Reactive oxygen species (ROS), reactive nitrogen species (RNS), and H2S-reactive sulfur species (RSS) collectively termed reactive oxygen, nitrogen, and sulfur species (RONSS), constitute a conglomerate of reactions that function as an energy dissipation mechanism, in addition to allowing environmental signals to be transduced into cellular information. This information, in the form of proteins with posttranslational modifications or signaling metabolites derived from RONSS, serves as an inducer of many processes for redoxtasis and metabolic adjustment to the changing environmental conditions to which plants are subjected. Although it is thought that the role of reactive chemical species was originally energy dissipation, during evolution they seem to form a cluster of RONSS that, in addition to dissipating excess excitation potential or reducing potential, also fulfils essential signaling functions that play a vital role in the stress acclimation of plants. Signaling occurs by synthesizing many biomolecules that modify the activity of transcription factors and through modifications in thiol groups of enzymes. The result is a series of adjustments in plants’ gene expression, biochemistry, and physiology. The action of RONSS as signaling agents in Biostimulation is discussed.
- RONSS
- ROS
- Reactive Oxygen Species
- Reactive Nitrogen Species
- Reactive Sulfur Species
- RNS
- Plant stress
1. RONSS Integration as a Metabolic Cluster
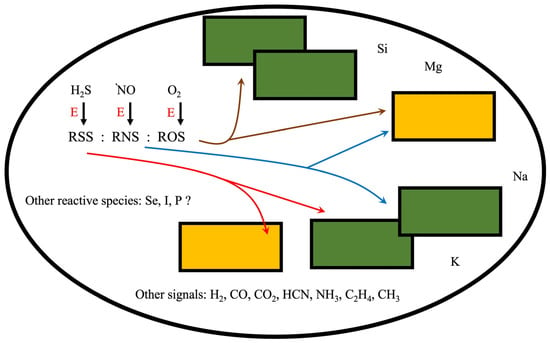
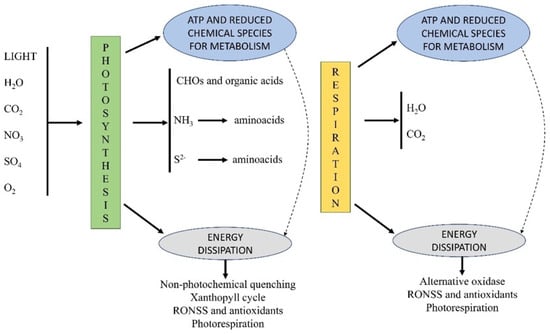
3. RONSS as Biostimulants
| Impact on the Plant | Reactive Species | Plant Species | Reference |
|---|---|---|---|
| Decreased absorption and/or toxicity of heavy metals | H2S, ·NO H2S, ·NO H2S, ·NO H2S, ·NO |
Medicago sativa Sesamum indicum Triticum aestivum Triticum aestivum |
[67] [68] [69] [51] |
| Increase in the concentration of essential elements | H2S, ·NO H2S, ·NO |
Triticum aestivum Sesamum indicum |
[69] [68] |
| Increase in Relative Growth Rate (RGR) and/or biomass | H2S, ·NO H2S, ·NO H2S, ·NO H2S, ·NO H2S, ·NO H2S, ·NO H2O2, ·NO H2O2, ·NO H2O2, ·NO |
Cynodon dactylon Medicago sativa Sesamum indicum Solanum lycopersicum Triticum aestivum Triticum aestivum Ocimum basilicum Oriza sativa Triticum aestivum |
[67] [70] [68] [71] [51] [69] [72] [73] [74] |
| Improved crop yield and/or quality | H2O2, ·NO | Ocimum basilicum | [72] |
| Increase in Relative Water Content (RWC) | H2S, ·NO H2O2, ·NO |
Triticum aestivum Fragaria × ananassa |
[51] [75] |
| Increment in stomatal conductance (gs) | H2S, ·NO H2S, ·NO |
Medicago sativa Triticum aestivum |
[50] [51] |
| Increase in the quantum efficiency of PSII (Fv/Fm) | H2S, ·NO H2S, ·NO H2O2, ·NO H2O2, ·NO |
Medicago sativa Triticum aestivum Citrus aurantium Fragaria × ananassa |
[50] [51] [76] [75] |
| Increase in CO2 assimilation (A) | H2O2, ·NO | Citrus aurantium | [76] |
| Increment in the concentration of photosynthetic pigments | H2S, ·NO H2S, ·NO H2O2, ·NO H2O2, ·NO H2O2, ·NO |
Sesamum indicum Triticum aestivum Citrus aurantium Fragaria × ananassa Ocimum basilicum |
[68] [69] [76] [75] [72] |
| Increased activity of antioxidant enzymes (e.g., SOD and CAT) and the ascorbate–glutathione (AsA–GSH) cycle | H2S, ·NO H2S, ·NO H2S, ·NO H2S, ·NO H2S, ·NO H2S, ·NO H2S, ·NO H2O2, ·NO |
Cynodon dactylon Medicago sativa Medicago sativa Medicago sativa Solanum lycopersicum Triticum aestivum Triticum aestivum Ocimum basilicum |
[70] [77] [67] [50] [71] [69] [51] [72] |
| Proteome reprogramming through reversible or irreversible posttranslational modifications (PTM) and changes in gene expression | H2S, ·NO H2S, ·NO H2O2, ·NO |
Citrus aurantium Citrus aurantium Citrus aurantium |
[78] [79] [80] |
| Mitigation of the relative electrolyte leakage under stress | H2S, ·NO H2O2, ·NO H2O2, ·NO H2O2, ·NO |
Cynodon dactylon Citrus aurantium Citrus aurantium Fragaria × ananassa |
[70] [80] [76] [75] |
| Mitigation of lipid peroxidation under stress | H2S, ·NO H2S, ·NO H2S, ·NO H2S, ·NO H2S, ·NO H2O2, ·NO |
Cynodon dactylon Medicago sativa Medicago sativa Solanum lycopersicum Triticum aestivum Fragaria × ananassa |
[70] [50] [67] [71] [69] [75] |
| Increased accumulation of proline and other osmolytes | H2S, ·NO H2O2, ·NO |
Medicago sativa Triticum aestivum |
[50] [74] |
This entry is adapted from the peer-reviewed paper 10.3390/plants11233203
References
- Ricard, J. Systems Biology and the Origins of Life? Part I. Are Biochemical Networks Possible Ancestors of Living Systems? Reproduction, Identity and Sensitivity to Signals of Biochemical Networks. Comptes Rendus Biol. 2010, 333, 761–768.
- Muchowska, K.B.; Varma, S.J.; Moran, J. Nonenzymatic Metabolic Reactions and Life’s Origins. Chem. Rev. 2020, 120, 7708–7744.
- Becerra, A. The Semi-Enzymatic Origin of Metabolic Pathways: Inferring a Very Early Stage of the Evolution of Life. J. Mol. Evol. 2021, 89, 183–188.
- Morowitz, H.J.; Deamer, D.W.; Smith, T. Biogenesis as an Evolutionary Process. J. Mol. Evol. 1991, 33, 207–208.
- Michaelian, K. Non-Equilibrium Thermodynamic Foundations of the Origin of Life. Foundations 2022, 2, 308–337.
- Olson, K.R. Reactive Oxygen Species or Reactive Sulfur Species: Why We Should Consider the Latter. J. Exp. Biol. 2020, 223, jeb196352.
- Jones, D.P.; Sies, H. The Redox Code. Antioxid. Redox Signal. 2015, 23, 734–746.
- Lotka, A.J. Contribution to the Energetics of Evolution. Proc. Natl. Acad. Sci. USA 1922, 8, 147–151.
- Corning, P.A. A Systems Theory of Biological Evolution. Biosystems 2022, 214, 104630.
- Suki, B. The Major Transitions of Life from a Network Perspective. Front. Physiol. 2012, 3, 1–13.
- Hancock, J.T. Hydrogen Sulfide and Environmental Stresses. Environ. Exp. Bot. 2019, 161, 50–56.
- Hancock, J.T.; Whiteman, M. Hydrogen Sulfide Signaling: Interactions with Nitric Oxide and Reactive Oxygen Species. Ann. N. Y. Acad. Sci. 2016, 1365, 5–14.
- De la Fuente, I.M. Elements of the Cellular Metabolic Structure. Front. Mol. Biosci. 2015, 2, 16.
- Heylighen, F.; Beigi, S.; Busseniers, E. The Role of Self-Maintaining Resilient Reaction Networks in the Origin and Evolution of Life. Biosystems 2022, 219, 104720.
- De Bianchi, S.; Ballottari, M.; Dall’Osto, L.; Bassi, R. Regulation of Plant Light Harvesting by Thermal Dissipation of Excess Energy. Biochem. Soc. Trans. 2010, 38, 651–660.
- Haupt-Herting, S.; Fock, H.P. Exchange of Oxygen and Its Role in Energy Dissipation during Drought Stress in Tomato Plants. Physiol. Plant 2000, 110, 489–495.
- Baluška, F.; Reber, A.S.; Miller, W.B. Cellular Sentience as the Primary Source of Biological Order and Evolution. Biosystems 2022, 218, 104694.
- Mahajan, A.S.; Oetjen, H.; Saiz-Lopez, A.; Lee, J.D.; McFiggans, G.B.; Plane, J.M.C. Reactive Iodine Species in a Semi-Polluted Environment. Geophys. Res. Lett. 2009, 36, L16803.
- Pasek, M.A.; Harnmeijer, J.P.; Buick, R.; Gull, M.; Atlas, Z. Evidence for Reactive Reduced Phosphorus Species in the Early Archean Ocean. PNAS 2013, 110, 10089–10094.
- Kharma, A.; Grman, M.; Misak, A.; Domínguez-Álvarez, E.; Nasim, M.J.; Ondrias, K.; Chovanec, M.; Jacob, C. Inorganic Polysulfides and Related Reactive Sulfur–Selenium Species from the Perspective of Chemistry. Molecules 2019, 24, 1359.
- Alché, J.d.D. A Concise Appraisal of Lipid Oxidation and Lipoxidation in Higher Plants. Redox Biol. 2019, 23, 101136.
- Kolbert, Z.; Barroso, J.B.; Brouquisse, R.; Corpas, F.J.; Gupta, K.J.; Lindermayr, C.; Loake, G.J.; Palma, J.M.; Petřivalský, M.; Wendehenne, D.; et al. A Forty Year Journey: The Generation and Roles of NO in Plants. Nitric Oxide 2019, 93, 53–70.
- Tola, A.J.; Jaballi, A.; Missihoun, T.D. Protein Carbonylation: Emerging Roles in Plant Redox Biology and Future Prospects. Plants 2021, 10, 1451.
- Olson, K.R.; Straub, K.D. The Role of Hydrogen Sulfide in Evolution and the Evolution of Hydrogen Sulfide in Metabolism and Signaling. Physiology 2015, 31, 60–72.
- Yamasaki, H.; Cohen, M.F. Biological Consilience of Hydrogen Sulfide and Nitric Oxide in Plants: Gases of Primordial Earth Linking Plant, Microbial and Animal Physiologies. Nitric Oxide 2016, 55–56, 91–100.
- Gumsley, A.P.; Chamberlain, K.R.; Bleeker, W.; Söderlund, U.; de Kock, M.O.; Larsson, E.R.; Bekker, A. Timing and Tempo of the Great Oxidation Event. PNAS 2017, 114, 1811–1816.
- Santolini, J.; Wootton, S.A.; Jackson, A.A.; Feelisch, M. The Redox Architecture of Physiological Function. Curr. Opin. Physiol. 2019, 9, 34–47.
- Allen, J.F.; Thake, B.; Martin, W.F. Nitrogenase Inhibition Limited Oxygenation of Earth’s Proterozoic Atmosphere. Trends Plant Sci. 2019, 24, 1022–1031.
- Jacob, F. Evolution and Tinkering. Science 1977, 196, 1161–1166.
- Macklem, P.T. Emergent Phenomena and the Secrets of Life. J. Appl. Physiol. 2008, 104, 1844–1846.
- Toyabe, S.; Sagawa, T.; Ueda, M.; Muneyuki, E.; Sano, M. Experimental Demonstration of Information-to-Energy Conversion and Validation of the Generalized Jarzynski Equality. Nat. Phys. 2010, 6, 988–992.
- Farooq, M.A.; Niazi, A.K.; Akhtar, J.; Saifullah; Farooq, M.; Souri, Z.; Karimi, N.; Rengel, Z. Acquiring Control: The Evolution of ROS-Induced Oxidative Stress and Redox Signaling Pathways in Plant Stress Responses. Plant Physiol. Biochem. 2019, 141, 353–369.
- Noctor, G.; Reichheld, J.-P.; Foyer, C.H. ROS-Related Redox Regulation and Signaling in Plants. Semin. Cell Dev. Biol. 2018, 80, 3–12.
- Antoniou, C.; Savvides, A.; Christou, A.; Fotopoulos, V. Unravelling Chemical Priming Machinery in Plants: The Role of Reactive Oxygen–Nitrogen–Sulfur Species in Abiotic Stress Tolerance Enhancement. Curr. Opin. Plant Biol. 2016, 33, 101–107.
- Corpas, F.J.; Carreras, A.; Valderrama, R.; Chaki, M.; Palma, J.M. Reactive Nitrogen Species and Nitrosative Stress in Plants. Plant Stress 2007, 1, 37–41.
- Ahmad, P.; Sarwat, M.; Sharma, S. Reactive Oxygen Species, Antioxidants and Signaling in Plants. J. Plant Biol. 2008, 51, 167–173.
- Gruhlke, M.C.H.; Slusarenko, A.J. The Biology of Reactive Sulfur Species (RSS). Plant Physiol. Biochem. 2012, 59, 98–107.
- Corpas, F.J.; Gupta, D.K.; Palma, J.M. Production Sites of Reactive Oxygen Species (ROS) in Organelles from Plant Cells. In Reactive Oxygen Species and Oxidative Damage in Plants Under Stress; Gupta, D.K., Palma, J.M., Corpas, F.J., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 1–22. ISBN 978-3-319-20421-5.
- Giles, G.I.; Nasim, M.J.; Ali, W.; Jacob, C. The Reactive Sulfur Species Concept: 15 Years On. Antioxidants 2017, 6, 38.
- Corpas, F.J.; Palma, J.M. Assessing Nitric Oxide (NO) in Higher Plants: An Outline. Nitrogen 2020, 1, 12–20.
- González-Morales, S.; López-Sánchez, R.C.; Juárez-Maldonado, A.; Robledo-Olivo, A.; Benavides-Mendoza, A. A Transcriptomic and Proteomic View of Hydrogen Sulfide Signaling in Plant Abiotic Stress. In Hydrogen Sulfide and Plant Acclimation to Abiotic Stresses; Khan, M.N., Siddiqui, M.H., Alamri, S., Corpas, F.J., Eds.; Plant in Challenging Environments; Springer International Publishing: Cham, Switzerland, 2021; pp. 161–186. ISBN 978-3-030-73678-1.
- Khanna, K.; Sharma, N.; Kour, S.; Ali, M.; Ohri, P.; Bhardwaj, R. Hydrogen Sulfide: A Robust Combatant against Abiotic Stresses in Plants. Hydrogen 2021, 2, 319–342.
- Mansoor, S.; Ali Wani, O.; Lone, J.K.; Manhas, S.; Kour, N.; Alam, P.; Ahmad, A.; Ahmad, P. Reactive Oxygen Species in Plants: From Source to Sink. Antioxidants 2022, 11, 225.
- Mittler, R.; Zandalinas, S.I.; Fichman, Y.; Van Breusegem, F. Reactive Oxygen Species Signalling in Plant Stress Responses. Nat. Rev. Mol. Cell Biol. 2022, 23, 663–679.
- Giles, G.I.; Tasker, K.M.; Jacob, C. Hypothesis: The Role of Reactive Sulfur Species in Oxidative Stress. Free Radic. Biol. Med. 2001, 31, 1279–1283.
- Del Río, L.A. ROS and RNS in Plant Physiology: An Overview. J. Exp. Bot. 2015, 66, 2827–2837.
- Weidinger, A.; Kozlov, A.V. Biological Activities of Reactive Oxygen and Nitrogen Species: Oxidative Stress versus Signal Transduction. Biomolecules 2015, 5, 472–484.
- Turkan, I. ROS and RNS: Key Signalling Molecules in Plants. J Exp Bot 2018, 69, 3313–3315.
- Olson, K.R. Hydrogen Sulfide, Reactive Sulfur Species and Coping with Reactive Oxygen Species. Free Radic. Biol. Med. 2019, 140, 74–83.
- Antoniou, C.; Xenofontos, R.; Chatzimichail, G.; Christou, A.; Kashfi, K.; Fotopoulos, V. Exploring the Potential of Nitric Oxide and Hydrogen Sulfide (NOSH)-Releasing Synthetic Compounds as Novel Priming Agents against Drought Stress in Medicago Sativa Plants. Biomolecules 2020, 10, 120.
- Ozfidan-Konakci, C.; Yildiztugay, E.; Elbasan, F.; Kucukoduk, M.; Turkan, I. Hydrogen Sulfide (H2S) and Nitric Oxide (NO) Alleviate Cobalt Toxicity in Wheat (Trriticum aestivum L.) by Modulating Photosynthesis, Chloroplastic Redox and Antioxidant Capacity. J. Hazard. Mater. 2020, 388, 122061.
- Palma, J.M.; Mateos, R.M.; López-Jaramillo, J.; Rodríguez-Ruiz, M.; González-Gordo, S.; Lechuga-Sancho, A.M.; Corpas, F.J. Plant Catalases as NO and H2S Targets. Redox Biol. 2020, 34, 101525.
- Tomar, R.S.; Kataria, S.; Jajoo, A. Behind the Scene: Critical Role of Reactive Oxygen Species and Reactive Nitrogen Species in Salt Stress Tolerance. J. Agron. Crop. Sci. 2021, 207, 577–588.
- Wani, K.I.; Naeem, M.; Castroverde, C.D.M.; Kalaji, H.M.; Albaqami, M.; Aftab, T. Molecular Mechanisms of Nitric Oxide (NO) Signaling and Reactive Oxygen Species (ROS) Homeostasis during Abiotic Stresses in Plants. Int. J. Mol. Sci. 2021, 22, 9656.
- Kumar, S.P.J.; Chintagunta, A.D.; Reddy, Y.M.; Rajjou, L.; Garlapati, V.K.; Agarwal, D.K.; Prasad, S.R.; Simal-Gandara, J. Implications of Reactive Oxygen and Nitrogen Species in Seed Physiology for Sustainable Crop Productivity under Changing Climate Conditions. Curr. Plant Biol. 2021, 26, 100197.
- Lushchak, V.I.; Lushchak, O. Interplay between Reactive Oxygen and Nitrogen Species in Living Organisms. Chem. Biol. Interact. 2021, 349, 109680.
- Savvides, A.; Ali, S.; Tester, M.; Fotopoulos, V. Chemical Priming of Plants Against Multiple Abiotic Stresses: Mission Possible? Trends Plant Sci. 2016, 21, 329–340.
- Ashraf, M.A.; Rasheed, R.; Hussain, I.; Iqbal, M.; Riaz, M.; Arif, M.S. Chemical Priming for Multiple Stress Tolerance. In Priming and Pretreatment of Seeds and Seedlings: Implication in Plant Stress Tolerance and Enhancing Productivity in Crop Plants; Hasanuzzaman, M., Fotopoulos, V., Eds.; Springer: Singapore, 2019; pp. 385–415. ISBN 9789811386251.
- Kaur, P.; Handa, N.; Verma, V.; Bakshi, P.; Kalia, R.; Sareen, S.; Nagpal, A.; Vig, A.P.; Mir, B.A.; Bhardwaj, R. Cross Talk Among Reactive Oxygen, Nitrogen and Sulfur During Abiotic Stress in Plants. In Reactive Oxygen, Nitrogen and Sulfur Species in Plants; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2019; pp. 857–871. ISBN 978-1-119-46867-7.
- Zhou, X.; Joshi, S.; Patil, S.; Khare, T.; Kumar, V. Reactive Oxygen, Nitrogen, Carbonyl and Sulfur Species and Their Roles in Plant Abiotic Stress Responses and Tolerance. J. Plant Growth Regul. 2021, 41, 119–142.
- Mangal, V.; Lal, M.K.; Tiwari, R.K.; Altaf, M.A.; Sood, S.; Kumar, D.; Bharadwaj, V.; Singh, B.; Singh, R.K.; Aftab, T. Molecular Insights into the Role of Reactive Oxygen, Nitrogen and Sulphur Species in Conferring Salinity Stress Tolerance in Plants. J. Plant Growth Regul. 2022, 1–21.
- Kaya, C.; Higgs, D.; Ashraf, M.; Alyemeni, M.N.; Ahmad, P. Integrative Roles of Nitric Oxide and Hydrogen Sulfide in Melatonin-Induced Tolerance of Pepper (Capsicum annuum L.) Plants to Iron Deficiency and Salt Stress Alone or in Combination. Physiol. Plant 2020, 168, 256–277.
- Kaya, C.; Ashraf, M.; Alyemeni, M.N.; Corpas, F.J.; Ahmad, P. Salicylic Acid-Induced Nitric Oxide Enhances Arsenic Toxicity Tolerance in Maize Plants by Upregulating the Ascorbate-Glutathione Cycle and Glyoxalase System. J. Hazard. Mater. 2020, 399, 123020.
- Kataria, S.; Jain, M.; Tripathi, D.K.; Singh, V.P. Involvement of Nitrate Reductase-Dependent Nitric Oxide Production in Magnetopriming-Induced Salt Tolerance in Soybean. Physiol. Plant 2020, 168, 422–436.
- Kaur, H.; Bhatla, S.C. Melatonin and Nitric Oxide Modulate Glutathione Content and Glutathione Reductase Activity in Sunflower Seedling Cotyledons Accompanying Salt Stress. Nitric Oxide 2016, 59, 42–53.
- Kaya, C.; Ashraf, M.; Al-Huqail, A.A.; Alqahtani, M.A.; Ahmad, P. Silicon Is Dependent on Hydrogen Sulphide to Improve Boron Toxicity Tolerance in Pepper Plants by Regulating the AsA-GSH Cycle and Glyoxalase System. Chemosphere 2020, 257, 127241.
- Li, L.; Wang, Y.; Shen, W. Roles of Hydrogen Sulfide and Nitric Oxide in the Alleviation of Cadmium-Induced Oxidative Damage in Alfalfa Seedling Roots. Biometals 2012, 25, 617–631.
- Amooaghaie, R.; Zangene-Madar, F.; Enteshari, S. Role of Two-Sided Crosstalk between NO and H2S on Improvement of Mineral Homeostasis and Antioxidative Defense in Sesamum Indicum under Lead Stress. Ecotoxicol. Environ. Saf. 2017, 139, 210–218.
- Kaya, C.; Ashraf, M.; Alyemeni, M.N.; Ahmad, P. Responses of Nitric Oxide and Hydrogen Sulfide in Regulating Oxidative Defence System in Wheat Plants Grown under Cadmium Stress. Physiol. Plant 2020, 168, 345–360.
- Shi, H.; Ye, T.; Chan, Z. Nitric Oxide-Activated Hydrogen Sulfide Is Essential for Cadmium Stress Response in Bermudagrass (Cynodon dactylon (L). Pers.). Plant Physiol. Biochem. 2014, 74, 99–107.
- Liang, Y.; Zheng, P.; Li, S.; Li, K.; Xu, H. Nitrate Reductase-Dependent NO Production Is Involved in H2S-Induced Nitrate Stress Tolerance in Tomato via Activation of Antioxidant Enzymes. Sci. Hortic. 2018, 229, 207–214.
- Gohari, G.; Alavi, Z.; Esfandiari, E.; Panahirad, S.; Hajihoseinlou, S.; Fotopoulos, V. Interaction between Hydrogen Peroxide and Sodium Nitroprusside Following Chemical Priming of Ocimum basilicum L. against Salt Stress. Physiol. Plant 2020, 168, 361–373.
- Uchida, A.; Jagendorf, A.T.; Hibino, T.; Takabe, T.; Takabe, T. Effects of Hydrogen Peroxide and Nitric Oxide on Both Salt and Heat Stress Tolerance in Rice. Plant Sci. 2002, 163, 515–523.
- Farooq, M.; Nawaz, A.; Chaudhary, M.A.M.; Rehman, A. Foliage-Applied Sodium Nitroprusside and Hydrogen Peroxide Improves Resistance against Terminal Drought in Bread Wheat. J. Agron. Crop. Sci. 2017, 203, 473–482.
- Christou, A.; Manganaris, G.A.; Fotopoulos, V. Systemic Mitigation of Salt Stress by Hydrogen Peroxide and Sodium Nitroprusside in Strawberry Plants via Transcriptional Regulation of Enzymatic and Non-Enzymatic Antioxidants. Environ. Exp. Bot. 2014, 107, 46–54.
- Tanou, G.; Filippou, P.; Belghazi, M.; Job, D.; Diamantidis, G.; Fotopoulos, V.; Molassiotis, A. Oxidative and Nitrosative-Based Signaling and Associated Post-Translational Modifications Orchestrate the Acclimation of Citrus Plants to Salinity Stress. Plant J. 2012, 72, 585–599.
- Wang, Y.; Li, L.; Cui, W.; Xu, S.; Shen, W.; Wang, R. Hydrogen Sulfide Enhances Alfalfa (Medicago sativa) Tolerance against Salinity during Seed Germination by Nitric Oxide Pathway. Plant Soil 2012, 351, 107–119.
- Ziogas, V.; Tanou, G.; Belghazi, M.; Filippou, P.; Fotopoulos, V.; Grigorios, D.; Molassiotis, A. Roles of Sodium Hydrosulfide and Sodium Nitroprusside as Priming Molecules during Drought Acclimation in Citrus Plants. Plant Mol. Biol. 2015, 89, 433–450.
- Tanou, G.; Job, C.; Rajjou, L.; Arc, E.; Belghazi, M.; Diamantidis, G.; Molassiotis, A.; Job, D. Proteomics Reveals the Overlapping Roles of Hydrogen Peroxide and Nitric Oxide in the Acclimation of Citrus Plants to Salinity. Plant J. 2009, 60, 795–804.
- Tanou, G.; Job, C.; Belghazi, M.; Molassiotis, A.; Diamantidis, G.; Job, D. Proteomic Signatures Uncover Hydrogen Peroxide and Nitric Oxide Cross-Talk Signaling Network in Citrus Plants. J. Proteome Res. 2010, 9, 5994–6006.
- Mukherjee, S.; Corpas, F.J. Crosstalk among Hydrogen Sulfide (H2S), Nitric Oxide (NO) and Carbon Monoxide (CO) in Root-System Development and Its Rhizosphere Interactions: A Gaseous Interactome. Plant Physiol. Biochem. 2020, 155, 800–814.
- Foyer, C.H.; Ruban, A.V.; Noctor, G. Viewing Oxidative Stress through the Lens of Oxidative Signalling Rather than Damage. Biochem. J. 2017, 474, 877–883.
- Cortese-Krott, M.M.; Koning, A.; Kuhnle, G.G.C.; Nagy, P.; Bianco, C.L.; Pasch, A.; Wink, D.A.; Fukuto, J.M.; Jackson, A.A.; van Goor, H.; et al. The Reactive Species Interactome: Evolutionary Emergence, Biological Significance, and Opportunities for Redox Metabolomics and Personalized Medicine. Antioxid. Redox Signal. 2017, 27, 684–712.
- Freschi, L. Nitric Oxide and Phytohormone Interactions: Current Status and Perspectives. Front. Plant Sci. 2013, 4, 398.
- He, H.; Garcia-Mata, C.; He, L.-F. Interaction between Hydrogen Sulfide and Hormones in Plant Physiological Responses. Plant Growth Regul 2019, 87, 175–186.
- Antoniou, C.; Chatzimichail, G.; Kashfi, K.; Fotopoulos, V. P77: Exploring the Potential of NOSH-Aspirin as a Plant Priming Agent against Abiotic Stress Factors. Nitric Oxide 2014, 39, S39.
- Shah, Z.H.; Rehman, H.M.; Akhtar, T.; Alsamadany, H.; Hamooh, B.T.; Mujtaba, T.; Daur, I.; Al Zahrani, Y.; Alzahrani, H.A.S.; Ali, S.; et al. Humic Substances: Determining Potential Molecular Regulatory Processes in Plants. Front. Plant Sci. 2018, 9, 263.
- Rai, K.K.; Pandey, N.; Rai, S.P. Salicylic Acid and Nitric Oxide Signaling in Plant Heat Stress. Physiol. Plant 2020, 168, 241–255.
- Tripathi, D.K.; Vishwakarma, K.; Singh, V.P.; Prakash, V.; Sharma, S.; Muneer, S.; Nikolic, M.; Deshmukh, R.; Vaculík, M.; Corpas, F.J. Silicon Crosstalk with Reactive Oxygen Species, Phytohormones and Other Signaling Molecules. J. Hazard. Mater. 2020, 408, 124820.
- González-Morales, S.; Solís-Gaona, S.; Valdés-Caballero, M.V.; Juárez-Maldonado, A.; Loredo-Treviño, A.; Benavides-Mendoza, A. Transcriptomics of Biostimulation of Plants under Abiotic Stress. Front. Genet. 2021, 12, 583888.
- Shan, C.; Dai, H.; Sun, Y. Hydrogen Sulfide Protects Wheat Seedlings against Copper Stress by Regulating the Ascorbate and Glutathione Metabolism in Leaves. Aust. J. Crop. Sci. 2012, 6, 248–254.
- Zhang, H.; Hu, L.-Y.; Hu, K.-D.; He, Y.-D.; Wang, S.-H.; Luo, J.-P. Hydrogen Sulfide Promotes Wheat Seed Germination and Alleviates Oxidative Damage against Copper Stress. J. Integr. Plant Biol. 2008, 50, 1518–1529.
- Khan, M.N.; Siddiqui, Z.H.; Naeem, M.; Abbas, Z.K.; Ansari, M.W. Chapter 8—Nitric Oxide and Hydrogen Sulfide Interactions in Plants under Adverse Environmental Conditions. In Emerging Plant Growth Regulators in Agriculture; Aftab, T., Naeem, M., Eds.; Academic Press: Cambridge, MA, USA, 2022; pp. 215–244. ISBN 978-0-323-91005-7.
- Pandey, N.; Verma, L. Nitric Oxide Alleviates Iron Toxicity by Reducing Oxidative Damage and Growth Inhibition in Wheat (Triticum aestivum L.) Seedlings. Int. J. Plant Environ. 2019, 5, 16–22.
- Habib, N.; Ali, Q.; Ali, S.; Javed, M.T.; Zulqurnain Haider, M.; Perveen, R.; Shahid, M.R.; Rizwan, M.; Abdel-Daim, M.M.; Elkelish, A.; et al. Use of Nitric Oxide and Hydrogen Peroxide for Better Yield of Wheat (Triticum aestivum L.) under Water Deficit Conditions: Growth, Osmoregulation, and Antioxidative Defense Mechanism. Plants 2020, 9, 285.
- Manai, J.; Kalai, T.; Gouia, H.; Corpas, F.J. Exogenous Nitric Oxide (NO) Ameliorates Salinity-Induced Oxidative Stress in Tomato (Solanum Lycopersicum) Plants. J. Soil Sci. Plant Nutr. 2014, 14, 433–446.
- Wang, H.; Xiao, W.; Niu, Y.; Jin, C.; Chai, R.; Tang, C.; Zhang, Y. Nitric Oxide Enhances Development of Lateral Roots in Tomato (Solanum lycopersicum L.) under Elevated Carbon Dioxide. Planta 2013, 237, 137–144.
- Ghorbani, A.; Pishkar, L.; Roodbari, N.; Pehlivan, N.; Wu, C. Nitric Oxide Could Allay Arsenic Phytotoxicity in Tomato (Solanum lycopersicum L.) by Modulating Photosynthetic Pigments, Phytochelatin Metabolism, Molecular Redox Status and Arsenic Sequestration. Plant Physiol. Biochem. 2021, 167, 337–348.
- Fierascu, R.C.; Temocico, G.; Fierascu, I.; Ortan, A.; Babeanu, N.E. Fragaria Genus: Chemical Composition and Biological Activities. Molecules 2020, 25, 498.
- Eum, H.-L.; Lee, S.-K. The Responses of Yukbo Strawberry (Fragaria Ananassa Duch.) Fruit to Nitric Oxide. Food Sci. Biotechnol. 2007, 16, 123–126.
- Wills, R.B.H.; Ku, V.V.V.; Leshem, Y.Y. Fumigation with Nitric Oxide to Extend the Postharvest Life of Strawberries. Postharvest Biol. Technol. 2000, 18, 75–79.
- Kaya, C.; Akram, N.A.; Ashraf, M. Influence of Exogenously Applied Nitric Oxide on Strawberry (Fragaria × Ananassa) Plants Grown under Iron Deficiency and/or Saline Stress. Physiol. Plant 2019, 165, 247–263.
- Kaya, C.; Ashraf, M. The Mechanism of Hydrogen Sulfide Mitigation of Iron Deficiency-Induced Chlorosis in Strawberry (Fragaria × Ananassa) Plants. Protoplasma 2019, 256, 371–382.
- Manafi, H.; Baninasab, B.; Gholami, M.; Talebi, M. Nitric Oxide Induced Thermotolerance in Strawberry Plants by Activation of Antioxidant Systems and Transcriptional Regulation of Heat Shock Proteins. J. Hortic. Sci. Biotechnol. 2021, 96, 783–796.
- Liu, S.; Yang, R.; Pan, Y.; Ren, B.; Chen, Q.; Li, X.; Xiong, X.; Tao, J.; Cheng, Q.; Ma, M. Beneficial Behavior of Nitric Oxide in Copper-Treated Medicinal Plants. J. Hazard. Mater. 2016, 314, 140–154.
- Alam, M.M.; Naeem, M.; Khan, M.M.M.A.; Uddin, M. Vincristine and Vinblastine Anticancer Catharanthus Alkaloids: Pharmacological Applications and Strategies for Yield Improvement. In Catharanthus roseus: Current Research and Future Prospects; Naeem, M., Aftab, T., Khan, M.M.A., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 277–307. ISBN 978-3-319-51620-2.
- Gupta, M.M.; Singh, D.V.; Tripathi, A.K.; Pandey, R.; Verma, R.K.; Singh, S.; Shasany, A.K.; Khanuja, S.P.S. Simultaneous Determination of Vincristine, Vinblastine, Catharanthine, and Vindoline in Leaves of Catharanthus Roseus by High-Performance Liquid Chromatography. J. Chromatogr. Sci. 2005, 43, 450–453.
- Weathers, P.J.; Towler, M.; Hassanali, A.; Lutgen, P.; Engeu, P.O. Dried-Leaf Artemisia Annua: A Practical Malaria Therapeutic for Developing Countries? World J. Pharm. 2014, 3, 39–55.
- Nomani, L.; Zehra, A.; Choudhary, S.; Wani, K.I.; Naeem, M.; Siddiqui, M.H.; Khan, M.M.A.; Aftab, T. Exogenous Hydrogen Sulphide Alleviates Copper Stress Impacts in Artemisia annua L.: Growth, Antioxidant Metabolism, Glandular Trichome Development and Artemisinin Biosynthesis. Plant Biol. 2022, 24, 642–651.
- Kim, W.-S.; Choi, W.J.; Lee, S.; Kim, W.J.; Lee, D.C.; Sohn, U.D.; Shin, H.-S.; Kim, W. Anti-Inflammatory, Antioxidant and Antimicrobial Effects of Artemisinin Extracts from Artemisia annua L. Korean J. Physiol. Pharm. 2015, 19, 21–27.
- Adamska, I.; Biernacka, P. Bioactive Substances in Safflower Flowers and Their Applicability in Medicine and Health-Promoting Foods. Int. J. Food Sci. 2021, 2021, e6657639.
- Amir, S.B.; Rasheed, R.; Ashraf, M.A.; Hussain, I.; Iqbal, M. Hydrogen Sulfide Mediates Defense Response in Safflower by Regulating Secondary Metabolism, Oxidative Defense, and Elemental Uptake under Drought. Physiol. Plant 2021, 172, 795–808.
- El-Beltagi, H.S.; Ahmed, O.K.; Hegazy, A.E. Molecular Role of Nitric Oxide in Secondary Products Production in Ginkgo biloba Cell Suspension Culture. Not. Bot. Horti Agrobot. Cluj-Napoca 2015, 43, 12–18.
- Hao, G.-P.; Du, X.-H.; Shi, R.-J. Exogenous nitric oxide accelerates soluble sugar, proline and secondary metabolite synthesis in Ginkgo biloba under drought stress. Zhi Wu Sheng Li Yu Fen Zi Sheng Wu Xue Xue Bao 2007, 33, 499–506.
- Post-White, J.; Ladas, E.J.; Kelly, K.M. Advances in the Use of Milk Thistle (Silybum marianum). Integr. Cancer 2007, 6, 104–109.
- Zangani, E.; Zehtab-Salmasi, S.; Andalibi, B.; Zamani, A.-A. Protective Effects of Nitric Oxide on Photosynthetic Stability and Performance of Silybum Marianum under Water Deficit Conditions. Agron. J. 2018, 110, 555–564.
- Zangani, E.; Zehtab-Salmasi, S.; Andalibi, B.; Zamani, A.A.; Hashemi, M. Exogenous Nitric Oxide Improved Production and Content of Flavonolignans in Milk Thistle Seeds under Water Deficit System. Acta Physiol. Plant 2021, 43, 87.
- Saller, R.; Meier, R.; Brignoli, R. The Use of Silymarin in the Treatment of Liver Diseases. Drugs 2001, 61, 2035–2063.
- Sunil, C.; Xu, B. An Insight into the Health-Promoting Effects of Taxifolin (Dihydroquercetin). Phytochemistry 2019, 166, 112066.
- Bai, L.; Gao, J.; Wei, F.; Zhao, J.; Wang, D.; Wei, J. Therapeutic Potential of Ginsenosides as an Adjuvant Treatment for Diabetes. Front. Pharmacol. 2018, 9, 423.
- Rahimi, S.; Kim, Y.-J.; Devi, B.S.R.; Oh, J.Y.; Kim, S.-Y.; Kwon, W.-S.; Yang, D.-C. Sodium Nitroprusside Enhances the Elicitation Power of Methyl Jasmonate for Ginsenoside Production in Panax Ginseng Roots. Res. Chem. Intermed. 2016, 42, 2937–2951.
- Farouk, S.; Al-Huqail, A.A. Sodium Nitroprusside Application Regulates Antioxidant Capacity, Improves Phytopharmaceutical Production and Essential Oil Yield of Marjoram Herb under Drought. Ind. Crops Prod. 2020, 158, 113034.
- Nasrin, F.; Fatemeh, N.; Ramezan, R. Comparison the Effects of Nitric Oxide and Spermidin Pretreatment on Alleviation of Salt Stress in Chamomile Plant (Matricaria recutita L.). J. Stress Physiol. Biochem. 2012, 8, 214–223.
- Murkowski, A. Heat Stress and Spermidine: Effect on Chlorophyll Fluorescence in Tomato Plants. Biol. Plant 2001, 44, 53–57.
- Hassan, W.; Noreen, H.; Rehman, S.; Gul, S.; Amjad Kamal, M.; Paul Kamdem, J.; Zaman, B.; da Rocha, J. Oxidative Stress and Antioxidant Potential of One Hundred Medicinal Plants. Curr. Top. Med. Chem. 2017, 17, 1336–1370.
- Banerjee, J.; Das, A.; Sinha, M.; Saha, S. Biological Efficacy of Medicinal Plant Extracts in Preventing Oxidative Damage. Oxidative Med. Cell. Longev. 2018, 2018, e7904349.
- Vranješ, M.; Štajner, D.; Vranješ, D.; Blagojevic, B.; Pavlović, K.; Milanov, D.; Popović, B.M. Medicinal Plants Extracts Impact on Oxidative Stress in Mice Brain Under the Physiological Conditions: The Effects of Corn Silk, Parsley, and Bearberry. Acta Chim. Slov. 2021, 68, 896–903.
- Gu, R.; Wang, Y.; Wu, S.; Wang, Y.; Li, P.; Xu, L.; Zhou, Y.; Chen, Z.; Kennelly, E.J.; Long, C. Three New Compounds with Nitric Oxide Inhibitory Activity from Tirpitzia Sinensis, an Ethnomedicinal Plant from Southwest China. BMC Chem. 2019, 13, 47.
- Singh, A.L.; Singh, S.; Kurella, A.; Verma, A.; Mahatama, M.K.; Venkatesh, I. Chapter 12—Plant Bio-Stimulants, Their Functions and Use in Enhancing Stress Tolerance in Oilseeds. In New and Future Developments in Microbial Biotechnology and Bioengineering; Singh, H.B., Vaishnav, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2022; pp. 239–259. ISBN 978-0-323-85579-2.
- keya Tudu, C.; Dey, A.; Pandey, D.K.; Panwar, J.S.; Nandy, S. Chapter 8—Role of Plant Derived Extracts as Biostimulants in Sustainable Agriculture: A Detailed Study on Research Advances, Bottlenecks and Future Prospects. In New and Future Developments in Microbial Biotechnology and Bioengineering; Singh, H.B., Vaishnav, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2022; pp. 159–179. ISBN 978-0-323-85579-2.
- Karthiga, D.; Chozhavendhan, S.; Gandhiraj, V.; Aniskumar, M. The Effects of Moringa Oleifera Leaf Extract as an Organic Bio-Stimulant for the Growth of Various Plants: Review. Biocatal. Agric. Biotechnol. 2022, 43, 102446.
- Yuniati, N.; Kusumiyati, K.; Mubarok, S.; Nurhadi, B. The Role of Moringa Leaf Extract as a Plant Biostimulant in Improving the Quality of Agricultural Products. Plants 2022, 11, 2186.
- Ertani, A.; Schiavon, M.; Muscolo, A.; Nardi, S. Alfalfa Plant-Derived Biostimulant Stimulate Short-Term Growth of Salt Stressed Zea mays L. Plants. Plant Soil 2013, 364, 145–158.
- Godlewska, K.; Ronga, D.; Michalak, I. Plant Extracts—Importance in Sustainable Agriculture. Ital. J. Agron. 2021, 16.
- Oliveira, H.C.; Gomes, B.C.R.; Pelegrino, M.T.; Seabra, A.B. Nitric Oxide-Releasing Chitosan Nanoparticles Alleviate the Effects of Salt Stress in Maize Plants. Nitric Oxide 2016, 61, 10–19.
- Silveira, N.M.; Seabra, A.B.; Marcos, F.C.C.; Pelegrino, M.T.; Machado, E.C.; Ribeiro, R.V. Encapsulation of S-Nitrosoglutathione into Chitosan Nanoparticles Improves Drought Tolerance of Sugarcane Plants. Nitric Oxide 2019, 84, 38–44.
- Fuentes-Lara, L.O.; Medrano-Macías, J.; Pérez-Labrada, F.; Rivas-Martínez, E.N.; García-Enciso, E.L.; González-Morales, S.; Juárez-Maldonado, A.; Rincón-Sánchez, F.; Benavides-Mendoza, A. From Elemental Sulfur to Hydrogen Sulfide in Agricultural Soils and Plants. Molecules 2019, 24, 2282.
- Hasanuzzaman, M.; Bhuyan, M.H.M.B.; Mahmud, J.A.; Nahar, K.; Mohsin, S.M.; Parvin, K.; Fujita, M. Interaction of Sulfur with Phytohormones and Signaling Molecules in Conferring Abiotic Stress Tolerance to Plants. Plant Signal. Behav. 2018, 13, e1477905.
- Mikkelsen, R.; Norton, R. Soil and Fertilizer Sulfur. Better Crops Plant Food 2013, 97, 7–9.
- Jahan, B.; AlAjmi, M.F.; Rehman, M.T.; Khan, N.A. Treatment of Nitric Oxide Supplemented with Nitrogen and Sulfur Regulates Photosynthetic Performance and Stomatal Behavior in Mustard under Salt Stress. Physiol. Plant 2020, 168, 490–510.
