The diterpene lactone andrographolide, isolated from Andrographis paniculata (Burm.f.) Wall. ex Nees that showed shows a plethora of biological activities, including not only anti-cancer activity, but also anti-inflammatory, anti-viral, anti-bacterial, neuroprotective, hepatoprotective, hypoglycemic, and immunomodulatory properties. Andrographolide has been shown to act as an anti-tumor drug by affecting specific molecular targets that play a part in the development and progression of several cancer types including breast, lung, colon, renal, and cervical cancer, as well as leukemia and hepatocarcinoma.
- cancer
- mechanism of action
- apoptosis
- autophagy
1. Introduction
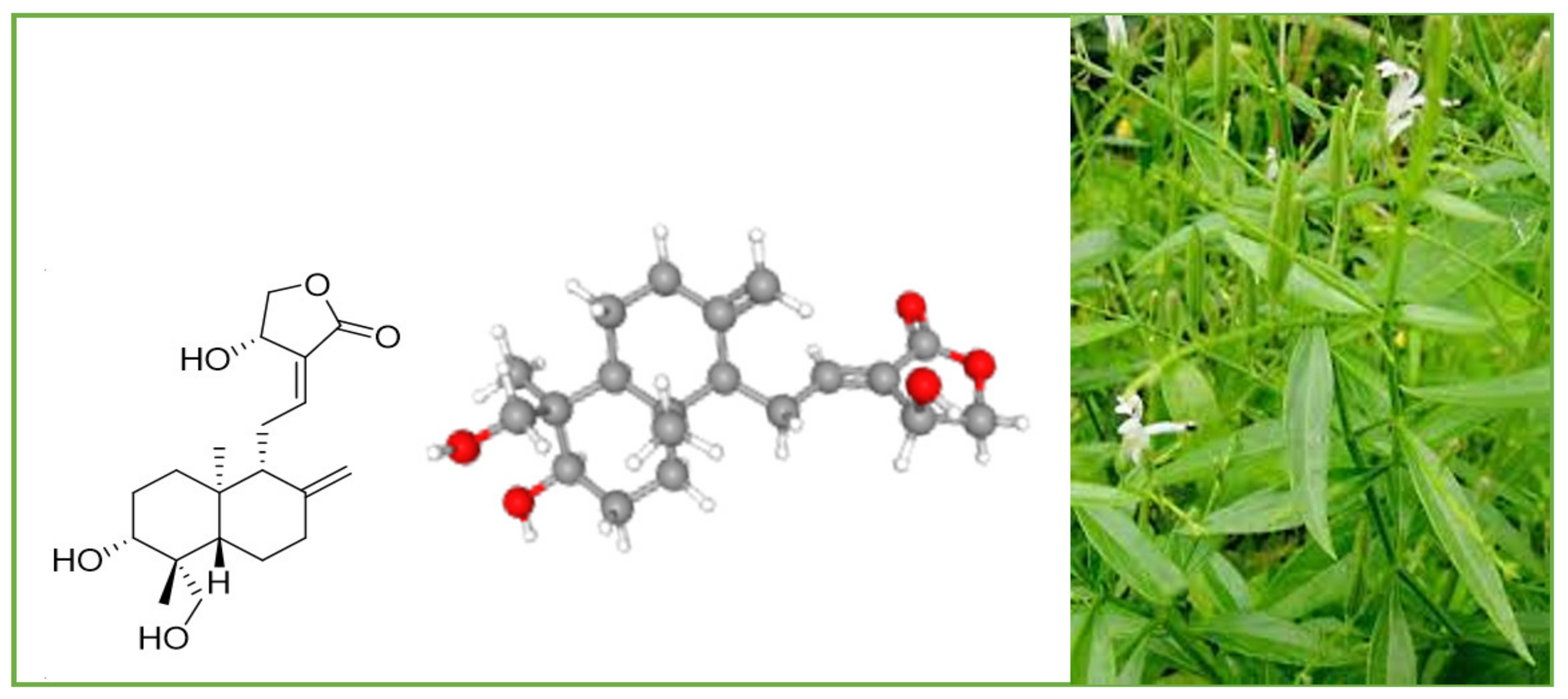
2. Occurrence and Chemistry of the Labdane Diterpenoid Andrographolide
4. Pharmacological Importance of Andrographolide and Its Derivatives
5. Anti-Cancer Properties and Mechanism of Action of Andrographolide and Its Derivatives
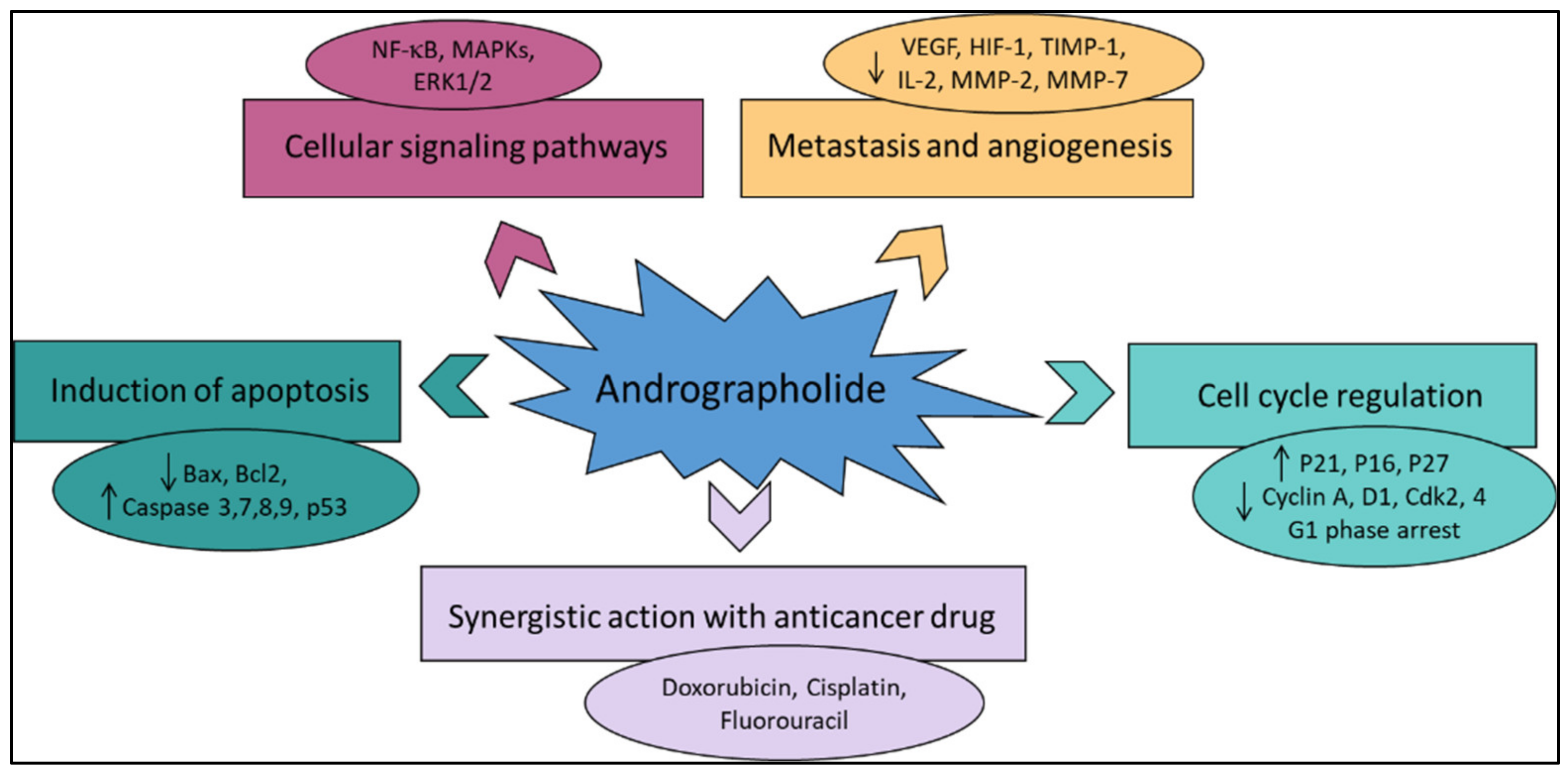
5.1. Induction of Apoptosis and Growth Inhibitory Activity
5.2. Inhibition of Tumor Angiogenesis
5.3. Anti-Proliferative Activity
5.4. Induction of Autophagy
5.5. Oxidative Stress and Antioxidant Properties of Andrographolide in Anti-Cancer Treatment
| Cancer Cell Line | Study Type | Main Effects | Ref. |
|---|---|---|---|
| Ovarian teratocarcinoma | In vitro | PA-1 cells—MTT test—IC50 3.7 µg/mL; induction of apoptosis | [31] |
| Lung cancer | In vitro | Suppression of autophagy and enhanced cisplatin-mediated apoptosis | [38] |
| Colon cancer | In vitro | HT-29 cells—induction of programed cell death and cell-cycle arrest through the increase of intracellular ROS level | [51] |
| Leukemia | In vitro | HL-60 cells—cell-cycle arrest and mitochondrial-mediated apoptosis | [57] |
| Prostate cancer | In vitro | PC-3 cells—caspase 8 and caspase 3 activation | [58] |
| Neuroblastoma | In vitro | Induction of p53- and caspase-independent cell death | [59] |
| Liver cancer | In vitro | HepG2 and Hep3B cells—sensitization of cancer cells to TRAIL-induced apoptosis via p53-mediated death receptor 4 upregulation | [61] |
| Cervical cancer | In vitro | HeLa cells—sensitization of cancer cells to TRAIL-induced apoptosis via p53-mediated death receptor 4 upregulation | [61] |
| Colorectal cancer | In vitro | HCT116 cells—sensitization of cancer cells to TRAIL-induced apoptosis via p53-mediated death receptor 4 upregulation | [61] |
| Prostate cancer | In vitro | LNCaP, DU145, and PC-3 cells—apoptotic cell death by reducing the mRNA and protein levels of IL6 | [62] |
| Colorectal carcinoma | In vitro | LoVo cells—induction of cell-cycle arrest and inhibition of cell proliferation | [63][64] |
| Brest cancer | In vitro | MCF-7 cells—G0/G1 phase arrest | [65] |
| Glioblastoma | U251 and U87 cells—arrest of the G2/M phase cell cycle phase | [67] | |
| Leukemia | K562 cells—arrest of the G2/M cell cycle phase | [17] | |
| Breast cancer | Arrest of the G2/M phase | [68][69] | |
| Prostate cancer | In vitro | LNCaP, C4-2b, and PC3 cells—inhibition of cell-cycle progression at the G2/M phase; decrease of tumor-specific angiogenesis | [71] |
| Osteosarcoma | In vitro | Inhibition of cell proliferation by arresting of the cell cycle at the G2/M phase | [72] |
| Osteosarcoma | In vivo | Female Balb/c-nu mice with HOS-Luc cells injected subcutaneously—induction of apoptosis via the ROS/JNK pathway | [72] |
| Colon cancer | In vitro | HT29 cell line—anti-invasive activity against colon cancer cells via inhibition of matrix metalloproteinase 2 (MMP2) | [73] |
| Cervical cancer | In vitro | CaSki cell line—MTT test—reduction of cell viability of 50% at 87.52 µM | [80] |
| Cervical cancer | In vitro | SiHa cell line—MTT test—reduction of cell viability of 50% at 85.59 µM | [80] |
| Cervical cancer | In vitro | C33A cell line—MTT test—reduction of cell viability of 50% at 96.05 µM | [80] |
| Breast cancer | In vitro | MCF-7 cell line—MTT test—IC50 32.90 µM | [81] |
| Breast cancer | In vitro | MDAMB-231 cell line—MTT test—IC50 37.56 µM | [81] |
| Monocytic leukemia | In vitro | THP-1 cell line—MTT test—IC50 31 µM | [82] |
| IgAkappa-producing multiple myeloma | In vitro | H929 cell line—MTT test—IC50 8 µM | [82] |
| Malignant melanoma | In vitro | A375 cell line—MTT test—IC50 12.07 µM | [83] |
| Malignant melanoma | In vitro | C8161 cell line—MTT test—IC50 10.92 µM | [83] |
| Osteosarcoma | In vitro | Suppression of phosphatidylinositol-3-kinase (PI3K)/Akt and the mammalian target of rapamycin (mTOR) signaling pathways; enhancement of the c-Jun N-terminal kinase (JNK) pathway | [90] |
| Mice | In vivo | Inhibition of PI3K/AKT-dependent NOX2 and iNOS expression | [97] |
| APP/PS1 transgenic mice | In vivo | Reduction of oxidative stress and protection of mitochondria | [99] |
| Male Wistar rats | In vivo | Reduction of the production of nitric oxide, carbonyl protein, and malondialdehyde, and enhancement of mitochondrial complex activities in the electron transport chain. Increased activity of CAT, SOD, glutathione peroxidase, glutathione reductase, and glutathione-S-transferase | [100] |
5.6. Enhanced Radio-Sensitivity
5.7. Different Signalling Pathways
This entry is adapted from the peer-reviewed paper 10.3390/plants12101969
References
- Hassanpour, S.H.; Dehghani, M. Review of cancer from perspective of molecular. J. Cancer Res. Pract. 2017, 4, 127–129.
- World Health Organization (WHO). Latest Global Cancer Data: Cancer Burden Rises to 19.3 Million New Cases and 10.0 Million Cancer Deaths in 2020. Available online: https://www.iarc.who.int/news-events/latest-global-cancer-data-cancer-burden-rises-to-19-3-million-new-cases-and-10–0-million-cancer-deaths-in-2020/ (accessed on 9 November 2022).
- Huang, M.; Lu, J.J.; Ding, J. Natural products in cancer therapy: Past, present and future. Nat. Prod. Bioprospect. 2021, 11, 5–13.
- Cragg, G.M.; Grothaus, P.G.; Newman, D.J. Impact of natural products on developing new anti-cancer agents. Chem. Rev. 2009, 109, 3012–3043.
- Saeed, M.E.M.; Boulos, J.C.; Elhaboub, G.; Rigano, D.; Saab, A.; Loizzo, M.R.; Hassan, L.E.A.; Sugimoto, Y.; Piacente, S.; Tundis, R.; et al. Cytotoxicity of cucurbitacin E from Citrullus colocynthis against multidrug-resistant cancer cells. Phytomedicine 2019, 62, 152945.
- Tundis, R.; Bonesi, M.; Deguin, B.; Loizzo, M.R.; Menichini, F.; Conforti, F.; Tillequin, F.; Menichini, F. Cytotoxic activity and inhibitory effect on Nitric Oxide production of triterpene saponins from the roots of Physospermum verticillatum (Waldst & Kit) (Apiaceae). Bioorg. Med. Chem. 2009, 17, 4542–4547.
- Loizzo, M.R.; Tundis, R.; Statti, G.A.; Menichini, F. Jacaranone: A cytotoxic constituent from Senecio ambiguus subsp. ambiguus (Biv.) DC. against renal adenocarcinoma ACHN and prostate carcinoma LNCaP cells. Arch. Pharm. Res. 2007, 30, 701–707.
- Nobili, S.; Lippi, D.; Witort, E.; Donnini, M.; Bausi, L.; Mini, E.; Capaccioli, S. Natural compounds for cancer treatment and prevention. Pharmacol. Res. 2009, 59, 365–378.
- Acquaviva, R.; Malfa, G.A.; Loizzo, M.R.; Xiao, J.; Bianchi, S.; Tundis, R. Advances on natural abietane, labdane and clerodane diterpenes as anti-cancer agents: Sources and mechanisms of action. Molecules 2022, 27, 4791.
- Mussard, E.; Cesaro, A.; Lespessailles, E.; Legrain, B.; Berteina-Raboin, S.; Toumi, H. Andrographolide, a natural antioxidant: An update. Antioxidants 2019, 8, 571.
- Zhang, X.F.; Ding, M.J.; Cheng, C.; Zhang, Y.; Xiang, S.Y.; Lu, J.; Liu, Z.B. Andrographolide attenuates oxidative stress injury in cigarette smoke extract exposed macrophages through inhibiting SIRT1/ERK signaling. Int. Immunopharmacol. 2020, 81, 106230.
- Ding, Y.; Chen, L.; Wu, W.; Yang, J.; Yang, Z.; Liu, S. Andrographolide inhibits influenza A virus-induced inflammation in a murine model through NF-kappaB and JAK-STAT signalling pathway. Microbes Infect. 2017, 19, 605–615.
- Zhang, B.; Yang, B.; Du, L.; Guo, Y. Nitric oxide donor andrographolide enhances humoral and cell-mediated immune responses. Cell. Mol. Biol. 2020, 66, 176.
- Su, H.; Mo, J.; Ni, J.; Ke, H.; Bao, T.; Xie, J.; Xu, Y.; Xie, L.; Chen, W. Andrographolide exerts antihyperglycemic effect through strengthening intestinal barrier function and increasing microbial composition of Akkermansia muciniphila. Oxid. Med. Cell. Longev. 2020, 2020, 6538930.
- Gupta, S.; Mishra, K.P.; Ganju, L. Broad-spectrum antiviral properties of andrographolide. Archiv. Virol. 2017, 162, 611–623.
- Panraksa, P.; Ramphan, S.; Khongwichit, S.; Smith, D.R. Activity of andrographolide against dengue virus. Antivir. Res. 2017, 139, 69–78.
- Shi, T.H.; Huang, Y.L.; Chen, C.C.; Pi, W.C.; Hsu, Y.L.; Lo, L.C.; Chen, W.-Y.; Fu, S.-L.; Lin, C.H. Andrographolide and its fluorescent derivative inhibit the main proteases of 2019-nCoV and SARS-CoV through covalent linkage. Biochem. Biophys. Res. Commun. 2020, 533, 467–473.
- Srivastava, N.; Garg, P.; Srivastava, P.; Seth, P.K. A molecular dynamics simulation study of the ACE2 receptor with screened natural inhibitors to identify novel drug candidate against COVID-19. PeerJ 2021, 9, e11171.
- Das, S.; Mishra, K.P.; Ganju, L.; Singh, S.B. Andrographolide-A promising therapeutic agent, negatively regulates glial cell derived neurodegeneration of prefrontal cortex, hippocampus and working memory impairment. J. Neuroimmunol. 2017, 313, 161–175.
- Chen, S.; Luo, Z.; Chen, X. Andrographolide mitigates cartilage damage via miR-27-3p-modulated matrix metalloproteinase13 repression. J. Gene Med. 2020, 22, e3187.
- Zhang, H.; Li, S.; Si, Y.; Xu, H. Andrographolide and its derivatives: Current achievements and future perspectives. Eur. J. Med. Chem. 2021, 224, 113710.
- Jiang, M.; Sheng, F.; Zhang, Z.; Ma, X.; Gao, T.; Fu, C.; Li, P. Andrographis paniculata (Burm.f.) Nees and its major constituent andrographolide as potential antiviral agents. J. Ethnopharmacol. 2021, 272, 113954.
- Tan, W.S.D.; Liao, W.; Zhou, S.; Wong, W.F. Is there a future for andrographolide to be an anti-inflammatory drug? Deciphering its major mechanisms of action. Biochem. Pharmacol. 2017, 139, 71–81.
- Uthirapandi, V.; Subramanian, S.R.; Ponnerulan, B.; Saminathan, E.; Narayanan, V.; Durairaj, K. Andrographolide production and enhanced antioxidant activity in Andrographis paniculata (Burm f.) Nees. promoted by seaweed liquid extracts. Braz. J. Bot. 2021, 44, 317–330.
- Lin, H.C.; Lii, C.K.; Chen, H.C.; Lin, A.H.; Yang, Y.C.; Chen, H.W. Andrographolide inhibits oxidized LDL-induced cholesterol accumulation and foam cell formation in macrophages. Am. J. Chin. Med. 2018, 46, 87–106.
- Hossain, S.; Urbi, Z.; Karuniawati, H.; Mohiuddin, R.B.; Moh Qrimida, A.; Allzrag, A.M.M.; Ming, L.C.; Pagano, E.; Capasso, R. Andrographis paniculata (Burm. F.) wall. Ex Nees: An updated review of phytochemistry, antimicrobial-pharmacology, and clinical safety and efficacy. Life 2021, 11, 348.
- Kumar, S.; Singh, B.; Bajpai, V. Andrographis paniculata (Burm.f.) Nees: Traditional uses, phytochemistry, pharmacological properties and quality control/quality assurance. J. Ethnopharmacol. 2021, 275, 114054.
- Gorter, M. The bitter constituent of Andrographis paniculata Nees. Rec. Trav. Chim. 1911, 30, 151–160.
- Kumar, G.; Singh, D.; Tali, J.A.; Dheer, D.; Shankar, R. Andrographolide: Chemical modification and its effect on biological activities. Bioorg. Chem. 2020, 95, 103511.
- Rafi, M.; Karomah, A.H.; Heryanto, R.; Septaningsih, D.A.; Kusuma, W.A.; Amran, M.B.; Rohman, A.; Prajogo, B. Metabolite profiling of Andrographis paniculata leaves and stem extract using UHPLC-Orbitrap-MS/MS. Nat. Prod. Res. 2022, 36, 625–629.
- Bhat, M.A.; Murthy, H.N. Isolation of andrographolide from Andrographis lineata Wall. ex Nees var. lawii C.B. clarke and its anticancer activity against human ovarian teratocarcinoma. Pharmacog. J. 2021, 13, 660–668.
- Zeng, B.; Wei, A.; Zhou, Q.; Yuan, M.; Lei, K.; Liu, Y.; Song, J.; Guo, L.; Ye, Q. Andrographolide: A review of its pharmacology, pharmacokinetics, toxicity and clinical trials and pharmaceutical researches. Phytother. Res. 2022, 36, 336–364.
- Jayakumar, T.; Hsieh, C.-Y.; Lee, J.-J.; Sheu, J.-R. Experimental and clinical pharmacology of Andrographis paniculata and its major bioactive phytoconstituent andrographolide. Evid. Based Complement Alternat. Med. 2013, 2013, 846740.
- Islam, M.T.; Ali, E.S.; Uddin, S.J.; Islam, M.A.; Shaw, S.; Khan, I.N.; Saravi, S.S.S.; Ahmad, S.; Rehman, S.; Gupta, V.K.; et al. Andrographolide, a diterpene lactone from Andrographis paniculata and its therapeutic promises in cancer. Cancer Lett. 2018, 420, 129–145.
- Kandanur, S.G.S.; Tamang, N.; Golakoti, N.R.; Nanduri, S. Andrographolide: A natural product template for the generation of structurally and biologically diverse diterpenes. Eur. J. Med. Chem. 2019, 176, 513–533.
- Kim, T.G.; Hwi, K.K.; Hung, C.S. Morphological and biochemical changes of andrographolide-induced cell death in human prostatic adenocarcinoma PC-3 cells. In Vivo 2005, 19, 551–557.
- Yang, S.; Evens, A.M.; Prachand, S.; Singh, A.T.K.; Bhalla, S.; David, K.; Gordon, L.I. Mitochondrial-mediated apoptosis in lymphoma cells by the diterpenoid lactone andrographolide, the active component of Andrographis paniculata. Clin. Cancer Res. 2010, 16, 4755–4768.
- Yuwen, D.; Mi, S.; Ma, Y.; Guo, W.; Xu, Q.; Shen, Y.; Shu, Y. Andrographolide enhances cisplatin-mediated anticancer effects in lung cancer cells through blockade of autophagy. Anti-Cancer Drugs 2017, 28, 967–976.
- Devendar, P.; Nayak, V.L.; Yadav, D.K.; Kumar, A.N.; Kumar, J.K.; Srinivas, K.S.; Sridhar, B.; Khan, F.; Sastry, K.P.; Ramakrishna, S. Synthesis and evaluation of anticancer activity of novel andrographolide derivatives. MedChemComm 2015, 6, 898–904.
- Lim, J.C.W.; Jeyaraj, E.J.; Sagineedu, S.R.; Wong, W.S.F.; Stanslas, J. SRS06, a new semisynthetic andrographolide derivative with improved anticancer potency and selectivity, inhibits nuclear factor-κB nuclear binding in the A549 non-small cell lung cancer cell line. Pharmacology 2015, 95, 70–77.
- Peng, Y.; Li, J.; Sun, Y.; Chan, J.Y.-W.; Sheng, D.; Wang, K.; Wei, P.; Ouyang, P.; Wang, D.; Lee, S.M.Y.; et al. SAR studies of 3, 14, 19-derivatives of andrographolide on anti-proliferative activity to cancer cells and toxicity to zebrafish: An in vitro and in vivo study. RSC Adv. 2015, 5, 22510–22526.
- Wanandi, S.I.; Limanto, A.; Yunita, E.; Syahrani, R.A.; Louisa, M.; Wibowo, A.E.; Arumsari, S. In silico and in vitro studies on the anti-cancer activity of andrographolide targeting survivin in human breast cancer stem cells. PLoS ONE 2020, 15, e0240020.
- Luo, W.; Jia, L.; Zhang, J.W.; Wang, D.J.; Ren, Q.; Zhang, W. Andrographolide against lung cancer-new pharmacological insights based on high-throughput metabolomics analysis combined with network pharmacology. Front. Pharmacol. 2021, 12, 596652.
- Vukmirovic, D.; Vo, N.T.K.; Seymour, C.; Rollo, D.; Mothersill, C. Influence of common dietary supplements (curcumin, andrographolide, and d-limonene) on the radiobiological responses of p53-competent colonic cancer epithelial cells. Int. J. Radiation Biol. 2021, 97, 341–347.
- Banerjee, V.; Sharda, N.; Huse, J.; Singh, D.; Sokolov, D.; Czinn, S.J.; Blanchard, T.G.; Banerjee, A. Synergistic potential of dual andrographolide and melatonin targeting of metastatic colon cancer cells: Using the Chou-Talalay combination index method. Eur. J. Pharmacol. 2021, 897, 173919.
- Bi, R.; Deng, Y.Y.; Tang, C.; Xuan, L.; Xu, B.; Du, Y.J.; Wang, C.; Wei, W. Andrographolide sensitizes human renal carcinoma cells to TRAILinduced apoptosis through upregulation of death receptor 4. Oncol. Rep. 2020, 44, 1939–1948.
- Pasha, A.; Kumbhakar, D.V.; Doneti, R.; Kumar, K.; Dharmapuri, G.; Poleboyina, P.K.; Heena, S.K.; Basavaraju, P.; Pasumarthi, D.; Annapurna, S.D.; et al. Inhibition of inducible nitric oxide synthase (iNOS) by andrographolide and in vitro evaluation of its antiproliferative and proapoptotic effects on cervical cancer. Oxid. Med. Cell. Longev. 2021, 2021, 6692628.
- Shi, L.; Zhang, G.Q.; Zheng, Z.Y.; Lu, B.; Ji, L.L. Andrographolide reduced VEGFA expression in hepatoma cancer cells by inactivating HIF-1 alpha: The involvement of JNK and MTA1/HDCA. Chem. Biol. Interact. 2017, 273, 228–236.
- Banerjee, M.; Chattopadhyay, S.; Choudhuri, T.; Bera, R.; Kumar, S.; Chakraborty, B.; Mukherjee, S.K. Cytotoxicity and cell cycle arrest induced by andrographolide lead to programmed cell death of MDA-MB-231 breast cancer cell line. J. Biomed. Sci. 2016, 23, 40.
- Dai, L.; Wang, G.; Pan, W. Andrographolide inhibits proliferation and metastasis of SGC7901 gastric cancer cells. Biomed. Res. Int. 2017, 2017, 6242103.
- Khan, I.; Khan, F.; Farooqui, A.; Ansari, I.A. Andrographolide exhibits anticancer potential against human colon cancer cells by inducing cell cycle arrest and programmed cell death via augmentation of intracellular reactive oxygen species level. Nutr. Cancer 2018, 70, 787–803.
- Chen, W.; Feng, L.; Nie, H.; Zheng, X. Andrographolide induces autophagic cell death in human liver cancer cells through cyclophilin D-mediated mitochondrial permeability transition pore. Carcinogenesis 2012, 33, 2190–2198.
- Das, S.; Rahaman, A.; Nath, R.; Talukdar, A.D.; Nath, D.; Bhattacharjee, S.; Mandal, D.P.; Choudhury, M.D.; Das, D.; Das, G.; et al. Effect of acetone fraction of Ottelia alismoides on the G2/M cell cycle arrest and apoptosis in the human carcinoma cell lines. J. Ethnopharmacol. 2023, 300, 115729.
- Xie, J.; Peng, L.-J.; Yang, M.-R.; Jiang, W.-W.; Mao, J.-Y.; Shi, C.-Y.; Tian, Y.; Sheng, J. Alkaloid extract of Moringa oleifera lam. Exerts antitumor activity in human non-small-cell lung cancer via modulation of the JAK2/STAT3 signaling pathway. Evid. Based Complement. Altern. Med. 2021, 2021, 5591687.
- Saqr, A.A.; Khafagy, E.-S.; Aldawsari, M.F.; Almansour, K.; Abu Lila, A.S. Screening of apoptosis pathway-mediated anti-proliferative activity of the phytochemical compound furanodienone against human non-small lung cancer A-549 cells. Life 2022, 12, 114.
- Sheeja, K.; Kuttan, G. Activation of cytotoxic T lymphocyte responses and attenuation of tumor growth in vivo by Andrographis paniculata extract and andrographolide. Immunopharmacol. Immunotoxicol. 2007, 29, 81–93.
- Cheung, H.-Y.; Cheung, S.-H.; Li, J.; Cheung, C.-S.; Lai, W.-P.; Fong, W.-F.; Leung, F.-M. Andrographolide isolated from Andrographis paniculata induces cell cycle arrest and mitochondrial-mediated apoptosis in human leukemic HL-60 cells. Planta Med. 2005, 71, 1106–1111.
- Kim, Y.S.; Milner, J.A. Targets for indole-3-carbinol in cancer prevention. J. Nutr. Biochem. 2005, 16, 65–73.
- Sukumari-Ramesh, S.; Bentley, J.N.; Laird, M.D.; Singh, N.; Vender, J.R.; Dhandapani, K.M. Dietary phytochemicals induce p53-and caspase-independent cell death in human neuroblastoma cells. Int. J. Develop. Neurosci. 2011, 29, 701–710.
- Wu, Y.; Zhou, B.P. Inflammation: A driving force speeds cancer metastasis. Cell Cycle 2009, 8, 3267–3273.
- Zhou, J.; Lu, G.-D.; Ong, C.-S.; Ong, C.-N.; Shen, H.-M. Andrographolide sensitizes cancer cells to TRAIL-induced apoptosis via p53-mediated death receptor 4 up-regulation. Mol. Cancer Ther. 2008, 7, 2170–2180.
- Chun, J.Y.; Tummala, R.; Nadiminty, N.; Lou, W.; Liu, C.; Yang, J.; Evans, C.P.; Zhou, Q.; Gao, A.C. Andrographolide, an herbal medicine, inhibits interleukin-6 expression and suppresses prostate cancer cell growth. Genes Cancer 2010, 1, 868–876.
- Shi, M.D.; Lin, H.H.; Lee, Y.C.; Chao, J.K.; Lin, R.A.; Chen, J.H. Inhibition of cell-cycle progression in human colorectal carcinoma Lovo cells by andrographolide. Chem. Biol. Interact. 2008, 174, 201–210.
- Satyanarayana, C.; Deevi, D.S.; Rajagopalan, R.; Srinivas, N.; Rajagopal, S. DRF 3188 a novel semi-synthetic analog of an-drographolide: Cellular response to MCF 7 breast cancer cells. BMC Cancer 2004, 4, 26–33.
- Rajagopal, S.; Kumar, R.A.; Deevi, D.S.; Satyanarayana, C.; Rajagopalan, R. Andrographolide, a potential cancer therapeutic agent isolated from Andrographis paniculate. J. Exp. Therapeut. Oncol. 2003, 3, 147–158.
- Yan, J.; Chen, Y.; He, C.; Yang, Z.-Z.; Lü, C.; Chen, X.-S. Andrographolide induces cell cycle arrest and apoptosis in human rheumatoid arthritis fibroblast-like synoviocytes. Cell Biol. Toxicol. 2012, 28, 47–56.
- Roy, P.; Das, S.; Mondal, A.; Chatterji, U.; Mukherjee, A. Nanoparticle engineering enhances anticancer efficacy of andrographolide in MCF-7 cells and mice bearing EAC. Curr. Pharm. Biotechnol. 2012, 13, 2669–2681.
- Kumar, S.; Patil, H.S.; Sharma, P.; Kumar, D.; Dasari, S.; Puranik, V.G.; Thulasiram, H.V.; Kundu, G.C. Andrographolide inhibits osteopontin expression and breast tumor growth through down regulation of PI3 kinase/Akt signaling pathway. Curr. Mol. Med. 2012, 12, 952–966.
- Zhang, Q.-Q.; Zhou, D.-L.; Ding, Y.; Liu, H.-Y.; Lei, Y.; Fang, H.-Y.; Yang, Y. Andrographolide inhibits melanoma tumor growth by inactivating the TLR4/NF-κB signaling pathway. Melanoma Res. 2014, 24, 545–555.
- Wong, C.C.; Lim, S.H.; Sagineedu, S.R.; Lajis, N.H.; Stanslas, J. SRJ09, a promising anticancer drug lead: Elucidation of mechanisms of antiproliferative and apoptogenic effects and assessment of in vivo antitumor efficacy. Pharmacol. Res. 2016, 107, 66–78.
- Sheeja, K.; Guruvayoorappan, C.; Kuttan, G. Antiangiogenic activity of Andrographis paniculata extract and andrographolide. Int. Immunopharmacol. 2007, 7, 211–221.
- Wang, S.; Li, H.; Chen, S.; Wang, Z.; Yao, Y.; Chen, T.; Ye, Z.; Lin, P. Andrographolide induces apoptosis in human osteosarcoma cells via the ROS/JNK pathway. Int. J. Oncol. 2020, 56, 1417–1428.
- Chao, H.-P.; Kuo, C.-D.; Chiu, J.-H.; Fu, S.-L. Andrographolide exhibits anti-invasive activity against colon cancer cells via inhibition of MMP2 activity. Planta Med. 2010, 76, 1827–1833.
- Lin, H.-H.; Tsai, C.-W.; Chou, F.-P.; Wang, C.-J.; Hsuan, S.-W.; Wang, C.-K.; Chen, J.-H. Andrographolide down-regulates hypoxia-inducible factor-1α in human non-small cell lung cancer A549 cells. Toxicol. Appl. Pharmacol. 2011, 250, 336–345.
- Xiao, X.-W.; Fu, H.-Z.; Luo, Y.-H.; Wei, X.-Y. Potential anti-angiogenic sulfates of andrographolide. J. Asian Nat. Prod. Res. 2013, 15, 809–818.
- Kajal, K.; Panda, A.K.; Bhat, J.; Chakraborty, D.; Bose, S.; Bhattacharjee, P.; Sarkar, T.; Chatterjee, S.; Kar, S.K.; Sa, G. Andrographolide binds to ATP-binding pocket of VEGFR2 to impede VEGFA-mediated tumor-angiogenesis. Sci. Rep. 2019, 9, 4073.
- Dai, J.; Lin, Y.; Duan, Y.; Li, Z.; Zhou, D.; Chen, W.; Wang, L.; Zhang, Q.Q. Andrographolide Inhibits Angiogenesis by Inhibiting the Mir-21-5p/TIMP3 Signaling Pathway. Int. J. Biol. Sci. 2017, 13, 660–668.
- Yadav, R.V.; Sadhukhan, S.; Saha, M.L.; Ghosh, S.; Das, M. Exploring the mechanism of andrographolide in the treatment of gastric cancer through network pharmacology and molecular docking. Sci. Rep. 2022, 12, 18413.
- Li, J.; Li, F.; Tang, F.; Zhang, J.; Li, R.; Sheng, D.; Lee, S.M.Y.; Zhou, G.C.; Leung, G.P.H. AGS-30, an andrographolide derivative, suppresses tumor angiogenesis and growth in vitro and in vivo. Biochem. Pharmacol. 2020, 171, 113694.
- Udomwan, P.; Pientong, C.; Tongchai, P.; Burassakarn, A.; Sunthamala, N.; Roytrakul, S.; Suebsasana, S.; Ekalaksananan, T. Proteomics analysis of andrographolide-induced apoptosis via the regulation of tumor suppressor p53 proteolysis in cervical cancer-derived human papillomavirus 16-positive cell lines. Int. J. Mol. Sci. 2021, 22, 6806.
- Tohkayomatee, R.; Reabroi, S.; Tungmunnithum, D.; Parichatikanond, W.; Pinthong, D. Andrographolide exhibits anticancer activity against breast cancer cells (MCF-7 and MDA-MB-231 Cells) through suppressing cell proliferation and inducing cell apoptosis via inactivation of ER-α receptor and PI3K/AKT/mTOR signaling. Molecules 2022, 27, 3544.
- Doi, H.; Matsui, T.; Dijkstra, J.M.; Ogasawara, A.; Higashimoto, Y.; Imamura, S.; Ohye, T.; Takematsu, H.; Katsuda, I.; Akiyama, H. Andrographolide, isolated from Andrographis paniculata, induces apoptosis in monocytic leukemia and multiple myeloma cells via augmentation of reactive oxygen species production. F1000Research 2022, 10, 542.
- Liu, G.; Chu, H. Andrographolide inhibits proliferation and induces cell cycle arrest and apoptosis in human melanoma cells. Oncol. Lett. 2018, 15, 5301–5305.
- He, C.; Klionsky, D.J. Regulation mechanisms and signaling pathways of autophagy. Ann. Rev. Genet. 2009, 43, 67–93.
- Codogno, P.; Meijer, A.J. Autophagy and signaling: Their role in cell survival and cell death. Cell Death Differ. 2005, 12, 1509–1518.
- Azad, M.B.; Chen, Y.; Gibson, S.B. Regulation of autophagy by reactive oxygen species (ROS): Implications for cancer progression and treatment. Antiox. Redox. Signal 2009, 11, 777–790.
- Corcelle, E.; Djerbi, N.; Mari, M.; Nebout, M.; Fiorini, C.; Fenichel, P.; Hofman, P.; Poujeol, P.; Mograbi, B. Control of the autophagy maturation step by the MAPK ERK and p38: Lessons from environmental carcinogens. Autophagy 2007, 3, 57–59.
- He, Z.J.; Zhu, F.Y.; Li, S.S.; Zhong, L.; Tan, H.Y.; Wang, K. Inhibiting ROS-NF-kappaB-dependent autophagy enhanced brazilin-induced apoptosis in head and neck squamous cell carcinoma. Food Chem. Toxicol. 2017, 101, 55–66.
- Heras-Sandoval, D.; Perez-Rojas, J.M.; Hernandez-Damian, J.; Pedraza-Chaverri, J. The role of PI3K/AKT/mTOR pathway in the modulation of autophagy and the clearance of protein aggregates in neurodegeneration. Cell Signal 2014, 26, 2694–2701.
- Liu, Y.; Zhang, Y.; Zou, J.; Yan, L.; Yu, X.; Lu, P.; Wu, X.; Li, Q.; Gu, R.; Zhu, D. Andrographolide induces autophagic cell death and inhibits invasion and metastasis of human osteosarcoma cells in an autophagy-dependent manner. Cell Physiol. BioChem 2017, 44, 1396–1410.
- Krithika, R.; Verma, R.J.; Shrivastav, P.S. Antioxidative and cytoprotective effects of andrographolide against CCl4-induced hepatotoxicity in HepG2 cells. Hum. Exp. Toxicol. 2013, 32, 530–543.
- Shen, Y.-C.; Chen, C.-F.; Chiou, W.-F. Andrographolide prevents oxygen radical production by human neutrophils: Possible mechanism(s) involved in its anti-inflammatory effect. Br. J. Pharmacol. 2002, 135, 399–406.
- Peng, S.; Gao, J.; Liu, W.; Jiang, C.; Yang, X.; Sun, Y.; Guo, W.; Xu, Q. Andrographolide ameliorates OVA-induced lung injury in mice by suppressing ROS-mediated NF- B signaling and NLRP3 inflammasome activation. Oncotarget 2016, 7, 80262–80274.
- Zhan, J.Y.-X.; Wang, X.-F.; Liu, Y.-H.; Zhang, Z.-B.; Wang, L.; Chen, J.-N.; Huang, S.; Zeng, H.-F.; Lai, X.-P. Andrographolide sodium bisulfate prevents UV-induced skin photoaging through inhibiting oxidative stress and inflammation. Mediat. Inflamm. 2016, 2016, 3271451.
- Thangathirupathi, A.; Ali, N.; Natarajan, P.; Ramesh Kumar, D. Molecular docking studies of andrographolide with xanthine oxidase. Asian J. Pharm. Clin. Res. 2013, 6, 295–297.
- Rahmi, E.P.; Kumolosasi, E.; Jalil, J.; Buang, F.; Jamal, J.A. Extracts of Andrographis paniculata (Burm.f.) nees leaves exert anti-gout effects by lowering uric acid levels and reducing monosodium urate crystalinduced inflammation. Front. Pharmacol. 2022, 12, 787125.
- Chern, C.-M.; Liou, K.-T.; Wang, Y.-H.; Liao, J.-F.; Yen, J.-C.; Shen, Y.-C. Andrographolide inhibits PI3K/AKT-dependent NOX2 and iNOS expression protecting mice against hypoxia/ischemia-induced oxidative brain injury. Planta Med. 2011, 77, 1669–1679.
- Liang, E.; Liu, X.; Du, Z.; Yang, R.; Zhao, Y. Andrographolide ameliorates diabetic cardiomyopathy in mice by blockage of oxidative damage and NF-B-mediated inflammation. Oxid. Med. Cell Longev. 2018, 2018, 9086747.
- Geng, J.; Liu, W.; Xiong, Y.; Ding, H.; Jiang, C.; Yang, X.; Li, X.; Elgehama, A.; Sun, Y.; Xu, Q.; et al. Andrographolide sulfonate improves Alzheimer-associated phenotypes and mitochondrial dysfunction in APP/PS1 transgenic mice. Biomed. Pharmacother. 2018, 97, 1032–1039.
- Das, S.; Gautam, N.; Dey, S.K.; Maiti, T.; Roy, S. Oxidative stress in the brain of nicotine-induced toxicity: Protective role of Andrographis paniculata Nees and vitamin E. Appl. Physiol. Nutr. Metab. 2009, 34, 124–135.
- Wong, D.P.W.; Ng, M.Y.; Leung, J.Y.; Boh, B.K.; Lim, E.C.; Tan, S.H.; Lim, S.; Seah, W.H.; Hu, C.Z.; Ho, B.C.; et al. Regulation of the NRF2 transcription factor by andrographolide and organic extracts from plant endophytes. PLoS ONE 2018, 13, e0204853.
- Fu, K.; Chen, H.; Wang, Z.; Cao, R. Andrographolide attenuates inflammatory response induced by LPS via activating Nrf2 signaling pathway in bovine endometrial epithelial cells. Res. Vet. Sci. 2021, 134, 36–41.
- Zhang, C.; Qiu, X. Andrographolide radiosensitizes human ovarian cancer SKOV3 xenografts due to an enhanced apoptosis and autophagy. Tumour Biol. 2015, 36, 8359–8365.
- Chen, D.; Song, Y.; Lu, Y.; Xue, X. Synthesis and in vitro cytotoxicity of andrographolide-19-oic acid analogues as anti-cancer agents. Bioorg. Med. Chem. Lett. 2013, 23, 3166–3169.
- Zhu, Y.-Y.; Yu, G.; Zhang, Y.; Xu, Z.; Wang, Y.-Q.; Yan, G.-R.; He, Q.-Y. A novel andrographolide derivative AL-1 exerts its cytotoxicity on K562 cells through a ROS-dependent mechanism. Proteomics 2013, 13, 169–178.
- Lu, H.; Zhang, X.-Y.; Wang, Y.-Q.; Zheng, X.-L.; Yin, Z.; Xing, W.-M.; Zhang, Q. Andrographolide sodium bisulfate-induced apoptosis and autophagy in human proximal tubular endothelial cells is a ROS-mediated pathway. Environm. Toxicol. Pharmacol. 2014, 37, 718–728.
- Wang, Z.M.; Kang, Y.H.; Yang, X.; Wang, J.F.; Zhang, Q.; Yang, B.X.; Zhao, K.L.; Xu, L.P.; Yang, L.P.; Ma, J.X.; et al. Andrographolide radiosensitizes human esophageal cancer cell line ECA109 to radiation in vitro. Dis. Esophagus 2016, 29, 54–61.
- Hung, S.K.; Hung, L.C.; Kuo, C.D.; Lee, K.Y.; Lee, M.S.; Lin, H.Y.; Chen, Y.J.; Fu, S.L. Andrographolide sensitizes Ras-transformed cells to radiation in vitro and in vivo. J. Radiat. Oncol. Biol. Phys. 2010, 77, 1232–1239.
- Yang, P.-Y.; Hsieh, P.-L.; Wang, T.H.; Yu, C.-C.; Lu, M.-Y.; Liao, Y.-W.; Lee, T.-H.; Peng, C.-Y. Andrographolide impedes cancer stemness and enhances radio-sensitivity in oral carcinomas via miR-218 activation. Oncotarget 2017, 8, 4196–4207.
- Li, X.; Tian, R.; Liu, L.; Wang, L.; He, D.; Cao, K.; Ma, J.K.; Huang, C. Andrographolide enhanced radiosensitivity by downregulating glycolysis via the inhibition of the PI3K-Akt-mTOR signaling pathway in HCT116 colorectal cancer cells. J. Int. Med. Res. 2020, 48, 300060520946169.
- Zhang, D.; Huang, Y.; Qiao, Y.; Xia, C.; Luo, Y.; Chen, Z. Antibacterial activity of inclusion complexes of andrographolide and 14-acetylandrographolide by hydroxypropyl-β-cyclodextrin. J. Nanjing Agric. Univ. 2016, 39, 318–324.
- Yuan, M.; Meng, W.; Liao, W.; Lian, S. Andrographolide Antagonizes TNF-α-Induced IL-8 via Inhibition of NADPH Oxidase/ROS/NF-κB and Src/MAPKs/AP-1 Axis in Human Colorectal Cancer HCT116 Cells. J. Agric. Food Chem. 2018, 66, 5139–5148.
- Zhou, J.; Ong, C.-N.; Hur, G.-M.; Shen, H.-M. Inhibition of the JAK-STAT3 pathway by andrographolide enhances chemosensitivity of cancer cells to doxorubicin. Biochem. Pharmacol. 2010, 79, 1242–1250.
- Bao, G.-Q.; Shen, B.-Y.; Pan, C.-P.; Zhang, Y.-J.; Shi, M.-M.; Peng, C.-H. Andrographolide causes apoptosis via inactivation of STAT3 and Akt and potentiates antitumor activity of gemcitabine in pancreatic cancer. Toxicol. Lett. 2013, 222, 23–35.
