The urgent necessity to fight antimicrobial resistance is universally recognized. In the search of new targets and strategies to face this global challenge, a promising approach resides in the study of the cellular response to antimicrobial exposure and on the impact of global cellular reprogramming on antimicrobial drugs’ efficacy. The metabolic state of microbial cells has been shown to undergo several antimicrobial-induced modifications and, at the same time, to be a good predictor of the outcome of an antimicrobial treatment. Metabolism is a promising reservoir of potential drug targets/adjuvants that has not been fully exploited to date. One of the main problems in unraveling the metabolic response of cells to the environment resides in the complexity of such metabolic networks. To solve this problem, modeling approaches have been developed, and they are progressively gaining in popularity due to the huge availability of genomic information and the ease at which a genome sequence can be converted into models to run basic phenotype predictions.
- metabolic modeling
- antimicrobial resistance
- bacterial metabolism
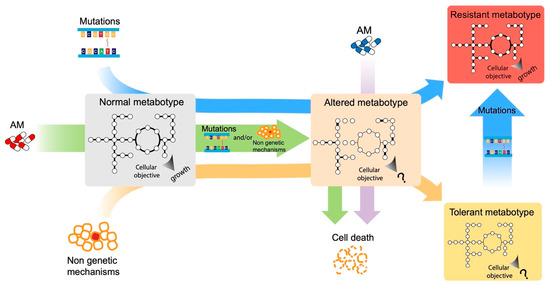
This entry is adapted from the peer-reviewed paper 10.3390/antibiotics12050896
References
- Murray, C.J.; Ikuta, K.S.; Sharara, F.; Swetschinski, L.; Aguilar, G.R.; Gray, A.; Han, C.; Bisignano, C.; Rao, P.; Wool, E.; et al. Global Burden of Bacterial Antimicrobial Resistance in 2019: A Systematic Analysis. Lancet 2022, 399, 629–655.
- Stokes, J.M.; Lopatkin, A.J.; Lobritz, M.A.; Collins, J.J. Bacterial Metabolism and Antibiotic Efficacy. Cell Metab. 2019, 30, 251–259.
- Roemhild, R.; Bollenbach, T.; Andersson, D.I. The Physiology and Genetics of Bacterial Responses to Antibiotic Combinations. Nat. Rev. Microbiol. 2022, 20, 478–490.
- Martínez, J.L.; Rojo, F. Metabolic Regulation of Antibiotic Resistance. FEMS Microbiol. Rev. 2011, 35, 768–789.
- Baquero, F.; Levin, B.R. Proximate and Ultimate Causes of the Bactericidal Action of Antibiotics. Nat. Rev. Microbiol. 2021, 19, 123–132.
- Gavaghan, C.L.; Holmes, E.; Lenz, E.; Wilson, I.D.; Nicholson, J.K. An NMR-Based Metabonomic Approach to Investigate the Biochemical Consequences of Genetic Strain Differences: Application to the C57BL10J and Alpk:ApfCD Mouse. FEBS Lett. 2000, 484, 169–174.
- Adolfsen, K.J.; Brynildsen, M.P. Futile Cycling Increases Sensitivity toward Oxidative Stress in Escherichia coli. Metab. Eng. 2015, 29, 26–35.
- Belenky, P.; Ye, J.D.; Porter, C.B.M.; Cohen, N.R.; Lobritz, M.A.; Ferrante, T.; Jain, S.; Korry, B.J.; Schwarz, E.G.; Walker, G.C.; et al. Bactericidal Antibiotics Induce Toxic Metabolic Perturbations That Lead to Cellular Damage. Cell Rep. 2015, 13, 968–980.
- Cho, H.; Uehara, T.; Bernhardt, T.G. Beta-Lactam Antibiotics Induce a Lethal Malfunctioning of the Bacterial Cell Wall Synthesis Machinery. Cell 2014, 159, 1300–1311.
- Dwyer, D.J.; Belenky, P.A.; Yang, J.H.; MacDonald, I.C.; Martell, J.D.; Takahashi, N.; Chan, C.T.Y.; Lobritz, M.A.; Braff, D.; Schwarz, E.G.; et al. Antibiotics Induce Redox-Related Physiological Alterations as Part of Their Lethality. Proc. Natl. Acad. Sci. USA 2014, 111, E2100–E2109.
- Foti, J.J.; Devadoss, B.; Winkler, J.A.; Collins, J.J.; Walker, G.C. Oxidation of the Guanine Nucleotide Pool Underlies Cell Death by Bactericidal Antibiotics. Science 2012, 336, 315–319.
- Hong, Y.; Zeng, J.; Wang, X.; Drlica, K.; Zhao, X. Post-Stress Bacterial Cell Death Mediated by Reactive Oxygen Species. Proc. Natl. Acad. Sci. USA 2019, 116, 10064–10071.
- Kohanski, M.A.; Dwyer, D.J.; Hayete, B.; Lawrence, C.A.; Collins, J.J. A Common Mechanism of Cellular Death Induced by Bactericidal Antibiotics. Cell 2007, 130, 797–810.
- Lobritz, M.A.; Belenky, P.; Porter, C.B.M.; Gutierrez, A.; Yang, J.H.; Schwarz, E.G.; Dwyer, D.J.; Khalil, A.S.; Collins, J.J. Antibiotic Efficacy Is Linked to Bacterial Cellular Respiration. Proc. Natl. Acad. Sci. USA 2015, 112, 8173–8180.
- Vatansever, F.; de Melo, W.C.M.A.; Avci, P.; Vecchio, D.; Sadasivam, M.; Gupta, A.; Chandran, R.; Karimi, M.; Parizotto, N.A.; Yin, R.; et al. Antimicrobial Strategies Centered around Reactive Oxygen Species—Bactericidal Antibiotics, Photodynamic Therapy, and Beyond. FEMS Microbiol. Rev. 2013, 37, 955–989.
- Wong, F.; Stokes, J.M.; Cervantes, B.; Penkov, S.; Friedrichs, J.; Renner, L.D.; Collins, J.J. Cytoplasmic Condensation Induced by Membrane Damage Is Associated with Antibiotic Lethality. Nat. Commun. 2021, 12, 2321.
- Wong, F.; Stokes, J.M.; Bening, S.C.; Vidoudez, C.; Trauger, S.A.; Collins, J.J. Reactive Metabolic Byproducts Contribute to Antibiotic Lethality under Anaerobic Conditions. Mol. Cell 2022, 82, 3499–3512.e10.
- Lin, X.; Kang, L.; Li, H.; Peng, X. Fluctuation of Multiple Metabolic Pathways Is Required for Escherichia coli in Response to Chlortetracycline Stress. Mol. Biosyst. 2014, 10, 901–908.
- Dahlberg, C.; Chao, L. Amelioration of the Cost of Conjugative Plasmid Carriage in Eschericha coli K12. Genetics 2003, 165, 1641–1649.
- Melnyk, A.H.; Wong, A.; Kassen, R. The Fitness Costs of Antibiotic Resistance Mutations. Evol. Appl. 2015, 8, 273–283.
- Levin, B.R.; Lipsitch, M.; Perrot, V.; Schrag, S.; Antia, R.; Simonsen, L.; Walker, N.M.; Stewart, F.M. The Population Genetics of Antibiotic Resistance. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 1997, 24 (Suppl. S1), S9–S16.
- Marciano, D.C.; Karkouti, O.Y.; Palzkill, T. A Fitness Cost Associated with the Antibiotic Resistance Enzyme SME-1 Beta-Lactamase. Genetics 2007, 176, 2381–2392.
- Zampieri, M.; Enke, T.; Chubukov, V.; Ricci, V.; Piddock, L.; Sauer, U. Metabolic Constraints on the Evolution of Antibiotic Resistance. Mol. Syst. Biol. 2017, 13, 917.
- Lopatkin, A.J.; Bening, S.C.; Manson, A.L.; Stokes, J.M.; Kohanski, M.A.; Badran, A.H.; Earl, A.M.; Cheney, N.J.; Yang, J.H.; Collins, J.J. Clinically Relevant Mutations in Core Metabolic Genes Confer Antibiotic Resistance. Science 2021, 371, eaba0862.
- Brauner, A.; Fridman, O.; Gefen, O.; Balaban, N.Q. Distinguishing between Resistance, Tolerance and Persistence to Antibiotic Treatment. Nat. Rev. Microbiol. 2016, 14, 320–330.
- Andersson, D.I.; Nicoloff, H.; Hjort, K. Mechanisms and Clinical Relevance of Bacterial Heteroresistance. Nat. Rev. Microbiol. 2019, 17, 479–496.
- Sulaiman, J.E.; Lam, H. Evolution of Bacterial Tolerance under Antibiotic Treatment and Its Implications on the Development of Resistance. Front. Microbiol. 2021, 12, 617412.
- Balaban, N.Q.; Helaine, S.; Lewis, K.; Ackermann, M.; Aldridge, B.; Andersson, D.I.; Brynildsen, M.P.; Bumann, D.; Camilli, A.; Collins, J.J.; et al. Definitions and Guidelines for Research on Antibiotic Persistence. Nat. Rev. Microbiol. 2019, 17, 441–448.
- Levin, B.R.; Rozen, D.E. Non-Inherited Antibiotic Resistance. Nat. Rev. Microbiol. 2006, 4, 556–562.
- Lopatkin, A.J.; Stokes, J.M.; Zheng, E.J.; Yang, J.H.; Takahashi, M.K.; You, L.; Collins, J.J. Bacterial Metabolic State More Accurately Predicts Antibiotic Lethality than Growth Rate. Nat. Microbiol. 2019, 4, 2109–2117.
- Srivatsan, A.; Wang, J.D. Control of Bacterial Transcription, Translation and Replication by (p)PpGpp. Curr. Opin. Microbiol. 2008, 11, 100–105.
- Pesavento, C.; Hengge, R. Bacterial Nucleotide-Based Second Messengers. Curr. Opin. Microbiol. 2009, 12, 170–176.
- Wu, J.; Long, Q.; Xie, J. (P)PpGpp and Drug Resistance. J. Cell. Physiol. 2010, 224, 300–304.
- Jain, V.; Kumar, M.; Chatterji, D. PpGpp: Stringent Response and Survival. J. Microbiol. Seoul Korea 2006, 44, 1–10.
- Lewis, K. Persister Cells. Annu. Rev. Microbiol. 2010, 64, 357–372.
- Balaban, N.Q.; Merrin, J.; Chait, R.; Kowalik, L.; Leibler, S. Bacterial Persistence as a Phenotypic Switch. Science 2004, 305, 1622–1625.
- Shah, D.; Zhang, Z.; Khodursky, A.; Kaldalu, N.; Kurg, K.; Lewis, K. Persisters: A Distinct Physiological State of E. Coli. BMC Microbiol. 2006, 6, 53.
- Prax, M.; Bertram, R. Metabolic Aspects of Bacterial Persisters. Front. Cell. Infect. Microbiol. 2014, 4, 148.
- Kim, W.; Killam, T.; Sood, V.; Surette, M.G. Swarm-Cell Differentiation in Salmonella enterica Serovar Typhimurium Results in Elevated Resistance to Multiple Antibiotics. J. Bacteriol. 2003, 185, 3111–3117.
- Overhage, J.; Bains, M.; Brazas, M.D.; Hancock, R.E.W. Swarming of Pseudomonas aeruginosa Is a Complex Adaptation Leading to Increased Production of Virulence Factors and Antibiotic Resistance. J. Bacteriol. 2008, 190, 2671–2679.
- Yeung, A.T.; Torfs, E.C.; Jamshidi, F.; Bains, M.; Wiegand, I.; Hancock, R.E.; Overhage, J. Swarming of Pseudomonas aeruginosa Is Controlled by a Broad Spectrum of Transcriptional Regulators, Including MetR. J. Bacteriol. 2009, 191, 5592–5602.
- Kim, W.; Surette, M.G. Swarming Populations of Salmonella Represent a Unique Physiological State Coupled to Multiple Mechanisms of Antibiotic Resistance. Biol. Proced. Online 2003, 5, 189–196.
- Irazoki, O.; Campoy, S.; Barbé, J. The Transient Multidrug Resistance Phenotype of Salmonella enterica Swarming Cells Is Abolished by Sub-Inhibitory Concentrations of Antimicrobial Compounds. Front. Microbiol. 2017, 8, 1360.
- Sauer, K.; Stoodley, P.; Goeres, D.M.; Hall-Stoodley, L.; Burmølle, M.; Stewart, P.S.; Bjarnsholt, T. The Biofilm Life Cycle: Expanding the Conceptual Model of Biofilm Formation. Nat. Rev. Microbiol. 2022, 20, 608–620.
- Ciofu, O.; Moser, C.; Jensen, P.Ø.; Høiby, N. Tolerance and Resistance of Microbial Biofilms. Nat. Rev. Microbiol. 2022, 20, 621–635.
- Jo, J.; Price-Whelan, A.; Dietrich, L.E.P. Gradients and Consequences of Heterogeneity in Biofilms. Nat. Rev. Microbiol. 2022, 20, 593–607.
- Fajardo, A.; Linares, J.F.; Martínez, J.L. Towards an Ecological Approach to Antibiotics and Antibiotic Resistance Genes. Clin. Microbiol. Infect. Off. Publ. Eur. Soc. Clin. Microbiol. Infect. Dis. 2009, 15 (Suppl. S1), 14–16.
- Corona, F.; Martinez, J.L. Phenotypic Resistance to Antibiotics. Antibiotics 2013, 2, 237–255.
- Bhagirath, A.Y.; Li, Y.; Patidar, R.; Yerex, K.; Ma, X.; Kumar, A.; Duan, K. Two Component Regulatory Systems and Antibiotic Resistance in Gram-Negative Pathogens. Int. J. Mol. Sci. 2019, 20, 1781.
- Dersch, P.; Khan, M.A.; Mühlen, S.; Görke, B. Roles of Regulatory RNAs for Antibiotic Resistance in Bacteria and Their Potential Value as Novel Drug Targets. Front. Microbiol. 2017, 8, 803.
- Kim, W.J.; Kim, H.U.; Lee, S.Y. Current State and Applications of Microbial Genome-Scale Metabolic Models. Curr. Opin. Syst. Biol. 2017, 2, 10–18.
- Arkin, A.P.; Cottingham, R.W.; Henry, C.S.; Harris, N.L.; Stevens, R.L.; Maslov, S.; Dehal, P.; Ware, D.; Perez, F.; Canon, S.; et al. KBase: The United States Department of Energy Systems Biology Knowledgebase. Nat. Biotechnol. 2018, 36, 566–569.
- Henry, C.S.; DeJongh, M.; Best, A.A.; Frybarger, P.M.; Linsay, B.; Stevens, R.L. High-Throughput Generation, Optimization and Analysis of Genome-Scale Metabolic Models. Nat. Biotechnol. 2010, 28, 977–982.
- Machado, D.; Andrejev, S.; Tramontano, M.; Patil, K.R. Fast Automated Reconstruction of Genome-Scale Metabolic Models for Microbial Species and Communities. Nucleic Acids Res. 2018, 46, 7542–7553.
- Kanehisa, M.; Goto, S. KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res. 2000, 28, 27–30.
- Sayers, E.W.; Bolton, E.E.; Brister, J.R.; Canese, K.; Chan, J.; Comeau, D.C.; Connor, R.; Funk, K.; Kelly, C.; Kim, S.; et al. Database Resources of the National Center for Biotechnology Information. Nucleic Acids Res. 2022, 50, D20–D26.
- King, Z.A.; Lu, J.; Dräger, A.; Miller, P.; Federowicz, S.; Lerman, J.A.; Ebrahim, A.; Palsson, B.O.; Lewis, N.E. BiGG Models: A Platform for Integrating, Standardizing and Sharing Genome-Scale Models. Nucleic Acids Res. 2016, 44, D515–D522.
- Passi, A.; Tibocha-Bonilla, J.D.; Kumar, M.; Tec-Campos, D.; Zengler, K.; Zuniga, C. Genome-Scale Metabolic Modeling Enables In-Depth Understanding of Big Data. Metabolites 2022, 12, 14.
- Orth, J.D.; Thiele, I.; Palsson, B.Ø. What Is Flux Balance Analysis? Nat. Biotechnol. 2010, 28, 245–248.
- Edwards, J.S.; Palsson, B.O. Systems Properties of the Haemophilus InfluenzaeRd Metabolic Genotype. J. Biol. Chem. 1999, 274, 17410–17416.
- Saini, D.K.; Rai, A.; Devi, A.; Pabbi, S.; Chhabra, D.; Chang, J.-S.; Shukla, P. A Multi-Objective Hybrid Machine Learning Approach-Based Optimization for Enhanced Biomass and Bioactive Phycobiliproteins Production in Nostoc sp. CCC-403. Bioresour. Technol. 2021, 329, 124908.
- Swayambhu, G.; Moscatello, N.; Atilla-Gokcumen, G.E.; Pfeifer, B.A. Flux Balance Analysis for Media Optimization and Genetic Targets to Improve Heterologous Siderophore Production. iScience 2020, 23, 101016.
- Chung, W.Y.; Zhu, Y.; Mahamad Maifiah, M.H.; Shivashekaregowda, N.K.H.; Wong, E.H.; Abdul Rahim, N. Novel Antimicrobial Development Using Genome-Scale Metabolic Model of Gram-Negative Pathogens: A Review. J. Antibiot. 2021, 74, 95–104.
- Zhu, Y.; Zhao, J.; Li, J. Genome-Scale Metabolic Modeling in Antimicrobial Pharmacology. Eng. Microbiol. 2022, 2, 100021.
- Sertbas, M.; Ulgen, K.O. Genome-Scale Metabolic Modeling for Unraveling Molecular Mechanisms of High Threat Pathogens. Front. Cell Dev. Biol. 2020, 8, 566702.
