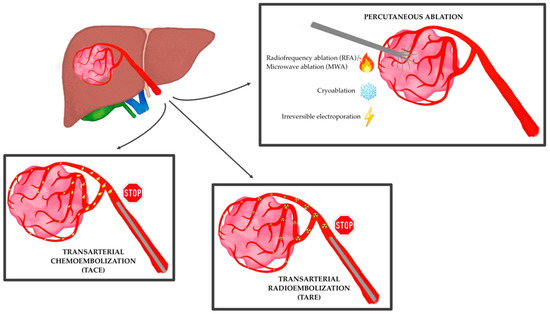
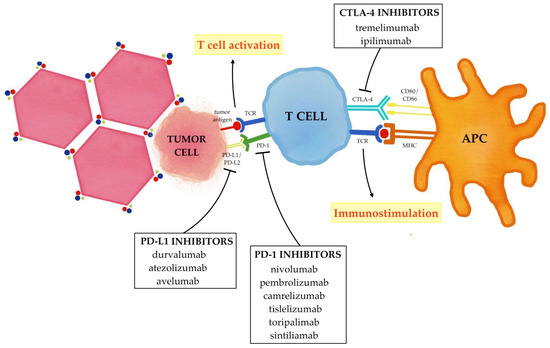
3. The Immune Modulation Effect of Locoregional Therapies
In several animal and human studies, locoregional treatments have been shown to induce immune responses in HCC patients, positively altering their tumor microenvironment [
84,
85]. The release of tumor antigens due to cell death and subsequent recruitment and activation of APCs and effector immune cells are the main processes responsible for the changes in anti-tumor immune responses after locoregional treatments [
84].
Immunogenic cell death involves the translocation of calreticulin on the cell surface, the secretion of ATP, and the release of the non-histone chromatin protein high-mobility group box 1 (HMGB1) and other immunostimulatory molecules that collectively facilitate the recruitment and activation of APCs into the tumor microenvironment, the engulfment of tumor antigens from dying tumor cells and, finally, the optimal antigen presentation to T cells [
85,
86,
87,
88,
89,
90]. Locoregional treatments can induce both apoptosis and necrosis of tumor cells. Necrosis is a form of cell death characterized by loss of plasma membrane integrity, culminating in the escape of cell contents into the extracellular space, including tumor specific antigens, thus is known to be immunogenic; conversely, apoptosis is a programmed cell death in which the plasma membrane is not disrupted and cellular contents are packaged and then released into apoptotic bodies, thus it is regarded as immunologically “silent” [
91,
92]; nevertheless, previous reports have also implicated that certain types of apoptosis could be immunogenic and therefore favor the immune response against the tumor [
93,
94].
A plethora of cytokines, chemokines, and inflammatory/cell stress molecular markers have been described following the execution of the majority of locoregional treatments for HCC, supporting the immune modulation effect of these techniques. The effect of MWA as a single therapy was one of the first to be investigated, demonstrating the activation of Tregs, CD4+ and CD8+ T cell and NK cells, as well as the release of IL-12 [
95,
96]. The evidence that ablative therapy can cause tumor-specific immune responses was observed also in patients who underwent RFA, which can increase the number of tumor-associated antigen-derived peptides in peripheral blood [
97], induce APCs activation and proliferation [
98] and stimulate the secretion of Th1 cytokines (such as IL-2, TNF-α and IFN-γ) that promote CD8+ T activity [
99]. Similarly, also TACE was reported to promote immunogenic cell death, as testified to by the increased serum levels of immunogenic cell death biomarkers following the procedure [
100]; moreover, TACE can also promote Th17 and CD8+ activation and reduce the number of Tregs [
101,
102]. More recently, infiltration of CD8+ T cells and NK cells and an increase in cytokines levels (especially IL-1, IL-6 and IL-8) was found after TARE with yttrium-90 [
103,
104,
105].
Locoregional therapy can promote systemic immune response by releasing neoantigens into blood circulation, but their effect alone might be too modest to prevent tumor recurrence and metastasis, even after successful treatments. Moreover, especially when incomplete, locoregional treatments can also induce immunosuppressive factors (such as IL-6, VEGF, HIF-1α, TGF-β, PD-1 and PD-L1), stimulate the accumulation of Tregs in the tumor and cause lymphopenia, leading to tumor progression in the end [
106,
107,
108,
109,
110]. Incomplete T cell restoration despite antigen clearance and immune-tolerant liver environment might also affect the attenuation of immune surveillance. Additionally, their immunological effects appear limited in time. Indeed, as demonstrated by a previous study, the memory phenotype and lifetime of tumor-specific T cells were not sufficient to prevent HCC recurrence completely after RFA [
97]. For all these reasons, the efficacy of locoregional treatments could be enhanced by their combination with immunotherapeutic drugs, which would guarantee the achievement of an immunologically more favorable tumor microenvironment [
111,
112]; at the same time, through a mutually beneficial and synergistic mechanism, the positive alteration of the tumor microenvironment derived from locoregional treatments may enhance ICI therapy efficacy (
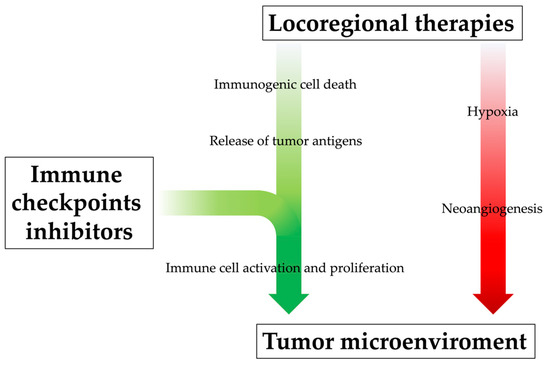
Figure 3) [
38].
Figure 3. The rationale behind combining locoregional therapies and immunotherapy. Locoregional therapies, especially when incomplete, can increase the level of pro-angiogenetic cytokines and thus promote neoangiogenesis of residual cancer cells and metastasis development; at the same time, however, they promote systemic immune response by releasing neoantigens into blood circulation, although this immunogenic effect might be too modest. The immunological efficacy of locoregional treatments could be enhanced by their combination with immunotherapeutic drugs, which would promote immune cell activation and proliferation, positively influencing the tumor microenvironment.
To date, there is no direct comparison between the different ablation or intra-arterial techniques, therefore it is not known whether one technique is superior to the others in inducing tumor-specific immune response [
113]. In a previous study, it was demonstrated that serum levels of Glypican-3, a carcinoembryonic antigen inducing tumor-specific activation of cytotoxic T cells, were increased in 55% of patients with HCC after RFA and in 44% of patients after TACE, although these results were non-significant [
114]. Interestingly, more recent evidence seems to suggest that TACE may have a greater immunogenic role than other locoregional treatments, possibly due to the potential immunogenic cell death induced by doxorubicin [
115]. Doxorubicin is the most used chemotherapeutic agent for TACE and, despite the absence of a proven superiority compared to other drugs (such as cisplatin, epirubicin and mitomycin), is the only one to have demonstrated to possess immunogenic properties and thus trigger a significant tumor-specific immunological response [
116]. In particular, anthracyclines such as doxorubicin seem to cause the post-transcriptional translocation of calreticulin from the endoplasmic reticulum, where it is involved in the maintenance of Ca
2+ homeostasis, to the plasma membrane of tumor cells; surface-exposed calreticulin then acts as an “eat me” signal for phagocytosis by neighboring APCs, which is required for subsequent antigen cross-presentation to cytotoxic T cells [
117]. Because chemotherapy is an integral part of TACE, these studies indicate that not only the immunogenic effects of embolization must be considered, but also the immune effects of the chemotherapy of choice. Therefore, if TACE is combined with immunotherapy, doxorubicin likely would lead to better outcomes compared to other chemotherapeutic agents.
4. The Current Evidence from Clinical Trials
The high risk of local and distant recurrence after locoregional treatments indicates the need for efficient adjuvant strategies to improve cure rates, even at very early and early stages. Features, such as large tumors, multinodularity, and vascular invasion (macroscopic or microscopic), are significantly related to higher recurrence rates in both ablative and intra-arterial therapies [
118,
119,
120]. With this perspective, the addition of immunotherapy after locoregional treatments could amplify the effect of these treatments against micro-metastatic residual disease, especially in patients with a high risk of recurrence or those who would present clinical or hepatic deterioration after treatment. Similarly, there is a rationale to integrate immunotherapy in the neoadjuvant setting as well, especially in intermediate and advanced stages. The pre-treatment administration of ICIs can indeed leverage the higher levels of tumor antigens and thus promote the expansion of tumor-specific T cells, increasing the chance of cure following locoregional treatments [
121,
122].
One of the first trials that investigated the role of ICIs in combination with locoregional treatments in HCC patients evaluated the safety and efficacy of tremelimumab plus subtotal conventional TACE, RFA or cryoablation in patients who were non-responders to sorafenib. In particular, the protocol was shown to be safe and feasible, with no clear trends in adverse events or dose-limited toxicity; moreover, this therapeutic combination resulted in objective tumor responses even outside of the ablated or embolized zone, indicating that the systemic effects brought by locoregional therapies indeed exist [
123]. The combination of tremelimumab plus ablation (RFA or cryoablation) or drug-eluting beads TACE (DEB-TACE) was also assessed in another study with HCC patients progressed on sorafenib therapy, proving the safety and efficacy of the protocol; in particular, the primary lesion kept shrinking and almost disappeared at 6 months and the untreated other intrahepatic lesions reduced in size gradually [
84]. The enhanced efficacy of anti-PD-1 and ablative combined therapy was later confirmed in another retrospective study, where patients who underwent RFA plus camrelizumab or sintilimab demonstrated a longer OS and a higher recurrence-free survival (RFS) compared to those treated with RFA alone (32.5% vs. 10.0% and 51 weeks vs. 47.6 weeks, respectively) [
124]. Similarly, a proof-of-concept clinical trial enrolling 50 patients with advanced HCC after sorafenib failure reported that additional RFA or MWA to anti-PD-1 therapy (nivolumab or pembrolizumab) increased the response rate from 10% to 24%. This latter study, moreover, documented that repeated ablations were also proved feasible and safe, reporting only common ablation-related complications that were easily managed as per the standard of care [
125].
Three different studies [
126,
127,
128] indicated that anti-PD-1 therapy (camrelizumab) plus TACE regimen is effective and safe, with effective tumor control, improved survival and manageable ICI-related adverse effects, leading to better outcomes than treatment with anti-PD-1 inhibitors alone; moreover, a longer interval between camrelizumab administration and TACE was related to the unsatisfying OS, whereas the timing of administration (before or after TACE) did not significantly influence the results. However, another study reported similar efficacy of TACE combined with camrelizumab compared to TACE alone, although the protocol was safe and tolerable [
129]. Among the most common adverse events, itching was the most common, and is often associated with dermatitis and increased liver transaminases; whereas the appearance of colitis, thyroiditis and pneumonia is rarer. An interesting study compared the efficacy and safety of conventional TACE + camrelizumab with DEB-TACE + camrelizumab with the aim of determining which technique was superior. Despite both protocols being safe and well-tolerated, DEB-TACE produced better tumor response and PFS (70.4% vs. 40.7% and 10 vs. 3 months, respectively); however, these results could have been influenced by the inclusion of patients with large and multiple HCCs, who are theoretically more susceptible to this type of intra-arterial procedure; thus, further studies are needed [
130].
Similar to TACE, even TARE in combination with nivolumab was demonstrated as a safe and effective treatment for HCC patients, showing a higher objective response rate (ORR) compared to both TARE alone and anti-PD-1 agents alone (30.6% vs. 20% vs. 15–23%, respectively) [
131]; of note, the ORR in patients without extrahepatic spread was 43.5%, suggesting that TARE followed by nivolumab should be further evaluated in patients with BCLC B or BCLC C with no extrahepatic spread. One small retrospective trial examined patients with advanced HCC but preserved liver function who had received TARE and nivolumab with or without ipilimumab, documenting the safety of this association; moreover, there were no differences in toxicities between patients who received both therapies within 30 days of each other and those who received both therapies within 30–90 days [
132]. The safety and efficacy of TARE plus anti-PD-1 therapy were also confirmed in other studies [
133,
134].
Despite this encouraging evidence, larger and comparative studies are needed to confirm the efficacy of immunotherapy combined with locoregional treatments in HCC patients. Currently, several other trials are exploring the role of ICIs in combination with locoregional treatments in HCC patients, with or without other drugs (such as tyrosine-kinase inhibitors), but participants are still being recruited or are receiving intervention, or data have yet to be analyzed. The role of numerous immunotherapeutic drugs is being tested in the adjuvant setting of patients who underwent ablative therapies, including nivolumab (the CheckMate 9DX trial, NCT03383458), atezolizumab plus bevacizumab (the IMbrave050 trial, NCT04102098) and pembrolizumab (the KEYNOTE-937 trial, NCT03867084); similarly, the use of nivolumab in both adjuvant and neoadjuvant settings after electroporation is currently being investigated (the NIVOLEP trial, NCT03630640). The number of studies that are evaluating the combination of immunotherapy with TACE and TARE in intermediate and advanced patients is even larger. The results of the LEAP-012 trial exploring TACE plus pembrolizumab plus lenvatinib in advanced HCC patients are eagerly awaited (NCT04246177), as are those of ongoing trials evaluating TACE plus atezolizumab plus bevacizumab (NCT04712643), TACE plus durvalumab plus bevacizumab (the EMERALD-1 trial, NCT03778957), TACE plus durvalumab plus bevacizumab plus tremelimumab (the EMERALD-3 trial, NCT05301842), TACE plus nivolumab (the TACE-3 trial, NCT04268888) and TACE plus nivolumab plus ipilimumab (the CheckMate 74W trial, NCT04340193). Similarly, the results that will emerge from trials combining TARE with yttrium-90 plus nivolumab (NCT03033446), pembrolizumab (NCT03099564) or durvalumab plus tremelimumab (the MEDI4736 trial, NCT04522544) are highly anticipated.