Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Camila Xu and Version 1 by Pier Giorgio Natali.
Tomato, the fruit providing the most comprehensive complex of prostate-health-preserving micronutrients, has been shown to be superior to its single-nutrient counterparts in decreasing the incidence of age-related prostate diseases.
- benign prostate hypertrophy
- food supplement
- prostate cancer
- tomato
1. Introduction
The presence of chronic inflammation in an aging prostate may lead to two main diseases, benign prostatic hyperplasia (BPH) and cancer (PCa) [1]. BPH may severely impair the quality of life in one third of subjects older than 50 years and is reported in about 90% of individuals reaching 80 years of age [2]. PCa is the most frequently diagnosed cancer in men, with an annual incidence of nearly 1.5 million new cases [3]. Although significant differences in PCa incidence and mortality across ethnic groups have been reported [4], this malignancy represents an alarming public health issue worldwide because of the increasing incidence in young patients and the growing frequency of advanced disease cases [5]. In view of the global augmented life expectancy, both BPH and PCa embody a growing economic burden for wealthy societies and for patients’ direct and indirect costs [6,7][6][7].
Because of their late age occurrence, BPH and PCa are suitable for preventive interventions since even a modest delay in their development can lead to a substantial reduction in the incidence of clinically identifiable lesions. Primary prevention of BPH and PCa relies on healthy choices, such as regular exercise, low fat intake, and high vegetable and fruit consumption [8]. The latter recommendations, often inclusively described as adherence to the Mediterranean diet [9], advocate the regular uptake of various antioxidants and anti-inflammatory complexes present in fresh vegetables and fruits [10,11][10][11].
2. Tomato and Olive Oil in Prostate Health
2.1. BPH’s Natural History and Current Medical Treatment
The molecular mechanisms underlying the pathogenesis of BPH leading to a symptomatic disease are still ill-defined [16][12]. Age-related endocrine [17][13] and vascular changes [18][14] as well as uncontrolled oxidative stress derived from acute and chronic inflammation have been proposed to induce the overgrowth of the stromal and cellular compartments in the transition zone of the gland [19,20,21][15][16][17]. However, because a low degree of proliferation of both epithelial and stromal cells has been detected, BPH has been interpreted as the result of impaired programmed cell death mechanisms [22,23][18][19]. Of interest, the recent molecular profiling of BPH has identified distinct subtypes, such as those with altered mTOR signaling linked to unfavorable prognosis [24][20], which are candidates for more targeted therapies. The diagnosis of BPH is usually driven by lower urinary tract obstructive symptoms (LUTSs), i.e., urinary hesitancy, urgency, frequency, and post-void dribble [25][21]. Patients with BPH show a higher incidence of “in situ” PCa [26[22][23],27], thus suggesting that a reduction in risk factors or BPH treatment can, in at least a fraction of patients, delay the onset of PCa. Chronic inflammation has also been shown to contribute to an increased risk of rapid disease progression because of a lower response to medical treatment [28][24]. Therefore, inflammation represents a key target of BPH treatment [29][25]. However, innovation in drug development for BPH is currently stalled, being confined to alpha-blockers, 5-alpha reductase inhibitors, and phosphodiesterase type 5 inhibitors [30,31][26][27] that, because of the underlying heterogeneity of BPH lesions [24[20][28],32], are often used in combination or successively [33][29]. Despite these strategies, LUTSs are not controlled in about one third of patients [34][30], and a number of responders’ side effects, such as decreased libido, erectile dysfunction, dizziness, and hypotension, may occur [33][29]. The failure of medical treatment invariably leads to surgical intervention to reduce LUTSs’ severity [35][31].2.2. Phytotherapies for BPH: The Role of Tomato and Olive Micronutrients
Guidelines on therapeutic options for BPH include phytotherapy as well. Indeed, phytocompounds, either as plant portions, derived extracts, or purified molecules, are increasingly used in the treatment of patients with moderate–severe BPH [36][32]. A large body of epidemiological, experimental, and clinical studies on BPH prevention or improvement of the associated symptoms [37,38][33][34] has focused on tomato lycopene, the red-colored, open-chain beta carotenoid devoid of retinoid activity, present in a variable concentration in different tomato-cultivar-derived foods. Lycopene, which is contained in all tomato dietary sources, mainly in the low bioavailable trans isomeric form [39][35], is endowed with a large range of biological activities, which are also retained by its metabolites [40][36]. Its bioavailability, on the other hand, can be affected by a number of factors [39,41,42][35][37][38] and, differently from the trans isomeric form [43][39], the biologically active cis lycopene, which is produced mainly by heating the fruit [44][40], concentrates on definite anatomical sites, which include the prostate [39][35]. However, comparative studies have clearly demonstrated that the healthy properties of tomatoes could not be not exclusively ascribed to their lycopene content [45,46][41][42]. Indeed, a number of compounds endowed with wide-ranging biological activity are present in the fruit or are newly formed during its heat processing [12,47,48][43][44][45]; thus, the overall complex contributes to the in vivo anti-proliferative, anti-inflammatory, and pro-apoptotic activities [47[44][46][47],49,50], as well as to the anti-androgenic properties of the fruit [51][48]. As a result, the consumption of whole tomatoes has been shown to provide healthier effects than lycopene alone in experimental [10,45][10][41] and clinical studies [46,52][42][49]. Olive oil is well proven to represent a source of chemo-preventive nutrients [13][50]. Its polyphenol content prevents PCa development and invasiveness [53][51], as these molecules can modulate the molecular signaling involved in the growth and proliferation of transformed cells in inflammation (e.g., MAPK, PI3K, and NF-κB) [54][52] and angiogenesis [55][53]. In particular, hydroxytyrosol has been reported to inhibit in vitro human PCa cell proliferation and induce apoptosis [56,57][54][55].2.3. Tomato and Olive Eco-Sustainability
Various phytochemicals endowed with prostate-healthy properties are widely present in nature [58][56]. WResearchers have concentrated outheir investigation on complexes of tomato and olive micronutrients, with outheir choice herein detailed. Tomato, which represents the second most popular crop worldwide [59][57], has a high global consumption rate [60][58]. The fruit, characterized by extensive biodiversity [61][59] and chemodiversity [62][60], with a high average nutritional yield [63][61], requires timely controlled irrigation and moderate soil tillage [64][62]. Tomatoes accumulate low levels of environmental contaminants [65][63] in the fruit and are thus considered to be an “excluder plant” since they concentrate soil contaminants in the roots, stems, and shoots [64][62]. Residues of pesticides, if present on the skin, are removed by washing and cooking [66][64]. In addition, tomato lycopene has been shown to decrease the risk associated with exposure to natural chemical toxins, including pesticides and herbicides [67][65]. The waste caused by tomatoes’ industrial processing, as well as their packaging, is highly recyclable [68][66]. Because of their unique culinary versatility in multiethnic cuisine, they have wide acceptance in various dietary regimens. Olive cultivation is another important agro-industrial sector [69][67] that also plays a role in maintaining biodiversity. Olive growth is possible under extreme environmental conditions, such as drought and high temperatures [70,71][68][69]. Epidemiological and experimental data have underscored the healthy properties of moderate dietary consumption of olive oil due to its antioxidant and inflammatory components [13,72][50][70].2.4. Development of a New Whole-Tomato-Based Food Supplement (WTFS)
Among the different preparations derived from roots, seeds, pollen, bark, or fruits [73[71][72],74], the tomato is particularly attractive for prostate health maintenance. The fruit’s anti-inflammatory and antioxidant molecules [12][43], by acting both systemically as well as in the gland, are potentially useful in also shielding prostate cells [75][73] from aging-related degenerative changes [58][56]. There is no scientific evidence of any interaction between tomato and drugs, a relevant aspect in older patients who are often undergoing multiple pharmacologic treatments. Because the bioavailability of lycopene, the tomato’s major antioxidant, is highly increased by cooking the fruit [42][38] and by the presence of fats, especially olive virgin oil [76][74], early attempts to improve tomato antioxidant activity used a concentrated whole ripe fruit puree heated at 95 °C for 5 min, with 10% extra-virgin olive oil added. This “food for special medical purposes” (FSMP), when administered to patients affected by chronic viral C hepatitis, significantly increased plasma lycopene concentrations compared to other tomato products and was thus effective in preventing carotenoid serum depletion and in improving the oxidative status during antiviral therapy [77][75]. When tested in a transgenic mouse model of human prostate carcinogenesis (TRAMP) [78][76], the supplementation of the animal’s diet with 10% of FSMP significantly decreased the appearance of poorly differentiated cancer and mortality [79][77]. In addition, an in-depth serological analysis of the animals during treatment demonstrated that the FSMP was able to reduce the levels of circulating inflammatory/angiogenic cytokines, such as vascular endothelial growth factor (VEGF), tumor necrosis factor alpha (TNF-α), and interleukin (IL)-6 [79][77]. These effects were dose-dependent since tomato-based supplemented diets with lower lycopene concentrations failed to modify the clinical course of the TRAMP mice’s cancer [80][78]. Along this line of investigation, and with the aim of producing an improved and standardized “whole tomato food supplement” (WTFS) of potential use in clinical studies, an innovative whole tomato, solvent-free processing protocol [81][79] described in Figure 1A has been developed.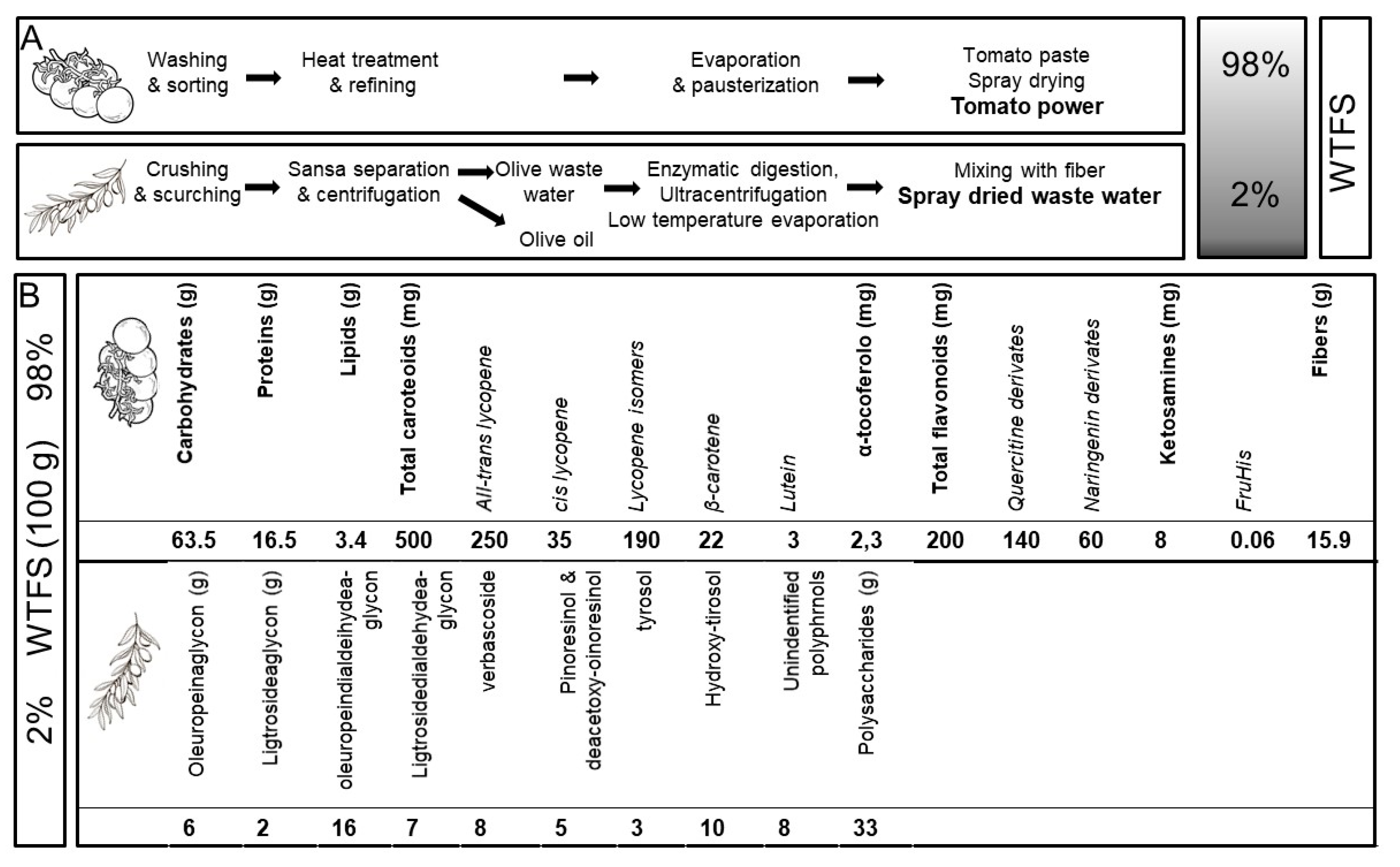
Figure 1.
(
A
) Protocol for “whole tomato food supplement” (WTFS) preparation. (
B
) Composition for 100 g of WTFS.
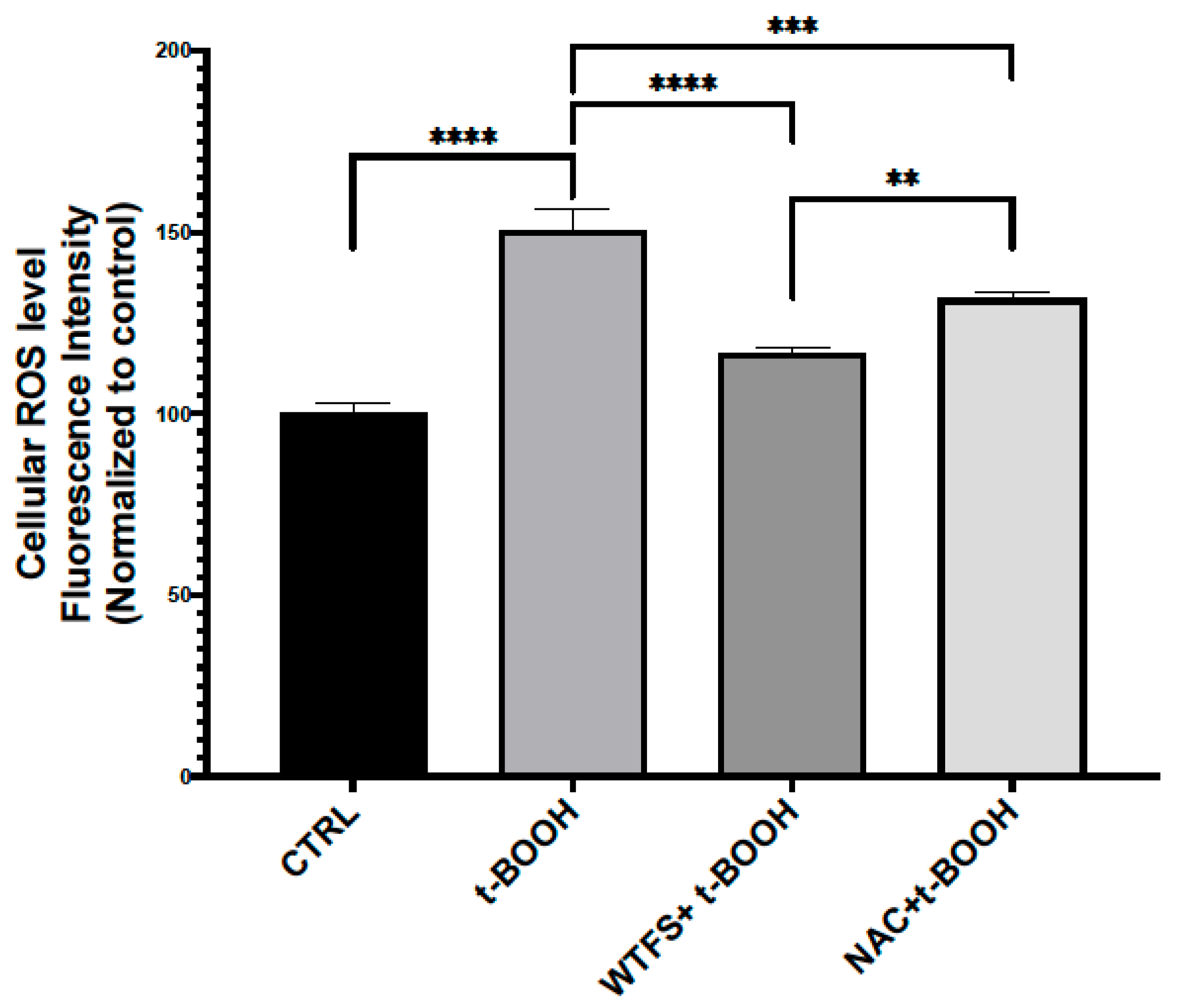
Figure 2. Reactive oxygen species (ROS) detection. To compare the antioxidant activity of WTFS and N-acetylcysteine (NAC), LNCaP (androgen-sensitive human prostate epithelial cell line) cells were pretreated for 4 h with 1 mg/mL of WTFS or 500 µM of NAC and then were stressed with 75 μM of tert-butyl hydroperoxide (t-BOOH) for 30 min. ROS were quantified using the CellROX Green Flow Cytometry Assay Kit (Thermo Fisher Scientific, Monza, Italy, cat. C10492) following the manufacturer’s instructions. Samples were analyzed using a BD Accuri C6 flow cytometer (BD Biosciences, Milano, Italy). The experiment was repeated three times with similar results, and the obtained values are presented as the mean and standard deviation. Statistical analysis was performed using GraphPad Prisma 8.2.1(279) software and ANOVA followed by Tukey’s post hoc test. Statistically significant differences (** p < 0.01; *** p < 0.001; **** p < 0.0001) are marked with asterisks and refer to the untreated LNCaP cells used as the control (CTRL).
2.5. WTFS in BPH
The availability of the standardized WTFS product allowed for its assessment in a clinical pilot study which demonstrated that following the daily assumption of 6 gr of the WTFS containing 22 mg of total carotenoids for two months, 80% of symptomatic BPH patients showed a decrease in LUTSs [84][82]. The patients’ compliance was high, and no side effects were reported. Therefore, a phase II prospective, randomized, double-blinded, placebo-controlled study was performed on patients with biopsy-proven BPH characterized by various degrees of inflammation [85][83]. The results of this trial indicated that following the same treatment schedule as the pilot study, WTFS significantly relieved LUTSs (p < 0.0002) and improved the quality of life (p < 0.0001). In this context, the WTFS uptake did not change free-prostate-specific antigen (PSA) and free/total PSA ratio values, but a trend in the decrease in free PSA in patients with baseline levels above 10 ng/mL was documented [86,87][84][85]. This is in line with data from early studies demonstrating that tomato supplementation reduces PSA levels, but just in PCa patients [88][86]. Only one patient left the study, and no side effects, often associated with the culinary use of tomato [89][87], were recorded. In view of the fact that metabolic syndrome and chronic inflammation, both considered to be risk factors for BHP, are frequently observed in HIV-infected patients [90][88] (in whom PCa incidence is expected to increase in the near future [91][89]), a validation study was performed on HIV+ patients with BPH using the treatment schedule. Additionally, in this trial, the daily WTFS consumption resulted in a significant amelioration in LUTSs, quality of life, free/total PSA ratio, and diastolic blood pressure, with a trend in the decrease in the IL-6 serum level [92][90].2.6. Links between BPH and Pca
BPH and PCa share genetic traits [93][91]. Epidemiologic and pathologic links between BPH and later PCa development have been reported, especially for Asian patients [94[92][93],95], that parallel those described by a European long-term, large cohort study [96][94]. As in BPH, PCa may be associated with metabolic syndrome and insulin resistance, suggesting a relationship with a dietary factor for this group of diseases [97][95]. This is also supported by the association between high body mass index, an element of metabolic syndrome, and PCa progression and specific mortality [98,99,100,101,102][96][97][98][99][100]. Although the understanding of the molecular basis of the two diseases is far from being fully outlined, common deranged molecular pathways include hormonal dependence [103][101], chronic inflammation, and downstream signaling pathways involving cytokines such as IL-12, TNF-α, IL-1, IL-1β, and IL-6 [104,105][102][103]; downstream signaling pathways mediated by NF-κB, which contributes to tumor progression [106][104]; IGF1R-modulating angiogenesis [107][105]; and IL-6/JAK/STAT signaling, which stimulates cell growth and impairs apoptosis [108,109][106][107]. An overview and detailed representation of the common pathways between BPH and PCa are represented in Figure 3.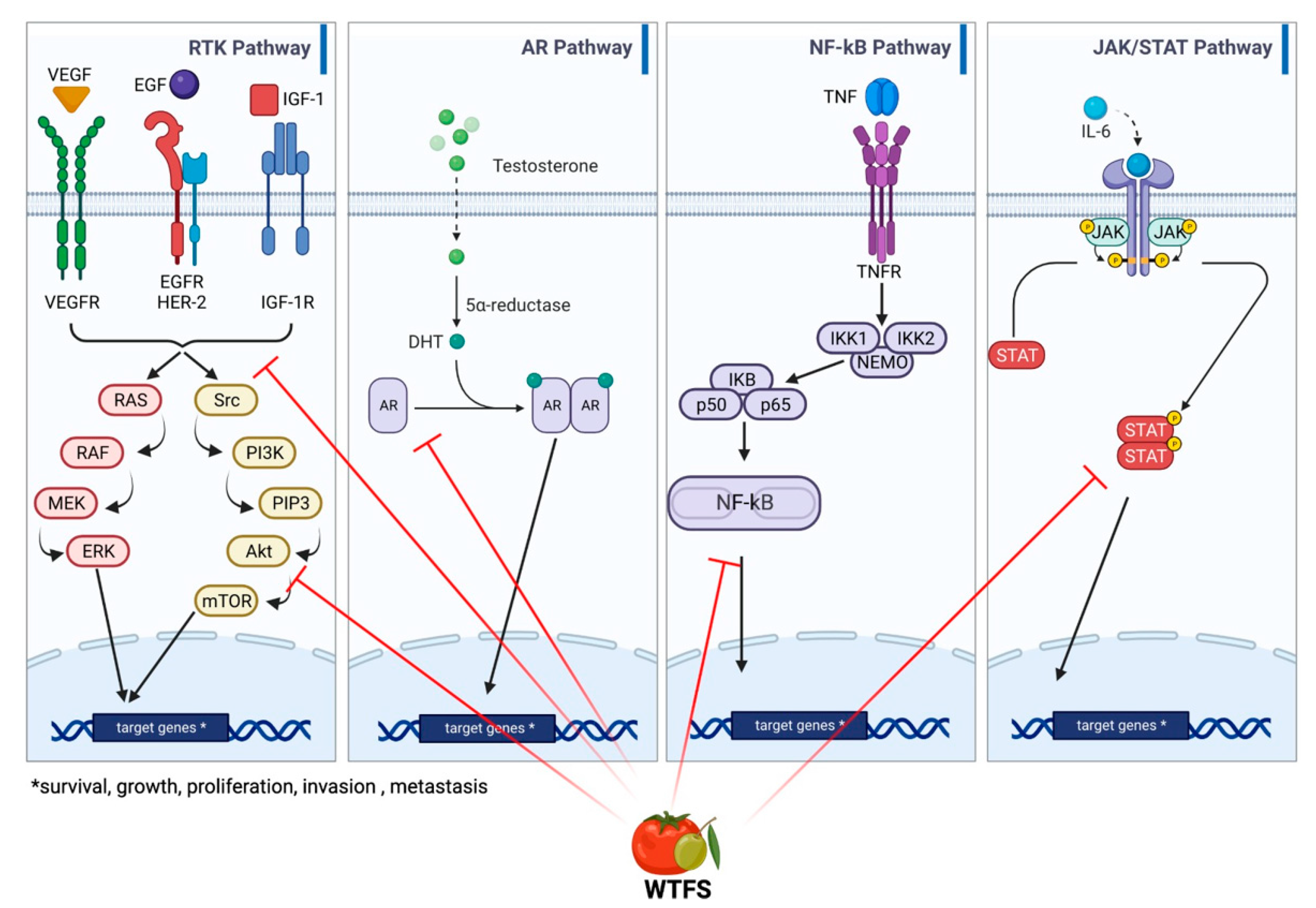
Figure 3. Main signaling pathways involved in BHP and PCa development: receptor tyrosine kinase (RKT), androgen receptor (AR), NF-κB, and JAK/STAT signaling. WTFS targets (also see Table 1) are highlighted by red lines.
2.7. Tomato Consumption and PCa
Due to the above relationships, phytotherapies are also gaining increasing attention in PCa management in preventive, therapeutic, and palliative settings [58][56]. A link between tomato and lycopene uptake and PCa risk was initially indicated by epidemiologic studies [88,111,112][86][109][110]. A prospective study of tomato products confirmed the inverse association between tomato sauce consumption and PCa risk. Notably, subjects consuming more than two servings/week of tomato compared to less than one serving/month exhibited a 66% decreased risk of PCa metastatic cancer [113][111]. More recently, a consistently high intake of tomato after PCa diagnosis was found to be associated with a significantly lower risk of specific mortality among patients diagnosed with high-risk tumors [114][112]. In addition, low lycopene concentrations in the prostate favor PCa onset in patients with high-risk prostatic intraepithelial tumors [115][113]. Conclusively, a meta-analysis confirmed that the beneficial effect of processed (cooked tomatoes and sauces) and raw tomato consumption on PCa risk are dose-dependent, and adherence to the PCa-specific dietary recommendations via constant tomato consumption is associated with a decreased risk of PCa [89][87].2.8. WTBS and Inhibition of PCa-Activated Molecular Pathways
Early evidence that WTBS can interfere with prostate carcinogenesis is derived from the TRAMP murine carcinoma model of progressive PCa that mirrors the stages of human disease, including the androgen-independent stage [79][77]. In these mice, the FSMP diet significantly increased overall survival (p < 0.01), delayed progression from prostatic intraepithelial neoplasia to adenocarcinoma, and decreased the incidence of poorly differentiated cancer. Biochemical studies disclosed a decrease in antioxidant activity in animal sera and a reduction in the circulating biomarkers of relevance to prostate carcinogenesis, such as IL-6 and TNF-α, which significantly correlated with PCa grade [116][114] and VEGF, known to be associated with prostate tumor grade, metastasis, and prognosis [117][115]. Indeed, the IL-6/STAT axis represents a link between inflammation and prostate carcinogenesis [105][103], which is the signal transducer and activator of transcription 3 (STAT3), a key modulator in the expression of a wide range of oncogenic genes [118][116] and a player in prostate cancer energy [119][117]. Furthermore, recent data created using the androgen-sensitive human prostate epithelial cell line LNCaP exposed to an environmental carcinogen have demonstrated that WTFS protects DNA from oxidative stress damage, blocks the pathways involved in PCa development—such as STAT3 activation androgen receptor signaling—and displays pro-apoptotic and anti-proliferative properties [120][118]. Table 1 summarizes the biological activities of the complex of micronutrients present in the WTFS. Table 1. Activity of WTBS’s single components.
| Compound | Activity | References |
|---|---|---|
| Lycopene | [87,88,121,122,123][85][86][119][120][121] | |
|
||
|
||
|
||
|
||
|
||
|
||
|
||
| Tyrosol/hydroxytirosol | [57,124,125,126][55][122][123][124] | |
|
||
|
||
|
||
|
||
|
||
| Tocopherol | [127,128,129,[126][127130][125]][128] | |
|
||
|
||
|
||
|
||
| Quercetin | [131,132,133][129][130][131] | |
|
||
|
||
|
||
|
||
|
||
| Secoiridoid aglycones (oleuropein, ligstroside) | [134,135,136,137][132][133][134][135] | |
|
||
|
||
| Verbascoside | [138,139,140][136][137][138] | |
|
||
|
||
|
||
| Pinoresinol | [141,142][139][140] | |
|
||
|
||
|
References
- Krušlin, B.; Tomas, D.; Džombeta, T.; Milković-Periša, M.; Ulamec, M. Inflammation in prostatic hyperplasia and carcinoma-basic scientific approach. Front. Oncol. 2017, 7, 77.
- Vuichoud, C.; Loughlin, K.R. Benign prostatic hyperplasia: Epidemiology, economics and evaluation. Can. J. Urol. 2015, 22 (Suppl. S1), 1–6.
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249.
- Ferlay, J.; Soerjomataram, I.; Ervik, M.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.; Forman, D.; Bray, F.; et al. 2012 v1.0, Cancer Incidence and Mortality Worldwide: Iarc Cancerbase No. 11; International Agency for Research on Cancer: Lyon, France, 2015; Available online: https://www.scirp.org/(S(351jmbntvnsjt1aadkposzje))/reference/ReferencesPapers.aspx?ReferenceID=1953319 (accessed on 12 January 2023).
- Ellinger, J.; Alajati, A.; Kubatka, P.; Giordano, F.A.; Ritter, M.; Costigliola, V.; Golubnitschaja, O. Prostate cancer treatment costs increase more rapidly than for any other cancer-how to reverse the trend? EPMA J. 2022, 13, 1–7.
- DeWitt-Foy, M.E.; Gill, B.C.; Ulchaker, J.C. Cost Comparison of benign prostatic hyperplasia treatment options. Curr. Urol. Rep. 2019, 20, 45.
- Zaorsky, N.G.; Lin, J.; Ba, D.; Segel, J.E.; Heath, B.; Mackley, H.B.; Drabick, J.; Liu, G.; Leslie, D. The cost of prostate cancer care to society and to patients in the United States. J. Clin. Oncol. 2019, 37, 116.
- Prostate Cancer. Available online: https://medlineplus.gov/prostatecancer.html (accessed on 12 January 2023).
- Schwingshackl, L.; Morze, J.; Hoffmann, G. Mediterranean diet and health status: Active ingredients and pharmacological mechanisms. Br. J. Pharmacol. 2020, 177, 1241–1257.
- Canene-Adams, K.; Campbell, J.K.; Zaripheh, S.; Jeffery, E.H.; Erdman, J.W., Jr. The tomato as a functional food. J. Nutr. 2005, 135, 1226–1230.
- Capurso, C.; Vendemiale, G. The Mediterranean diet reduces the risk and mortality of the prostate cancer: A narrative review. Front. Nutr. 2017, 4, 38.
- Middleton, L.W.; Shen, Z.; Varma, S.; Pollack, A.S.; Gong, X.; Zhu, S.; Zhu, C.; Foley, J.W.; Vennam, S.; Sweeney, R.T.; et al. Genomic analysis of benign prostatic hyperplasia implicates cellular re-landscaping in disease pathogenesis. JCI Insight 2019, 5, e129749.
- Araujo, A.B.; Wittert, G.A. Endocrinology of the aging male. Best Pract. Res. Clin. Endocrinol. Metab. 2011, 25, 303–319.
- Moreau, K.L. Modulatory influence of sex hormones on vascular aging. Am. J. Physiol. Heart Circ. Physiol. 2019, 316, H522–H526.
- Bushman, W. Etiology, epidemiology, and natural history of benign prostatic hyperplasia. Urol. Clin. N. Am. 2009, 36, 403–415.
- Chughtai, B.; Lee, R.; Te, A.; Kaplan, S. Role of inflammation in benign prostatic hyperplasia. Rev. Urol. 2011, 13, 147–150.
- Ficarra, V.; Rossanese, M.; Zazzara, M.; Giannarini, G.; Abbinante, M.; Bartoletti, R.; Mirone, V.; Scaglione, F. The role of inflammation in lower urinary tract symptoms (LUTS) due to benign prostatic hyperplasia (BPH) and its potential impact on medical therapy. Curr. Urol. Rep. 2014, 15, 463–469.
- Roehrborn, C.G.; McConnell, J. Etiology, pathophysiology, epidemiology and natural history of benign prostatic hyperplasia. In Campbell’s Urology, 8th ed.; Walsh, P., Retik, A., Vaughan, E., Wein, A., Eds.; Saunders: Philadelphia, PA, USA, 2002; pp. 1297–1333.
- Untergasser, G.; Madersbacher, S.; Berger, P. Benign prostatic hyperplasia: Age-related tissue-remodeling. Exp. Gerontol. 2005, 40, 121–128.
- Liu, D.; Shoag, J.E.; Poliak, D.; Goueli, R.S.; Ravikumar, V.; Redmond, D.; Vosoughi, A.; Fontugne, J.; Pan, H.; Lee, D.; et al. Integrative multiplatform molecular profiling of benign prostatic hyperplasia identifies distinct subtypes. Nat. Commun. 2020, 24, 1987.
- Badía, X.; García-Losa, M.; Dal-Ré, R. Ten-language translation and harmonization of the International Prostate Symptom Score: Developing a methodology for multinational clinical trials. Eur. Urol. 1997, 31, 129–140.
- Bostwick, D.G.; Cooner, W.H.; Denis, L.; Jones, G.W.; Scardino, P.T.; Murphy, G.P. The association of benign prostatic hyperplasia and cancer of the prostate. Cancer 1992, 70, 291–301.
- Ornstein, D.K.; Rao, G.S.; Smith, D.S.; Andriole, G.L. The impact of systematic prostate biopsy on prostate cancer incidence in men with symptomatic benign prostatic hyperplasia undergoing transurethral resection of the prostate. J. Urol. 1997, 157, 880–883.
- Mishra, V.C.; Allen, D.J.; Nicolaou, C.; Sharif, H.; Hudd, C.; Karim, O.M.; Motiwala, H.G.; Laniado, M.E. Does intraprostatic inflammation have a role in the pathogenesis and progression of benign prostatic hyperplasia? BJU Int. 2007, 100, 327–331.
- De Nunzio, C.; Salonia, A.; Gacci, M.; Ficarra, V. Inflammation is a target of medical treatment for lower urinary tract symptoms associated with benign prostatic hyperplasia. World J. Urol. 2020, 38, 2771–2779.
- Sarma, A.V.; Wei, J.T. Clinical practice. Benign prostatic hyperplasia and lower urinary tract symptoms. N. Engl. J. Med. 2012, 367, 248–257.
- Bechis, S.K.; Otsetov, A.G.; Ge, R.; Olumi, A.F. Personalized medicine for the management of benign prostatic hyperplasia. J. Urol. 2014, 192, 16–23.
- Strand, D.W.; Costa, D.N.; Francis, F.; Ricke, W.A.; Roehrborn, C.G. Targeting phenotypic heterogeneity in benign prostatic hyperplasia. Differentiation 2017, 96, 49–61.
- Yu, Z.J.; Yan, H.L.; Xu, F.H.; Chao, H.C.; Deng, L.H.; Xu, X.D.; Huang, J.B.; Zeng, T. Efficacy and side effects of drugs commonly used for the treatment of lower urinary tract symptoms associated with benign prostatic hyperplasia. Front. Pharmacol. 2020, 11, 658.
- McConnell, J.D.; Roehrborn, C.G.; Bautista, O.M.; Andriole, G.L., Jr.; Dixon, C.M.; Kusek, J.W.; Lepor, H.; McVary, K.T.; Nyberg, L.M., Jr.; Clarke, H.S.; et al. The long-term effect of doxazosin, finasteride, and combination therapy on the clinical progression of benign prostatic hyperplasia. N. Engl. J. Med. 2003, 349, 2387–2398.
- Nair, S.M.; Pimentel, M.A.; Gilling, P.J. Evolving and investigational therapies for benign prostatic hyperplasia. Can. J. Urol. 2015, 22 (Suppl. S1), 82–87.
- Csikós, E.; Horváth, A.; Ács, K.; Papp, N.; Balázs, V.L.; Dolenc, M.S.; Kenda, M.; Kočevar Glavač, N.; Nagy, M.; Protti, M.; et al. Treatment of benign prostatic hyperplasia by natural drugs. Molecules 2021, 26, 7141.
- Ilic, D.; Misso, M. Lycopene for the prevention and treatment of benign prostatic hyperplasia and prostate cancer: A systematic review. Maturitas 2012, 72, 269–276.
- Wertz, K.; Siler, U.; Goralczyk, R. Lycopene: Modes of action to promote prostate health. Arch. Biochem. Biophys. 2004, 430, 127–134.
- Boileau, T.W.; Boileau, A.C.; Erdman, J.W., Jr. Bioavailability of all-trans and cis-isomers of lycopene. Exp. Biol. Med. (Maywood) 2002, 227, 914–919.
- Mein, J.R.; Lian, F.; Wang, X.-D. Biological activity of lycopene metabolites: Implications for cancer prevention. Nutr. Rev. 2008, 66, 667–683.
- Bohn, T.; Desmarchelier, C.; Dragsted, L.O.; Nielsen, C.S.; Stahl, W.; Rühl, R.; Keijer, J.; Borel, P. Host-related factors explaining interindividual variability of carotenoid bioavailability and tissue concentrations in humans. Mol. Nutr. Food Res. 2017, 61, 1600685.
- Amorim, A.D.G.N.; Vasconcelos, A.G.; Souza, J.; Oliveira, A.; Gullón, B.; de Souza de Almeida Leite, J.R.; Pintado, M. Bio-availability, anticancer potential, and chemical data of lycopene: An overview and technological prospecting. Antioxidants 2022, 11, 360.
- Marquez, C.S.; Reis Lima, M.-J.; Oliveira, J.; Teixeira-Lemos, E. Tomato lycopene: Functional proprieties and health benefits. Int. J. Agric. Biol. Eng. 2015, 9, 1089–1099.
- Dewanto, V.; Wu, X.; Adom, K.K.; Liu, R.H. Thermal processing enhances the nutritional value of tomatoes by increasing total antioxidant activity. J. Agric. Food Chem. 2002, 50, 3010–3014.
- Canene-Adams, K.; Lindshield, B.L.; Wang, S.; Jeffery, E.H.; Clinton, S.K.; Erdman, J.W., Jr. Combinations of tomato and broccoli enhance antitumor activity in dunning r3327-h prostate adenocarcinomas. Cancer Res. 2007, 67, 836–843.
- Rowles, J.L., 3rd; Erdman, J.W., Jr. Carotenoids and their role in cancer prevention. Biochim. Biophys. Acta Mol. Cell. Biol. Lipids 2020, 1865, 158613.
- Chaudhary, P.; Sharma, A.; Singh, B.; Nagpal, A.K. Bioactivities of phytochemical present in tomato. J. Food Sci. Technol. 2018, 55, 2833–2849.
- Mohri, S.; Takahashi, H.; Sakai, M.; Takahashi, S.; Waki, N.; Aizawa, K.; Suganuma, H.; Ara, T.; Matsumura, Y.; Shibata, D.; et al. Wide-range screening of anti-inflammatory compounds in tomato using LC-MS and elucidating the mechanism of their functions. PLoS ONE 2018, 13, e0191203.
- Mossine, V.V.; Chopra, P.; Mawhinney, T.P. Interaction of tomato lycopene and ketosamine against rat prostate tumorigenesis. Cancer Res. 2008, 68, 4384–4391.
- Kim, H.S.; Bowen, P.; Chen, L.; Duncan, C.; Ghosh, L.; Sharifi, R.; Christov, K. Effects of tomato sauce consumption on apoptotic cell death in prostate benign hyperplasia and carcinoma. Nutr. Cancer 2003, 47, 40–47.
- Applegate, C.C.; Rowles, J., 3rd; Miller, R.; Wallig, M.; Clinton, S.; O’Brien, W.; Erdman, J., Jr. Dietary tomato, but not lycopene supplementation, impacts molecular outcomes of castration-resistant prostate cancer in the TRAMP model (P05-015-19). Curr. Dev. Nutr. 2019, 3, 438.
- Applegate, C.C.; Rowles, J.L., 3rd; Erdman, J.W., Jr. Can lycopene impact the androgen axis in prostate cancer?: A systematic review of cell culture and animal studies. Nutrients 2019, 11, 633.
- Linnewiel-Hermoni, K.; Khanin, M.; Danilenko, M.; Zango, G.; Amo, Y.; Levy, J.; Sharoni, Y. The anti-cancer effects of carotenoids and other phytonutrients resides in their combined activity. Arch. Biochem. Biophys. 2015, 572, 28–35.
- Piroddi, M.; Albini, A.; Fabiani, R.; Giovannelli, L.; Luceri, C.; Natella, F.; Rosignoli, P.; Rossi, T.; Taticchi, A.; Servili, M.; et al. Nutrigenomics of extra-virgin olive oil: A review. Biofactors 2017, 43, 17–41.
- Weng, C.J.; Yen, G.C. Chemopreventive effects of dietary phytochemicals against cancer invasion and metastasis: Phenolic acids, monophenol, polyphenol, and their derivatives. Cancer Treat. Rev. 2012, 38, 76–87.
- Albini, A.; Indraccolo, S.; Noonan, D.M.; Pfeffer, U. Functional genomics of endothelial cells treated with anti-angiogenic or angiopreventive drugs. Clin. Exp. Metastasis 2010, 27, 419–439.
- Baci, D.; Gallazzi, M.; Cascini, C.; Tramacere, M.; De Stefano, D.; Bruno, A.; Noonan, D.M.; Albini, A. Downregulation of pro-inflammatory and pro-angiogenic pathways in prostate cancer cells by a polyphenol-rich extract from olive mill wastewater. Int. J. Mol. Sci. 2019, 20, 307.
- Luo, C.; Li, Y.; Wang, H.; Cui, Y.; Feng, Z.; Li, H.; Li, Y.; Wang, Y.; Wurtz, K.; Weber, P.; et al. Hydroxytyrosol promotes superoxide production and defects in autophagy leading to anti-proliferation and apoptosis on human prostate cancer cells. Curr. Cancer Drug Targets 2013, 13, 625–639.
- Zubair, H.; Bhardwaj, A.; Ahmad, A.; Srivastava, S.K.; Khan, M.A.; Patel, G.K.; Singh, S.; Singh, A.P. Hydroxytyrosol induces apoptosis and cell cycle arrest and suppresses multiple oncogenic signaling pathways in prostate cancer cells. Nutr. Cancer 2017, 69, 932–942.
- Mazurakova, A.; Samec, M.; Koklesova, L.; Biringer, K.; Kudela, E.; Al-Ishaq, R.K.; Pec, M.; Giordano, F.A.; Büsselberg, D.; Kubatka, P.; et al. Anti-prostate cancer protection and therapy in the framework of predictive, preventive and personalised medicine–comprehensive effects of phytochemicals in primary, secondary and tertiary care. EPMA J. 2022, 13, 461–486.
- Ritchie, H.; Rosado, P.; Roser, M. Agricultural Production. Available online: https://ourworldindata.org/agricultural-production (accessed on 12 January 2023).
- Branthôme, F.-X. Worldwide Consumption of Tomato Products, 2018/2019 (Part 1). François-Xavier 2020 WPTC Congress. Available online: https://www.tomatonews.com/en/worldwide-consumption-of-tomato-products-20182019-part-1_2_994.html (accessed on 12 January 2023).
- Bhattarai, K.; Sharma, S.; Panthee, D.R. Diversity among modern tomato genotypes at different levels in fresh-market breeding. Int. J. Agron. 2018, 2018, 4170432.
- Frusciante, L.; Carli, P.; Ercolano, M.R.; Pernice, R.; Di Matteo, A.; Fogliano, V.; Pellegrini, N. Antioxidant nutritional quality of tomato. Mol. Nutr. Food Res. 2007, 51, 609–617.
- Erika, C.; Ulrich, D.; Naumann, M.; Smit, I.; Horneburg, B.; Pawelzik, E. Flavor and other quality traits of tomato cultivars bred for diverse production systems as revealed in organic low-input management. Front. Nutr. 2022, 9, 916642.
- Sainju, U.M.; Singh, B.P.; Rahman, S.; Reddy, V.R. Tillage, Cover Cropping, and Nitrogen Fertilization Influence Tomato Yield and Nitrogen Uptake. HortScience 2000, 35, 217–221.
- Salem, N.M.; Albanna, L.S.; Awwad, A.M. Toxic heavy metals accumulation in tomato plant (solanum lycopersicum). ARPN J. Agric. Biol. Sci. 2016, 11, 399–404.
- Abou-Arab, A.A.K. Behavior of pesticides in tomatoes during commercial and home preparation. Food Chem. 1999, 4, 509–514.
- Hedayati, N.; Naeini, M.B.; Nezami, A.; Hosseinzadeh, H.; Wallace Hayes, A.; Hosseini, S.; Imenshahidi, M.; Karimi, G. Protective effect of lycopene against chemical and natural toxins: A review. Biofactors 2019, 45, 5–23.
- Trombino, S.; Cassano, R.; Procopio, D.; Di Gioia, M.L.; Barone, E. Valorization of tomato waste as a source of carotenoids. Molecules 2021, 26, 5062.
- Vossen, P. Olive oil: History, production, and characteristics of the world’s classic oils. HortScience 2007, 42, 1093–1100.
- Fraga, H.; Moriondo, M.; Leolini, L.; Santos, J.A. Mediterranean olive orchards under climate change: A review of future impacts and adaptation strategies. Agronomy 2021, 11, 56.
- Mafrica, R.; Piscopo, A.; De Bruno, A.; Poiana, M. Effects of climate on fruit growth and development on olive oil quality in cultivar carolea. Agriculture 2021, 11, 147.
- Psaltopoulou, T.; Kosti, R.I.; Haidopoulos, D.; Dimopoulos, M.; Panagiotakos, D.B. Olive oil intake is inversely related to cancer prevalence: A systematic review and a meta-analysis of 13,800 patients and 23,340 controls in 19 observational studies. Lipids Health Dis. 2011, 10, 127.
- Cai, T.; Luciani, L.G.; Caola, I.; Mondaini, N.; Malossini, G.; Lanzafame, P.; Mazzoli, S.; Bartoletti, R. Effects of pollen extract in association with vitamins (Deprox 500) for pain relief in patients affected by chronic prostatitis/chronic pelvic pain syndrome: Results from a pilot study. Urologia. 2013, 80 (Suppl. S22), 5–10.
- Pagano, E.; Laudato, M.; Griffo, M.; Capasso, R. Phytotherapy of benign prostatic hyperplasia. A minireview. Phytother. Res. 2014, 28, 949–955.
- Widjaja, G.; Doewes, R.I.; Rudiansyah, M.; Sultan, M.Q.; Ansari, M.J.; Izzat, S.E.; Al Jaber, M.S.; Kzar, H.H.; Mustafa, Y.F.; Hammid, A.T.; et al. Effect of tomato consumption on inflammatory markers in health and disease status: A systematic review and meta-analysis of clinical trials. Clin. Nutr. ESPEN 2022, 50, 93–100.
- Vallverdú-Queralt, A.; Regueiro, J.; de Alvarenga, J.F.; Torrado, X.; Lamuela-Raventos, R.M. Carotenoid profile of tomato sauces: Effect of cooking time and content of extra virgin olive oil. Int. J. Mol. Sci. 2015, 16, 9588–9599.
- Vitaglione, P.; Fogliano, V.; Stingo, S.; Scalfi, L.; Caporaso, N.; Morisco, F. Development of a tomato-based food for special medical purposes as therapy adjuvant for patients with HCV infection. Eur. J. Clin. Nutr. 2007, 61, 906–915.
- Greenberg, N.M.; DeMayo, F.; Finegold, M.J.; Medina, D.; Tilley, W.D.; Aspinall, J.O.; Cunha, G.R.; Donjacour, A.A.; Matusik, R.J.; Rosen, J.M. Prostate cancer in a transgenic mouse. Proc. Natl. Acad. Sci. USA 1995, 92, 3439–3443.
- Pannellini, T.; Iezzi, M.; Liberatore, M.; Sabatini, F.; Iacobelli, S.; Rossi, C.; Alberti, S.; Di Ilio, C.; Vitaglione, P.; Fogliano, V.; et al. A dietary tomato supplement prevents prostate cancer in TRAMP mice. Cancer Prev. Res. 2010, 3, 1284–1291.
- Conlon, L.E.; Wallig, M.A.; Erdman, J.W., Jr. Low-lycopene containing tomato powder diet does not protect against prostate cancer in TRAMP mice. Nutr. Res. 2015, 35, 882–890.
- Fogliano, V.; Iacobelli, S.; Piantelli, M. Euro Patent 3 052 113 B1, Italian Health Ministry (registration n. 68843, 2018–2019). Available online: https://worldwide.espacenet.com/patent/search/family/049226079/publication/EP3052113A1?q=3052113 (accessed on 17 February 2023).
- Peroulis, N.; Androutsopoulos, V.P.; Notas, G.; Koinaki, S.; Giakoumaki, E.; Spyros, A.; Manolopoulou, Ε.; Kargaki, S.; Tzardi, M.; Moustou, E.; et al. Significant metabolic improvement by a water extract of olives: Animal and human evidence. Eur. J. Nutr. 2019, 58, 2545–2560.
- Alda, L.M.; Gogoaşă, I.; Bordean, D.-M.; Gergen, I.; Alda, S.; Moldovan, C.; Niţă, L. Lycopene content of tomatoes and tomato products. J. Agroaliment. Process. Technol. 2009, 15, 540–542. Available online: https://www.yumpu.com/en/document/view/50513977/lycopene-content-of-tomatoes-and-tomato-products-journal-of- (accessed on 12 January 2023).
- Cellini, A.; Natali, P.G.; Iezzi, M.; Piantelli, M.; Fogliano, V.; Iacobelli, S. Efficacy and safety of Lycoprozen®, a novel tomato-based food supplement in patients with benign prostatic hyperplasia. Int. J. Nutr. 2018, 3, 1–5.
- Irani, J.; Levillain, P.; Goujon, J.M.; Bon, D.; Doré, B.; Aubert, J. Inflammation in benign prostatic hyperplasia: Correlation with prostate specific antigen value. J. Urol. 1997, 157, 1301–1303.
- Cormio, L.; Calò, B.; Falagario, U.; Iezzi, M.; Lamolinara, A.; Vitaglione, P.; Silecchia, G.; Carrieri, G.; Fogliano, V.; Iacobelli, S.; et al. Improvement of urinary tract symptoms and quality of life in benign prostate hyperplasia patients associated with consumption of a newly developed whole tomato-based food supplement: A phase II prospective, randomized double-blinded, placebo-controlled study. J. Transl. Med. 2021, 19, 24.
- Schwarz, S.; Obermuüller-Jevic, U.C.; Hellmis, E.; Koch, W.; Jacobi, G.; Biesalski, H.K. Lycopene inhibits disease progression in patients with benign prostate hyperplasia. J. Nutr. 2008, 138, 49–53.
- Chen, L.; Stacewicz-Sapuntzakis, M.; Duncan, C.; Sharifi, R.; Ghosh, L.; van Breemen, R.; Ashton, D.; Bowen, P.E. Oxidative DNA damage in prostate cancer patients consuming tomato sauce-based entrees as a whole-food intervention. J. Natl. Cancer Inst. 2001, 93, 1872–1879.
- Salehi, B.; Sharifi-Rad, R.; Sharopov, F.; Namiesnik, J.; Roointan, A.; Kamle, M.; Kumar, P.; Martins, N.; Sharifi-Rad, J. Beneficial effects and potential risks of tomato consumption for human health: An overview. Nutrition 2019, 62, 201–208.
- Bloch, M.; John, M.; Smith, D.; Rasmussen, T.A.; Wright, E. Managing HIV-associated inflammation and ageing in the era of modern ART. HIV Med. 2020, 21, 2–16.
- Shiels, M.S.; Islam, J.Y.; Rosenberg, P.S.; Hall, H.I.; Jacobson, E.; Engels, E.A. Projected cancer incidence rates and burden of incident Cancer Cases in HIV-infected adults in the United States through 2030. Ann. Intern. Med. 2018, 168, 866–873.
- Quiros-Roldan, E.; Carriero, C.; Paghera, S.; Degli Antoni, M.; Fiorini, C.; Quaresima, V.; Castelli, F.; Imberti, L. Symptoms and quality of life in HIV-infected patients with benign prostatic hyperplasia are improved by the consumption of a newly developed whole tomato-based food supplement. A phase II prospective, randomized double-blinded, placebo-controlled study. J. Funct. Foods 2021, 82, 104495.
- Glaser, A.; Shi, Z.; Wei, J.; Lanman, N.A.; Ladson-Gary, S.; Vickman, R.E.; Franco, O.E.; Crawford, S.E.; Lilly Zheng, S.; Hayward, S.W.; et al. Shared Inherited genetics of benign prostatic hyperplasia and prostate cancer. Eur. Urol. Open Sci. 2022, 43, 54–61.
- Alcaraz, A.; Hammerer, P.; Tubaro, A.; Schröder, F.H.; Castro, R. Is there evidence of a relationship between benign prostatic hyperplasia and prostate cancer? Findings of a literature review. Eur. Urol. 2009, 55, 864–873.
- Dai, X.; Fang, X.; Ma, Y.; Xianyu, J. Benign Prostatic Hyperplasia and the Risk of Prostate Cancer and Bladder Cancer: A Meta-Analysis of Observational Studies. Medicine 2016, 95, e3493.
- Ørsted, D.D.; Bojesen, S.E.; Nielsen, S.F.; Nordestgaard, B.G. Association of clinical benign prostate hyperplasia with prostate cancer incidence and mortality revisited: A nationwide cohort study of 3,009,258 men. Eur. Urol. 2011, 60, 691–698.
- Kaiser, A.; Haskins, C.; Siddiqui, M.M.; Hussain, A.; D’Adamo, C. The evolving role of diet in prostate cancer risk and progression. Curr. Opin. Oncol. 2019, 31, 222–229.
- Ma, J.; Li, H.; Giovannucci, E.; Mucci, L.; Qiu, W.; Nguyen, P.L.; Gaziano, J.M.; Pollak, M.; Stampfer, M.J. Prediagnostic body-mass index, plasma C-peptide concentration, and prostate cancer-specific mortality in men with prostate cancer: A long-term survival analysis. Lancet Oncol. 2008, 9, 1039–1047.
- Joshu, C.E.; Mondul, A.M.; Menke, A.; Meinhold, C.; Han, M.; Humphreys, E.B.; Freedland, S.J.; Walsh, P.C.; Platz, E.A. Weight gain is associated with an increased risk of prostate cancer recurrence after prostatectomy in the PSA era. Cancer Prev. Res. 2011, 4, 544–551.
- Ploussard, G.; de la Taille, A.; Bayoud, Y.; Durand, X.; Terry, S.; Xylinas, E.; Allory, Y.; Vacherot, F.; Abbou, C.C.; Salomon, L. The risk of upstaged disease increases with body mass index in low-risk prostate cancer patients eligible for active surveillance. Eur. Urol. 2012, 61, 356–362.
- Bonn, S.E.; Wiklund, F.; Sjölander, A.; Szulkin, R.; Stattin, P.; Holmberg, E.; Grönberg, H.; Bälter, K. Body mass index and weight change in men with prostate cancer: Progression and mortality. Cancer Causes Control 2014, 25, 933–943.
- Lavalette, C.; Trétarre, B.; Rebillard, X.; Lamy, P.J.; Cénée, S.; Menegaux, F. Abdominal obesity and prostate cancer risk: Epidemiological evidence from the EPICAP study. Oncotarget 2018, 9, 34485–34494.
- Ramalingam, S.; Ramamurthy, V.P.; Njar, V.C.O. Dissecting major signaling pathways in prostate cancer development and progression: Mechanisms and novel therapeutic targets. J. Steroid Biochem. Mol. Biol. 2017, 166, 16–27.
- Nunzio, C.; Presicce, F.; Tubaro, A. Inflammatory mediators in the development and progression of benign prostatic hyperplasia. Nat. Rev. Urol. 2016, 13, 613–626.
- Ene, C.V.; Nicolae, I.; Geavlete, B.; Geavlete, P.; Ene, C.D. IL-6 Signaling link between inflammatory tumor microenvironment and prostatic tumorigenesis. Anal. Cell. Pathol. 2022, 2022, 5980387.
- Thomas-Jardin, S.E.; Dahl, H.; Nawas, A.F.; Bautista, M.; Delk, N.A. NF-κB signaling promotes castration-resistant prostate cancer initiation and progression. Pharmacol. Ther. 2020, 211, 107538.
- Holly, J.M.P.; Biernacka, K.; Perks, C.M. The role of insulin-like growth factors in the development of prostate cancer. Expert Rev. Endocrinol. Metab. 2020, 15, 237–250.
- Lin, S.-R.; Yeh, H.-L.; Liu, Y.-N. Interplay of epidermal growth factor receptor and signal transducer and activator of transcription 3 in prostate cancer: Beyond androgen receptor transactivation. Cancers 2021, 13, 3452.
- Johnson, D.E.; O’Keefe, R.A.; Grandis, J.R. Targeting the IL-6/JAK/STAT3 signalling axis in cancer. Nat. Rev. Clin. Oncol. 2018, 15, 234–248.
- Lin, S.R.; Chang, C.H.; Hsu, C.F.; Tsai, M.J.; Cheng, H.; Leong, M.K.; Sung, P.J.; Chen, J.C.; Weng, C.F. Natural compounds as potential adjuvants to cancer therapy: Preclinical evidence. Br. J. Pharmacol. 2020, 177, 1409–1423.
- Bowen, P.; Chen, L.; Stacewicz-Sapuntzakis, M.; Duncan, C.; Sharifi, R.; Ghosh, L.; Kim, H.S.; Christov-Tzelkov, K.; van Breemen, R. Tomato sauce supplementation and prostate cancer: Lycopene accumulation and modulation of biomarkers of carcinogenesis. Exp. Biol. Med. 2002, 227, 886–893.
- Giovannucci, E.; Ascherio, A.; Rimm, E.B.; Stampfer, M.J.; Colditz, G.A.; Willett, W.C. Intake of carotenoids and retinol in relation to risk of prostate cancer. J. Natl. Cancer Inst. 1995, 87, 1767–1776.
- Giovannucci, E.; Rimm, E.B.; Liu, Y.; Stampfer, M.J.; Willett, W.C. A prospective study of tomato products, lycopene, and prostate cancer risk. J. Natl. Cancer Inst. 2002, 94, 391–398.
- Wang, Y.; Jacobs, E.J.; Newton, C.C.; McCullough, M.L. Lycopene, tomato products and prostate cancer-specific mortality among men diagnosed with nonmetastatic prostate cancer in the cancer prevention study II nutrition cohort. Int. J. Cancer 2016, 138, 2846–2855.
- Mariani, S.; Lionetto, L.; Cavallari, M.; Tubaro, A.; Rasio, D.; De Nunzio, C.; Hong, G.M.; Borro, M.; Simmaco, M. Low prostate concentration of lycopene is associated with development of prostate cancer in patients with high-grade prostatic intraepithelial neoplasia. Int. J. Mol Sci. 2014, 15, 1433–1440.
- Zhou, J.; Chen, H.; Wu, Y.; Shi, B.; Ding, J.; Qi, J. Plasma IL-6 and TNF-α levels correlate significantly with grading changes in localized prostate cancer. Prostate 2022, 82, 531–539.
- Bruno, A.; Pagani, A.; Magnani, E.; Rossi, T.; Noonan, D.M.; Cantelmo, A.R.; Albini, A. Inflammatory angiogenesis and the tumor microenvironment as targets for cancer therapy and prevention. Cancer Treat. Res. 2014, 59, 401–426.
- Tesoriere, A.; Dinarello, A.; Argenton, F. The roles of post-translational modifications in STAT3 biological activities and functions. Biomedicines 2021, 9, 956.
- Marrocco, I.; Altieri, F.; Rubini, E.; Paglia, G.; Chichiarelli, S.; Giamogante, F.; Macone, A.; Perugia, G.; Magliocca, F.M.; Gurtner, A.; et al. Shmt2: A Stat3 signaling new player in prostate cancer energy metabolism. Cells 2019, 8, 1048.
- Rubini, E.; Minacori, M.; Paglia, G.; Macone, A.; Chichiarelli, S.; Altieri, F.; Eufemi, M. Tomato and olive bioactive compounds: A natural shield against the cellular effects induced by β-hexachlorocyclohexane-Activated Signaling Pathways. Molecules 2021, 26, 7135.
- Mirahmadi, M.; Azimi-Hashemi, S.; Saburi, E.; Kamali, H.; Pishbin, M.; Hadizadeh, F. Potential inhibitory effect of lycopene on prostate cancer. Biomed. Pharmacother. 2020, 129, 110459.
- Sahin, K.; Yenice, E.; Tuzcu, M.; Orhan, C.; Mizrak, C.; Ozercan, I.H.; Sahin, N.; Yilmaz, B.; Bilir, B.; Ozpolat, B.; et al. Lycopene protects against spontaneous ovarian cancer formation in laying hens. J. Cancer Prev. 2018, 23, 25–36.
- Park, B.; Lim, J.W.; Kim, H. Lycopene treatment inhibits activation of Jak1/Stat3 and Wnt/β-catenin signaling and attenuates hyperproliferation in gastric epithelial cells. Nutr. Res. 2019, 70, 70–81.
- Marković, A.K.; Torić, J.; Barbarić, M.; Brala, C.J. Hydroxytyrosol, tyrosol and derivatives and their potential effects on human health. Molecules 2019, 24, 2001.
- Warleta, F.; Quesada, C.S.; Campos, M.; Allouche, Y.; Beltrán, G.; Gaforio, J.J. Hydroxytyrosol protects against oxidative DNA damage in human breast cells. Nutrients 2011, 3, 839–857.
- Calahorra, J.; Martínez-Lara, E.; Granadino-Roldán, J.M.; Martí, J.M.; Cañuelo, A.; Blanco, S.; Oliver, F.J.; Siles, E. Crosstalk between hydroxytyrosol, a major olive oil phenol, and HIF-1 in MCF-7 breast cancer cells. Sci. Rep. 2020, 10, 6361.
- Harris, A.; Devaraj, S.; Jialal, I. Oxidative stress, alpha-tocopherol therapy, and atherosclerosis. Curr. Atheroscler. Rep. 2002, 4, 373–380.
- Huang, H.; He, Y.; Cui, X.X.; Goodin, S.; Wang, H.; Du, Z.Y.; Li, D.; Zhang, K.; Tony Kong, A.N.; Dipaola, R.S.; et al. Potent inhibitory effect of β-tocopherol on prostate cancer cells cultured in vitro and grown as xenograft tumors in vivo. J. Agric. Food Chem. 2014, 62, 10752–10758.
- Das Gupta, S.; Suh, N. Tocopherols in cancer: An update. Mol. Nutr. Food Res. 2016, 60, 1354–1363.
- Dobrzynski, M.; Kuropka, P.; Léskow, A.; Herman, K.; Tarnowska, M.; Wiglusz, R. Co-expression of the aryl hydrocarbon receptor and estrogen receptor in the developing teeth of rat offspring after rat mothers’ exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin and the protective action of α-tocopherol and acetylsalicylic acid. Adv. Clin. Exp. Med. 2019, 28, 973–980.
- Michaud-Levesque, J.; Bousquet-Gagnon, N.; Béliveau, R. Quercetin abrogates IL-6/STAT3 signaling and inhibits glioblastoma cell line growth and migration. Exp. Cell Res. 2012, 318, 925–935.
- Xing, N.; Chen, Y.; Mitchell, S.H.; Young, C.Y.F. Quercetin inhibits the expression and function of the androgen receptor in LNCaP prostate cancer cells. Carcinogenesis 2001, 22, 409–414.
- Xing, N.; Yang, F.; Song, L.; Wang, H.; Wang, J.; Xu, Z.; Xing, N. Quercetin in prostate cancer: Chemotherapeutic and chemopreventive effects, mechanisms and clinical application potential (review). Oncol. Rep. 2015, 33, 2659–2668.
- Hassen, I.; Casabianca, H.; Hosni, K. Biological activities of the natural antioxidant oleuropein: Exceeding the expectation—A mini-review. J. Funct. Foods 2015, 18, 926–940.
- Menendez, J.A.; Joven, J.; Aragonès, G.; Barrajón-Catalán, E.; Beltrán-Debón, R.; Borrás-Linares, I.; Camps, J.; Corominas-Faja, B.; Cufí, S.; Fernández-Arroyo, S.; et al. Xenohormetic and anti-aging activity of secoiridoid polyphenols present in extra virgin olive oil: A new family of gerosuppressant agents. Cell Cycle 2013, 12, 555–578.
- Rigacci, S.; Stefani, M. Nutraceutical properties of olive oil polyphenols. An itinerary from cultured cells through animal models to humans. Int. J. Mol. Sci. 2016, 17, 843.
- Fabiani, R. Anti-cancer properties of olive oil secoiridoid phenols: A systematic review of in vivo studies. Food Funct. 2016, 7, 4145–4159.
- Zhang, Y.; Yuan, Y.; Wu, H.; Xie, Z.; Wu, Y.; Song, X.; Wang, J.; Shu, W.; Xu, J.; Liu, B.; et al. Effect of verbascoside on apoptosis and metastasis in human oral squamous cell carcinoma. Int. J. Cancer 2018, 143, 980–991.
- Jia, W.Q.; Wang, Z.T.; Zou, M.M.; Lin, J.H.; Li, Y.H.; Zhang, L.; Xu, R.X. Verbascoside inhibits glioblastoma cell proliferation, migration and invasion while promoting apoptosis through upregulation of protein tyrosine phosphatase SHP-1 and inhibition of STAT3 phosphorylation. Cell. Physiol. Biochem. 2018, 47, 1871–1882.
- Fabiani, R.; Rosignoli, P.; De Bartolomeo, A.; Fuccelli, R.; Servili, M.; Montedoro, G.F.; Morozzi, G. Oxidative DNA damage is prevented by extracts of olive oil, hydroxytyrosol, and other olive phenolic compounds in human blood mononuclear cells and HL60 cells. J. Nutr. 2008, 138, 1411–1416.
- López-Biedma, A.; Sánchez-Quesada, C.; Beltrán, G.; Delgado-Rodríguez, M.; Gaforio, J.J. Phytoestrogen (+)-pinoresinol exerts antitumor activity in breast cancer cells with different oestrogen receptor statuses. BMC Complement. Altern. Med. 2016, 16, 350.
- Fini, L.; Hotchkiss, E.; Fogliano, V.; Graziani, G.; Romano, M.; De Vol, E.B.; Qin, H.; Selgrad, M.; Boland, C.R.; Ricciardiello, L. Chemopreventive properties of pinoresinol-rich olive oil involve a selective activation of the ATM-p53 cascade in colon cancer cell lines. Carcinogenesis 2008, 29, 139–146.
More
