Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Conner Chen and Version 1 by Maria felice Brizzi.
The correlation between diabetes mellitus and infectious diseases is widely recognized. DM patients are characterized by the impaired function of the immune system. This translates into the occurrence of a variety of infections, including urinary tract, skin and surgical site infections, pneumonia, tuberculosis, and, more recently, SARS-CoV-2.
- Diabetes Mellitus
- infection
- tuberculosis
1. Introduction
Diabetes mellitus (DM) is a chronic disease characterized by abnormal blood glucose levels resulting from impaired insulin action and/or insulin secretion, usually both [1]. DM can be classified as type 1 diabetes, type 2, and gestational diabetes [2]. According to the 10th edition of the IDF Diabetes Atlas, 536.6 million people are currently diagnosed with DM worldwide with a prevalence of 10.5%, and this number is expected to increase to 783.2 million in 2045. The prevalence of DM increases with age, while the incidence of type 2 diabetes is expected to decrease or remain stable in high-income countries [3]. In addition to macro- and microvascular complications, an increased risk of infection is commonly associated with DM [4]. Individuals with DM are at a greater risk of hospitalization and mortality due to viral, bacterial, and fungal infections [5]. However, recent evidence indicates that DM does not represent a significant risk factor for poor survival in patients with sepsis, regardless of intensive care unit (ICU) admission [6,7][6][7]. Nevertheless, DM and sepsis remain important causes of morbidity and mortality worldwide, and DM patients represent the largest population experiencing post-sepsis complications [8]. This is mainly due to immunosuppression and uncontrolled hyperglycemia. In fact, high blood glucose impairs innate and adaptive immunity through various mechanisms [9].
Poor glycemic control increases the risk for skin, bone, eye, ear, gastrointestinal, urinary tract, and respiratory infections [10]. Moreover, impaired healing of diabetic wounds, which affects approximately 25% of all DM patients, is associated with an increased risk of limb amputation, thereby representing a crucial economic and psychosocial issue [11]. Uncommon life-threatening infections are also more frequent among DM patients. These include invasive otitis externa, rhino-cerebral mucormycosis, and emphysematous infections of the gall bladder, kidney, and urinary bladder [12]. Evidence has also been provided on the prevalence of drug resistance in DM patients [13], however, an increased prevalence of resistance to commonly used antibiotics in DM patients is still debated [14]. More recently, several infections have been supposed to rely on newly introduced therapies. As an example, sodium-glucose co-transport 2 inhibitors (SGLT2-i) are associated with the occurrence of urinary and genital tract infections [15,16,17][15][16][17]. Moreover, among infections commonly associated with hospitalization, including urinary tract and skin infections, pneumonias, and surgical infections, the presence of DM confers an increased risk (Figure 1).
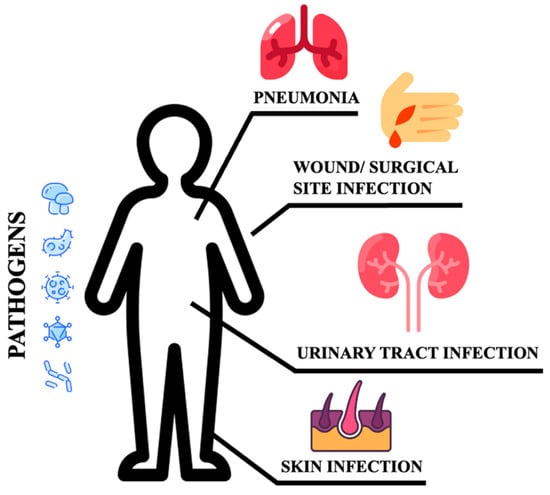
Figure 1.
Sites of infections.
These patients are more susceptible to urinary tract infections than non-DM individuals. They have a higher risk of developing asymptomatic bacteriuria, cystitis, acute pyelonephritis, and complications such as emphysematous pyelonephritis [9,18][9][18]. Bacteria isolated from DM patients with an urinary tract infection (UTI) do not differ from those found in non-DM patients with complicated UTI. E. coli are the most common pathogens, while Klebsiella spp., Enterobacter spp., Proteus spp., Group B Streptococci, and Enterococcus faecalis are the most frequently isolated pathogens [19]. DM patients are more likely to develop skin and soft tissue infections, including cellulitis and osteomyelitis [14]. Staphylococcus aureus and Pseudomonas [20] are the most common isolated gram-positive and negative bacteria respectively. Methicillin-resistant Staphylococcus aureus (MRSA) and other antibiotic-resistant pathogens generally account for skin and soft tissue infections in the diabetic foot compared to other tissue sites and populations [14]. DM increases the susceptibility to different respiratory infections, thereby representing an independent risk factor for lower respiratory tract infections, particularly influenza and pneumonia [20,21][20][21]. More importantly, DM individuals are at higher risk of pulmonary infections caused by microorganisms such as Mycobacterium tuberculosis, Staphylococcus aureus, gram-negative bacteria, and fungi, and have a high risk of hospitalization upon influenza or flu-like infections. Additionally, infections caused by Streptococcus pneumonia or influenza virus are characterized by high morbidity and mortality rates [20]. Furthermore, pulmonary infections in elderly DM patients remain occult. Advanced age, comorbidities (senile dementia, hypothyroidism), and prolonged bed rest are indeed considered independent risk factors for occult pneumonia [22], resulting in long-term hospitalization and increased mortality. DM also increases community-acquired pneumonia (CAP) compared to non-diabetic individuals, and gram-negative bacteria such as K. pneumoniae and S. aureus are much more commonly isolated [9]. The susceptibility to fungal infections caused by Mucorales has been estimated at 75% in this population. Aspergillus is an additional microorganism causing infections in these patients [23]. It has been reported that S. pneumoniae, Enterobacter, K. pneumoniae, Serratia, E. coli, S. aureus, Proteus, and Haemophilus influenzae are the most common bacteria causing hospital-acquired pneumonia (HAP) during the first four days of hospitalization, while Acinetobacter, MRSA, E. coli, L. pneumophila, Pseudomonas aeruginosa, and K. pneumonia are more prevalent after day five [20,24][20][24]. Tuberculosis infections are also frequent and increased with a mortality rate corresponding to 50% [25]. Hyperglycemia also predisposes to superinfection of the surgical site following surgery (SSI), and the association between pre- and post-surgery hyperglycemia remains a significant risk factor for SSI [26]. In conclusion, the risk and mortality associated with infectious diseases are high in DM patients, implying that infections should be considered among the most common DM complications [12].
2. Immunity Impairment in DM
Several pre-clinical and clinical studies have revealed a significant defect in both innate and adaptive immunity. Although some mechanisms are glycemia-independent [27], most of them rely on hyperglycemia and its metabolic effects, such as non-enzymatic glycation, generation of reactive oxygen species, and hyperactivity of the polyol pathway [28].2.1. Neutrophils
Neutrophils are recognized as key elements to counteract infection, and DM impairs their recruitment as well as their killing capability. Several mechanisms also account for the dysfunction of adhesion, rolling, and chemotaxis [29,30,31][29][30][31]. CXCR2, a chemokine receptor expressed on neutrophils, was found downregulated during sepsis, thereby impairing neutrophil recruitment [27,32][27][32]. Moreover, since CXCR2 also controls the expression of the intracellular adhesion molecule-1 (ICAM) on endothelial cells (ECs), its decreased expression further weakens neutrophil recruitment at the inflammatory site [33]. Regarding phagocytosis, the main recognized abnormality is related to C3-mediated opsonization owing to complement glycation [34]. Both intracellular and extracellular killing mechanisms (involving the production of intracellular ROS [35[35][36],36], enzymatic degranulation [37], and inhibition of neutrophil extracellular traps (NET) formation [38]) are impaired in the hyperglycemic condition.2.2. Macrophages
Chronic hyperglycemia also weakens macrophage adhesion and chemotaxis, antibacterial activity, and phagocytosis, damaging both the FCy receptor and the complement cascade [39,40][39][40]. Furthermore, the low-grade inflammation caused by hyperglycemia, insulin resistance, and obesity promotes macrophage differentiation towards their anti-inflammatory M2 phenotype, activating the IL-6 signaling pathway [41]. The homeostatic action of IL-6 in limiting inflammation represents a relevant impediment to the control of infections [42]. Figure 2 summarizes the most relevant DM-associated immune cell impairment.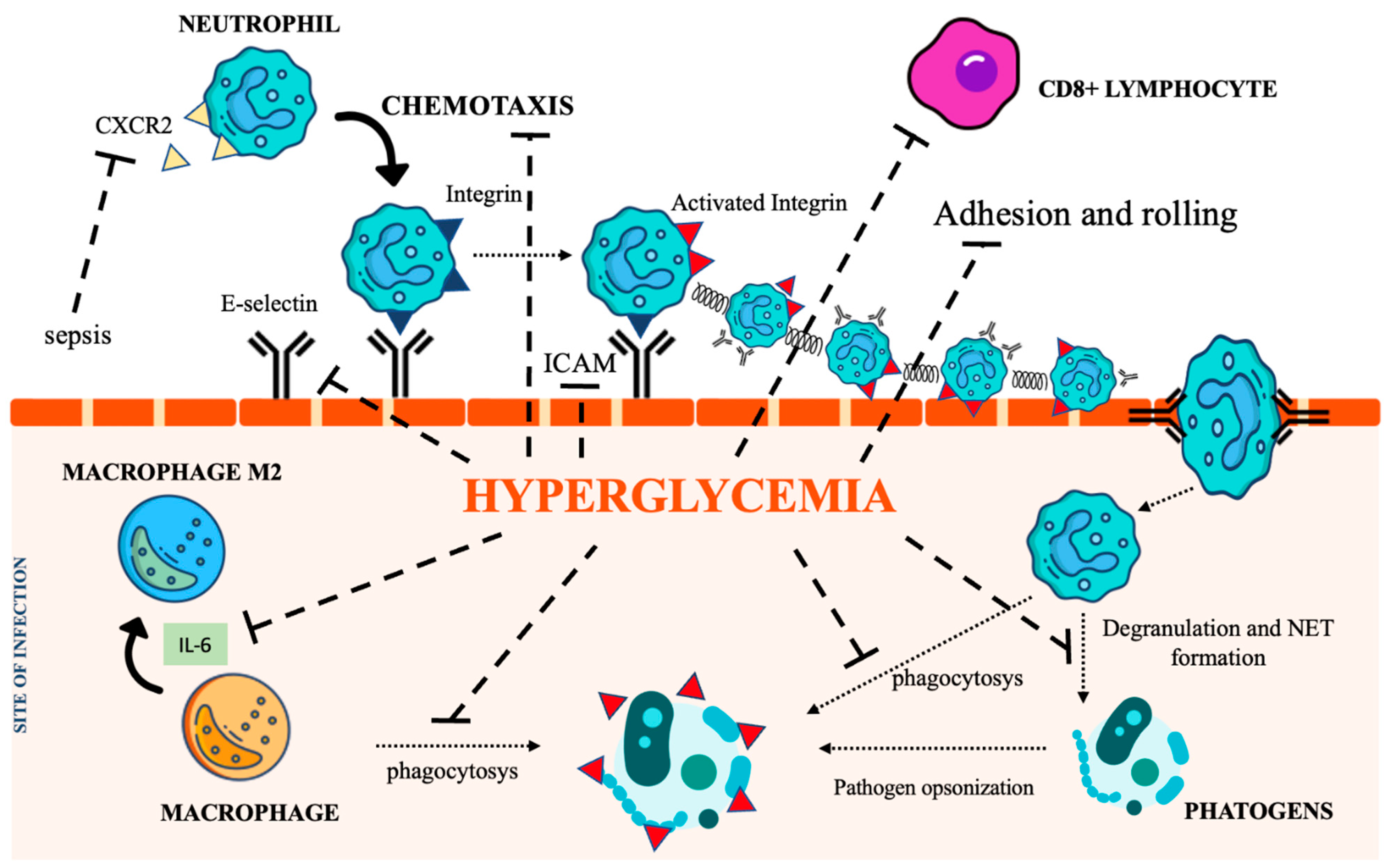
Figure 2. The immunological pathways under the control of hyperglycemia. CXCR2 (C-X-C Motif Chemokine Receptor 2), IL-6 (interleukin 6), NET (Neutrophil extracellular traps), ICAM (intercellular adhesion molecules). Icon design by icons8.
2.3. Natural Killer Cells
Hyperglycemia-induced oxidative stress also compromises natural killer (NK) cell activity [43]. Importantly, a strong inverse linear relationship between their activity and HbA1c level has been reported [44,45][44][45].2.4. Adaptive Immunity
The impact of DM on adaptive immunity is still debated since a few studies on T lymphocyte dysfunction have reported conflicting results. Preclinical studies have shown that the decreased expression of ICAM and E selectin in ECs impairs the recruitment of cytotoxic CD8+ T lymphocytes to sites of infection, resulting in more severe disease [46]. Dysregulation of the complement cascade has also been reported. As mentioned earlier, non-enzymatic glycation of C3 and C4 decreases opsonization [34]. In addition, the glycation of immunoglobulins can inhibit antigen recognition [47]. In conclusion, DM significantly impacts the immune system, resulting in a higher risk of infections. Several studies have shown defects in both innate and adaptive immunity. Hyperglycemia and insulin resistance are the most relevant factors contributing to the dysfunction of the immune system, which mainly involves neutrophils, macrophages, and natural killer cells.References
- Petersmann, A.; Müller-Wieland, D.; Müller, U.A.; Landgraf, R.; Nauck, M.; Freckmann, G.; Heinemann, L.; Schleicher, E. Definition, Classification and Diagnosis of Diabetes Mellitus. Exp. Clin. Endocrinol. Diabetes 2019, 127, S1–S7.
- Aschner, P.; Basit, A.; Fawwad, A.; Guariguata, L.; James, S.; Karuranga, S.; Malanda, B.; Mbanya, J.C.; O’neill, S.; Ogle, G.; et al. IDF Guide for Diabetes Epidemiology Studies IDF Guide for Diabetes Epidemiology Studies i Acknowledgements Authors. Available online: https://diabetesatlas.org/idf-guide-for-epidemiology-studies/ (accessed on 28 January 2023).
- IDF Diabetes Atlas 10th Edition. Available online: www.diabetesatlas.org (accessed on 28 January 2023).
- Schuetz, P.; Castro, P.; Shapiro, N.I. Diabetes and Sepsis: Preclinical Findings and Clinical Relevance. Diabetes Care. 2011, 34, 771–778.
- Erener, S. Diabetes, Infection Risk and COVID-19. Mol. Metab. 2020, 39, 101044.
- Akinosoglou, K.; Kapsokosta, G.; Mouktaroudi, M.; Rovina, N.; Kaldis, V.; Stefos, A.; Kontogiorgi, M.; Giamarellos-Bourboulis, E.; Gogos, C. Diabetes on Sepsis Outcomes in Non-ICU Patients: A Cohort Study and Review of the Literature. J Diabetes Complicat. 2021, 35, 107765.
- Jiang, L.; Cheng, M. Impact of Diabetes Mellitus on Outcomes of Patients with Sepsis: An Updated Systematic Review and Meta-Analysis. Diabetol. Metab. Syndr. 2022, 14, 1–17.
- Costantini, E.; Carlin, M.; Porta, M.; Brizzi, M.F. Type 2 Diabetes Mellitus and Sepsis: State of the Art, Certainties and Missing Evidence. Acta Diabetol. 2021, 58, 1139–1151.
- Akash, M.S.H.; Rehman, K.; Fiayyaz, F.; Sabir, S.; Khurshid, M. Diabetes-Associated Infections: Development of Antimicrobial Resistance and Possible Treatment Strategies. Arch. Microbiol. 2020, 202, 953–965.
- Critchley, J.A.; Carey, I.M.; Harris, T.; DeWilde, S.; Hosking, F.J.; Cook, D.G. Glycemic Control and Risk of Infections among People with Type 1 or Type 2 Diabetes in a Large Primary Care Cohort Study. Diabetes Care 2018, 41, 2127–2135.
- Burgess, J.L.; Wyant, W.A.; Abujamra, B.A.; Kirsner, R.S.; Jozic, I. Diabetic Wound-Healing Science. Medicina 2021, 57, 1072.
- Shah, B.R.; Hux, J.E. Quantifying the Risk of Infectious Diseases for People with Diabetes. Available online: http://diabetesjournals.org/care/article-pdf/26/2/510/648543/dc0203000510.pdf (accessed on 28 January 2023).
- Toniolo, A.; Cassani, G.; Puggioni, A.; Rossi, A.; Colombo, A.; Onodera, T.; Ferrannini, E. The Diabetes Pandemic and Associated Infections: Suggestions for Clinical Microbiology. Rev. Res. Med. Microbiol. 2019, 30, 1–17.
- Polk, C.; Sampson, M.M.; Roshdy, D.; Davidson, L.E. Skin and Soft Tissue Infections in Patients with Diabetes Mellitus. In Infectious Disease Clinics of North America; W.B. Saunders: Philadelphia, PA, USA, 2021; pp. 183–197.
- Chowdhury, T.; Gousy, N.; Bellamkonda, A.; Dutta, J.; Zaman, C.F.; Zakia, U.B.; Tasha, T.; Dutta, P.; Deb Roy, P.; Gomez, A.M.; et al. Fournier’s Gangrene: A Coexistence or Consanguinity of SGLT-2 Inhibitor Therapy. Cureus 2022, 14, e27773.
- Ellegård, L.; Prytz, M. Fournier’s Gangrene under SGLT-2 Inhibitor Therapy: A Literature Review and Case Report. Int. J. Surg. Case Rep. 2020, 77, 692–694.
- Zhang, X.L.; Zhu, Q.Q.; Chen, Y.H.; Li, X.L.; Chen, F.; Huang, J.A.; Xu, B. Cardiovascular Safety, Long-Term Noncardiovascular Safety, and Efficacy of Sodium-Glucose Cotransporter 2 Inhibitors in Patients with Type 2 Diabetes Mellitus: A Systemic Review and Meta-Analysis with Trial Sequential Analysis. J. Am. Heart Assoc. 2018, 7, e007165.
- Kamei, J.; Yamamoto, S. Complicated Urinary Tract Infections with Diabetes Mellitus. J. Infect. Chemother. 2021, 27, 1131–1136.
- Nitzan, O.; Elias, M.; Chazan, B.; Saliba, W. Urinary Tract Infections in Patients with Type 2 Diabetes Mellitus: Review of Prevalence, Diagnosis, and Management. Diabetes Metab. Syndr. Obes. Targets Ther. 2015, 8, 129–136.
- Klekotka, R.B.; Mizgała, E.; Król, W. The Etiology of Lower Respiratory Tract Infections in People with Diabetes. Pneumonol. I Alergol. Pol. 2015, 83, 401–408.
- Knapp, S. Diabetes and Infection: Is There a Link?—A Mini-Review. Gerontology 2013, 59, 99–104.
- Hua, J.; Huang, P.; Liao, H.; Lai, X.; Zheng, X. Prevalence and Clinical Significance of Occult Pulmonary Infection in Elderly Patients with Type 2 Diabetes Mellitus. Biomed. Res. Int. 2021, 2021, 3187388.
- Jones, R.N. Microbial Etiologies of Hospital-Acquired Bacterial Pneumonia and Ventilator-Associated Bacterial Pneumonia. Clin. Infect. Dis. 2010, 51, S81–S87.
- Zhang, X.; Wang, R.; Di, X.; Liu, B.; Liu, Y. Different Microbiological and Clinical Aspects of Lower Respiratory Tract Infections between China and European/American Countries. J. Thorac. Dis. 2014, 6, 134–142.
- Casqueiro, J.; Casqueiro, J.; Alves, C. Infections in Patients with Diabetes Mellitus: A Review of Pathogenesis. Indian J. Endocrinol. Metab. 2012, 16, 27.
- Martin, E.T.; Kaye, K.S.; Knott, C.; Nguyen, H.; Santarossa, M.; Evans, R.; Bertran, E.; Jaber, L. Diabetes and Risk of Surgical Site Infection: A Systematic Review and Meta-Analysis. Infect. Control. Hosp. Epidemiol. 2016, 37, 88–99.
- Carlos, D.; Spiller, F.; Souto, F.O.; Trevelin, S.C.; Borges, V.F.; de Freitas, A.; Alves-Filho, J.C.; Silva, J.S.; Ryffel, B.; Cunha, F.Q. Histamine H 2 Receptor Signaling in the Pathogenesis of Sepsis: Studies in a Murine Diabetes Model. J. Immunol. 2013, 191, 1373–1382.
- Alba-Loureiro, T.C.; Munhoz, C.D.; Martins, J.O.; Cerchiaro, G.A.; Scavone, C.; Curi, R.; Sannomiya, P. Neutrophil Function and Metabolism in Individuals with Diabetes Mellitus. Volume 40. Available online: www.bjournal.com.br (accessed on 28 January 2023).
- Andersen, B.; Goldsmith, G.H.; Spagnuolo, P.J. Neutrophil Adhesive Dysfunction in Diabetes Mellitus: The Role of Cellular and Plasma Factors. J. Lab. Clin. Med. 1988, 111, 267–274.
- Delamaire, M.; Maugendre, D.; Moreno, M.; le Goff, M.C.; Allannic, H.; Genetet, B. Impaired Leucocyte Functions in Diabetic Patients. Diabet. Med. 1997, 14, 29–34.
- Tater, D.; Tepaut, B.; Bercovici, J.P.; Youinou, P. Polymorphonuclear cell derangements in type i diabetes. Horm. Metab. Res. 1987, 19, 642–647.
- Rios-Santos, F.; Alves-Filho, J.C.; Souto, F.O.; Spiller, F.; Freitas, A.; Lotufo, C.M.C.; Soares, M.B.P.; dos Santos, R.R.; Teixeira, M.M.; Cunha, F.D.Q. Down-Regulation of CXCR2 on Neutrophils in Severe Sepsis Is Mediated by Inducible Nitric Oxide Synthase-Derived Nitric Oxide. Am. J. Respir. Crit. Care Med. 2007, 175, 490–497.
- Vieira, S.M.; Lemos, H.P.; Grespan, R.; Napimoga, M.H.; Dal-Secco, D.; Freitas, A.; Cunha, T.M.; Verri, W.A.; Souza, D.A.; Jamur, M.C.; et al. A Crucial Role for TNF-α in Mediating Neutrophil Influx Induced by Endogenously Generated or Exogenous Chemokines, KC/CXCL1 and LIX/CXCL5: RESEARCH PAPER. Br. J. Pharmacol. 2009, 158, 779–789.
- Hair, P.S.; Echague, C.G.; Rohn, R.D.; Krishna, N.K.; Nyalwidhe, J.O.; Cunnion, K.M. Hyperglycemic Conditions Inhibit C3-Mediated Immunologic Control of Staphylococcus Aureus. J. Transl. Med. 2012, 10, 1–16.
- Chao, W.C.; Yen, C.L.; Wu, Y.H.; Chen, S.Y.; Hsieh, C.Y.; Chang, T.C.; Ou, H.Y.; Shieh, C.C. Increased Resistin May Suppress Reactive Oxygen Species Production and Inflammasome Activation in Type 2 Diabetic Patients with Pulmonary Tuberculosis Infection. Microbes Infect. 2015, 17, 195–204.
- Shah, S.V.; Wallin, J.D.; Eilenj, S.D. Chemiluminescence and superoxide anion production by leukocytes from diabetic patients. J. Clin. Endocrinol. Metab. 1983, 57, 402–409.
- Stegenga, M.E.; van der Crabben, S.N.; Blümer, R.M.; Levi, M.; Meijers, J.C.; Serlie, M.J.; Tanck, M.W.; Sauerwein, H.P.; van der Poll, T. Hyperglycemia Enhances Coagulation and Reduces Neutrophil Degranulation, Whereas Hyperinsulinemia Inhibits Fibrinolysis during Human Endotoxemia. Blood J. Am. Soc. Hematol. 2008, 112, 82–89.
- Joshi, M.B.; Lad, A.; Bharath Prasad, A.S.; Balakrishnan, A.; Ramachandra, L.; Satyamoorthy, K. High Glucose Modulates IL-6 Mediated Immune Homeostasis through Impeding Neutrophil Extracellular Trap Formation. FEBS Lett. 2013, 587, 2241–2246.
- Restrepo, B.I.; Twahirwa, M.; Rahbar, M.H.; Schlesinger, L.S. Phagocytosis via Complement or Fc-Gamma Receptors Is Compromised in Monocytes from Type 2 Diabetes Patients with Chronic Hyperglycemia. PLoS ONE 2014, 9, e92977.
- Pavlou, S.; Lindsay, J.; Ingram, R.; Xu, H.; Chen, M. Sustained High Glucose Exposure Sensitizes Macrophage Responses to Cytokine Stimuli but Reduces Their Phagocytic Activity. BMC Immunol. 2018, 19, 24.
- Liu, H.F.; Zhang, H.J.; Hu, Q.X.; Liu, X.Y.; Wang, Z.Q.; Fan, J.Y.; Zhan, M.; Chen, F.L. Altered Polarization, Morphology, and Impaired Innate Immunity Germane to Resident Peritoneal Macrophages in Mice with Long-Term Type 2 Diabetes. J. Biomed. Biotechnol. 2012, 2012, 867023.
- Mauer, J.; Chaurasia, B.; Goldau, J.; Vogt, M.C.; Ruud, J.; Nguyen, K.D.; Theurich, S.; Hausen, A.C.; Schmitz, J.; Brönneke, H.S.; et al. Signaling by IL-6 Promotes Alternative Activation of Macrophages to Limit Endotoxemia and Obesity-Associated Resistance to Insulin. Nat. Immunol. 2014, 15, 423–430.
- Zhang, K.; Kaufman, R.J. From Endoplasmic-Reticulum Stress to the Inflammatory Response. Nature 2008, 454, 455–462.
- Whalen, M.M. Inhibition of Human Natural Killer Cell Function in Vitro by Glucose Concentrations Seen in Poorly Controlled Diabetes. Cell. Physiol. Biochem. 1997, 7, 53–60.
- Kim, J.H.; Park, K.; Lee, S.B.; Kang, S.; Park, J.S.; Ahn, C.W.; Nam, J.S. Relationship between Natural Killer Cell Activity and Glucose Control in Patients with Type 2 Diabetes and Prediabetes. J. Diabetes Investig. 2019, 10, 1223–1228.
- Kumar, M.; Roe, K.; Nerurkar, P.V.; Orillo, B.; Thompson, K.S.; Verma, S.; Nerurkar, V.R. Reduced Immune Cell Infiltration and Increased Pro-Inflammatory Mediators in the Brain of Type 2 Diabetic Mouse Model Infected with West Nile Virus. 2014. Available online: http://www.jneuroinflammation.com/content/11/1/80 (accessed on 28 January 2023).
- Lapolla, A.; Tonani, R.; Fedele, D.; Garbeglio, M.; Senesi, A.; Seraglia, R.; Favretto, D.; Traldi, P. Non-Enzymatic Glycation of IgG: An In Vivo Study. Horm. Metab. Res. 2002, 34, 260–264.
More
