Pulmonary fibrosis (PF) is an interstitial lung disease characterized by the destruction of the pulmonary parenchyma caused by excessive extracellular matrix deposition. Despite the well-known etiological factors such as senescence, aberrant epithelial cell and fibroblast activation, and chronic inflammation, PF has recently been recognized as a metabolic disease and abnormal lipid signature was observed both in serum and bronchoalveolar lavage fluid (BALF) of PF patients and mice PF model. Clinically, observational studies suggest a significant link between high-fat diet (HFD) and PF as manifested by high intake of saturated fatty acids (SFAs) and meat increases the risk of PF and mice lung fibrosis. However, the possible mechanisms between HFD and PF remain unclear. In tThe current review weis research emphasizes the diversity effects of the epigenetic dysregulation induced by HFD on the fibrotic factors such as epithelial cell injury, abnormal fibroblast activation and chronic inflammation. Finally, we discuss tThe potential ways for patients to improve their conditions and emphasize the prospect of targeted therapy based on epigenetic regulation for scientific researchers or drug developers was discussed here.
- high fat diet
- lipid biomolecule
- epigenetic regulation
- pulmonary fibrosis
1. Introduction
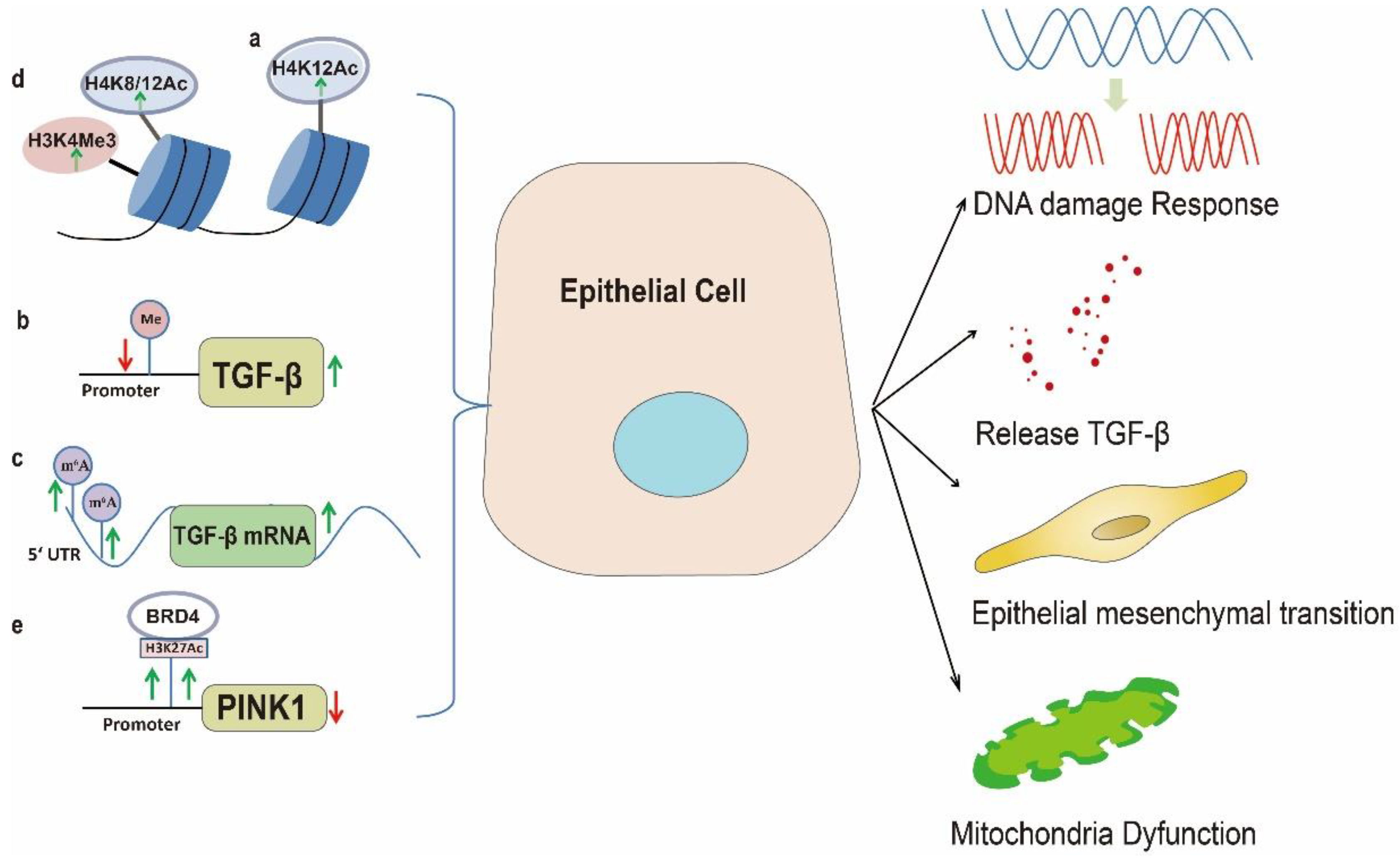
Pulmonary fibrosis (PF) is an interstitial lung disease characterized by inflammation and destruction of lung parenchyma caused by accelerated extra cellular matrix (ECM) deposition [1,2][1][2] which impaired gas exchange thus decreased quality of life [3]. Despite the well-known etiological factors such as senescence and aging, PF is recently recognized as a metabolic disease [4,5][4][5] and abnormal lipid signature has been observed both in serum and bronchoalveolar lavage fluid (BALF) of PF patients and mice model suggesting disturbed lipid metabolism [6,7,8][6][7][8]. Currently there is no cure for PF except for lung transplantation therefore revealing the potential pathogenic factors and possible mechanisms would contribute to the prevention and treatment of this deadly disease. A high-fat diet (HFD) is well accepted as a critical factor for obesity, a major global health issue [9,10,11][9][10][11] and is associated with poor outcomes in respiratory disease such as acute respiratory distress syndrome and PF [12,13,14,15,16][12][13][14][15][16]. HFD could induce various potentially harmful effects to the lungs, including the decreased number and function of mitochondria and increased lung inflammation and abnormal epithelial stem cell proliferation [17]. Observational studies found a significant link between HFD and PF [14,18][14][18] as manifested by high intake of SFAs and meat increased the risk of PF and mice lung fibrosis [19,20,21,22][19][20][21][22]. The exposure to lipids rich diets worsen the airway responsiveness to challenging agent [22,23][22][23] and further increased the incidence of PF [24,25][24][25]. The involvement of dietary lipids in PF was further supported by the fact that alterations in lipid metabolism enzymes could exacerbate PF progression [26] and that PF patients presented decreased mitochondrial β-oxidation capacity [27]. Taken together these studies reinforced the notion that dietary lipids are direct causative factors of PF which indicated that HFD was tightly associated with the initiation and progression of PF. Nevertheless, the mechanisms that individuals with HFD are susceptible to PF remained unclear. Since genomic mutations induced by HFD are very low [28], it is highly probable that epigenetic changes might contribute to HFD related PF. In tThe current review we researchers discussed the effects of HFD induced epigenetic dysregulation on the progression of PF such as epithelial cell injury, abnormal fibroblast activation and chronic inflammation and discussed the possible intervention methods to reduce the risk of HFD related PF. Therefore, this reviewsearch not only unveiled novel mechanisms but also provided with possible intervention methods to improve the condition of HFD related PF2. Epithelial Cell Injury and Abnormal Activation
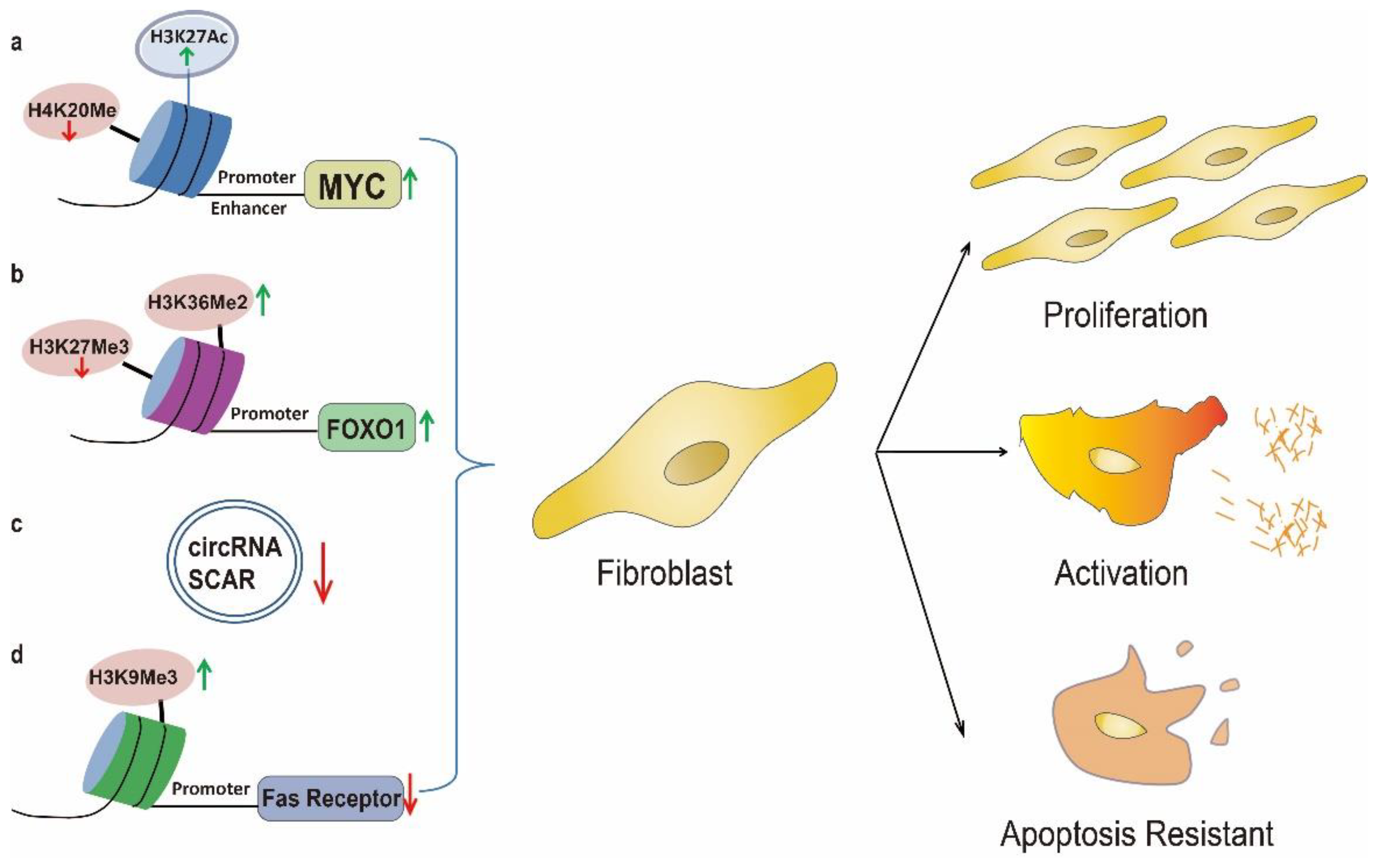
3. Uncontrolled Fibroblast Activation
4. Chronic Inflammation
Chronic inflammation played important roles in the progression of PF [106][96]. In addition to the above mentioned repair mechanisms: activation of lung epithelium stem cells and fibroblast upon injury, the immune response was simultaneously triggered to protect the tissue from further damages [107][97]. However, this process is out of control with chronic repeated injuries (typically observed in lung fibrosis) [108,109][98][99]. Due to continuous inflammation, secretion of pro-inflammatory cytokines such as IL-6 and tumor necrosis factor α (TNFα) will be increased which could further augment wound healing process by facilitating matrix deposition and subsequently resulted in fibrotic progression [103,104][93][94]. Therefore identification of factors participating in the onset and progression of inflammation is vital for comprehensively understanding inflammation related disorders such as PF [106,110][96][100]. Nutritional factors contributed to the formation of pro-inflammatory niche [111][101] and chronic low-grade inflammation induced by altered metabolic homeostasis appeared to be vital for the pathogenesis of organ fibrosis [112][102]. Chronic HFD has been linked with low-grade systemic inflammation in obesity [113][103]. Particularly, the consumption of western type HFD could provoke chronic metabolic inflammation which subsequently contributed to the progression of chronic diseases such as nonalcoholic steatohepatitis and lung fibrosis [114][104]. In HFD related obesity, adipocytes and macrophages in the adipose tissue generated pro-inflammatory cytokines such as TNFα and IL-6 which could provoke systemic inflammation and contribute to the progression of lung fibrosis [115,116][105][106]. A recent report showed that mice displayed increased pulmonary neutrophile accumulation and collagen deposition by feeding with HFD [25] and mice feed on HFD exhibited granulomatous lung inflammations which subsequently lead to progressive lung fibrosis [117][107]. However, the mechanisms through which HFD provoked inflammation response remained unclear. It has been well established that epigenetic modifications upon environmental factor stimulation played a fundamental role in regulation of inflammatory gene transcription [110,118,119,120][100][108][109][110]. Worse still, the adverse effects of the inflammatory state may induce epigenetic changes that perpetuate inflammation [121][111]. Therefore, wthe researchers postulated that epigenetic signature alterations induced by HFD may exacerbate inflammatory responses, thereby influencing progression of chronic inflammatory disease such as lung fibrosis [101,110,118][91][100][108]. In support of this, integrative epigenome wide association study showed that promoter methylation of TNFA were decreased with consumption of dietary fat [122][112] suggesting a nutrient epigenomic regulation of pro-inflammatory factors [123][113]. Accordingly high cholesterol and HFD would lead to low-grade pulmonary inflammation through activating TLR4/NFκB signaling [45][35] while significant lower methylation of CpGs in the first exon of the TLR4 were observed in obese individuals, indicating epigenetic regulation of TLR4 expression in obesity [48][38]. Mice fed on HFD exhibited significant reduced DNA methylation at the promoter of Pparγ1 which was critical for pro-inflammatory macrophages activation [124][114]. In adipose tissue, the DNMT3a methyltransferase was markedly increased which was accompanied by elevated expression of inflammatory cytokines such as TNFα and MCP-1, implying the role of DNMT3a in obesity related inflammation [125][115]. Similarly, increased expression of DNMT3b was found in adipose tissue macrophages and involved in the polarization of macrophage and inflammation [126][116]. In mice, HFD led to hypermethylation of the Ankrd26 which in turn contributed to enhanced secretion of pro-inflammatory factors [127][117]. Consistently, epigenetic silencing of the ANKRD26 by promoter methylation was related to pro-inflammatory state in obese individuals [128][118]. In addition to DNA methylation, significant association between expression of histone deacetylases and inflammation status was demonstrated in obese individuals [129][119]. Upon HFD treatment the levels of sphingosine-1-phosphate were increased [130][120] which subsequently inhibited histone deacetylases activity and increased histone acetylation at H3K9, H4K8 and H3K18, thereby promoting pro-inflammatory cytokines in BALF [131][121]. Accordingly, sphingosine-1-phosphate was reported to be increased in IPF patients and could facilitate disease progression [132,133][122][123]. Moreover ncRNAs, due to their versatile roles in the regulation of gene expression, are widely involved in HFD induced chronic inflammation. Consumption of high-fat or high calorie (rich in fat) diet was shown to increase inflammatory response by altering miRNA expression [134,135][124][125] and bioinformatics study showed that a miRNAs network significantly associated with obesity related inflammation [136][126] and deregulated circulating inflammatory miRNAs contributed to the elevated inflammatory state in obesity [137][127]. Besides, adipocyte-secreted exosomal miR-34 was progressively increased with the development of dietary obesity and subsequent systemic inflammation [138][128]. In the same way, HFD could increase miR-155 in adipocyte-derived microvesicles which could induce M1 macrophage polarization, thereby causing chronic inflammation [139][129]. HFD could further down regulate miR-30 by DNA methylation which facilitated M1 macrophages polarization [140][130]. Although the pro-inflammatory M1 macrophages are usually regarded as anti-fibrotic in lung fibrosis, they could exacerbate the inflammatory status of the lung injury and evoke the fibrotic response in lung fibrosis patients through activation of the TLR4 signaling [141][131]. Worse still, the maternal HFD could further hinder the lung development and function of offspring by epigenetic modulations [142,143][132][133] for example, maternal HFD could lead to offspring tissue inflammation through down regulation of miR-706 [144][134]. Since HFD could also suppress the expression of miR-26a and stimulate expression of pro-inflammatory cytokines such as TNFα [145][135] while decreasing TNFα was demonstrated to improve lung function of PF patient [146][136] therefore, this provide us with novel target for treating HFD related PF. Taken together the above evidence highlighted the crucial roles of epigenetic regulated inflammation in HFD induced lung fibrosis (Figure 3).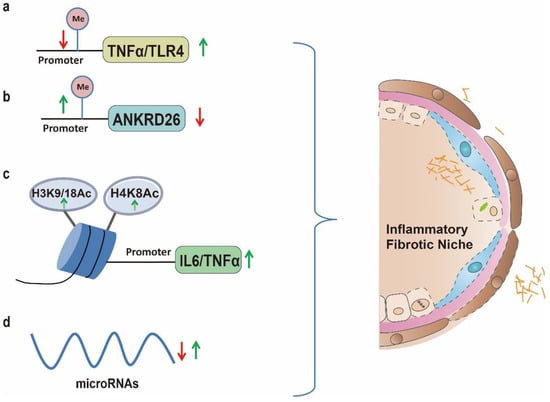
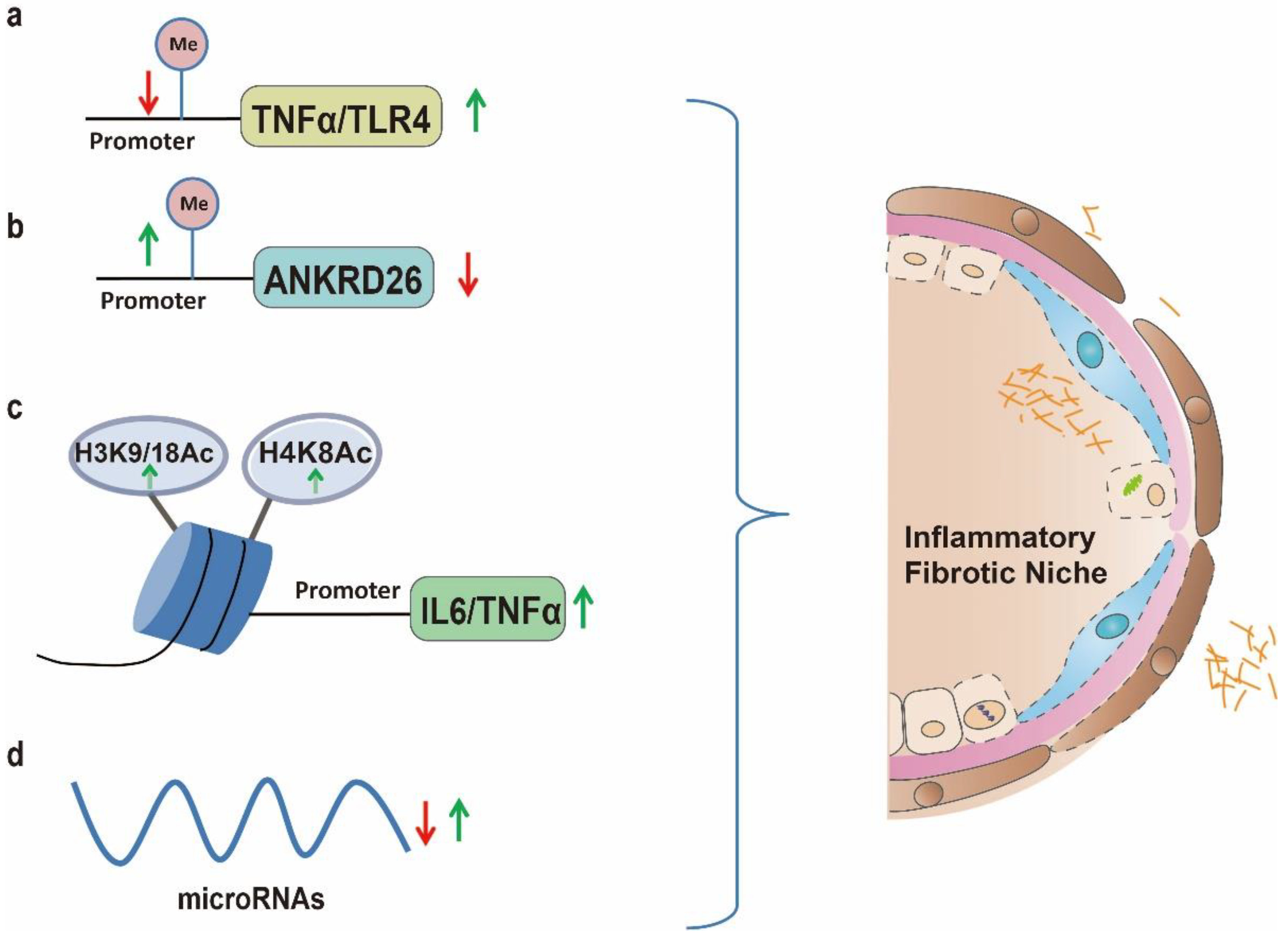 Figure 3. Epigenetically activation of inflammatory related genes play significant roles in fostering inflammation niche by HFD, thereby facilitating lung fibrosis progression. (a): HFD promotes TLR4 expression through demethylation of CpGs in the first exon whereby lead to low-grade pulmonary inflammation; (b): HFD led to hypermethylation of the Ankrd26 promoter region which in turn contribute to enhanced secretion of pro-inflammatory factors; (c): HFD increases histone acetylation at H3K9, H4K8, H3K18 which promoted pro-inflammatory cytokines in BALF; (d): Consumption of high-fat or high calorie diet (rich in fat) is shown to increase inflammatory response by altering miRNA expression such as increasing miR-155 and down regulating miR-30 could induce M1 macrophage polarization, thereby causing chronic inflammation.
Figure 3. Epigenetically activation of inflammatory related genes play significant roles in fostering inflammation niche by HFD, thereby facilitating lung fibrosis progression. (a): HFD promotes TLR4 expression through demethylation of CpGs in the first exon whereby lead to low-grade pulmonary inflammation; (b): HFD led to hypermethylation of the Ankrd26 promoter region which in turn contribute to enhanced secretion of pro-inflammatory factors; (c): HFD increases histone acetylation at H3K9, H4K8, H3K18 which promoted pro-inflammatory cytokines in BALF; (d): Consumption of high-fat or high calorie diet (rich in fat) is shown to increase inflammatory response by altering miRNA expression such as increasing miR-155 and down regulating miR-30 could induce M1 macrophage polarization, thereby causing chronic inflammation.
5. Clinical Perspectives
In tThe current review weresearchers emphasized the important roles of epigenetic regulation in HFD related lung fibrosis. HFD is well accepted as a critical factor leading to the obesity [11]. Paradoxically, previous multicenter study showed that body weight loss predicted worse survival of PF patients [147][137]. This issue was due to the currently used body weight measurement which neglected the body mass composition [148][138]. Actually individuals with the same body mass may varied in composition including fat mass (FM) and fat-free mass (FFM; or lean mass) which played different roles in health outcomes [148,149][138][139]. Large prospective cohort studies demonstrated that increased FM could significantly increase the risk while FFM reduced the risk of inflammation related and respiratory diseases [149,150][139][140] which mean that hidden loss of FFM or lean mass rather than weight loss was related to increased systemic inflammatory [151][141] since elevated expression of inflammation related genes were induced by HFD related FM increase [152][142]. On the contrary increased proportion of FFM was associated with better lung condition [153][143] which could be attributed to lower inflammation. It is well established that high intake of polyunsaturated fatty acids (PUFAs) has been associated with reduction of adiposity and increases in lean body mass [154][144]. However in the last decades, the daily diets FA intake has dramatically changed from monounsaturated and PUFAs rich pattern to a westernized pattern characterized by a high content in SFAs [155][145]. Accordingly, a previous comparative study showed that SFAs intake could increase the risk of PF [13] while the beneficial effects of PUFAs on mitigating lung fibrosis have been demonstrated in many studies. Intake of fish oil rich in eicosapentaenoic acid decreased bleomycin induced lung hydroxyproline accumulation [156][146]. Furthermore, the mitigation of lung fibrosis has been demonstrated with long-chain ω-3 PUFA docosahexaenoic acid [157][147] and short-chain ω-3 PUFA [158,159][148][149]. A relevant case showed that maternal diet supplied with docosahexaenoic acid could alleviate lung fibrosis and improve lung function in offspring by reducing collagen deposition and lessening inflammation [160][150]. These anti-fibrotic properties of PUFA could be mediated through inhibiting EMT in human AEC2s [161][151] and through activating PPARγ signaling [162][152]. Since HFD is critical contributor to fat body mass increase and obesity [11,152][11][142] while intake of PUFAs has been associated with reduction of adiposity and increases in fat-free body mass [154][144] therefore, an adequate dietary PUFAs intake might reduce the risk of HFD related lung fibrosis. Indeed observational data from a cohort of 104 Japanese patients showed that SFAs intake may be an independent risk factor for PF [13] while consumption of fruit was associated with a reduced risk [163][153]. Therefore a shift of dietary habit should be recommended for individuals with a high fat mass to avoid the occurrence of PF. Alternatively, an relative easy way for HFD individual to reduce the risk of lung fibrosis might be exercise since a latest study demonstrated that aerobic exercise could alleviate PF by ameliorating HFD induced inflammatory response and neutrophil infiltration [164][154].6. Conclusions
Recently, lung fibrosis is recognized as a metabolic disease and abnormal lipid signature was observed both in serum and BALF of PF patients and mice model, suggesting that lipid metabolism was unbalanced in lung fibrosis. Consistently clinical observation and animal studies showed that HFD was associated with the progression of lung fibrosis [14,18,21,24,25][14][18][21][24][25]. However, the mechanisms of individuals with HFD are susceptible to lung fibrosis remained unclear. Since genomic mutation induced by HFD is very low, it is highly probable that epigenetic changes might contribute to HFD related lung fibrosis. In tThe current review we researchers highlight the vital roles of epigenetic dysregulation in HFD induced PF from the perspective of epithelial cell injury, abnormal fibroblast activation and chronic inflammation. This knowledge opens new possibilities for a potential use of epigenetic signatures as biomarkers for diagnosis and targets for PF management [110][100]. Currently, there is no cure for PF except for lung transplantation therefore, revealing the potential pathogenic factors and possible mechanisms would contribute to the prevention and treatment of this deadly disease. Due to the reversible nature, intervention methods targeting dysregulated epigenetic regulation represented a promising way to treat lung fibrosis [165,166,167][155][156][157]. For a long time the weresearchers have studied on the therapeutic effects of miRNAs mimics in treating lung fibrosis [168,169][158][159]. Recently, wthe researchers generated MRG-229, a next-generation miR-29 mimic with improved stability and potential for targeted delivery which showed significant anti-fibrotic effects on human precision cut lung slices and mice lung fibrosis model and showed no adverse effects on non-human primates cynomolgus monkeys [168][158]. Accordingly, delivering circRNA SCAR using nanoparticle could suppress fibroblast activation in HFD treated mice [102][92]. The above evidence demonstrated the vital roles of targeting abnormal epigenetic regulation in ameliorating PF progression. In summary, ourthe review search not only unveil the important roles of epigenetic regulation in HFD mediated PF but also provide potential ways to deal with this issue. For patients they could change their diet habitat and do more aerobic exercise [158,159,164][148][149][154] while for scientific researchers or drug developers, unveiling the epigenetic mechanism of HFD related lung fibrosis will provide novel targets to treat this deadly disease.References
- Moss, B.J.; Ryter, S.W.; Rosas, I.O. Pathogenic Mechanisms Underlying Idiopathic Pulmonary Fibrosis. Annu. Rev. Pathol. 2022, 17, 515–546.
- Wang, C.; Yang, J. Mechanical forces: The missing link between idiopathic pulmonary fibrosis and lung cancer. Eur. J. Cell Biol. 2022, 101, 151234.
- Wijsenbeek, M.; Suzuki, A.; Maher, T.M. Interstitial lung diseases. Lancet 2022, 400, 769–786.
- Zhao, X.; Kwan, J.Y.Y.; Yip, K.; Liu, P.P.; Liu, F.F. Targeting metabolic dysregulation for fibrosis therapy. Nat. Rev. Drug Discov. 2020, 19, 57–75.
- Bueno, M.; Calyeca, J.; Rojas, M.; Mora, A.L. Mitochondria dysfunction and metabolic reprogramming as drivers of idiopathic pulmonary fibrosis. Redox Biol. 2020, 33, 101509.
- Yang, X.H.; Wang, F.F.; Chi, X.S.; Wang, X.M.; Cong, J.P.; Hu, Y.; Zhang, Y.Z. Disturbance of serum lipid metabolites and potential biomarkers in the Bleomycin model of pulmonary fibrosis in young mice. BMC Pulm. Med. 2022, 22, 176.
- Deng, X.; Hao, C.; Li, Y.; Guo, Y.; Si, H.; He, J.; Deng, M.; Niu, Z.; Wang, C.; Xu, X.; et al. Lysophosphatidylcholine acyltransferase 1 alleviates silica-induced pulmonary fibrosis by modulating lipid metabolism. Biomed. Pharmacother. 2022, 155, 113638.
- Nambiar, S.; Tan, D.B.A.; Clynick, B.; Bong, S.H.; Rawlinson, C.; Gummer, J.; Corte, T.J.; Glaspole, I.; Moodley, Y.P.; Trengove, R. Untargeted metabolomics of human plasma reveal lipid markers unique to chronic obstructive pulmonary disease and idiopathic pulmonary fibrosis. Proteomics. Clin. Appl. 2021, 15, e2000039.
- Malik, V.S.; Willet, W.C.; Hu, F.B. Nearly a decade on—Trends, risk factors and policy implications in global obesity. Nat. Reviews. Endocrinol. 2020, 16, 615–616.
- Bendor, C.D.; Bardugo, A.; Pinhas-Hamiel, O.; Afek, A.; Twig, G. Cardiovascular morbidity, diabetes and cancer risk among children and adolescents with severe obesity. Cardiovasc. Diabetol. 2020, 19, 79.
- Shin, A.C.; MohanKumar, S.M.; Sirivelu, M.P.; Claycombe, K.J.; Haywood, J.R.; Fink, G.D.; MohanKumar, P.S. Chronic exposure to a high-fat diet affects stress axis function differentially in diet-induced obese and diet-resistant rats. Int. J. Obes. 2010, 34, 1218–1226.
- Peters, U.; Suratt, B.T.; Bates, J.H.T.; Dixon, A.E. Beyond BMI: Obesity and Lung Disease. Chest 2018, 153, 702–709.
- Miyake, Y.; Sasaki, S.; Yokoyama, T.; Chida, K.; Azuma, A.; Suda, T.; Kudoh, S.; Sakamoto, N.; Okamoto, K.; Kobashi, G.; et al. Dietary fat and meat intake and idiopathic pulmonary fibrosis: A case-control study in Japan. Int. J. Tuberc. Lung Dis. Off. J. Int. Union Against Tuberc. Lung Dis. 2006, 10, 333–339.
- Guo, X.; Sunil, C.; Qian, G. Obesity and the Development of Lung Fibrosis. Front. Pharmacol. 2021, 12, 812166.
- Gong, M.N.; Bajwa, E.K.; Thompson, B.T.; Christiani, D.C. Body mass index is associated with the development of acute respiratory distress syndrome. Thorax 2010, 65, 44–50.
- Anderson, M.R.; Kim, J.S.; Allison, M.; Giles, J.T.; Hoffman, E.A.; Ding, J.; Barr, R.G.; Podolanczuk, A. Adiposity and Interstitial Lung Abnormalities in Community-Dwelling Adults: The MESA Cohort Study. Chest 2021, 160, 582–594.
- Hegab, A.E.; Ozaki, M.; Meligy, F.Y.; Kagawa, S.; Ishii, M.; Betsuyaku, T. High fat diet activates adult mouse lung stem cells and accelerates several aging-induced effects. Stem Cell Res. 2018, 33, 25–35.
- Han, H.; Chung, S.I.; Park, H.J.; Oh, E.Y.; Kim, S.R.; Park, K.H.; Lee, J.H.; Park, J.W. Obesity-induced Vitamin D Deficiency Contributes to Lung Fibrosis and Airway Hyperresponsiveness. Am. J. Respir. Cell Mol. Biol. 2021, 64, 357–367.
- Thompson, J.A.; Johnston, R.A.; Price, R.E.; Hubbs, A.F.; Kashon, M.L.; McKinney, W.; Fedan, J.S. High-fat Western diet consumption exacerbates silica-induced pulmonary inflammation and fibrosis. Toxicol. Rep. 2022, 9, 1045–1053.
- Hegab, A.E.; Ozaki, M.; Kagawa, S.; Fukunaga, K. Effect of High Fat Diet on the Severity and Repair of Lung Fibrosis in Mice. Stem Cells Dev. 2021, 30, 908–921.
- Chu, S.G.; Villalba, J.A.; Liang, X.; Xiong, K.; Tsoyi, K.; Ith, B.; Ayaub, E.A.; Tatituri, R.V.; Byers, D.E.; Hsu, F.F.; et al. Palmitic Acid-Rich High-Fat Diet Exacerbates Experimental Pulmonary Fibrosis by Modulating Endoplasmic Reticulum Stress. Am. J. Respir. Cell Mol. Biol. 2019, 61, 737–746.
- Park, Y.H.; Oh, E.Y.; Han, H.; Yang, M.; Park, H.J.; Park, K.H.; Lee, J.H.; Park, J.W. Insulin resistance mediates high-fat diet-induced pulmonary fibrosis and airway hyperresponsiveness through the TGF-beta1 pathway. Exp. Mol. Med. 2019, 51, 1–12.
- Ye, Z.; Huang, Y.; Liu, D.; Chen, X.; Wang, D.; Huang, D.; Zhao, L.; Xiao, X. Obesity induced by neonatal overfeeding worsens airway hyperresponsiveness and inflammation. PLoS ONE 2012, 7, e47013.
- Ge, X.N.; Greenberg, Y.; Hosseinkhani, M.R.; Long, E.K.; Bahaie, N.S.; Rao, A.; Ha, S.G.; Rao, S.P.; Bernlohr, D.A.; Sriramarao, P. High-fat diet promotes lung fibrosis and attenuates airway eosinophilia after exposure to cockroach allergen in mice. Exp. Lung Res. 2013, 39, 365–378.
- Vedova, M.C.D.; Soler Garcia, F.M.; Munoz, M.D.; Fornes, M.W.; Gomez Mejiba, S.E.; Gomez, N.N.; Ramirez, D.C. Diet-Induced Pulmonary Inflammation and Incipient Fibrosis in Mice: A Possible Role of Neutrophilic Inflammation. Inflammation 2019, 42, 1886–1900.
- Sunaga, H.; Matsui, H.; Ueno, M.; Maeno, T.; Iso, T.; Syamsunarno, M.R.; Anjo, S.; Matsuzaka, T.; Shimano, H.; Yokoyama, T.; et al. Deranged fatty acid composition causes pulmonary fibrosis in Elovl6-deficient mice. Nat. Commun. 2013, 4, 2563.
- Zhao, Y.D.; Yin, L.; Archer, S.; Lu, C.; Zhao, G.; Yao, Y.; Wu, L.; Hsin, M.; Waddell, T.K.; Keshavjee, S.; et al. Metabolic heterogeneity of idiopathic pulmonary fibrosis: A metabolomic study. BMJ Open Respir. Res. 2017, 4, e000183.
- Li, J.; Huang, J.; Li, J.S.; Chen, H.; Huang, K.; Zheng, L. Accumulation of endoplasmic reticulum stress and lipogenesis in the liver through generational effects of high fat diets. J. Hepatol. 2012, 56, 900–907.
- Burgoyne, R.A.; Fisher, A.J.; Borthwick, L.A. The Role of Epithelial Damage in the Pulmonary Immune Response. Cells 2021, 10, 2763.
- Kobayashi, Y.; Tata, A.; Konkimalla, A.; Katsura, H.; Lee, R.F.; Ou, J.; Banovich, N.E.; Kropski, J.A.; Tata, P.R. Persistence of a regeneration-associated, transitional alveolar epithelial cell state in pulmonary fibrosis. Nat. Cell Biol. 2020, 22, 934–946.
- Kathiriya, J.J.; Wang, C.; Zhou, M.; Brumwell, A.; Cassandras, M.; Le Saux, C.J.; Cohen, M.; Alysandratos, K.D.; Wang, B.; Wolters, P.; et al. Human alveolar type 2 epithelium transdifferentiates into metaplastic KRT5(+) basal cells. Nat. Cell Biol. 2022, 24, 10–23.
- Katzen, J.; Beers, M.F. Contributions of alveolar epithelial cell quality control to pulmonary fibrosis. J. Clin. Investig. 2020, 130, 5088–5099.
- Hwang, S.; Chung, K.W. Targeting fatty acid metabolism for fibrotic disorders. Arch. Pharmacal Res. 2021, 44, 839–856.
- Buren, J.; Ericsson, M.; Damasceno, N.R.T.; Sjodin, A. A Ketogenic Low-Carbohydrate High-Fat Diet Increases LDL Cholesterol in Healthy, Young, Normal-Weight Women: A Randomized Controlled Feeding Trial. Nutrients 2021, 13, 814.
- Fang, Y.; Wang, S.; Zhu, T.; Zhang, Y.; Lian, X. Atherogenic high cholesterol/high fat diet induces TLRs-associated pulmonary inflammation in C57BL/6J mice. Inflamm. Res. 2017, 66, 39–47.
- Li, X.; Ma, L.; Wei, Y.; Gu, J.; Liang, J.; Li, S.; Cui, Y.; Liu, R.; Huang, H.; Yang, C.; et al. Cabozantinib ameliorates lipopolysaccharide-induced lung inflammation and bleomycin--induced early pulmonary fibrosis in mice. Int. Immunopharmacol. 2021, 101, 108327.
- Dowson, C.; O’Reilly, S. DNA methylation in fibrosis. Eur. J. Cell Biol. 2016, 95, 323–330.
- Remely, M.; Aumueller, E.; Jahn, D.; Hippe, B.; Brath, H.; Haslberger, A.G. Microbiota and epigenetic regulation of inflammatory mediators in type 2 diabetes and obesity. Benef. Microbes 2014, 5, 33–43.
- Strunz, M.; Simon, L.M.; Ansari, M.; Kathiriya, J.J.; Angelidis, I.; Mayr, C.H.; Tsidiridis, G.; Lange, M.; Mattner, L.F.; Yee, M.; et al. Alveolar regeneration through a Krt8+ transitional stem cell state that persists in human lung fibrosis. Nat. Commun. 2020, 11, 3559.
- Auyeung, V.C.; Downey, M.S.; Thamsen, M.; Wenger, T.A.; Backes, B.J.; Sheppard, D.; Papa, F.R. IRE1alpha drives lung epithelial progenitor dysfunction to establish a niche for pulmonary fibrosis. Am. J. Physiol. Lung Cell. Mol. Physiol. 2022, 322, L564–L580.
- Schuliga, M.; Kanwal, A.; Read, J.; Blokland, K.E.C.; Burgess, J.K.; Prele, C.M.; Mutsaers, S.E.; Grainge, C.; Thomson, C.; James, A.; et al. A cGAS-dependent response links DNA damage and senescence in alveolar epithelial cells: A potential drug target in IPF. Am. J. Physiol. Lung Cell. Mol. Physiol. 2021, 321, L859–L871.
- Millan-Zambrano, G.; Burton, A.; Bannister, A.J.; Schneider, R. Histone post-translational modifications—Cause and consequence of genome function. Nat. Reviews. Genet. 2022, 23, 563–580.
- Shvedunova, M.; Akhtar, A. Modulation of cellular processes by histone and non-histone protein acetylation. Nat. Reviews. Mol. Cell Biol. 2022, 23, 329–349.
- Han, X.; Tian, M.; Shliaha, P.V.; Zhang, J.; Jiang, S.; Nan, B.; Alam, M.N.; Jensen, O.N.; Shen, H.; Huang, Q. Real-world particulate matters induce lung toxicity in rats fed with a high-fat diet: Evidence of histone modifications. J. Hazard. Mater. 2021, 416, 126182.
- Peng, L.; Wen, L.; Shi, Q.F.; Gao, F.; Huang, B.; Meng, J.; Hu, C.P.; Wang, C.M. Scutellarin ameliorates pulmonary fibrosis through inhibiting NF-kappaB/NLRP3-mediated epithelial-mesenchymal transition and inflammation. Cell Death Dis. 2020, 11, 978.
- Wang, L.T.; Liu, K.Y.; Jeng, W.Y.; Chiang, C.M.; Chai, C.Y.; Chiou, S.S.; Huang, M.S.; Yokoyama, K.K.; Wang, S.N.; Huang, S.K.; et al. PCAF-mediated acetylation of ISX recruits BRD4 to promote epithelial-mesenchymal transition. EMBO Rep. 2020, 21, e48795.
- Uthaya Kumar, D.B.; Motakis, E.; Yurieva, M.; Kohar, V.; Martinek, J.; Wu, T.C.; Khoury, J.; Grassmann, J.; Lu, M.; Palucka, K.; et al. Bronchial epithelium epithelial-mesenchymal plasticity forms aberrant basaloid-like cells in vitro. Am. J. Physiol. Lung Cell. Mol. Physiol. 2022, 322, L822–L841.
- Tsukui, T.; Sun, K.H.; Wetter, J.B.; Wilson-Kanamori, J.R.; Hazelwood, L.A.; Henderson, N.C.; Adams, T.S.; Schupp, J.C.; Poli, S.D.; Rosas, I.O.; et al. Collagen-producing lung cell atlas identifies multiple subsets with distinct localization and relevance to fibrosis. Nat. Commun. 2020, 11, 1920.
- Qiu, X.; Bajinka, O.; Wang, L.; Wu, G.; Tan, Y. High-fat diet promotes epithelial-mesenchymal transition through enlarged growth of opportunistic pathogens and the intervention of saturated hydrogen. Am. J. Transl. Res. 2021, 13, 6016–6030.
- Yi, S.J.; Lee, H.; Lee, J.; Lee, K.; Kim, J.; Kim, Y.; Park, J.I.; Kim, K. Bone Remodeling: Histone Modifications as Fate Determinants of Bone Cell Differentiation. Int. J. Mol. Sci. 2019, 20, 3147.
- Kim, J.; Lee, H.; Yi, S.J.; Kim, K. Gene regulation by histone-modifying enzymes under hypoxic conditions: A focus on histone methylation and acetylation. Exp. Mol. Med. 2022, 54, 878–889.
- Wan, Q.L.; Meng, X.; Wang, C.; Dai, W.; Luo, Z.; Yin, Z.; Ju, Z.; Fu, X.; Yang, J.; Ye, Q.; et al. Histone H3K4me3 modification is a transgenerational epigenetic signal for lipid metabolism in Caenorhabditis elegans. Nat. Commun. 2022, 13, 768.
- Hu, L.; Yu, Y.; Huang, H.; Fan, H.; Hu, L.; Yin, C.; Li, K.; Fulton, D.J.; Chen, F. Epigenetic Regulation of Interleukin 6 by Histone Acetylation in Macrophages and Its Role in Paraquat-Induced Pulmonary Fibrosis. Front. Immunol. 2016, 7, 696.
- Liang, Z.L.; Wu, D.D.; Yao, Y.; Yu, F.Y.; Yang, L.; Tan, H.W.; Hylkema, M.N.; Rots, M.G.; Xu, Y.M.; Lau, A.T.Y. Epiproteome profiling of cadmium-transformed human bronchial epithelial cells by quantitative histone post-translational modification-enzyme-linked immunosorbent assay. J. Appl. Toxicol. JAT 2018, 38, 888–895.
- Phan, T.H.G.; Paliogiannis, P.; Nasrallah, G.K.; Giordo, R.; Eid, A.H.; Fois, A.G.; Zinellu, A.; Mangoni, A.A.; Pintus, G. Emerging cellular and molecular determinants of idiopathic pulmonary fibrosis. Cell. Mol. Life Sci. CMLS 2021, 78, 2031–2057.
- Wu, H.; Yu, Y.; Huang, H.; Hu, Y.; Fu, S.; Wang, Z.; Shi, M.; Zhao, X.; Yuan, J.; Li, J.; et al. Progressive Pulmonary Fibrosis Is Caused by Elevated Mechanical Tension on Alveolar Stem Cells. Cell 2020, 180, 107–121.e17.
- Izquierdo, V.; Palomera-Avalos, V.; Pallas, M.; Grinan-Ferre, C. Resveratrol Supplementation Attenuates Cognitive and Molecular Alterations under Maternal High-Fat Diet Intake: Epigenetic Inheritance over Generations. Int. J. Mol. Sci. 2021, 22, 1453.
- Feng, Y.; Dong, H.; Sun, B.; Hu, Y.; Yang, Y.; Jia, Y.; Jia, L.; Zhong, X.; Zhao, R. METTL3/METTL14 Transactivation and m6A-Dependent TGF-beta1 Translation in Activated Kupffer Cells. Cell. Mol. Gastroenterol. Hepatol. 2021, 12, 839–856.
- Pandey, A.; Goru, S.K.; Kadakol, A.; Malek, V.; Sharma, N.; Gaikwad, A.B. H2AK119 monoubiquitination regulates Angiotensin II receptor mediated macrophage infiltration and renal fibrosis in type 2 diabetic rats. Biochimie 2016, 131, 68–76.
- Siddeek, B.; Mauduit, C.; Chehade, H.; Blin, G.; Liand, M.; Chindamo, M.; Benahmed, M.; Simeoni, U. Long-term impact of maternal high-fat diet on offspring cardiac health: Role of micro-RNA biogenesis. Cell Death Discov. 2019, 5, 71.
- Thannickal, V.J.; Horowitz, J.C. Evolving concepts of apoptosis in idiopathic pulmonary fibrosis. Proc. Am. Thorac. Soc. 2006, 3, 350–356.
- Stancil, I.T.; Michalski, J.E.; Davis-Hall, D.; Chu, H.W.; Park, J.A.; Magin, C.M.; Yang, I.V.; Smith, B.J.; Dobrinskikh, E.; Schwartz, D.A. Pulmonary fibrosis distal airway epithelia are dynamically and structurally dysfunctional. Nat. Commun. 2021, 12, 4566.
- Carraro, G.; Mulay, A.; Yao, C.; Mizuno, T.; Konda, B.; Petrov, M.; Lafkas, D.; Arron, J.R.; Hogaboam, C.M.; Chen, P.; et al. Single-Cell Reconstruction of Human Basal Cell Diversity in Normal and Idiopathic Pulmonary Fibrosis Lungs. Am. J. Respir. Crit. Care Med. 2020, 202, 1540–1550.
- Jones, S.F.; Infante, J.R. Molecular Pathways: Fatty Acid Synthase. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2015, 21, 5434–5438.
- Plataki, M.; Fan, L.; Sanchez, E.; Huang, Z.; Torres, L.K.; Imamura, M.; Zhu, Y.; Cohen, D.E.; Cloonan, S.M.; Choi, A.M. Fatty acid synthase downregulation contributes to acute lung injury in murine diet-induced obesity. JCI Insight 2019, 5, e127823.
- Ng, M.Y.W.; Wai, T.; Simonsen, A. Quality control of the mitochondrion. Dev. Cell 2021, 56, 881–905.
- Mu, J.; Zhang, D.; Tian, Y.; Xie, Z.; Zou, M.H. BRD4 inhibition by JQ1 prevents high-fat diet-induced diabetic cardiomyopathy by activating PINK1/Parkin-mediated mitophagy in vivo. J. Mol. Cell. Cardiol. 2020, 149, 1–14.
- Knoell, J.; Chillappagari, S.; Knudsen, L.; Korfei, M.; Dartsch, R.; Jonigk, D.; Kuehnel, M.P.; Hoetzenecker, K.; Guenther, A.; Mahavadi, P. PACS2-TRPV1 axis is required for ER-mitochondrial tethering during ER stress and lung fibrosis. Cell. Mol. Life Sci. CMLS 2022, 79, 151.
- Dobrinskikh, E.; Hennessy, C.E.; Kurche, J.S.; Kim, E.; Estrella, A.M.; Cardwell, J.; Yang, I.V.; Schwartz, D.A. Epithelial ER Stress Enhances the Risk of Muc5b Associated Lung Fibrosis. Am. J. Respir. Cell Mol. Biol. 2023, 68, 62–74.
- Hernandez, T.L.; Sutherland, J.P.; Wolfe, P.; Allian-Sauer, M.; Capell, W.H.; Talley, N.D.; Wyatt, H.R.; Foster, G.D.; Hill, J.O.; Eckel, R.H. Lack of suppression of circulating free fatty acids and hypercholesterolemia during weight loss on a high-fat, low-carbohydrate diet. Am. J. Clin. Nutr. 2010, 91, 578–585.
- Staab-Weijnitz, C.A. Fighting the Fiber: Targeting Collagen in Lung Fibrosis. Am. J. Respir. Cell Mol. Biol. 2022, 66, 363–381.
- Kaufman, J.; Graf, B.A.; Leung, E.C.; Pollock, S.J.; Koumas, L.; Reddy, S.Y.; Blieden, T.M.; Smith, T.J.; Phipps, R.P. Fibroblasts as sentinel cells: Role of the CDcd40-CDcd40 ligand system in fibroblast activation and lung inflammation and fibrosis. Chest 2001, 120, 53S–55S.
- Qian, G.; Adeyanju, O.; Sunil, C.; Huang, S.K.; Chen, S.Y.; Tucker, T.A.; Idell, S.; Guo, X. Dedicator of Cytokinesis 2 (DOCK2) Deficiency Attenuates Lung Injury Associated with Chronic High-Fat and High-Fructose Diet-Induced Obesity. Am. J. Pathol. 2022, 192, 226–238.
- Sun, K.; Park, J.; Gupta, O.T.; Holland, W.L.; Auerbach, P.; Zhang, N.; Goncalves Marangoni, R.; Nicoloro, S.M.; Czech, M.P.; Varga, J.; et al. Endotrophin triggers adipose tissue fibrosis and metabolic dysfunction. Nat. Commun. 2014, 5, 3485.
- Kikuchi, T.; Sugiura, H.; Koarai, A.; Ichikawa, T.; Minakata, Y.; Matsunaga, K.; Nakanishi, M.; Hirano, T.; Akamatsu, K.; Yanagisawa, S.; et al. Increase of 27-hydroxycholesterol in the airways of patients with COPD: Possible role of 27-hydroxycholesterol in tissue fibrosis. Chest 2012, 142, 329–337.
- Sehlmeyer, K.; Ruwisch, J.; Roldan, N.; Lopez-Rodriguez, E. Alveolar Dynamics and Beyond—The Importance of Surfactant Protein C and Cholesterol in Lung Homeostasis and Fibrosis. Front. Physiol. 2020, 11, 386.
- Yao, X.; Gordon, E.M.; Figueroa, D.M.; Barochia, A.V.; Levine, S.J. Emerging Roles of Apolipoprotein E and Apolipoprotein A-I in the Pathogenesis and Treatment of Lung Disease. Am. J. Respir. Cell Mol. Biol. 2016, 55, 159–169.
- Yildirim, M.; Kayalar, O.; Atahan, E.; Oztay, F. Atorvastatin attenuates pulmonary fibrosis in mice and human lung fibroblasts, by the regulation of myofibroblast differentiation and apoptosis. J. Biochem. Mol. Toxicol. 2022, 36, e23074.
- Gu, X.; Han, Y.Y.; Yang, C.Y.; Ji, H.M.; Lan, Y.J.; Bi, Y.Q.; Zheng, C.; Qu, J.; Cheng, M.H.; Gao, J. Activated AMPK by metformin protects against fibroblast proliferation during pulmonary fibrosis by suppressing FOXM1. Pharmacol. Res. 2021, 173, 105844.
- Rashkovan, M.; Albero, R.; Gianni, F.; Perez-Duran, P.; Miller, H.I.; Mackey, A.L.; Paietta, E.M.; Tallman, M.S.; Rowe, J.M.; Litzow, M.R.; et al. Intracellular Cholesterol Pools Regulate Oncogenic Signaling and Epigenetic Circuitries in Early T-cell Precursor Acute Lymphoblastic Leukemia. Cancer Discov. 2022, 12, 856–871.
- Labbe, D.P.; Zadra, G.; Yang, M.; Reyes, J.M.; Lin, C.Y.; Cacciatore, S.; Ebot, E.M.; Creech, A.L.; Giunchi, F.; Fiorentino, M.; et al. High-fat diet fuels prostate cancer progression by rewiring the metabolome and amplifying the MYC program. Nat. Commun. 2019, 10, 4358.
- Qin, H.; Tang, Y.; Mao, Y.; Zhou, X.; Xu, T.; Liu, W.; Su, X. C-MYC induces idiopathic pulmonary fibrosis via modulation of miR-9-5p-mediated TBPL1. Cell. Signal. 2022, 93, 110274.
- Buras, E.D.; Converso-Baran, K.; Davis, C.S.; Akama, T.; Hikage, F.; Michele, D.E.; Brooks, S.V.; Chun, T.H. Fibro-Adipogenic Remodeling of the Diaphragm in Obesity-Associated Respiratory Dysfunction. Diabetes 2019, 68, 45–56.
- Schmidt, R.; Meier, U.; Markart, P.; Grimminger, F.; Velcovsky, H.G.; Morr, H.; Seeger, W.; Gunther, A. Altered fatty acid composition of lung surfactant phospholipids in interstitial lung disease. Am. J. Physiol. Lung Cell. Mol. Physiol. 2002, 283, L1079–L1085.
- Kumar, S.; Pamulapati, H.; Tikoo, K. Fatty acid induced metabolic memory involves alterations in renal histone H3K36me2 and H3K27me3. Mol. Cell. Endocrinol. 2016, 422, 233–242.
- Yu, B.; Jin, X.Q.; Yu, W.Y.; Dong, Y.Y.; Ying, H.Z.; Yu, C.H. 1beta-Hydroxyalantolactone from Inulae Flos alleviated the progression of pulmonary fibrosis via inhibiting JNK/FOXO1/NF-kappaB pathway. Int. Immunopharmacol. 2021, 101, 108339.
- Ferst, J.G.; Glanzner, W.G.; Gutierrez, K.; de Macedo, M.P.; Ferreira, R.; Gasperin, B.G.; Duggavathi, R.; Goncalves, P.B.; Bordignon, V. Supplementation of oleic acid, stearic acid, palmitic acid and beta-hydroxybutyrate increase H3K9me3 in endometrial epithelial cells of cattle cultured in vitro. Anim. Reprod. Sci. 2021, 233, 106851.
- Huang, S.K.; Scruggs, A.M.; Donaghy, J.; Horowitz, J.C.; Zaslona, Z.; Przybranowski, S.; White, E.S.; Peters-Golden, M. Histone modifications are responsible for decreased Fas expression and apoptosis resistance in fibrotic lung fibroblasts. Cell Death Dis. 2013, 4, e621.
- Pessoa Rodrigues, C.; Chatterjee, A.; Wiese, M.; Stehle, T.; Szymanski, W.; Shvedunova, M.; Akhtar, A. Histone H4 lysine 16 acetylation controls central carbon metabolism and diet-induced obesity in mice. Nat. Commun. 2021, 12, 6212.
- Zhang, X.; Liu, H.; Zhou, J.Q.; Krick, S.; Barnes, J.W.; Thannickal, V.J.; Sanders, Y.Y. Modulation of H4K16Ac levels reduces pro-fibrotic gene expression and mitigates lung fibrosis in aged mice. Theranostics 2022, 12, 530–541.
- Xue, T.; Qiu, X.; Liu, H.; Gan, C.; Tan, Z.; Xie, Y.; Wang, Y.; Ye, T. Epigenetic regulation in fibrosis progress. Pharmacol. Res. 2021, 173, 105910.
- Zhao, Q.; Liu, J.; Deng, H.; Ma, R.; Liao, J.Y.; Liang, H.; Hu, J.; Li, J.; Guo, Z.; Cai, J.; et al. Targeting Mitochondria-Located circRNA SCAR Alleviates NASH via Reducing mROS Output. Cell 2020, 183, 76–93.e22.
- Weiskirchen, R.; Weiskirchen, S.; Tacke, F. Organ and tissue fibrosis: Molecular signals, cellular mechanisms and translational implications. Mol. Asp. Med. 2019, 65, 2–15.
- Wynn, T.A.; Ramalingam, T.R. Mechanisms of fibrosis: Therapeutic translation for fibrotic disease. Nat. Med. 2012, 18, 1028–1040.
- Meng, X.M.; Nikolic-Paterson, D.J.; Lan, H.Y. TGF-beta: The master regulator of fibrosis. Nat. Reviews. Nephrol. 2016, 12, 325–338.
- Hey, J.; Paulsen, M.; Toth, R.; Weichenhan, D.; Butz, S.; Schatterny, J.; Liebers, R.; Lutsik, P.; Plass, C.; Mall, M.A. Epigenetic reprogramming of airway macrophages promotes polarization and inflammation in muco-obstructive lung disease. Nat. Commun. 2021, 12, 6520.
- Shenderov, K.; Collins, S.L.; Powell, J.D.; Horton, M.R. Immune dysregulation as a driver of idiopathic pulmonary fibrosis. J. Clin. Investig. 2021, 131, e143226.
- Ptasinski, V.A.; Stegmayr, J.; Belvisi, M.G.; Wagner, D.E.; Murray, L.A. Targeting Alveolar Repair in Idiopathic Pulmonary Fibrosis. Am. J. Respir. Cell Mol. Biol. 2021, 65, 347–365.
- Lee, S.M.; Shin, Y.J.; Go, R.E.; Bae, S.H.; Kim, C.W.; Kim, S.; Kim, M.S.; Choi, K.C. Inhalation exposure by cigarette smoke: Effects on the progression of bleomycin- and lipopolysaccharide-induced lung injuries in rat models. Toxicology 2021, 451, 152695.
- Ramos-Lopez, O.; Milagro, F.I.; Riezu-Boj, J.I.; Martinez, J.A. Epigenetic signatures underlying inflammation: An interplay of nutrition, physical activity, metabolic diseases, and environmental factors for personalized nutrition. Inflamm. Res. 2021, 70, 29–49.
- Ramallal, R.; Toledo, E.; Martinez, J.A.; Shivappa, N.; Hebert, J.R.; Martinez-Gonzalez, M.A.; Ruiz-Canela, M. Inflammatory potential of diet, weight gain, and incidence of overweight/obesity: The SUN cohort. Obesity 2017, 25, 997–1005.
- Richter, K.; Konzack, A.; Pihlajaniemi, T.; Heljasvaara, R.; Kietzmann, T. Redox-fibrosis: Impact of TGFbeta1 on ROS generators, mediators and functional consequences. Redox Biol. 2015, 6, 344–352.
- Ellulu, M.S.; Patimah, I.; Khaza’ai, H.; Rahmat, A.; Abed, Y. Obesity and inflammation: The linking mechanism and the complications. Arch. Med. Sci. AMS 2017, 13, 851–863.
- Christ, A.; Lauterbach, M.; Latz, E. Western Diet and the Immune System: An Inflammatory Connection. Immunity 2019, 51, 794–811.
- O’Rourke, R.W. Inflammation in obesity-related diseases. Surgery 2009, 145, 255–259.
- Tilg, H.; Moschen, A.R. Adipocytokines: Mediators linking adipose tissue, inflammation and immunity. Nat. Reviews. Immunol. 2006, 6, 772–783.
- Samokhin, A.O.; Buhling, F.; Theissig, F.; Bromme, D. ApoE-deficient mice on cholate-containing high-fat diet reveal a pathology similar to lung sarcoidosis. Am. J. Pathol. 2010, 176, 1148–1156.
- Stylianou, E. Epigenetics of chronic inflammatory diseases. J. Inflamm. Res. 2019, 12, 1–14.
- Bayarsaihan, D. Epigenetic mechanisms in inflammation. J. Dent. Res. 2011, 90, 9–17.
- Zhu, X.; Chen, Z.; Shen, W.; Huang, G.; Sedivy, J.M.; Wang, H.; Ju, Z. Inflammation, epigenetics, and metabolism converge to cell senescence and ageing: The regulation and intervention. Signal Transduct. Target. Ther. 2021, 6, 245.
- Ahmed, M.; de Winther, M.P.J.; Van den Bossche, J. Epigenetic mechanisms of macrophage activation in type 2 diabetes. Immunobiology 2017, 222, 937–943.
- Gonzalez-Jaramillo, V.; Portilla-Fernandez, E.; Glisic, M.; Voortman, T.; Ghanbari, M.; Bramer, W.; Chowdhury, R.; Nijsten, T.; Dehghan, A.; Franco, O.H.; et al. Epigenetics and Inflammatory Markers: A Systematic Review of the Current Evidence. Int. J. Inflamm. 2019, 2019, 6273680.
- Hermsdorff, H.H.; Mansego, M.L.; Campion, J.; Milagro, F.I.; Zulet, M.A.; Martinez, J.A. TNF-alpha promoter methylation in peripheral white blood cells: Relationship with circulating TNFalpha, truncal fat and n-6 PUFA intake in young women. Cytokine 2013, 64, 265–271.
- Wang, X.; Cao, Q.; Yu, L.; Shi, H.; Xue, B.; Shi, H. Epigenetic regulation of macrophage polarization and inflammation by DNA methylation in obesity. JCI Insight 2016, 1, e87748.
- Kamei, Y.; Suganami, T.; Ehara, T.; Kanai, S.; Hayashi, K.; Yamamoto, Y.; Miura, S.; Ezaki, O.; Okano, M.; Ogawa, Y. Increased expression of DNA methyltransferase 3a in obese adipose tissue: Studies with transgenic mice. Obesity 2010, 18, 314–321.
- Yang, X.; Wang, X.; Liu, D.; Yu, L.; Xue, B.; Shi, H. Epigenetic regulation of macrophage polarization by DNA methyltransferase 3b. Mol. Endocrinol. 2014, 28, 565–574.
- Raciti, G.A.; Spinelli, R.; Desiderio, A.; Longo, M.; Parrillo, L.; Nigro, C.; D’Esposito, V.; Mirra, P.; Fiory, F.; Pilone, V.; et al. Specific CpG hyper-methylation leads to Ankrd26 gene down-regulation in white adipose tissue of a mouse model of diet-induced obesity. Sci. Rep. 2017, 7, 43526.
- Desiderio, A.; Longo, M.; Parrillo, L.; Campitelli, M.; Cacace, G.; de Simone, S.; Spinelli, R.; Zatterale, F.; Cabaro, S.; Dolce, P.; et al. Epigenetic silencing of the ANKRD26 gene correlates to the pro-inflammatory profile and increased cardio-metabolic risk factors in human obesity. Clin. Epigenetics 2019, 11, 181.
- Shanaki, M.; Omidifar, A.; Shabani, P.; Toolabi, K. Association between HDACs and pro-inflammatory cytokine gene expressions in obesity. Arch. Physiol. Biochem. 2022, 128, 880–886.
- Nuno, D.W.; Lamping, K.G. Dietary Fatty Acid Saturation Modulates Sphingosine-1-Phosphate-Mediated Vascular Function. J. Diabetes Res. 2019, 2019, 2354274.
- Fu, P.; Ebenezer, D.L.; Ha, A.W.; Suryadevara, V.; Harijith, A.; Natarajan, V. Nuclear lipid mediators: Role of nuclear sphingolipids and sphingosine-1-phosphate signaling in epigenetic regulation of inflammation and gene expression. J. Cell. Biochem. 2018, 119, 6337–6353.
- Suryadevara, V.; Ramchandran, R.; Kamp, D.W.; Natarajan, V. Lipid Mediators Regulate Pulmonary Fibrosis: Potential Mechanisms and Signaling Pathways. Int. J. Mol. Sci. 2020, 21, 4257.
- Huang, L.S.; Berdyshev, E.; Mathew, B.; Fu, P.; Gorshkova, I.A.; He, D.; Ma, W.; Noth, I.; Ma, S.F.; Pendyala, S.; et al. Targeting sphingosine kinase 1 attenuates bleomycin-induced pulmonary fibrosis. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2013, 27, 1749–1760.
- Zheng, J.; Zhang, Q.; Mul, J.D.; Yu, M.; Xu, J.; Qi, C.; Wang, T.; Xiao, X. Maternal high-calorie diet is associated with altered hepatic microRNA expression and impaired metabolic health in offspring at weaning age. Endocrine 2016, 54, 70–80.
- Ding, Y.; Li, J.; Liu, S.; Zhang, L.; Xiao, H.; Li, J.; Chen, H.; Petersen, R.B.; Huang, K.; Zheng, L. DNA hypomethylation of inflammation-associated genes in adipose tissue of female mice after multigenerational high fat diet feeding. Int. J. Obes. 2014, 38, 198–204.
- Zhang, X.M.; Guo, L.; Chi, M.H.; Sun, H.M.; Chen, X.W. Identification of active miRNA and transcription factor regulatory pathways in human obesity-related inflammation. BMC Bioinform. 2015, 16, 76.
- Hijmans, J.G.; Diehl, K.J.; Bammert, T.D.; Kavlich, P.J.; Lincenberg, G.M.; Greiner, J.J.; Stauffer, B.L.; DeSouza, C.A. Influence of Overweight and Obesity on Circulating Inflammation-Related microRNA. MicroRNA 2018, 7, 148–154.
- Pan, Y.; Hui, X.; Hoo, R.L.C.; Ye, D.; Chan, C.Y.C.; Feng, T.; Wang, Y.; Lam, K.S.L.; Xu, A. Adipocyte-secreted exosomal microRNA-34a inhibits M2 macrophage polarization to promote obesity-induced adipose inflammation. J. Clin. Investig. 2019, 129, 834–849.
- Zhang, Y.; Mei, H.; Chang, X.; Chen, F.; Zhu, Y.; Han, X. Adipocyte-derived microvesicles from obese mice induce M1 macrophage phenotype through secreted miR-155. J. Mol. Cell Biol. 2016, 8, 505–517.
- Miranda, K.; Yang, X.; Bam, M.; Murphy, E.A.; Nagarkatti, P.S.; Nagarkatti, M. MicroRNA-30 modulates metabolic inflammation by regulating Notch signaling in adipose tissue macrophages. Int. J. Obes. 2018, 42, 1140–1150.
- Zhang, L.; Wang, Y.; Wu, G.; Xiong, W.; Gu, W.; Wang, C.Y. Macrophages: Friend or foe in idiopathic pulmonary fibrosis? Respir. Res. 2018, 19, 170.
- Heyob, K.M.; Mieth, S.; Sugar, S.S.; Graf, A.E.; Lallier, S.W.; Britt, R.D., Jr.; Rogers, L.K. Maternal high-fat diet alters lung development and function in the offspring. Am. J. Physiol. Lung Cell. Mol. Physiol. 2019, 317, L167–L174.
- Snow, S.J.; Phillips, P.M.; Ledbetter, A.; Johnstone, A.F.M.; Schladweiler, M.C.; Gordon, C.J.; Kodavanti, U.P. The influence of maternal and perinatal high-fat diet on ozone-induced pulmonary responses in offspring. J. Toxicol. Environ. Health. Part A 2019, 82, 86–98.
- Alfaradhi, M.Z.; Kusinski, L.C.; Fernandez-Twinn, D.S.; Pantaleao, L.C.; Carr, S.K.; Ferland-McCollough, D.; Yeo, G.S.; Bushell, M.; Ozanne, S.E. Maternal Obesity in Pregnancy Developmentally Programs Adipose Tissue Inflammation in Young, Lean Male Mice Offspring. Endocrinology 2016, 157, 4246–4256.
- Xie, Q.; Wei, M.; Kang, X.; Liu, D.; Quan, Y.; Pan, X.; Liu, X.; Liao, D.; Liu, J.; Zhang, B. Reciprocal inhibition between miR-26a and NF-kappaB regulates obesity-related chronic inflammation in chondrocytes. Biosci. Rep. 2015, 35, e00204.
- Zuo, Y.; Liu, J.; Xu, H.; Li, Y.; Tao, R.; Zhang, Z. Pirfenidone inhibits cell fibrosis in connective tissue disease-associated interstitial lung disease by targeting the TNF-alpha/STAT3/KL6 pathway. J. Thorac. Dis. 2022, 14, 2089–2102.
- Nakatsuka, Y.; Handa, T.; Kokosi, M.; Tanizawa, K.; Puglisi, S.; Jacob, J.; Sokai, A.; Ikezoe, K.; Kanatani, K.T.; Kubo, T.; et al. The Clinical Significance of Body Weight Loss in Idiopathic Pulmonary Fibrosis Patients. Respir. Int. Rev. Thorac. Dis. 2018, 96, 338–347.
- Dai, H.; Xiang, J.; Hou, Y.; Xuan, L.; Wang, T.; Li, M.; Zhao, Z.; Xu, Y.; Lu, J.; Chen, Y.; et al. Fat mass to fat-free mass ratio and the risk of non-alcoholic fatty liver disease and fibrosis in non-obese and obese individuals. Nutr. Metab. 2021, 18, 21.
- Lee, D.H.; Keum, N.; Hu, F.B.; Orav, E.J.; Rimm, E.B.; Willett, W.C.; Giovannucci, E.L. Predicted lean body mass, fat mass, and all cause and cause specific mortality in men: Prospective US cohort study. Bmj 2018, 362, k2575.
- Iliodromiti, S.; Celis-Morales, C.A.; Lyall, D.M.; Anderson, J.; Gray, S.R.; Mackay, D.F.; Nelson, S.M.; Welsh, P.; Pell, J.P.; Gill, J.M.R.; et al. The impact of confounding on the associations of different adiposity measures with the incidence of cardiovascular disease: A cohort study of 296 535 adults of white European descent. Eur. Heart J. 2018, 39, 1514–1520.
- Ionescu, A.A.; Evans, W.D.; Pettit, R.J.; Nixon, L.S.; Stone, M.D.; Shale, D.J. Hidden depletion of fat-free mass and bone mineral density in adults with cystic fibrosis. Chest 2003, 124, 2220–2228.
- Yang, X.F.; Qiu, Y.Q.; Wang, L.; Gao, K.G.; Jiang, Z.Y. A high-fat diet increases body fat mass and up-regulates expression of genes related to adipogenesis and inflammation in a genetically lean pig. J. Zhejiang Univ. Sci. B 2018, 19, 884–894.
- Nagy, R.; Gede, N.; Ocskay, K.; Dobai, B.M.; Abada, A.; Vereczkei, Z.; Pazmany, P.; Kato, D.; Hegyi, P.; Parniczky, A. Association of Body Mass Index with Clinical Outcomes in Patients with Cystic Fibrosis: A Systematic Review and Meta-analysis. JAMA Netw. Open 2022, 5, e220740.
- Monnard, C.R.; Dulloo, A.G. Polyunsaturated fatty acids as modulators of fat mass and lean mass in human body composition regulation and cardiometabolic health. Obes. Rev. Off. J. Int. Assoc. Study Obes. 2021, 22 (Suppl. S2), e13197.
- Cordain, L.; Eaton, S.B.; Sebastian, A.; Mann, N.; Lindeberg, S.; Watkins, B.A.; O’Keefe, J.H.; Brand-Miller, J. Origins and evolution of the Western diet: Health implications for the 21st century. Am. J. Clin. Nutr. 2005, 81, 341–354.
- Kennedy, J.I., Jr.; Chandler, D.B.; Fulmer, J.D.; Wert, M.B.; Grizzle, W.E. Dietary fish oil inhibits bleomycin-induced pulmonary fibrosis in the rat. Exp. Lung Res. 1989, 15, 315–329.
- Chen, J.; Zeng, T.; Zhao, X.; Xiea, K.; Bi, Y.; Zhong, Z.; Zhao, X. Docosahexaenoic acid (DHA) ameliorates paraquat-induced pulmonary fibrosis in rats possibly through up-regulation of Smad 7 and SnoN. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2013, 57, 330–337.
- Lawrenz, J.; Herndon, B.; Kamal, A.; Mehrer, A.; Dim, D.C.; Baidoo, C.; Gasper, D.; Nitz, J.; Molteni, A.; Baybutt, R.C. Dietary Flaxseed Oil Protects against Bleomycin-Induced Pulmonary Fibrosis in Rats. Pulm. Med. 2012, 2012, 457031.
- Abidi, A.; Kourda, N.; Feki, M.; Ben Khamsa, S. Protective Effect of Tunisian Flaxseed Oil against Bleomycin-Induced Pulmonary Fibrosis in Rats. Nutr. Cancer 2020, 72, 226–238.
- Velten, M.; Britt, R.D., Jr.; Heyob, K.M.; Tipple, T.E.; Rogers, L.K. Maternal dietary docosahexaenoic acid supplementation attenuates fetal growth restriction and enhances pulmonary function in a newborn mouse model of perinatal inflammation. J. Nutr. 2014, 144, 258–266.
- Zheng, S.; Wang, Q.; D’Souza, V.; Bartis, D.; Dancer, R.; Parekh, D.; Gao, F.; Lian, Q.; Jin, S.; Thickett, D.R. ResolvinD(1) stimulates epithelial wound repair and inhibits TGF-beta-induced EMT whilst reducing fibroproliferation and collagen production. Lab. Investig. A J. Tech. Methods Pathol. 2018, 98, 130–140.
- Mercader-Barcelo, J.; Truyols-Vives, J.; Rio, C.; Lopez-Safont, N.; Sala-Llinas, E.; Chaplin, A. Insights into the Role of Bioactive Food Ingredients and the Microbiome in Idiopathic Pulmonary Fibrosis. Int. J. Mol. Sci. 2020, 21, 6051.
- Miyake, Y.; Sasaki, S.; Yokoyama, T.; Chida, K.; Azuma, A.; Suda, T.; Kudoh, S.; Sakamoto, N.; Okamoto, K.; Kobashi, G.; et al. Vegetable, fruit, and cereal intake and risk of idiopathic pulmonary fibrosis in Japan. Ann. Nutr. Metab. 2004, 48, 390–397.
- Wang, X.; Yi, X.; Tang, D. Aerobic Exercise Improves Pulmonary Fibrosis by Improving Insulin Resistance and Inflammation in Obese Mice. Front. Physiol. 2021, 12, 785117.
- Tang, X.; Peng, R.; Phillips, J.E.; Deguzman, J.; Ren, Y.; Apparsundaram, S.; Luo, Q.; Bauer, C.M.; Fuentes, M.E.; DeMartino, J.A.; et al. Assessment of Brd4 inhibition in idiopathic pulmonary fibrosis lung fibroblasts and in vivo models of lung fibrosis. Am. J. Pathol. 2013, 183, 470–479.
- Bartczak, K.; Bialas, A.J.; Kotecki, M.J.; Gorski, P.; Piotrowski, W.J. More than a Genetic Code: Epigenetics of Lung Fibrosis. Mol. Diagn. Ther. 2020, 24, 665–681.
- Davies, E.R.; Haitchi, H.M.; Thatcher, T.H.; Sime, P.J.; Kottmann, R.M.; Ganesan, A.; Packham, G.; O’Reilly, K.M.; Davies, D.E. Spiruchostatin A inhibits proliferation and differentiation of fibroblasts from patients with pulmonary fibrosis. Am. J. Respir. Cell Mol. Biol. 2012, 46, 687–694.
- Chioccioli, M.; Roy, S.; Newell, R.; Pestano, L.; Dickinson, B.; Rigby, K.; Herazo-Maya, J.; Jenkins, G.; Ian, S.; Saini, G.; et al. A lung targeted miR-29 mimic as a therapy for pulmonary fibrosis. EBioMedicine 2022, 85, 104304.
- Montgomery, R.L.; Yu, G.; Latimer, P.A.; Stack, C.; Robinson, K.; Dalby, C.M.; Kaminski, N.; van Rooij, E. MicroRNA mimicry blocks pulmonary fibrosis. EMBO Mol. Med. 2014, 6, 1347–1356.
