White adipose tissue (WAT) represents an endocrinologically and immunologically active tissue whose primary role is energy storage and homeostasis. Breast WAT is involved in the secretion of hormones and proinflammatory molecules that are associated with breast cancer development and progression.
- breast cancer
- obesity
- inflammation
- metabolism
1. Introduction
2. Immunometabolic Changes in Obese Adipose Tissue
Under normal physiological conditions, in adipose tissue of individuals with healthy BMI, there is homeostasis between anti-inflammatory and proinflammatory molecules that maintains adipose tissue functions [13][15]. Adipose tissue is classified into white and brown adipose tissue (WAT and BAT, respectively) which differ functionally and morphologically. WAT, distributed subcutaneously and viscerally, constitutes 20% of the body weight and 80% of total adipose tissue of a normal adult [14][16]. WAT is the largest store of energy, whilst BAT plays a key role in thermogenesis [14][16]. Under conditions of nutritional excess and during development of obesity, adipocytes undergo structural alterations, such as adipocyte hypertrophy [15][17]. Then adipocytes become dysfunctional, undergo cell death and secrete cytokines that contribute to adipose tissue inflammation and recruitment of pre-adipocytes, leading to adipose tissue hyperplasia [16][17][18,19]. Apart from structural changes, adipose tissue also undergoes functional changes such as mitochondrial dysfunction and endoplasmic reticulum stress [18][20]. In obesity, there is a significant reduction in mitochondrial gene expression leading to downregulation of mitochondrial biogenesis in subcutaneous tissue associated with insulin resistance and inflammation in obese monozygotic twins compared with their leaner co-twins [19][21]. Furthermore, free fatty acid-mediated generation of reactive oxygen species is correlated with endoplasmic reticulum stress and upregulation of pro-inflammatory gene signatures in adipose tissue [20][21][22,23]. Overall, these changes in the adipose tissue result in the activation of proinflammatory signalling pathways, leading to chronic low-grade adipose tissue inflammation mediated by macrophage infiltration, neovascularisation and increase in extracellular matrix [22][23][24][25][24,25,26,27] (Table 1). BC arises in an adipose rich environment and therefore its tumour microenvironment could be impacted by these factors.| Immune Changes | Metabolic Changes | ||
|---|---|---|---|
| Upregulation of proinflammatory signalling pathways [22] | Upregulation of proinflammatory signalling pathways [24] | Increased insulin and IGF levels [26] | Increased insulin and IGF levels [28] |
| Increased immune cell infiltration [27] | Increased immune cell infiltration [29] | Insulin resistance [18][25] | Insulin resistance [20,27] |
| Upregulation of WNT signalling [28] | Upregulation of WNT signalling [30] | Elevates leptin levels [29] | Elevates leptin levels [31] |
| Increased synthesis of arachidonic acid and PGE2 [30] | Increased synthesis of arachidonic acid and PGE2 [32] | Increases oestrogen and androgen levels [31][32] | Increases oestrogen and androgen levels [33,34] |
| Downregulation of response to antigen and mitogen stimulation [33][34] | Downregulation of response to antigen and mitogen stimulation [35,36] | Anti-apoptotic, promotes stemness [35] | Anti-apoptotic, promotes stemness [37] |
3. Innate Immunity in Obese Adipose Tissue
During weight gain, adipocytes increase the storage of lipids, resulting in structural changes, such as adipocyte hypertrophy, and adipocyte death. The mechanism of adipocyte death is still not clear, although it has been attributed to either inflammatory programmed cell death (pyroptosis) or necrosis [36][37][38,39]. Adipose tissue macrophages (ATMs) scavenge the debris of the necrotic adipocytes, which in turn activate ATMs via the initiation of inflammatory signalling pathways [38][39][40][41][40,41,42,43]. ATMs that are metabolically activated by fatty acids or inflammatory mediators that are released from necrotic adipocytes are recruited or proliferate in situ, and encircle adipocytes forming crown-like structures (CLSs) (Figure 1) [42][43][44,45].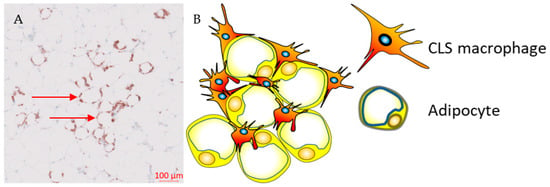
4. Adaptive Immunity in Obese Adipose Tissue
During the development of obesity, the composition of adaptive immune cells resident in adipose tissue changes, with an increase in the CD8+ to CD4+ T-cell ratio, but a reduction in the number of regulatory T-cells (Tregs) within the adipose tissue [48][50]. Both CD4+ and CD8+ T-cells play a vital role in the recruitment and activation of ATM via secretion of cytokines such as IFN-γ [27][29]. Flow-cytometric and immunohistochemical analyses demonstrated higher numbers of CD8+ effector T-cells and lower numbers of CD4+ helper T-cells in obese murine epididymal adipose tissue compared to lean mice on a normal diet [27][29]. In addition, CD8+ T-cells were found within CLS in obese epididymal adipose tissue whereas there was no association was found between CD4+ T-cells and CLS [27][29]. A time course evaluation of immune cells in adipose tissue in C57BL/6 mice during a high-fat diet showed that CD8+ T-cell infiltration preceded the recruitment of macrophages [27][29]. In contrast, the number of CD4+CD8- helper T-cells and CD4+CD25+FoxP3+ Tregs decreased, suggesting that CD8+ T-cell infiltration is a crucial event during inflammation in adipose tissue [27][29]. This was further validated by depleting CD8+ T-cells in C57BL/6 mice using anti-CD8 antibody, which resulted in reduction of M1-like macrophages and CLSs without affecting M2-like macrophages [27][29]. In addition, high-fat diet did not increase levels of IL-6 and TNF-α mRNA in CD8-deficient mice, whereas adoptive transfer of CD8+ T-cells into CD8-deficient mice increased M1-like macrophage infiltration [27][29]. These findings suggest that Tregs maintain immune homeostasis by suppressing inflammation induced by pro-inflammatory macrophages in adipose tissue under physiological conditions. CD8+ T-cell infiltration is required for adipose tissue inflammation in obesity as it precedes macrophage accumulation in adipose tissue and plays a vital role in macrophage polarisation and infiltration. Obesity-induced metabolic dysregulation may interfere in the interplay between macrophages and T-cell immune populations [27][29]. Differences in the immunophenotype of ATMs between non-obese and obese subjects may be attributed to their different immunometabolic functions influenced by metabolic stress and chronic inflammation, which is promoted by enlarged or necrotic adipocytes.5. Adipose Tissue Macrophages and Breast Cancer
CLSs are correlated with a proinflammatory environment and represent an index of WAT inflammation and metabolic dysregulation such as dyslipidemia, increased glucose and glycated hemoglobin (HbA1) levels [42][43][49][50][44,45,51,52]. Observational studies in patients with early breast cancer demonstrated that chronic systemic inflammation, as defined by elevated serum proinflammatory cytokines, is associated with CLSs, especially in individuals with obesity or who are overweight [49][51][52][51,53,54]. Previous reports have demonstrated an inconsistency in survival, with three out of five studies reporting improved outcomes (Table 2), which can be partly explained by biological and methodological heterogeneity [43][49][53][54][45,51,55,56]. ReseaOurchers' recent study showed that the presence of CLSs expressing the inhibitory FcγRIIB (CD32B) at the tumour border was associated with worse clinical outcomes in patients with HER2+ breast cancer treated with trastuzumab compared to trastuzumab-naïve patients [43][45]. The underlying biological mechanism that links the presence of CD32B+ CLS and resistance to trastuzumab is currently unclear and further investigation is required.| Study | Sample Size | CLS− (n) | CLS+ (n) | CLS Marker | RFS, CLS+ vs. CLS− | OS, CLS+ vs. CLS− | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Iyengar N (cohort 2) (2016) [55] | Iyengar N (cohort 2) (2016) [58] |
127 | 75 | 52 | CD68 | 1.83 | (1.07–3.13) | a,c | not reported | ||
| Koru-Sengul T | f (2016) [54] | (2016) [56] | 134 | NR | NR | CD40 | 5.87 (0.73–47.23) | a,c | 13.59 | (1.56–118.16) | a,c |
| Koru-Sengul T | f (2016) [54] | (2016) [56] | 134 | NR | NR | CD163 | 2.21 (0.65–7.59) | a,c | 2.42 (0.54–10.89) | a,c | |
| Koru-Sengul T | f (2016) [54] | (2016) [56] | 134 | NR | NR | CD206 | 1.17 (0.09–15.35) | a,c | 0.74 (0.04–15.55) | a,c | |
| Cha YJ (2018) | g [53] | [55] | 140 | 122 | 18 | CD163 | 105 (94–116) vs. 124 (118–131) | b,d | 105 (94–116) vs. 130 (124–136) | b,d | |
| Cha YJ (2018) | g [53] | [55] | 140 | 115 | 25 | CD68 | 106 (97–114) vs. 124 (117–131) | b,d | 106 (99–114) vs. 130 (124–136) | b,d | |
| Cha YJ (2018) | g [53] | [55] | 56 | 49 | 7 | CD68 | 76 (56–96) vs. 120 (108–132) | b,d,e | 79 (63–96) vs. 125 (114–136) | b,d,e | |
| Maliniak M (2020) [56] | Maliniak M (2020) [59] | 319 | 223 | 96 | CD68 | 1.05 (0.64–1.72) | a,c | 1.02 (0.55–1.87) | a,c | ||
| Birts C (2022) [43] | Birts C (2022) [45] | 117 | 47 | 61 | CD32B | 4.2 | (1.01–17.4) | a,c | not reported |
The role of adiposity and inflammation was also demonstrated in ER− BC. A systematic review and meta-analysis that included 13 observational studies of patients with triple negative BC (TNBC) with baseline BMI measurements, showed that BMI ≥ 25 was associated with worse disease-free and overall survival compared to patients with healthy BMI [74]. In a study of 1779 patients with primary invasive BC, patients with triple-negative disease had a 3-fold risk of being overweight and of having raised CRP compared to luminal A subjects [70]. Recent studies proposed potential mechanisms underlying the association between obesity and TNBC. These include the activation of Akt/mTOR signalling pathway by insulin, which is elevated in patients with obesity-induced insulin resistance [76]. Activation of the Akt/mTOR pathway is associated with aggressive molecular and glycolytic phenotypes that promote tumour growth in TNBC [77,78]. Secondly, obesity-mediated inflammation has been associated with activation-signalling pathways that are involved in tumour invasion and metastasis in TNBC [79]. Thirdly, this chronic inflammatory environment was reported to be correlated with reduced tumour-infiltrating immune cells both in a 4T1 TNBC model and in human triple negative tumours [80].
These findings suggest that hyperadiposity can induce WAT inflammation and metabolic dysregulation. In ER+ breast tumours, this is potentially mediated via a paracrine interaction between macrophages and preadipocytes, leading to elevated aromatase expression and secretion of pro-inflammatory adipokines in the breast adipose tissue in patients with high BMI [42][44]. In ER− BC obesity is positively associated with inflammatory and aggressive molecular phenotypes. In contrast, in HER2+ breast tumours, WAT inflammation can induce trastuzumab resistance via activation of MAPK or PI3K pathways [57][60].