Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Biology
Imeglimin enhances glucose-stimulated insulin secretion and inhibits the apoptosis of β-cells in the pancreas by the maintaining function and structure of mitochondria and the endoplasmic reticulum in β-cells. Further, imeglimin inhibits hepatic glucose production and ameliorates insulin sensitivity.
- endoplasmic reticulum
- glucose-stimulated insulin secretion
- imeglimin
1. An Improvement in Mitochondrial Function
Mitochondria are organelles found in the cells of animals. Mitochondria use aerobic respiration to generate ATP. Imeglimin is a new oral hypoglycemic agent with a unique mechanism for the actions targeting mitochondrial bioenergetics. Mitochondrial dysfunction is a prominent pathological feature of type 2 diabetes, which contributes to worse plasma glucose control via β-cell mass reduction and insulin resistance [25,26,27,28,29,30,31]. Vial et al. characterized the anti-diabetic effects of imeglimin using a high-fat, high-sucrose diet (HFHSD) mice [22]. Imeglimin significantly reduces plasma glucose, and improves insulin sensitivity in HFHSD mice; however, body weight and food intake do not change in such mice. An improvement in glucose metabolism and insulin sensitivity may be induced by an increase in insulin-stimulated protein kinase B phosphorylation in the liver and skeletal muscles. In mitochondria, imeglimin redirects substrate flux towards complex II, inhibits complex I, restores complex III activity, and promotes fatty acid (FA) oxidation. Imeglimin also decreases ROS production and increases mitochondrial DNA. Furthermore, the effects of imeglimin on mitochondrial phospholipid composition may contribute to the improvement in mitochondrial function.
2. An Improvement in Function of Endoplasmic Reticulum (ER)
ER is a continuous membrane system that plays an essential role in the folding and processing of secretory proteins such as insulin. The excess accumulation of “poorly folded proteins”, which marks the induction of pathogenic ER stress in pancreatic β-cells, largely contributes to the pathogenesis of diabetes [32]. In isolated mouse islets, imeglimin modulates the expression of ER-related various molecules in β-cells under ER stress, and restores protein synthesis in β-cells [33]. Imeglimin significantly decreases apoptotic β-cells and increases β-cell mass in Akita mice. Imeglimin-mediated improvement in structural integrity and homeostasis of ER largely contributes to an enhancement of GSIS [33].
3. An Inhibition of Hepatic Glucose Production
Imeglimin dose-dependently inhibits hepatic glucose production by increasing mitochondrial redox potential and by decreasing membrane potential in rat hepatocytes [34]. In another study, the authors observed that imeglimin inhibits glucose production in hepatocytes isolated from Wistar rats [35]. Imeglimin markedly inhibits the gluconeogenesis by reducing the phosphoenolpyruvate carboxykinase (PEPCK) and glucose-6-phosphatase (G6Pase) in rat hepatocytes [35].
4. An Improvement in GSIS
An improvement in mitochondrial dysfunction via imeglimin induces anti-diabetic effects such as the amplification of GSIS and preservation of β-cell mass [33]. Imeglimin improves the markers for β-cell function, such as proinsulin/insulin in humans [36].
Nicotinamide phosphoribosyltransferase (NAMPT) regulates the intracellular nicotinamide adenine dinucleotide (NAD) pool. NAD is an essential coenzyme which is necessary for cellular redox reactions [37]. In metabolic disorders, the levels of NAD are decreased. NAMPT can change the pathogenesis of obesity and type 2 diabetes by modifying oxidative stress response, apoptosis, lipid and glucose metabolism, inflammation, and insulin resistance [37]. Imeglimin acutely and directly amplifies GSIS in rodents with type 2 diabetes. The elevation of cellular NAD via the salvage pathway and the induction of NAMPT with the enhancement of glucose-induced ATP levels may amplify GSIS [38]. The conversion of NAD into a cyclic ADP ribose is required for GSIS, suggesting a significant association between elevated NAD and augmented glucose-induced Ca2+ mobilization which induces the exocytosis of insulin granule [38]. An imeglimin-mediated increase in intracellular calcium may enhance insulin secretion [38]. Further, imeglimin reduces β-cell apoptosis by lowering the glucotoxicity via a mitochondrial-improvement-dependent mechanism [39]. Imeglimin increases β-cell mass by inhibiting the permeability transition pores (PTP) of mitochondria [39]. Imeglimin increases insulin secretion in a glucose-dependent manner, and also exerts beneficial effects on β-cell mitochondrial integrity in type 2 diabetic mice [40]. The improvement in β-cell mitochondria may facilitate ATP production, enhancing the synthesis and secretion of insulin.
5. An Improvement in β-Cell Function
Imeglimin has a favorable effect on the preservation of the number of insulin granules, the recovery of mitochondrial structure, and the reduction in apoptosis [40]. How could imeglimin improve pancreatic β-cell function? The reduced expression of apoptosis- and inflammation-associated factors such as inflammatory cytokines may prevent β-cell apoptosis. A decrease in oxidative stress by imeglimin may also lead to reduceed β-cell apoptotic cell death and to improved β-cell function. The decrease in β-cell death by imeglimin may be closely linked with the amelioration of β-cell function. In the situation with the induction of apoptotic β-cell death, it is hard for β-cells to preserve the synthesis and secretion of insulin. Although further studies should be performed, the imeglimin-mediated prevention of apoptotic β-cell death may improve β-cell function. Briefly, the improvement in β-cell mitochondrial structure is likely to facilitate ATP production, enhancing β-cell function. Furthermore, imeglimin-mediated improvement in structural integrity and homeostasis of ER may largely contribute to an improvement in β-cell function [33].
6. An Enhancement of Glucose Uptake by the Skeletal Muscles
Skeletal muscle is the major tissue involved in insulin-mediated glucose disposal. The decreased uptake of glucose by muscle due to insulin resistance is an important factor in the development of type 2 diabetes. Acute administration of imeglimin significantly stimulates glucose uptake by muscle cells in a dose-dependent manner [35]. Muscle glucose uptake is reduced in the streptozotocin-treated rats compared to the control rats. Chronic administration of imeglimin (45 days) increases glucose uptake by such muscles. An improvement in mitochondrial dysfunction by imeglimin induces an improvement in insulin signaling in skeletal muscle [35]. An improvement in insulin resistance by imeglimin is expected to increase glucose transporter 4 (GLUT4) expression and improve GLUT4 function in muscle, but this has not yet been proven.
7. An Improvement in Oxidative Stress and Insulin Resistance
Oxidative stress has a crucial role in the pathogenesis of diabetes and its complications [41,42]. Oxidative stress can induce insulin resistance by impairing various insulin signaling [42]. Imeglimin has antioxidative properties which enable it to ameliorate free radical generation and readjust the redox state [22]. Imeglimin reduces oxidative stress by suppressing the mitochondrial free radical generation, which improves glucose homeostasis [23].
Vial et al. show that imeglimin improves insulin sensitivity in high-fat diet mice [22]. Pacini et al. demonstrate that imeglimin improves insulin sensitivity in β-cells in type 2 diabetic patients [43]. They suggest that imeglimin can increase peripheral insulin sensitivity in patients with diabetes [43]; however, the mechanism for this remains largely unknown.
The summary of glucose-lowering mechanisms of imeglimin was shown in Figure 1. Imeglimin improves mitochondrial function, which ameliorates insulin sensitivity in liver and skeletal muscle. As a result, FA oxidation increases in liver and skeletal muscle, hepatic glucose production decreases, and glucose uptake increases in skeletal muscle. Furthermore, imgelimin improves mitochondrial and ER function, which increases GSIS and β-cell mass. Such improvements in insulin sensitivity in metabolically active organs may improve systemic insulin resistance, which may change the insulin resistance milieu in adipose tissue in patients with type 2 diabetes, such as via an increase in adiponectin and a decrease in inflammatory cytokines.
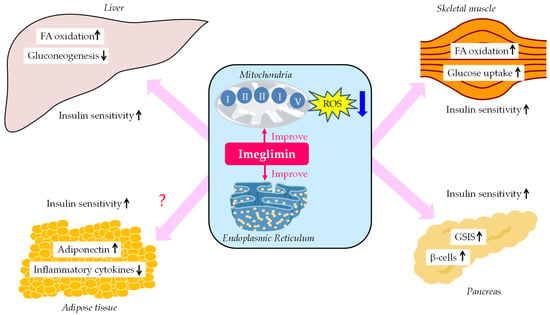
Figure 1. The summary of glucose-lowering mechanisms of imeglimin. FA, fatty acid; GSIS, glucose-stimulated insulin secretion; ROS, reactive oxygen species.
This entry is adapted from the peer-reviewed paper 10.3390/biology12050726
This entry is offline, you can click here to edit this entry!
