silver nanoparticles and PEDOT:PSS film. Sens. Actuators B Chem. 2018, 255, 2264–2273.
- Liu, S.; Yu, Y.; Ni, K.; Liu, T.; Gu, M.; Wu, Y.; Du, G.; Ran, X. Construction of a novel electrochemical sensor based on biomass material nanocellulose and its detection of acetaminophen. RSC Adv. 2022, 12, 27736–27745.
- Lebon, E.; Fau, P.; Comtat, M.; Kahn, M.L.; Sournia-Saquet, A.; Temple-Boyer, P.; Dubreuil, B.; Behra, P.; Fajerwerg, K. In Situ Metalorganic Deposition of Silver Nanoparticles on Gold Substrate and Square Wave Voltammetry: A Highly Efficient Combination for Nanomolar Detection of Nitrate Ions in Sea Water. Chemosensors 2018, 6, 50.
- Buledi, J.A.; Amin, S.; Haider, S.I.; Bhanger, M.I.; Solangi, A.R. A review on detection of heavy metals from aqueous media using nanomaterial-based sensors. Environ. Sci. Pollut. Res. 2020, 28, 58994–59002.
- Nayak, S.; Goveas, L.C.; Kumar, P.S.; Selvaraj, R.; Vinayagam, R. Plant-mediated gold and silver nanoparticles as detectors of heavy metal contamination. Food Chem. Toxicol. 2022, 167, 113271.
- Wani, I.A. Review—Recent Advances in Biogenic Silver Nanoparticles & NanoComposite Based Plasmonic-Colorimetric and Electrochemical Sensors. ECS J. Solid State Sci. Technol. 2021, 10, 047003.
- Milardovicć, S. Synthesis and Electrochemical Characterization of AgNP Ink Suitable for Inkjet Printing. Int. J. Electrochem. Sci. 2018, 13, 11136–11149.
- Raymundo-Pereira, P.A.; Campos, A.M.; Vicentini, F.C.; Janegitz, B.C.; Mendonça, C.D.; Furini, L.N.; Boas, N.V.; Calegaro, M.L.; Constantino, C.J.; Machado, S.A.; et al. Sensitive detection of estriol hormone in creek water using a sensor platform based on carbon black and silver nanoparticles. Talanta 2017, 174, 652–659.
- Wong, A.; Santos, A.M.; Fatibello-Filho, O. Simultaneous determination of paracetamol and levofloxacin using a glassy carbon electrode modified with carbon black, silver nanoparticles and PEDOT:PSS film. Sens. Actuators B Chem. 2018, 255, 2264–2273.
- Ma, C.; Qian, Y.; Zhang, S.; Song, H.; Gao, J.; Wang, S.; Liu, M.; Xie, K.; Zhang, X. Temperature-controlled ethanolamine and Ag-nanoparticle dual-functionalization of graphene oxide for enhanced electrochemical nitrite determination. Sens. Actuators B Chem. 2018, 274, 441–450.
- Verma, D.; Dhiman, T.K.; Das Mukherjee, M.; Solanki, P.R. Electrophoretically Deposited Green Synthesized Silver Nanoparticles Anchored in Reduced Graphene Oxide Composite Based Electrochemical Sensor for Detection of Bisphenol A. J. Electrochem. Soc. 2021, 168, 097504.
- Shivakumar, M.; Nagashree, K.L.; Manjappa, S.; Dharmaprakash, M.S. Electrochemical Detection of Nitrite Using Glassy Carbon Electrode Modified with Silver Nanospheres (AgNS) Obtained by Green Synthesis Using Pre-hydrolysed Liquor. Electroanalysis 2017, 29, 1434–1442.
- Shivakumar, M.; Dharmaprakash, M.S.; Manjappa, S.; Nagashree, K.L. Green synthesis of silver nanoparticles (SNPs)-modified electrode for electrochemical detection of nitrobenzene. J. Iran. Chem. Soc. 2020, 17, 893–900.
- Ahmad, N.; Al-Fatesh, A.S.; Wahab, R.; Alam, M.; Fakeeha, A.H. Synthesis of silver nanoparticles decorated on reduced graphene oxide nanosheets and their electrochemical sensing towards hazardous 4-nitrophenol. J. Mater. Sci. Mater. Electron. 2020, 31, 11927–11937.
- Laghrib, F.; Houcini, H.; Khalil, F.; Liba, A.; Bakasse, M.; Lahrich, S.; El Mhammedi, M.A. Synthesis of Silver Nanoparticles Using Chitosan as Stabilizer Agent: Application towards Electrocatalytical Reduction of p-Nitrophenol. Chemistryselect 2020, 5, 1220–1227.
- Sookhakian, M.; Basirun, W.; Goh, B.T.; Woi, P.M.; Alias, Y. Molybdenum disulfide nanosheet decorated with silver nanoparticles for selective detection of dopamine. Colloids Surfaces B Biointerfaces 2019, 176, 80–86
- Naseri, M.; Mohammadniaei, M.; Ghosh, K.; Sarkar, S.; Sankar, R.; Mukherjee, S.; Pal, S.; Dezfouli, E.A.; Halder, A.; Qiao, J.; et al. A Robust Electrochemical Sensor Based on Butterfly-shaped Silver Nanostructure for Concurrent Quantification of Heavy Metals in Water Samples. Electroanalysis 2022, 35, e202200114.
- Zhu, Y. Synthesis of Ag Nanoparticles Decorated Carbon Nanotubes as an Electrochemical Sensor for Determination of Phenolic Compounds in Shale Gas Wastewater. Int. J. Electrochem. Sci. 2021, 16, 21074.
- Salunke, R.S.; Chavan, P.G.; Shirale, D.J. Anodic stripping voltammetry studies of electrochemically engineered silver nanoparticles over single polypyrrole nanowire device for tracing of arsenic(III): An environmental perspective. Nanotechnol. Environ. Eng. 2018, 3, 12.
- Regasa, M.B.; Nyokong, T. Design and fabrication of electrochemical sensor based on molecularly imprinted polymer loaded onto silver nanoparticles for the detection of 17-β-estradiol. J. Mol. Recognit. 2022, 35, e2978.
- Zhao, K.; Ge, L.; Wong, T.I.; Zhou, X.; Lisak, G. Gold-silver nanoparticles modified electrochemical sensor array for simultaneous determination of chromium(III) and chromium(VI) in wastewater samples. Chemosphere 2021, 281, 130880.
- Meenakshi, S.; Devi, S.; Pandian, K.; Chitra, K.; Tharmaraj, P. Aniline-mediated synthesis of carboxymethyl cellulose protected silver nanoparticles modified electrode for the differential pulse anodic stripping voltammetry detection of mercury at trace level. Ionics 2019, 25, 3431–3441.
- Kaladevi, G.; Meenakshi, S.; Pandian, K.; Wilson, P. Synthesis of Well-Dispersed Silver Nanoparticles on Polypyrrole/Reduced Graphene Oxide Nanocomposite for Simultaneous Detection of Toxic Hydrazine and Nitrite in Water Sources. J. Electrochem. Soc. 2017, 164, B620–B631.
- Shen, Y.; Ma, C.; Zhang, S.; Li, P.; Zhu, W.; Zhang, X.; Gao, J.; Song, H.; Chen, D.; Pang, D.; et al. Nanosilver and protonated carbon nitride co-coated carbon cloth fibers based non-enzymatic electrochemical sensor for determination of carcinogenic nitrite. Sci. Total. Environ. 2020, 742, 140622.
- Shivakumar, M.; Dharmaprakash, M.S.; Manjappa, S.; Nagashree, K.L. Green synthesis of silver nanoparticles (SNPs)-modified electrode for electrochemical detection of nitrobenzene. J. Iran. Chem. Soc. 2020, 17, 893–900.
- Amalraj, A.J.J.; Murthy, U.N.; Wang, S.-F. Silver-capped selenium explored as an electro-catalyst for simultaneous detection of nitro-aromatic drugs in different aqueous samples. J. Ind. Eng. Chem. 2022, 108, 243–253.
- Laghrib, F.; Houcini, H.; Khalil, F.; Liba, A.; Bakasse, M.; Lahrich, S.; El Mhammedi, M.A. Synthesis of Silver Nanoparticles Using Chitosan as Stabilizer Agent: Application towards Electrocatalytical Reduction of p-Nitrophenol. Chemistryselect 2020, 5, 1220–1227.
- Liu, S.; Yu, Y.; Ni, K.; Liu, T.; Gu, M.; Wu, Y.; Du, G.; Ran, X. Construction of a novel electrochemical sensor based on biomass material nanocellulose and its detection of acetaminophen. RSC Adv. 2022, 12, 27736–27745.
- Baghayeri, M.; Mahdavi, B.; Abadi, Z.H.; Farhadi, S. Green synthesis of silver nanoparticles using water extract of Salvia leriifolia: Antibacterial studies and applications as catalysts in the electrochemical detection of nitrite. Appl. Organomet. Chem. 2018, 32, 9.
- Eksin, E.; Erdem, A.; Fafal, T.; Kıvçak, B. Eco-friendly Sensors Developed by Herbal Based Silver Nanoparticles for Electrochemical Detection of Mercury (II) Ion. Electroanalysis 2019, 31, 1075–1082.
- Bibi, S.; Zaman, M.I.; Niaz, A.; Rahim, A.; Nawaz, M.; Arian, M.B. Voltammetric determination of nitrite by using a multiwalled carbon nanotube paste electrode modified with chitosan-functionalized silver nanoparticles. Microchim. Acta 2019, 186, 595.
- Zamarchi, F.; Vieira, I.C. Determination of paracetamol using a sensor based on green synthesis of silver nanoparticles in plant extract. J. Pharm. Biomed. Anal. 2021, 196, 113912.
- Dash, S.R.; Bag, S.S.; Golder, A.K. Synthesis of highly structured spherical Ag@Pt core-shell NPs using bio-analytes for electrocatalytic Pb(II) sensing. Sens. Actuators B Chem. 2020, 314, 128062.
- Kokab, T.; Shah, A.; Khan, M.A.; Nisar, J.; Ashiq, M.N. Electrochemical sensing platform for the simultaneous femtomolar detection of amlodipine and atorvastatin drugs. RSC Adv. 2021, 11, 27135–27151.
- Sebastian, M.; Aravind, A.; Mathew, B. Green silver-nanoparticle-based dual sensor for toxic Hg(II) ions. Nanotechnology 2018, 29, 355502.
- Vyas, G.; Bhatt, S.; Paul, P. Synthesis of Calixarene-Capped Silver Nanoparticles for Colorimetric and Amperometric Detection of Mercury (HgII, Hg0). ACS Omega 2019, 4, 3860–3870.
- Punnoose, M.S.; Bijimol, D.; Abraham, T.; Plathanam, N.J.; Mathew, B. Green Synthesized Unmodified Silver Nanoparticles as Reproducible Dual Sensor for Mercuric Ions and Catalyst to Abate Environmental Pollutants. Bionanoscience 2021, 11, 739–754.
- George, J.M.; Mathew, B. Curcuma longa rhizome extract mediated unmodified silver nanoparticles as multisensing probe for Hg(II) ions. Mater. Res. Express 2019, 6, 1150h5.
- Aravind, A.; Sebastian, M.; Mathew, B. Green silver nanoparticles as a multifunctional sensor for toxic Cd(ii) ions. New J. Chem. 2018, 42, 15022–15031.
- Sebastian, M.; Aravind, A.; Mathew, B. Green Silver Nanoparticles Based Multi-Technique Sensor for Environmental Hazardous Cu(II) Ion. Bionanoscience 2019, 9, 373–385.
- Aravind, A.; Sebastian, M.; Mathew, B. Green synthesized unmodified silver nanoparticles as a multi-sensor for Cr(iii) ions. Environ. Sci. Water Res. Technol. 2018, 4, 1531–1542.
- Wen, S.-H.; Liang, R.-P.; Zhang, L.; Qiu, J.-D. Multimodal Assay of Arsenite Contamination in Environmental Samples with Improved Sensitivity through Stimuli-Response of Multiligands Modified Silver Nanoparticles. ACS Sustain. Chem. Eng. 2018, 6, 6223–6232.
- Suherman, A.L.; Ngamchuea, K.; Tanner, E.E.L.; Sokolov, S.V.; Holter, J.; Young, N.P.; Compton, R.G. Electrochemical Detection of Ultratrace (Picomolar) Levels of Hg2+ Using a Silver Nanoparticle-Modified Glassy Carbon Electrode. Anal. Chem. 2017, 89, 7166–7173.
- Regasa, M.B.; Nyokong, T. Synergistic recognition and electrochemical sensing of 17β-Estradiol using ordered molecularly imprinted polymer-graphene oxide-silver nanoparticles composite films. J. Electroanal. Chem. 2022, 922, 116713.
- Liao, G.; Fang, J.; Li, Q.; Li, S.; Xu, Z.; Fang, B. Ag-Based nanocomposites: Synthesis and applications in catalysis. Nanoscale 2019, 11, 7062–7096.
- Ma, C.; Qian, Y.; Zhang, S.; Song, H.; Gao, J.; Wang, S.; Liu, M.; Xie, K.; Zhang, X. Temperature-controlled ethanolamine and Ag-nanoparticle dual-functionalization of graphene oxide for enhanced electrochemical nitrite determination. Sens. Actuators B Chem. 2018, 274, 441–450.
- Kalaiyarasi, G.M.; Elakkiya, R.; Kundu, M.; Jin, W.; Sasidharan, M.; Maduraiveeran, G. Uncapped Silver Nanoclusters as Potential Catalyst for Enhanced Direct-Electrochemical Oxidation of 4-Nitrophenol. J. Clust. Sci. 2019, 30, 393–402.
- Jebril, S.; Fdhila, A.; Dridi, C. Nanoengineering of eco-friendly silver nanoparticles using five different plant extracts and development of cost-effective phenol nanosensor. Sci. Rep. 2021, 11, 22060.
- Tang, J.; Liu, Y.; Hu, J.; Zheng, S.; Wang, X.; Zhou, H.; Jin, B. Co-based metal-organic framework nanopinnas composite doped with Ag nanoparticles: A sensitive electrochemical sensing platform for simultaneous determination of dopamine and acetaminophen. Microchem. J. 2020, 155, 104759.
- Karthik, R.; Govindasamy, M.; Chen, S.-M.; Cheng, Y.-H.; Muthukrishnan, P.; Padmavathy, S.; Elangovan, A. Biosynthesis of silver nanoparticles by using Camellia japonica leaf extract for the electrocatalytic reduction of nitrobenzene and photocatalytic degradation of Eosin-Y. J. Photochem. Photobiol. B Biol. 2017, 170, 164–172.
- Bao, Z.-L.; Zhong, H.; Li, X.-R.; Zhang, A.-R.; Liu, Y.-X.; Chen, P.; Cheng, Z.-P.; Qian, H.-Y. Core-shell Au@Ag nanoparticles on carboxylated graphene for simultaneous electrochemical sensing of iodide and nitrite. Sens. Actuators B Chem. 2021, 345, 130319.
- Zhao, X.; Li, N.; Jing, M.; Zhang, Y.; Wang, W.; Liu, L.; Xu, Z.; Liu, L.; Li, F.; Wu, N. Monodispersed and spherical silver nanoparticles/graphene nanocomposites from gamma-ray assisted in-situ synthesis for nitrite electrochemical sensing. Electrochim. Acta 2019, 295, 434–443.
- Ahmad, R.; Mahmoudi, T.; Ahn, M.-S.; Yoo, J.-Y.; Hahn, Y.-B. Fabrication of sensitive non-enzymatic nitrite sensor using silver-reduced graphene oxide nanocomposite. J. Colloid Interface Sci. 2018, 516, 67–75.
- Kaladevi, G.; Meenakshi, S.; Pandian, K.; Wilson, P. Synthesis of Well-Dispersed Silver Nanoparticles on Polypyrrole/Reduced Graphene Oxide Nanocomposite for Simultaneous Detection of Toxic Hydrazine and Nitrite in Water Sources. J. Electrochem. Soc. 2017, 164, B620–B631.
- Kaladevi, G.; Wilson, P.; Pandian, K. Simultaneous and Selective Electrochemical Detection of Sulfite and Nitrite in Water Sources Using Homogeneously Dispersed Ag Nanoparticles over PANI/rGO Nanocomposite. J. Electrochem. Soc. 2020, 167, 027514.
- Salagare, S.; Adarakatti, P.S.; Yarradoddappa, V. Facile synthesis of silver nanoparticle-decorated zinc oxide nanocomposite-based pencil graphite electrode for selective electrochemical determination of nitrite. Carbon Lett. 2021, 31, 1273–1286.
- Ghanei-Motlagh, M.; Taher, M.A. A novel electrochemical sensor based on silver/halloysite nanotube/molybdenum disulfide nanocomposite for efficient nitrite sensing. Biosens. Bioelectron. 2018, 109, 279–285.
- Pang, D.; Ma, C.; Chen, D.; Shen, Y.; Zhu, W.; Gao, J.; Song, H.; Zhang, X.; Zhang, S. Silver nanoparticle-functionalized poly (3, 4-ethylenedioxythiophene): Polystyrene film on glass substrate for electrochemical determination of nitrite. Org. Electron. 2019, 75, 105374.
- Wan, Y.; Zheng, Y.F.; Wan, H.T.; Yin, H.Y.; Song, X.C. A novel electrochemical sensor based on Ag nanoparticles decorated multi-walled carbon nanotubes for applied determination of nitrite. Food Control. 2017, 73, 1507–1513.
- Bezerra, A.P.; Santos, A.O.; Abrantes-Coutinho, V.E.; Filho, E.D.S.; Soares, J.M.; Castro, S.S.; Oliveira, T.M. Theoretical and experimental findings regarding the electroanalysis of dienestrol in natural waters using a silver nanoparticles/single-walled carbon nanotubes-based amperometric sensor. J. Electroanal. Chem. 2021, 880, 114821.
- Donini, C.A.; da Silva, M.K.L.; Simões, R.P.; Cesarino, I. Reduced graphene oxide modified with silver nanoparticles for the electrochemical detection of estriol. J. Electroanal. Chem. 2018, 809, 67–73.
- Sudhakara, S.M.; Devendrachari, M.C.; Kotresh, H.M.N.; Khan, F. Silver nanoparticles decorated phthalocyanine doped polyaniline for the simultaneous electrochemical detection of hydroquinone and catechol. J. Electroanal. Chem. 2021, 884, 115071.
- Abraham, T.; Rejil, K.R.; George, J.M.; Antony, A.; Pillai, S.C.; Hinder, S.J.; Mathew, B. Magnetic Fe3O4–reduced graphene oxide composite decorated with Ag nanoparticles as electrochemical sensor and self-cleaning material for organic pollutants. J. Porous Mater. 2020, 27, 303–318.
- Hwa, K.-Y.; Sharma, T.S.K.; Ganguly, A. Design strategy of rGO–HNT–AgNPs based hybrid nanocomposite with enhanced performance for electrochemical detection of 4-nitrophenol. Inorg. Chem. Front. 2020, 7, 1981–1994.
- Faisal, M.; Alam, M.M.; Ahmed, J.; Asiri, A.M.; Alsaiari, M.; Alruwais, R.S.; Madkhali, O.; Rahman, M.M.; Harraz, F.A. Efficient Detection of 2,6-Dinitrophenol with Silver Nanoparticle-Decorated Chitosan/SrSnO3 Nanocomposites by Differential Pulse Voltammetry. Biosensors 2022, 12, 976.
- Rani, S.; Dilbaghi, N.; Kumar, S.; Varma, R.S.; Malhotra, R. Rapid redox sensing of p-nitrotoluene in real water samples using silver nanoparticles. Inorg. Chem. Commun. 2020, 120, 108157.
- Pandiyarajan, C.; Rameshkumar, P.; Murugesan, S.; Selvaraj, M. Silver nanoparticles-supported graphitic-like carbon nitride for the electrochemical sensing of nitrobenzene and its derivatives. J. Mater. Sci. Mater. Electron. 2021, 32, 19912–19924.
- Manivannan, S.; Jeong, J.; Kang, D.-K.; Kim, K. One-step Synthesis of AuAg Alloy Nanodots and its Electrochemical Studies towards Nitrobenzene Reduction and Sensing. Electroanalysis 2018, 30, 57–66.
- Legrand, D.C.; Barus, C.; Garçon, V. Square Wave Voltammetry Measurements of Low Concentrations of Nitrate Using Au/AgNPs Electrode in Chloride Solutions. Electroanalysis 2017, 29, 2882–2887.
- Amali, R.; Lim, H.; Ibrahim, I.; Zainal, Z.; Ahmad, S. Silver nanoparticles-loaded copper (II)-terephthalate framework nanocomposite as a screen-printed carbon electrode modifier for amperometric nitrate detection. J. Electroanal. Chem. 2022, 918, 116440.
- Hajisafari, M.; Nasirizadeh, N. An electrochemical nanosensor for simultaneous determination of hydroxylamine and nitrite using oxadiazole self-assembled on silver nanoparticle-modified glassy carbon electrode. Ionics 2017, 23, 1541–1551.
- Zhao, J.; Wang, X.; Hoang, S.A.; Bolan, N.S.; Kirkham, M.; Liu, J.; Xia, X.; Li, Y. Silver nanoparticles in aquatic sediments: Occurrence, chemical transformations, toxicity, and analytical methods. J. Hazard. Mater. 2021, 418, 126368.
- Dash, S.R.; Bag, S.S.; Golder, A.K. Synthesis of highly structured spherical Ag@Pt core-shell NPs using bio-analytes for electrocatalytic Pb(II) sensing. Sens. Actuators B Chem. 2020, 314, 128062.
- Dou, N.; Zhang, S.; Qu, J. Simultaneous detection of acetaminophen and 4-aminophenol with an electrochemical sensor based on silver–palladium bimetal nanoparticles and reduced graphene oxide. RSC Adv. 2019, 9, 31440–31446.
- Mahmoudian, M.R.; Basirun, W.J.; Zalnezhad, E.; Ladan, M.; Alias, Y. l-Glutamine-assisted synthesis of flower-like NiO and ball-flower-like NiO/Ag as an electrochemical sensor for lead(ii) detection. RSC Adv. 2017, 7, 30870–30878.
- Navratil, R.; Kotzianova, A.; Halouzka, V.; Opletal, T.; Triskova, I.; Trnkova, L.; Hrbac, J. Polymer lead pencil graphite as electrode material: Voltammetric, XPS and Raman study. J. Electroanal. Chem. 2016, 783, 152–160.
- McCreery, R.L. Advanced Carbon Electrode Materials for Molecular Electrochemistry. Chem. Rev. 2008, 108, 2646–2687.
- Hajisafari, M.; Nasirizadeh, N. An electrochemical nanosensor for simultaneous determination of hydroxylamine and nitrite using oxadiazole self-assembled on silver nanoparticle-modified glassy carbon electrode. Ionics 2017, 23, 1541–1551.
- Su, S.; Chen, S.; Fan, C. Recent advances in two-dimensional nanomaterials-based electrochemical sensors for environmental analysis. Green Energy Environ. 2018, 3, 97–106.
- Boroujerdi, R.; Paul, R. Graphene-Based Electrochemical Sensors for Psychoactive Drugs. Nanomaterials 2022, 12, 2250.
- Singh, A.K.; Agrahari, S.; Gautam, R.K.; Tiwari, I. Fabrication of an innovative electrochemical sensor based on graphene-coated silver nanoparticles decorated over graphitic carbon nitride for efficient determination of estradiol. Environ. Sci. Pollut. Res. 2022.
- Kwon, D.; Kim, J. Ag metal organic frameworks nanocomposite modified electrode for simultaneous electrochemical detection of copper (II) and lead (II). J. Appl. Electrochem. 2021, 51, 1207–1216.
- Sookhakian, M.; Basirun, W.; Goh, B.T.; Woi, P.M.; Alias, Y. Molybdenum disulfide nanosheet decorated with silver nanoparticles for selective detection of dopamine. Colloids Surfaces B Biointerfaces 2019, 176, 80–86.
- Baciu, A.; Manea, F.; Pop, A.; Pode, R.; Schoonman, J. Simultaneous voltammetric detection of ammonium and nitrite from groundwater at silver-electrodecorated carbon nanotube electrode. Process. Saf. Environ. Prot. 2017, 108, 18–25.
- Goulart, L.A.; Gonçalves, R.; Correa, A.A.; Pereira, E.C.; Mascaro, L.H. Synergic effect of silver nanoparticles and carbon nanotubes on the simultaneous voltammetric determination of hydroquinone, catechol, bisphenol A and phenol. Microchim. Acta 2018, 185, 9.
- Hassan, K.M.; Elhaddad, G.M.; AbdelAzzem, M. Voltammetric determination of cadmium(II), lead(II) and copper(II) with a glassy carbon electrode modified with silver nanoparticles deposited on poly(1,8-diaminonaphthalene). Microchim. Acta 2019, 186, 440.
- Kaladevi, G.; Wilson, P.; Pandian, K. Simultaneous and Selective Electrochemical Detection of Sulfite and Nitrite in Water Sources Using Homogeneously Dispersed Ag Nanoparticles over PANI/rGO Nanocomposite. J. Electrochem. Soc. 2020, 167, 027514.
- Li, P.; Li, X.; Chen, W. Recent advances in electrochemical sensors for the detection of 2,4 6-trinitrotoluene. Curr. Opin. Electrochem. 2019, 17, 16–22.
- Laghrib, F.; Farahi, A.; Bakasse, M.; Lahrich, S.; El Mhammedi, M. Chemical synthesis of nanosilver on chitosan and electroanalysis activity against the p-nitroaniline reduction. J. Electroanal. Chem. 2019, 845, 111–118.
- Ganash, A.A.; Alghamdi, R.A. Fabrication of a novel polyaniline/green-synthesized, silver-nanoparticle-modified carbon paste electrode for electrochemical sensing of lead ions. JCCS 2021, 68, 2312–2325.
- Kariuki, J.; Ervin, E.; Olafson, C. Development of a Novel, Low-Cost, Disposable Wooden Pencil Graphite Electrode for Use in the Determination of Antioxidants and Other Biological Compounds. Sensors 2015, 15, 18887–18900.
- Sonkoue, B.M.; Tchekwagep, P.M.S.; Nanseu-Njiki, C.P.; Ngameni, E. Electrochemical Determination of Arsenic Using Silver Nanoparticles. Electroanalysis 2018, 30, 2738–2743.
- Mejri, A.; Mars, A.; Elfil, H.; Hamzaoui, A.H. Voltammetric simultaneous quantification of p-nitrophenol and hydrazine by using magnetic spinel FeCo2O4 nanosheets on reduced graphene oxide layers modified with curcumin-stabilized silver nanoparticles. Microchim. Acta 2019, 186, 561.
- Balram, D.; Lian, K.-Y.; Sebastian, N.; Al-Mubaddel, F.S.; Noman, M.T. Bi-functional renewable biopolymer wrapped CNFs/Ag doped spinel cobalt oxide as a sensitive platform for highly toxic nitroaromatic compound detection and degradation. Chemosphere 2022, 291, 132998.
- hao, K.; Ge, L.; Wong, T.I.; Zhou, X.; Lisak, G. Gold-silver nanoparticles modified electrochemical sensor array for simultaneous determination of chromium(III) and chromium(VI) in wastewater samples. Chemosphere 2021, 281, 130880.
- Pang, D.; Ma, C.; Chen, D.; Shen, Y.; Zhu, W.; Gao, J.; Song, H.; Zhang, X.; Zhang, S. Silver nanoparticle-functionalized poly (3, 4-ethylenedioxythiophene): Polystyrene film on glass substrate for electrochemical determination of nitrite. Org. Electron. 2019, 75, 105374.
- Wang, J.; Diao, P. Simultaneous detection of ammonia and nitrate using a modified electrode with two regions. Microchem. J. 2020, 154, 104649.
- Zhou, M.; Han, L.; Deng, D.; Zhang, Z.; He, H.; Zhang, L.; Luo, L. 4-mercaptobenzoic acid modified silver nanoparticles-enhanced electrochemical sensor for highly sensitive detection of Cu2+. Sens. Actuators B Chem. 2019, 291, 164–169.
