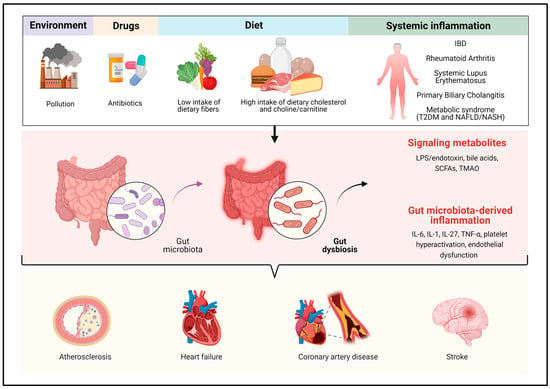
Cardiovascular disease (CVD) is a leading cause of death and morbidity worldwide. Identification of possible preventive strategies is of central interest to avoid CVD onset and progression. In this regard, intensive medical and scientific activity is currently employed in the management of traditional risk factors, but despite maximum medical therapy, there is still a residual risk of undetermined etiology. The gut microbiota is gaining increasing interest as one of the potentially modifiable factors that are involved in the pathogenesis of several diseases, including CVD. A multitude of microorganisms lives symbiotically with the human host; 10–100 trillion microbes among bacteria, viruses, fungi, and helminths are located in the gut. They serve a multitude of functions, which include maintenance of intestinal homeostasis and defence against external aggressive agents, modulation of the immune response, and production of metabolites.
- gut microbiota
- cardiovascular disease
- systemic inflammation
- dysbiosis
- atherosclerosis
- non-alcoholic fatty liver disease
1. Relationship between Gut Microbiota and Cardiovascular Risk Factors
2. Pathogenic Role of the Gut Microbiota in CVD: Focus on Metabolites and Inflammation
2.1. Trimethylamine N-Oxide
2.2. Short-Chain Fatty Acids
2.3. Bile Acids
2.4. Coprostanol
2.5. Phenylacetylglutamine
2.6. Vitamin K2
2.7. Gut Microbiota-Derived Inflammation
4. Gut Microbiota Composition in CVD: What We Know So Far
4.1. Atherosclerosis and Coronary Artery Disease
The close correlation between atherosclerosis and gut microbiota has been extensively described in literature. Dysbiosis and microbial metabolites (i.e., TMAO) play a role in the pathogenic mechanisms of atherosclerosis, such as systemic inflammation, endothelial dysfunction, and lipid homeostasis, and are associated with the severity of the disease. The increase in Enterobacteriaceae is associated with larger coronary plaque fibrotic area and more severe coronary atherosclerosis [62]. Recent studies have shown a correlation between specific intestinal bacteria (i.e., Dysgonomonas, Paraprevotella, Succinatimonas, and Bacillus) and plaque vulnerability, intended as the presence of a thin-cap fibroatheroma, lipid-rich plaque with necrotic core, macrophages, microvessels, cholesterol crystals, and large plaque burden [63]. Moreover, marked overexpression of TMA-producing intestinal microbial enzymes was observed in patients with coronary artery disease compared with healthy controls [64].
4.2. Heart Failure
The gut microbiota of patients with chronic heart failure is characterized by a decreased abundance of beneficial butyrate-producing bacteria and an increase in pathogenic bacteria, including Campylobacter, Salmonella, Shigella, Yersinia enterocolitica, and Candida species [1]. Patients with chronic heart failure and cardiac cachexia also show intestinal wall congestion and oedema, impaired microcirculation, and increased intestinal permeability; this results in gut dysbiosis with a predominance of Firmicutes, Bacteroidetes, and Proteobacteria, as well as in the translocation of bacteria and their metabolites with potential effects on cardiovascular health [65].
4.3. Stroke
It has been observed that a gut microbiota enriched with SCFAs-producing bacteria, such as Akkermansia, Victivallis, Ruminococcaceae, and Odoribacter, may lead to increased risk of cerebrovascular events and correlates with their severity. Conversely, stroke has been linked to gut dysbiosis and intestinal barrier dysfunction [66]. After stroke, a lower blood supply leads to ischemic intestinal damage and this results in the production of excessive nitrate through free radical reactions, but also in changes in the gut microbial community, with the increased abundance of Enterobacteriaceae. A recent animal study investigated the association between acute ischemic stroke and gut dysbiosis, showing how Enterobacteriaceae represent a biomarker of primary poor outcome; functionally, this is explained by increased systemic inflammation, worsening brain ischemia. The authors also highlighted that the administration of aminoguanidine or superoxide dismutase could be useful in counteracting brain injury by restoring gut dysbiosis [67]. Moreover, after stroke, the production of neurotransmitters is altered and the release of noradrenalin leads to changes in intestinal permeability, causing the activation of corticotropin-releasing hormones and glucocorticoid hormones, and promotes bacterial translocation [68]. Finally, TMAO promotes endothelial dysfunction, vascular inflammation, and changes in small cerebral arteries including lipohyalinosis, fibrinoid necrosis, and microaneurysm formation, thus favoring the onset of hemorrhagic stroke. In a Chinese study involving 622 patients with a first stroke, a significant correlation was found between the TMAO serum level and first hemorrhagic stroke [69].
5. Dysbiosis as a Condition Predisposing to CVD
Many inflammatory and metabolic diseases associated with gut dysbiosis also have a close association with CVD.
Several meta-analyses of cohort studies have reported associations between inflammatory bowel diseases (IBDs) and CVD [70,71]. In particular, increased carotid intimal thickness, wall stiffness, and endothelial dysfunction [72,73], a 4-to-5 fold increase in homocysteine serum level [74], and a higher prevalence of acute myocardial infarction (AMI) have been observed in patients with IBD compared with those without inflammatory intestinal diseases, with the highest risk of AMI in young women aged 30–34 years [75]. A Danish cohort study has also reported that patients affected by IBD have an increased risk of stroke [76,77], as well as a higher rate of atrial fibrillation and hospitalization for heart failure, even in young patients, and especially during acute flares or in case of persistently active disease [78,79]. It is well-established that IBDs are associated with gut dysbiosis [80,81] and several studies have shown a generalized decrease in microbial diversity and a reduction in specific beneficial bacterial taxa, including Lactobacillus, Eubacterium, and butyrate-producing bacteria such as Faecalibacterium prausnitzii [82,83,84,85,86,87,88]. Non-alcoholic fatty liver disease (NAFLD), which can be considered the hepatic manifestation of metabolic syndrome, also finds its roots in dysbiosis and increased intestinal permeability [89]. Several studies have identified a reduced gut microbiota diversity and various changes in gut microbiota composition in patients with NAFLD [90,91,92]. Dysbiosis, in turn, is responsible for the dysregulation of intestinal endothelial and vascular barrier function, with enhanced translocation of bacteria and their products (PAMPs: endotoxins, LPS, peptidoglycan) and molecules released from damaged intestinal cells (DAMPs); after reaching the liver through the portal circulation or the bloodstream through mesenteric lymph nodes, PAMPs and DAMPs trigger various cellular signaling pathways that induce a systemic inflammatory response [93,94]. Alterations in the gut microbiota were found in patients with coronary artery disease and NAFLD, mainly characterized by a decrease in Parabacterioides and Colinsella [95]. A prospective study has also shown that TMAO concentrations were higher in patients with NAFLD, being significantly and independently associated with an increased risk of all-cause mortality [96]. Intriguingly, this association was not present in subjects without NAFLD. This confirms that altered gut dysbiosis may influence progression of metabolic syndrome-associated diseases as NAFLD [97]. Another interesting setting are patients with PBC, an autoimmune cholestatic liver disease that is characterized by hypercholesterolemia and affects middle-aged women. However, there is limited data on the incidence of atherosclerosis and CVD in these patients, with only recent studies showing an increased risk [98]. Patients with PBC have a doubled prevalence of lower extremity arterial disease (LEAD) compared with age-matched general female population, and the gut microbiota seems to be associated with this finding [99]. Indeed, vascular adhesion molecule-1 (VCAM-1) and TNF-α were independent predictors of LEAD, and Acidaminococcus, a bacterial genus highly abundant in PBC women, was positively correlated with serum levels of TNF-α. A final remark should be made on rheumatic diseases such as rheumatoid arthritis (RA) and systemic lupus erythematous (SLE). RA has been associated with periodontitis [100], and high serum titers of Porphyromonas gingivalis antibodies have been found in patients with more severe disease activity and functional impairment [101,102]. Interestingly, P. gingivalis DNA was detected also in atherosclerotic plaques of subjects with periodontitis. Clinical and animal studies have pointed out that P. gingivalis accelerates atherosclerosis [103,104]; the mechanisms can be multiple, and include: (a) intracellular influx of oxidized-LDL and its conversion to cholesterol crystals, via increased expression of CD36 and fatty acid binding protein 4 (FABP4) on macrophages [105,106]; (b) activation of NLRP3 inflammasomes by cholesterol crystals’ damage to the phagolysosomes and P. gingivalis-induced production of reactive oxygen species, with consequent activation of the inflammatory cascade [107,108]; (c) down-regulation of the cholesterol transporters ATP-binding cassettes (ABCA1) on macrophages, which promotes cholesterol accumulation [109]. The pathogenesis of SLE has been associated with dysbiosis, which is mainly characterized by a lower Firmicutes/Bacteroides ratio and overabundance of Ruminococcus gnavus, Enterococcus gallinarum, Streptococcus anginosus, Streptococcus dispar, Veillonella, and Campylobacter, and contributes to disease development and progression through pro-inflammatory stimulation and production of anti-dsDNA antibodies [110,111,112]. The pro-inflammatory milieu interferes with blood pressure regulatory mechanisms, such as the renin–angiotensin system and the sympathetic nervous system [113]; in particular, circulating TNF-α, which is increased in patients with SLE and correlates with disease activity, is involved in the development of hypertension; indeed, in female murine models of SLE, a decrease in mean arterial pressure has been observed after treatment with the anti-TNF-α antibody etanercept [114,115]. Furthermore, LPS increases the expression of TLR4 in blood vessels, which results in increased nicotinamide adenine dinucleotide phosphate (NADPH) oxidase-dependent superoxide production, inflammation, and endothelial dysfunction [116,117]. Elevated levels of plasma LPS have been reported in both patients and hypertensive mice affected by SLE [118,119]. Lastly, in patients with SLE, intestinal dysbiosis is associated with an altered production of SCFAs [120].
In summary, dysbiosis is involved in the pathogenesis of several diseases that are also associated with cardiovascular dysfunction. Microbial-derived inflammation but also metabolites are the driving force of this link, but it is not possible to exclude further mechanisms that have not been clarified to date.
6. Evidence on the Impact of Gut Microbiota Modulation in Reducing Cardiovascular Risk
The recognition of the gut microbiota as a key player in the pathogenesis of CVD suggests that specific therapeutic interventions aimed at its modulation may potentially reduce cardiovascular risk. Several studies have focused on this topic; however, they have shown mixed results.
6.1. Dietary Intervention
Diet has multiple effects on the gut microbiota, being able to modify its composition and function [121]; these effects are not immediate and have greater impact if maintained for a long period [122]. A diet rich in plant products modulates the intestinal bacteria community favoring the growth of species able to ferment fibers, resulting in increased production of SCFAs and phosphatidylcholine. Conversely, a high-fat diet leads to unfavorable changes in the gut microbiota, fecal metabolomic profile, and systemic inflammation [123]. All of these changes are associated with adverse effects on human health, because in the long term they lead to increase the risk of obesity, metabolic syndrome, and cardiovascular risk [124,125]. A controlled-feeding trial conducted in China on 217 healthy volunteers highlighted that a high-fat diet modifies the gut microbiota composition, increasing Bacteroides and Alistipes, more abundant in patients with type 2 diabetes mellitus (T2DM), and reducing Faecalibacterium, a butyrate-producing microorganism. Conversely, in the low-fat diet group, an increased abundance of Faecalibacterium and Blautia was found, which favorably affect lipid metabolism [126]. Changes in the gut microbiota community reflect in fecal metabolomic profiles. In fact, a high-fat diet leads to a reduction in SCFAs and an increase in arachidonic acid and LPS biosynthesis, with consequent elevation in circulating pro-inflammatory factors (i.e., plasminogen activator inhibitor-1, IL-1, and TNF-α mRNA) [127]. In a randomized, controlled, crossover study conducted in subjects with ischemic heart disease, a 4-week consumption of a diet rich in plant-based products (vegetarian diet), has been shown to reduce oxidized low-density lipoprotein cholesterol and to change the relative abundance of intestinal bacteria, in particular of Ruminococcaceae and Barnesiella, and their metabolites compared with Mediterranean diet. An improvement in cardiovascular risk profile derived from reduction in oxidized LDL cholesterol was reported in the vegetarian diet group, but no significant difference in TMAO production was observed [128]. Another study showed that a Mediterranean diet has anti-inflammatory properties, with a negative correlation between SCFAs production and the expression of inflammatory cytokines such as vascular-endothelial growth factor (VEGF), monocyte chemoattractant protein-1 (MCP-1), IFN-γ-induced protein 10 (IP-10), IL-17, and IL-12. In addition, Mediterranean diet increased the abundance of Enterorhabdus, Lachnoclostridium, and Parabacteroides [129]. Another observational study conducted by Dong D. Wang et al. supports the hypothesis that a controlled diet may produce beneficial changes in the gut microbiota composition in prevention of CVD [130]. Long-term adherence to a Mediterranean diet exerts a gradual selective pressure on the adult gut microbiota, resulting in a relative abundance of fiber-metabolizing bacterial species (Faecalibacterium prausnitzii, Bacteroides cellulosilyticus, and Eubacterium eligens), compared to other pathogens mainly associated with a Western-type diet and red meat intake (such as Ruminococcus torques, Clostridium leptum, and Collinsella aerofaciens). For example, the selection of particular taxa responsible for the conversion of BAs (i.e., C. aerofaciens) may lead to adverse cardiometabolic effects by a dysregulation of the BAs pool; in fact, such compounds behave as hormones that interact with nuclear and G protein-coupled receptors interfering with certain metabolic processes. Other authors also refer to Prevotella copri, the role of which has not yet been sufficiently clarified, but seems to be associated with an increase in the biosynthesis of branched-chain amino acids, which are linked to an increased cardiovascular risk through insulin resistance in humans [131]. The adoption of a Mediterranean diet, according to Dong D. Wang et al., is able to mitigate this risk, as it is likely that such subjects do not acquire or retain P. copri. Another hypothesis is that the Mediterranean diet exerts its beneficial effects on CVD prevention only in P. copri non-carriers [130]. Many of the clinical trials currently available in literature have the major limitation of being restricted to a limited period; therefore, whether dietary changes can be maintained over time is still a matter of debate, and future studies are needed to test the resilience of the gut microbiota. The intake of particular foods within a balanced diet can produce a benefit for human health. Plant-based omega-3 fatty acids, α-linolenic acid, and polyunsaturated fatty acids (PUFAs) have been shown to exert benefits on the cardiovascular system. Notably, several of these substances can be classified as prebiotics, which are substrates selectively used by host microorganisms conferring a health benefit [132]. Walnuts are a main source of these and many other compounds, such as hydrolyzable tannins and fibers, which can be metabolized by gut bacteria and confer additional benefits in term of cardiometabolic risk. A recent randomized controlled trial involving 42 patients at cardiovascular risk (defined as overweight and obese middle-aged men and women, with dyslipidemia and hypertension) showed that a whole walnut-based diet and a walnut fatty acid–matched diet are able to change the intestinal bacterial composition, in particular increasing the abundance of Roseburia, a butyrate-producing bacteria. Moreover, ellagitannins naturally contained in walnuts, are metabolized by gut bacteria to form urolithins, which may provide additional cardiovascular benefit [133]. Dietary PUFAs are also associated with multiple cardiovascular effects mediated by changes in the gut microbiota; in fact, omega-3 PUFAs increase the abundance of several SCFAs-producing bacteria, decreasing those associated with TMA production. Additionally, omega-3 PUFAs would help maintain intestinal barrier integrity, thereby preventing the translocation of intestinal products into systemic circulation and reducing the production of pro-inflammatory cytokines [134]. A similar effect was also described for the polyphenol hesperidin, found in citrus fruits. Polyphenols are natural products of plant extraction potentially used as prebiotics. Dietary supplementation of polyphenols has proven to have cardiovascular benefits, because they reduce blood pressure and improve endothelial dysfunction and lipid profile [135]. Hesperidin promotes the growth of beneficial bacteria, such as Lactobacillus and Bifidobacterium, stimulates the production of SCFAs, and lowers plasma levels of pro-inflammatory cytokines such as IL-1β, TNF-α, and IL-6 [136]. Finally, there are scant results from human intervention studies with SCFAs in order to reduce blood pressure; Roshanravan et al. have demonstrated in a randomized, double-blind, placebo-controlled trial that oral butyrate supplements tend to significantly lower blood pressure in patients with metabolic syndrome [137].
6.2. Probiotics
The role of probiotics in reducing cardiovascular risk is an issue that is gathering scientific interest, especially in light of new scientific evidence supporting a role for the gut microbiota in the pathogenesis of CVD. A pilot study including 21 men with stable coronary artery disease showed that a 6-week daily supplementation with Lactobacillus plantarum 299v (Lp299v) has a favorable impact on CVD inducing changes in gut microbiome-derived circulating metabolites. Supplementation with Lp299v can improve endothelium-dependent vasodilation of the brachial artery, through the increase in nitric oxide bioavailability, and reduce systemic inflammation. These effects appear to be independent of traditional cardiovascular risk factors, and not related to TMAO serum levels [138]. Some studies show that Bacteroides depletion in humans is associated with higher incidence of symptomatic atherosclerosis. Nevertheless, in animal models, oral supplementation of Bacteroides vulgatus and Bacteroides dorei reduced atherosclerotic plaque inflammation and slowed its formation. In particular, Bacteroides supplementation in atherosclerosis-prone mice reduces LPS production, and successfully ameliorates endotoxemia, suppressing pro-inflammatory immune responses [139]. Akkermansia muciniphila is a component of the gut microbiota that exerts favorable effects on the pathogenesis of CVD and arterial hypertension [4]. Probiotic bacteria in milk, yogurt bacteria, and cheese starter bacteria are able to produce bioactive peptides with antihypertensive function. A study has shown that Lactobacillus helveticus produces angiotensin converting enzyme (ACE) inhibitory tripeptides, which play an antihypertensive role in renin-angiotensin system [140]. Studies conducted on spontaneous hypertensive rats reported that oral supplementation of high doses of Lactobacillus casei strain C1 led to a significant reduction in systolic and diastolic blood pressure at 8 weeks [141]. Trials conducted in humans underline that the reduction of systolic and diastolic blood pressure by probiotic supplements is modest but significant; the main limits on the effectiveness are the duration of treatment, dosage, age of the subjects, and type of strain used [142]. Probiotics may also be a therapeutic opportunity for the treatment of stroke. A recent study conducted in mice models of ischemic stroke showed that ischemic brain injury was reduced by 52% and the neurological outcome ameliorated after treatment with probiotic bacteria (such as Bifidobacterium breve, Lactobacillus bulgaricus, Lactobacillus casei, and Actobacillus acidophilus); the neuroprotective effect was probably due to the anti-inflammatory properties and modulation of oxidative stress damage [143].
6.3. Drugs
Many non-antibiotic medications can modulate microbiota composition and function, influencing health outcomes. For example, proton pump inhibitors are among the most widely used drugs that can modify the gut microbiota, leading to a decreased colonization-resistance to enteric infections (i.e., Clostridium difficile infection) and to the oralization of the colonic microbiota [144,145]. Several antidiabetic drugs, such as metformin and liraglutide, exert their therapeutic effects and additional metabolic benefits by changing the gut microbiota composition and metabolism [146]. Among the novel sodium/glucose cotransporter 2 inhibitors (SGLT2i), empagliflozin also shows an effect in increasing the richness and diversity of the gut microbiota, improving inflammatory parameters. In a recent study, empagliflozin was able to promote a selection of SCFAs-producing bacteria, such as Roseburia, Faecalibacterium, and Eubacterium over potentially harmful bacteria, including Escherichia-Shigella, Bilophila, and Hungatella [147]. In another study, a 28-day treatment with dapagliflozin, another SGLT2i, significantly improved cardiac function in the non-diabetic myocardial infarction mice model and modified the gut microbiota composition, increasing the abundance of beneficial bacteria such as Lactobacillaceae [148]. Muribaculaceae and Lactobacillaceae were the main components of the intestinal microbial community after treatment with dapagliflozin, while Muribaculaceae and Erysipelotrichaceae were the ones associated with myocardial infarction. Antihypertensive medications, such as the angiotensin-converting enzyme inhibitor captopril, have shown beneficial effects on hypertension-associated gut pathology, in particular reducing intestinal permeability, thickness of the muscularis layer and increasing the length of villi by 55% [149]. Pharmacological effect of cholesterol-lowering drugs is partly impaired by gut microbiota, and dysbiosis can generate further pharmacological variability [150]. In an animal model, it has been shown that the hypolipidemic effect of statins, particularly simvastatin, is partially reduced with concomitant administration of antibiotics [151]. Other studies focused on the modulating effect of statins on the gut microbiota composition; for example, in animals the administration of rosuvastatin increased the abundance of Lachnospiraceae and Erysipelotrichaceae and decreased the abundance of Proteobacteria, Coriobacteriaceae and Akkermansia [152]. Finally, antibiotic treatment disrupts gut microbiota homeostasis, and leads to potentially harmful dysbiosis. Rifaximin exerts anti-inflammatory and eubiotic effects, producing a positive modulation of the gut microbiota and reducing intestinal bacteria adherence, internalization, and translocation [153]; however, its effects on cardiovascular risk reduction have not yet been investigated. At present, the use of broad-spectrum antibiotics in reducing cardiovascular risk remains controversial, due to the potential side effects and the induction of bacterial resistance [1]. The concept of pharmacomicrobiomics, which means the impact of the gut microbiota on drug bioavailability, bioactivity, or toxicity by direct and indirect mechanisms, is currently emerging [154]. The role of the gut microbiota in influencing the effectiveness of a therapeutic treatment has already been investigated in various diseases such as ulcerative colitis, Crohn’s disease, and RA. Pharmacomicrobiomics is also applied in the cardiovascular field; in fact, it is known that digoxin, a drug used in heart failure, is ineffective in 1 in 10 patients because it is likely converted into an inactive form, dihydrodigoxin, by Eggerthella lenta [155]. In summary, pharmacomicrobiomics emphasizes the importance of pursuing a personalized medicine that focuses on the microbiota.
6.4. Small Molecule Antimicrobial Enzyme Therapeutics
Blocking microbial TMA production is a potential therapeutic strategy for the prevention and treatment of atherosclerosis. Selective enzymes structurally similar to choline have been developed and are being studied to reduce cardiovascular risk. In particular, 3,3-dimethyl-1-butanol (DMB) can inhibit microbial TMA-lyase and reduce both TMA production and serum levels of TMAO in mice fed a high carnitine or choline diet. DMB showed direct effects on atherosclerosis, inhibited dietary choline-dependent accumulation of cholesteryl ester in macrophages (foam cell formation) and development of aortic root atherosclerotic plaque [156]. New choline TMA-lyase inhibitors, including iodomethylcholine (IMC) and fluoromethylcholine (FMC), have been recently developed, but data are currently limited [17]. Other studies focused on the suppression of the FMO3 in animal models through an antisense oligonucleotide-based approach, highlighted a marker reduction of diet-enhanced atherosclerosis paralleled by a decrease in TMAO serum levels [157].
6.5. Faecal Microbiota Transplantation
Fecal microbiota transplantation (FMT) is an effective therapeutic strategy in multiple gastrointestinal pathologies and consists of transferring a structured community of intestinal bacteria derived from a stool donor in the affected subject. Although the possible implications of FMT in CVD have not yet been investigated in humans, preliminary results are encouraging. In particular, a recent study reported that the gut microbiota of spontaneously hypertensive rats was characterized by increased abundance of Turicibacter, which was positively associated with arterial hypertension, and by an altered T helper-17/regulatory T cells (Th17/Tregs) balance in mesenteric lymph nodes. When transplanted in normotensive rats, this dysbiotic microbiota induced endothelial dysfunction and hypertension, through both T cell activation and IL-17 production. Conversely, FMT from normotensive rats in spontaneously hypertensive rats improved systolic blood pressure, endothelial dysfunction, oxidative stress, and vascular inflammation, as well as the imbalance between Th17/Tregs [158].
7. Conclusions
Over the last decade, several studies have strengthened the concept of gut microbiota as a dynamic living entity, which can generate, sustain, and worsen various pathological processes, but at the same time influenced by diet, drugs and other stimuli from the external environment. CVD is a global health problem affecting millions of people, and it is of paramount interest to identify effective prevention and treatment strategies to reduce health care costs. Currently, scientific evidence proves the existence of a two-way relationship between the gut microbiota and CVD. The mechanisms involved in this relationship are multiple and extremely complex because they concern immune regulation, inflammatory response, gastrointestinal barrier integrity, metabolic pathways, and much more. Many of these effects are mediated by bacterial-derived products that play a significant role in generating and sustaining chronic inflammation. Human and animal studies have attempted to characterize dysbiosis in CVD, effectively identifying the species most frequently involved, and have analyzed different therapeutic approaches (i.e., dietary intervention, probiotics, prebiotics, drugs, FMT), with encouraging results. Our review sheds light on the complex relationship between intestinal microbiota and CVD by reviewing the latest scientific evidence, focusing on some metabolic diseases. However, further research is needed in order to identify effective microbiome-based preventive and therapeutic approaches to be adopted as additional weapons in the management of CVD.
This entry is adapted from the peer-reviewed paper 10.3390/ijms24109087
