Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Stamatis Gregoriou and Version 2 by Rita Xu.
Eccrine porocarcinoma (EPC) constitutes a rare malignant adnexal tumor, which accounts for about 0.005–0.01% of all cutaneous malignancies. It may develop de novo or arise from an eccrine poroma, after a latency period of years or even decades.
- porocarcinoma
- adnexal tumor
- skin appendages
1. Introduction
Adnexal malignant tumors of the eccrine sweat glands comprise an heterogenous group of rare cutaneous malignancies, which are generally considered fairly indolent, even though cases of aggressive disease course have been reported in the literature. Among them, eccrine porocarcinoma (EPC) constitutes the most common malignant adnexal tumor, which originates from the intraepidermal section of the excretory duct of the eccrine sweat gland, i.e., the acrosyringium, and accounts for about 0.005–0.01% of all cutaneous malignancies [1]. It was firstly described as “epidermotropic eccrine carcinoma” by Pinkus and Mehregan in 1963, while a few years later, Mishima and Morioka addressed the term “eccrine porocarcinoma”, which is now most frequently used and was included in the 2018 World Health Organization (WHO) classification of skin tumors [2][3][4][2,3,4]. This classification is based on clinical, histopathological and molecular genetic features of each appendageal carcinoma, classifying them into three types: apocrine-eccrine, follicular and sebaceous [2]. Other terms reported so far in the literature are “malignant eccrine poroma” or “malignant hidroacanthoma simplex”.
2. Pathogenesis—Risk Factors
The pathogenesis of EPC is not fully understood. It may develop de novo or arise from its benign counterpart, eccrine poroma, after a latency period of years or even decades [5][14]. This has been supported by published case series with long-term follow up, as well as the results of a clinicopathologic study of 69 cases reporting that 18% of EPCs demonstrated adjacent features of benign poroma [6][7][15,16]. Moreover, the presence of shared oncogenic gene fusions, i.e., hybrid genes formed by the translocation, inversion or deletion of two independent genes, has been observed in both EPC and eccrine poromas, accounting for the production of proteins with oncogenic functions [8][9][10][17,18,19]. Rarely, eccrine poromas may arise within nevus sebaceous and they should be considered in the differential diagnosis of secondary tumors developing on sebaceous nevi [11][20]. Further reported risk factors comprise chronic ultraviolet (UV) exposure, as well as immunosuppression, while the occurrence of EPC in pre-damaged skin (radiotherapy, trauma) has also been described [12][13][21,22]. Although the precise mechanisms behind tumorigenesis in EPC are yet to be elucidated, published studies support the involvement of implicating factors, such as specific oncogenic drivers, including epidermal growth factor receptor (EGFR), tumor protein p53 (TP53) and cyclin-dependent kinase inhibitor 2A (CDKN2A), as well as signaling pathways, including the mitogen-activated protein kinase (MAPK) pathway, on EPC development [14][15][16][23,24,25]. Merilainen et al. investigated the presence of Merkel cell polyoma virus (MCPyV) in eccrine poromas and EPC by means of immunohistochemistry and quantitative polymerase chain reaction (PCR), to evaluate if MCPyV infection may drive the oncogenesis of EPC, as in MCC. The results indicated that MCPyV may not serve as an oncogenic driver for EPC [17][26]. In another study, Puttonen et al. investigated the distribution and significance of tumor infiltrating lymphocytes (TILs) in the EPC microenvironment, assuming that an altered immunological environment could explain the involvement of chronic UV radiation in the tumorigenesis of EPC. Unlike the known association of TILs with positive prognosis in various skin tumors, such as melanoma and MCC, the authors observed a negative correlation between UV damage and TIL density in EPCs, supporting the immunosuppressive effect of UV radiation on the microenvironment of these tumors [18][27]. To further elucidate the EPC biology, Denisova et al. investigated the mutational landscape of EPC by performing whole-exome sequencing on 14 EPCs and matched healthy surrounding tissue. The authors observed a high overall median mutation rate attributed to UV exposure, with the most common mutation being the TP53, a cell cycle regulator and tumor suppressor gene, suggesting that inactivation of the p53 pathway plays a significant role in both tumorigenesis and tumor progression. Moreover, the identified mutational processes were found to be comparable to those observed in melanoma, BCC and SCC, suggesting common mechanisms of tumorigenesis [19][28]. In agreement with this study, Westphal et al. assessed molecular pathway alterations in EPC and revealed high overall mutational burden, mostly attributable to UV-induced mutational signatures, while they found altered protein expression of p53, as well as significant expression of epidermal growth factor receptor (EGFR) protein and programmed death-ligand-1 (PD-L1) [20][29]. The functional relevance of these findings was supported by reports of EPCs responding to anti-PD-1 and anti-EGFR therapy, suggesting that these pathways may serve as oncogenic drivers in EPC [21][22][30,31].3. Diagnosis
Diagnosis of EPC is challenging, as it is characterized by variable and non-specific clinical and histopathological findings, leading to diagnostic delay in most cases. Interestingly, the mean interval between tumor development and diagnosis has been reported to be five to nine years, but it may vary from days to even 60 years, according to the published literature [6][7][23][24][25][5,15,16,32,33]. Clinical differential diagnoses comprise benign or malignant lesions, such as pyogenic granuloma, seborrheic keratosis, squamous cell carcinoma (SCC), basal cell carcinoma (BCC), Bowen’s disease, Merkel cell carcinoma (MCC), Paget disease, lymphoma, hidradenoma, etc. Overall, diagnosis should be based on the combination of clinical, dermoscopical, histopathological and immunohistochemical findings.3.1. Clinical Presentation
The clinical presentation of EPC is highly variable. Usually, it manifests as an erythematous, violaceous nodular lesion or, more rarely, as a polypoid plaque of violet or erythematous color, growing over weeks to months (Figure 1). It may be asymptomatic or present with itching, ulceration and spontaneous bleeding. The latter should be clinically regarded as signs of malignant transformation, and it has been found to represent a significantly worsening prognostic factor [5][26][27][6,14,34]. The tumor size at time of diagnosis has been reported to range from 1–130 mm, having a mean diameter of 23.88 mm [28][7]. Behbahani et al. sought to correlate the tumor stage with the disease outcome. Except for the strong association of metastatic disease with worse prognosis, a larger tumor size was also independently associated with decreased overall survival [28][7]. This also applies to non-melanoma and non-Merkel cell skin cancers, in which a tumor diameter greater than two centimeters is correlated with worse prognosis according to the guidelines of the American Joint Committee on Cancer (AJCC) [29][35].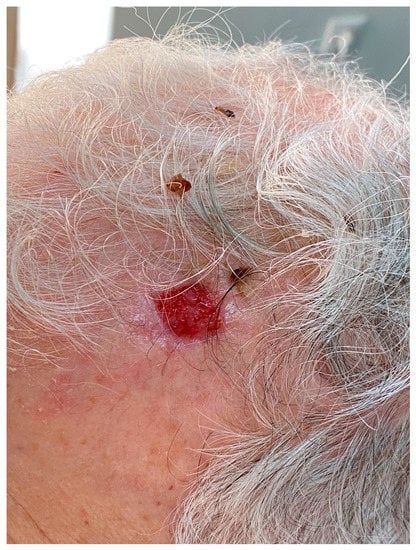
Figure 1.
Clinical presentation of eccrine porocarcinoma in a 75-year-old patient, showing an ulcerated lesion of 15 mm in diameter on the left temporal area.
3.2. Imaging
There have only been a few reports addressing the dermoscopic findings of EPC. An atypical vascular pattern with a diffuse arrangement of focused and unfocused polymorphous vessels as well, as pink-whitish ovoid areas, constitute the most commonly reported features (Figure 2) [33][39]. However, cases exhibiting fine scaling and erosions, as well as structureless pink-whitish areas with no apparent vasculature, have also been described [34][40]. Pinheiro et al. provided reflectance confocal microscopy (RCM) findings of a relapsing EPC. Aggregates of poroid cells clustered in nests, surrounded by elongated and tortous canalicular vessels and a dark stroma were observed. In addition, roundish dark structures corresponding to areas of ductal differentiation within the nests were also seen [33][39].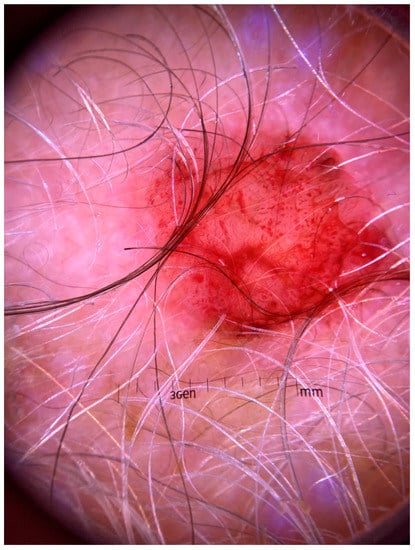
Figure 2. Dermoscopic picture of eccrine porocarcinoma exhibiting an atypical vascular pattern with polymorphous vessels and pink-whitish ovoid areas.
3.3. Histopathology
The histopathological characteristics of EPC in hematoxylin and eosin staining are diverse and may pose difficulties in the histopathological differential diagnosis of EPC, mainly from SCC. In most cases, large poromatous basaloid epithelial cells exhibiting ductal differentiation and cytologic atypias are observed [25][33]. This cytologic pleomorphism, along with the increased mitotic activity, tumor necrosis and infiltrative growth pattern, comprise histopathological markers, which enable the differentiation of EPC from benign eccrine poroma. Interestingly, EPC cells may exhibit squamous cell, clear cell and spindle cell differentiation or even melanocyte colonization [36][42]. In a meta-analysis of 120 EPCs, 25% and 23.4% of cases showed squamous and clear cell differentiation, respectively, while in another study of 33 cases, squamous cell differentiation was observed in 42% and melanocyte colonization in 21% of EPCs [25][26][6,33]. Duct formation constitutes another significant indicator for the diagnosis of EPC and differentiation from SCC. Histopathological studies have shown that the majority of cases exhibit mature ducts, characterized by cuboidal epithelial cells with an eosinophilic cuticle, even though poorly differentiated EPCs may only show less-developed ducts, which should not be overlooked [6][31][15,37]. Further histopathological findings that have been observed in EPC cases are the comedonecrosis; i.e., tumor cells containing central necrosis with an inflammatory reaction similar to a comedone, and a bowenoid pattern, characterized by pleomorphic cells and some multinucleated monster cells (Figure 3) [25][31][33,37].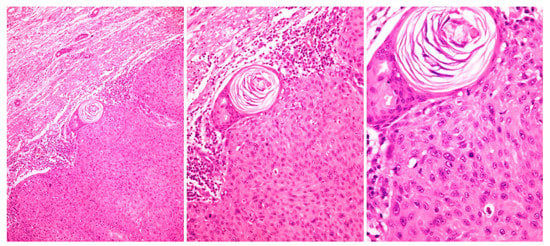
Figure 3. Histopathological picture of eccrine porocarcinoma exhibiting poromatous basaloid epithelial cells and duct formation (hematoxylin and eosin staining, 40×, 100×, 200×).
