Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Peter Mikuš and Version 2 by Lindsay Dong.
There are over fifty examples in which the inner coordination spheres about the Pt(II) atoms of the Pt(η3-X3L)(PL) type are formed by variable combinations of donor atoms of tridentate ligands. Each η3-ligand creates two metallocyclic rings. The complexes based on membered metallocyclic rings can be divided into four groups: 1. 6+6-Membered Metallocyclic Rings, 2. 6+5-Membered Metallocyclic Rings, 3. 5+6-Membered Metallocyclic Rings, and 4. 5+5-Membered Metallocyclic Rings.
- structure
- Pt(η3-X3L)(PL)
- distortion
- trans-effect
1. 6+6-Membered Metallocyclic Rings
There are only three examples in which a η3-ligand creates such rings (Table 1). In [Pt(η3-C22H11F6N3O2–O1,N1,O2)(PPh3)] (at 173 K) [1][4], the η3-ligand forms a metallocyclic ring of the O1C3N1C3O2 type with common ligating N1 atoms. The values of the chelate L-Pt-L angles are 90.6° (O1-Pt-N1) and 90.2° (N1-Pt-O2). The O1C2NN1C3O2 type with the respective chelate angles of 88.2° (O1-Pt-N1) and 90.0° (N1-Pt-O2) was found in [Pt(η3-C14H10N2O3–O1,N1,O2)(PPh3)] (at 150 K) [2][5]. The remaining L-Pt-L angles open in the following order (mean values): 88.1° (O2-Pt-P) < 89.0° (O1-Pt-P) < 176.0° (N1-Pt-P) < 177.7° (O1-Pt-O2). The monodentate PPh3 displayed square-planar geometry about each Pt(II) atom. The Pt-L bond distance increased in the following order (mean values): 1.995 Å (Pt-O1 trans to O2) < 1.996 Å (Pt-O2) < 2.010 Å (Pt-N1) < 2.254 Å (Pt-P).
Table 1.
Structural data for Pt(η
3
-X
3
)(Y) derivatives.
a
—6+6-membered metallocyclic rings.
| Complex | Chromophore Chelate Rings τ | 4 | b | Pt -L | c | (Å) |
L-Pt-L | c |
|---|
1
,S
1
)(PPh
)] toluene [8], [Pt(η
3
-C
9
H
9
N
3
OS–O
1
,N
1
,S
1
)(PPh
3
)] (at 100 K) (
) [9], and [Pt(η
3
-C
18
H
16
N
2
OS
2
–O
1
,N
1
,S
1
)(PPh
)] [7] (
Table 2
). In each of them, the η
3
-ligand creates six- and five-membered metallocyclic rings with a common ligating N
1
atom of the O
1
C
3
N
1
NCS
1
type. The values of the respective chelate angles (mean values) are 92.3° (O
1
-Pt-N
1
) and 84.6° (N
1
-Pt-S
1
). The remaining L-Pt-L bond angles open in the following order (mean values): 90.7° (O
1
-Pt-P) < 92.4° (S
1
-Pt-P) < 175.8° (N
1
-Pt-P) < 175.9° (O
1
-Pt-S
1
). Interestingly, the mean values of both trans-O
1
-Pt-S
1
and N
1
-Pt-P angles are equal. The Pt-L bond distance increases (mean values) in the following order: 2.028 Å (Pt-O
1
trans to S
1
) < 2.035 Å (Pt-N
1
trans to P) < 2.244 Å (Pt-S
1
) < 2.259 Å (Pt-P).

Table 2. Structural data for Pt(η3-X3)(Y) derivatives. a—6+5-membered metallocyclic rings.
| Complex | Chromophore Chelate Rings τ | 4 | b | Pt -L | c | (Å) (°) |
L-Pt-L Ref. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| c | (°) | Ref. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| [Pt(η | 3 | -C | 22 | H | 11 | F | 6 | N | 3 | O | 2 | -O | 1 | ,N | 1 | ,O | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| [Pt(η | 32 | )(PPh | 3 | )] (at 173 K) |
-C | 16 | H | 14 | N | 2 | OS | 2 | -O | 1 | ,N | 1 | ,SPtO | 1 | N | 1 | O | 2 | P (O | 1 | C | 3 | N | 1 | C | 3 | O | 2 | ) 0.032 |
1 | )(PPh | 3 | )] | PtO | 1 | N | 1 | S | 1 | P (O | 1 | C | 3 | N | 1 | NCS | 1 | ) 0.016O | 1 | 1.994(2) N | 1 | 2.021(2) O | 2 | 2.004(2) P 2.256(2) |
O | 1 | 1.992 N | 1 | 2.034 S | 1 | 2.245 P 2.258O | 1 | ,N | 1 | 90.6 | d | N | 1 | ,O | 2 | 90.2 | d | O | 1 | ,O | 2 | 179.0 O | 1 | ,P 90.6 O | 2 | ,P 87.5 N | 1 | ,P 177.0 | O | 1 | ,N | 1 | 91.2 | d | N | 1 | ,S | 1 | 85.0 | e | O | 1 | ,S | 1 | 176.0 O | 1 | ,P 89.0 S | 1 | ,P 93.1 N | 1 | ,P 178.1 | [1] | [4] |
| [ | 4 | ] | [ | 7] | [Pt(η | 3 | -C | 14 | H | 10 | N | 2 | O | 3 | -O | 1 | ,N | 1 | ,O | 2 | )(PPh | 3 | )] (at 150 K) |
PtO | 1 | N | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| [Pt(η | 3 | -C | 16 | H | 13 | N | 3 | O | 3 | S | 2 | -O | 1 | ,N | 1 | ,S | 1 | )(PPh | 3 | )] (at 200 K) | 1 | O | 2 | P (O | 1 | C | 2 | NN | 1 | C | 3 | O | 2 | ) 0.024 |
PtO | 1 | N | 1 | S | 1 | P (O | 1 | C | 3 | N | 1 | NCS | 1 | ) 0.018O | 1 | 1.995(2) N | 1 | 2.000(2) O | 2 | 1.988(2) P 2.251(2) |
O | 1 | 2.001 NO | 1 | ,N | 1 | 88.2 | d | N | 1 | ,O | 2 | 90.0 | d | O | 1 | ,O | 2 | 176.5 O | 1 | ,P 89.0 O | 2 | ,P 90.7 N | 1 | ,P 175.0 | [2] | [5] | ||||||||||||||||||||||||||||||
| [Pt{η | 3 | -C | 12 | H | 24 | S | 3 | -S | 1 | ,S | 2 | ,S | 3 | }(PPh | 3 | )]BF | 4 | PtS | 1 | S | 2 | S | 3 | P (S | 1 | C | 3 | S | 2 | C | 3 | S | 3 | ) 0.035 |
S | 1 | 2.330(2) S | 2 | 2.339(2) S | 3 | 2.336(2) P 2.332(2) |
S | 1 | ,S | 2 | 87.1(2) | d | S | 2 | ,S | 3 | 89.5(2) | d | S | 1 | ,S | 3 | 176.3(2) S | 1 | ,P 91.1(2) S | 3 | ,P 92.3(1) S | 2 | ,P 171.0(2) | [3] | [6] |
(a) Where more than one chemically equivalent distance or angle is present, the mean value is tabulated. The number in parentheses is the e.s.d. (b) Parameter τ4, degree of distortion. (c) The chemical identity of the coordinated atom/ligand is specific to these columns. (d) Six-membered metallocyclic ring.
For the complex [Pt{η3-C12H24S3-S1,S2,S3}(PPh3)]BF4, the η3-ligand creates a pair of six-membered metallocyclic rings of the S1C3S2C3S3 type (as shown in Figure 1) [3][6]. The values of the chelate angles are 87.1° (S1-Pt-S2) and 89.5° (S2-Pt-S3). The remaining L-Pt-L bond angles open in the following order: 91.1° (S1-Pt-P) < 92.3° (S3-Pt-P) < 171.0° (S2-Pt-P) < 176.3° (S1-Pt-S3). The Pt-L bond distance increases in the following order: 2.330 Å (Pt-S1) < 2.332 Å (Pt-P) < 2.336 Å (Pt-S3) < 2.339 Å (Pt-S2 trans to P). Noticeably, the trans-X1-Pt-X3 bond angles are somewhat bigger than the trans-X2-Pt-P bond angles (Table 1).
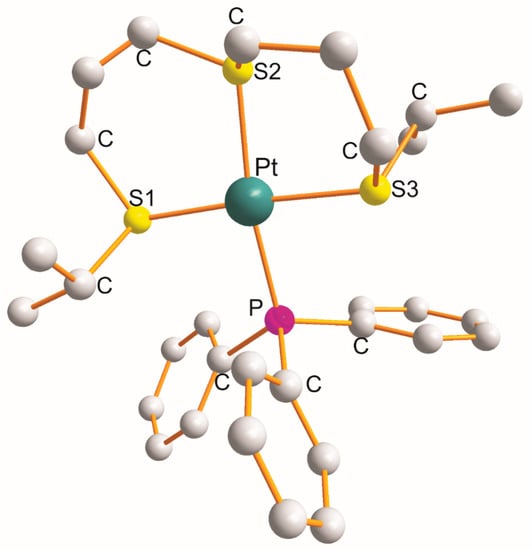

Figure 1.
Structure of [Pt{η
3
-C
12
H
24
S
3
-S
1
,S
2
,S
3
}(PPh
)] [6].
2. 6+5-Membered Metallocyclic Rings
There are five examples namely [Pt(η
3
-C
16
H
14
N
2
OS
2
–O
1
,N
1
,S
1
)(PPh
)] [7], [Pt(η
3
-C
16
H
13
N
3
O
3
S
2
–O
1
,N
1
,S
1
)(PPh
)] (at 200K) [7], [Pt(η
3
-C
8
H
8
N
3
OS–O
1
,N
| 1 |
| 2.041 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| S | 1 | 2.239 P 2.248 |
O | 1 | ,N | 1 | 92.6 | d | N | 1 | ,S | 1 | 85.3 | e | O | 1 | ,S | 1 | 177.6 O | 1 | ,P 89.0 S | 1 | ,P 93.3 N | 1 | ,P 176.0 | [4] | [7] | ||||||||||||||||||||||||||||||||||||
| [Pt(η | 3 | -C | 8 | H | 8 | N | 3 | OS-O | 1 | ,N | 1 | ,S | 1 | )(PPh | 3 | )].toluene | PtO | 1 | N | 1 | S | 1 | P (O | 1 | C | 3 | N | 1 | NCS | 1 | ) 0.020 |
O | 1 | 2.015 N | 1 | 2.031 S | 1 | 2.234 P 2.257 |
O | 1 | ,N | 1 | 93.1 | d | N | 1 | ,S | 1 | 83.8 | e | O | 1 | ,S | 1 | 176.6 O | 1 | ,P 89.9 S | 1 | ,P 93.3 N | 1 | ,P 176.3 | [5] | [8] |
| [Pt(η | 3 | -C | 9 | H | 9 | N | 3 | OS-O | 1 | ,N | 1 | ,S | 1 | )(PPh | 3 | )] (at 103 K) |
PtO | 1 | N | 1 | S | 1 | P (O | 1 | C | 3 | N | 1 | NCS | 1 | ) 0.024 |
O | 1 | 2.085 N | 1 | 2.036 S | 1 | 2.257 P 2.260 |
O | 1 | ,N | 1 | 92.5 | d | |||||||||||||||||||
| S | 1 | ,P 91.2 N | 1 | ,P 173.1 | [4] | [7] |
(a) Where more than one chemically equivalent distance or angle is present, the mean value is tabulated. The number in parentheses is the e.s.d. (b) Parameter τ4, degree of distortion. (c) The chemical identity of the coordinated atom/ligand is specific to these columns. (d) Six-membered metallocyclic ring. (e) Five-membered metallocyclic ring.
3. 5+6-Membered Metallocyclic Rings
There are four complexes mentioned in this section, namely [Pt(η3
-C12
H10
N4
–N1
,N2
,N3
)(PPh)] (at 100 K) [10], [Pt(η3
-C13
H9
NO2
–O1
,N1
,O2
)(PPh)] [11], [Pt(η3
-C12
H16
N2
O4
Se2
–Se1
,N1
,Se2
){P(η1
-C11
H19
O5
)(Ph)}] [12], and [Pt(η3
-C29
H20
F6
S2
O–S1
,S2
,O1
)(PPh)] (at 100 K) [13], and their structural parameters are gathered in Table 3
. The structure of [Pt(η3
-C12
H10
N4
–N1
,N2
,N3
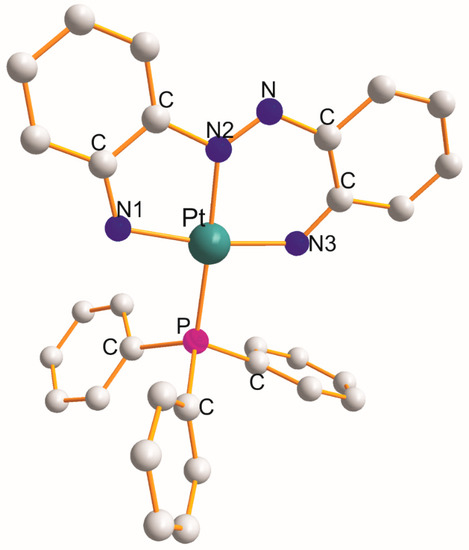
)(PPh)] [10] is shown in Figure 3
as an example. Each η3
-ligand creates five and six metallocyclic rings. The donor atoms of the respective η3
-ligands play a role in the size of the L-Pt-L chelate angles. These angles increase in the following sequences:

Figure 3. Structure of [Pt(η3-C12H10N4–N1,N2,N3)(PPh3)] [7][10].
Table 3. Structural data for Pt(η3-X3)(Y) derivatives. a—5+6-membered metallocyclic rings.
| Complex | Chromophore Chelate Rings τ | 4 | b | Pt -L | c | (Å) |
L-Pt-L | c | (°) |
Ref. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| [Pt(η | 3 | -C | 12 | H | 10 | N | 4 | -N | 1 | ,N | 2 | ,N | 3 | )(PPh | 3 | )] (at 100 K) |
Pt N | 1 | N | 2 | N | 3 | P (N | 1 | C | 2 | N | 2 | NC | 2 | N | 3 | ) 0.034 |
N | 1 | 1.984 N | 2 | 2.025 N | 3 | 1.964 P 2.255 |
N | 1 | ,N | 2 | 81.7 | e | N | 2 | ,N | 3 | 89.6 | d | N | 1 | ,N | 3 | 170.6 N | 1 | ,P 93.0 N | 3 | ,P 96.3 N | 2 | ,P 177.2 | [7] | [10] | ||||||||||||||||||
| [Pt(η | 3 | -C | 13 | H | 9 | NO | 2 | -O | 1 | ,N | 1 | ,O | 2 | )(PPh | 3 | )] | Pt O | 1 | N | 1 | O | 2 | P (O | 1 | C | 2 | N | 1 | C | 3 | O | 2 | ) 0.034 |
O | 1 | 1.975(9) N | 1 | 2.064(12) O | 2 | 1.996(9) P 2.248 |
O | 1 | ,N | 1 | 82.4(4) | e | N | 1 | ,O | 2 | 94.8(4) | d | O | 1 | ,O | 2 | 176.4(4) O | 1 | ,P 91.5(3) O | 2 | ,P 91.5(3) N | 1 | ,P 172.4 | [8] | [11] | ||||||||||||||||||
| [Pt(η | 3 | -C | 12 | H | 16 | N | 2 | O | 4 | Se | 2 | -Se | 1 | ,N | 1 | ,Se | 2 | ){P(η | 1 | -C | 11 | H | 19 | O | 5 | )(Ph) | 2 | }] | Pt Se | 1 | N | 1 | Se | 2 | (Se | 1 | C | 2 | N | 1 | NC | 2 | Se | 2 | ) 0.036 |
Se | 1 | 2.394 N | 1 | 2.078 Se | 2 | 2.349 P 2.259 |
Se | 1 | ,N | 1 | 83.3 | e | N | 1 | ,Se | 2 | 98.3 | d | Se | 1 | ,Se | 3 | 176.3 Se | 1 | ,P 87.2 Se | 2 | ,P 90.7 N | 1 | ,P 170.9 | [9] | [12] | ||||||
| [Pt(η | 3 | -C | 29 | H | 20 | F | 6 | O | 4 | S | 2 | O-S | 1 | ,S | 2 | ,O | 1 | )(PPh | 3 | )] (at 100 K) |
Pt S | 1 | S | 2 | O | 1 | P (S | 1 | C | 2 | S | 2 | C | 3 | O | 1 | ) 0.059 |
S | 1 | 2.268 S | 2 | 2.277 O | 1 | 2.066 P 2.253 N | 1 | ,S | 1 | 85.1 | e | O | 1 | ,S | 1 | 175.7 O | 1 | ,P 91.5 S | 1 | ,P 91.0 N | 1 | ,P 175.6 | S | 1 | ,S | 2 | 90.2 | e | S | 1 | ,O | 1 | 99.2 | d | S | 1 | ,O | 1 | 169.6 S | 1 | ,P 89.2 O | 1 | ,P 99.2 S | 2[6] | [9] |
| P 169.4 | [ | 10 | ] | [13] | [Pt(η | 3 | -C | 18 | H | 16 | N | 2 | OS | 2 | -O | 1 | ,N | 1 | ,S | 1 | )(PPh | 3 | )] | PtO | 1 | N | 1 | S | 1 | P (O | 1 | C | 3 | N | 1 | NCS | 1 | ) 0.037 |
O | 1 | 2.045 N | 1 | 2.029 S | 1 | 2.246 P 2.269 |
O | 1 | ,N | 1 | 92.3 | d | N | 1 | ,S | 1 | 83.2 | e | O | 1 | ,S | 1 | 173.6 O | 1 | ,P 93.1 |
(a) Where more than one chemically equivalent distance or angle is present, the mean value is tabulated. The number in parentheses is the e.s.d. (b) Parameter τ4, degree of distortion. (c) The chemical identity of the coordinated atom/ligand is specific to these columns. (d) Six-membered metallocyclic ring. (e) Five-membered metallocyclic ring.
N1C2N2NC2N3—81.7° (N1-Pt-N2) and 89.6° (N2-Pt-N3);
O1C2N1C3O2—2.4° (O1-Pt-N1) and 94.8° (N1-Pt-O2);
Se1C2N1NC2Se2—83.3° (Se1-Pt-N1) and 98.3° (N1-Pt-Se2);
S1C2S2C3O1—90.2° (S1-Pt-S2) and 99.2° (S2-Pt-O1).
The monodentate PL displayed distorted square-planar geometry about Pt(II) atoms. The Pt-L bond distance to PL increased in the following order: 2.025 Å (Pt-N
2
) < 2.064 Å (Pt-N1
) < 2.078 Å (Pt-N1
) < 2.277 Å (Pt-S2
). The order follows the above-mentioned sentence for the Pt-L (L is a common central ligating atom between five and six-rings).
4. 5+5-Membered Metallocyclic Rings
There are thirty-nine compounds in which each η3
-ligand creates two five-membered metallocyclic rings. These complexes based on variable combinations of atoms involved in the chelate angles can be divided into twelve groups.
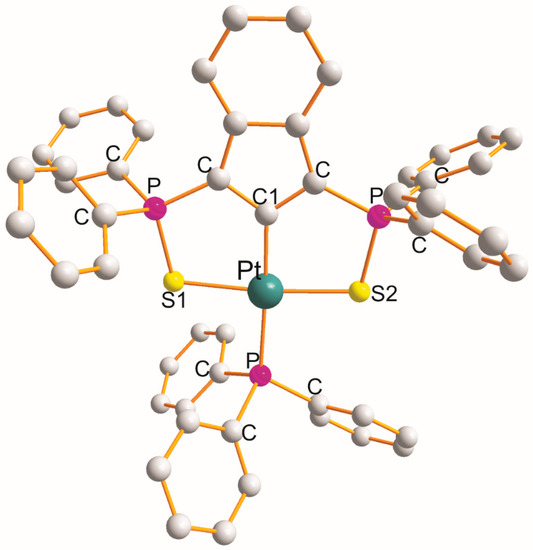
The structure of [Pt(η3
-C33
H24
P2
S2
–S1
,C1
,S2
)(PPh3
)].CH2
Cl [14] is shown in Figure 4
. The η3
-ligand creates two five-membered metallocyclic rings with a common C1
atom of the S1
PCC1
CPS2
type with chelate angles of 87.9° (S1
-Pt-C1
) and 87.7° (C1
-Pt-S2
). This is the only example of this type. The PPh3
demonstrated distorted square-planar geometry about Pt(II) atoms. The remaining L-Pt-L bond angles open in the following order: 89.7° (S1
-Pt-P) < 94.2° (S2
-Pt-P) < 173.8° (S1
-Pt-S2
) < 176.9° (C1
-Pt-P). The Pt-L bond distance increases in the following order: 2.020 Å (Pt-C1
) < 2.316 Å (Pt-S2
) < 2.332 Å (Pt-S1
) < 2.322 Å (Pt-P).