The capacity of the skin to heal a wound is the result of a highly intricate process that involves several different processes, such as vascular response, blood coagulation, fibrin network creation, re-epithelialisation, collagen maturation, and connective tissue remodelling. Wound healing may be controlled with topical antiseptics, topical antibiotics, herbal remedies, and cellular initiators. In order to effectively eradicate infections and shorten the healing process, contemporary antimicrobial treatments that include antibiotics or antiseptics must be investigated. A variety of delivery systems were described, including innovative delivery systems, hydrogels, microspheres, gold and silver nanoparticles, vesicles, emulsifying systems, nanofibres, artificial dressings, three-dimensional printed skin replacements, dendrimers and carbon nanotubes. It may be inferred that enhanced local delivery methods might be used to provide wound healing agents for faster healing of skin wounds.
- wound
- physiology of wound healing
- strategies towards wound healing
- local delivery systems
1. Introduction
A wound is the result of the “disruption of normal anatomic structure and function,” claims the Wound Healing Society. Depending on how long a wound takes to heal, attention might be given to the acute and chronic kinds. Acute wounds are typically treated effectively with a good possibility of success within a few weeks and are primarily caused by mechanical trauma or surgical procedures [2][1]. The location, size, depth, and type of an acute wound all affect its nature. The chain of events necessary for wound healing can be disrupted by a variety of disease processes, leading to chronic, non-healing wounds that cause the patient great suffering and need a tremendous number of resources from the medical system. The coagulation cascade, inflammatory pathways, and the cellular components of the immune system are all activated during wound healing, which causes a significant modification of all skin compartments [3][2]. For expediting in vivo wound healing and minimising scar formation, cellular scaffolds containing fibroblasts, keratinocytes, stem/progenitor cells, or reprogrammed cells have shown promising outcomes.
2. Physiology of Wound Healing Process
Skin wound healing is an intriguing biological process that has served mammals well throughout evolution. Skin wound healing is a crucial phase for survival that culminates in wound closure because of its essential roles as a physical, chemical, and bacterial barrier. The process of cellular, humoral, and molecular mechanisms involved in skin wound healing is dynamic, tightly regulated, and can take years to complete [12][7]. The epithelialisation of skin wounds depends on the details of the lesion, including its location, depth, size, microbial contamination, patient-related medical problems, genetics, and epigenetics. Following a skin injury, the exposed sub-endothelium, collagen, and tissue factor will stimulate platelet aggregation, which in turn causes degranulation and releases chemotactic factors (chemokines) and growth factors to form the clot [12][7]. By following all of the aforementioned steps, successful haemostasis will be achieved. A skin wound can heal completely through regeneration or repair. Skin repair shows an unspecific kind of healing in which the lesion heals by fibrosis and scar formation, in contrast to regeneration, which depicts the specific substitution of the tissue, such as the superficial epidermis, mucosa, or foetal skin. Unfortunately, the latter is the primary mode of adult skin wound healing [13][8]. The pathophysiology of chronic wounds is still poorly understood; however, it is known that they remain in the inflammation stage of the healing process rather than progressing further. Critical obstacles to the physiologic healing of chronic wounds include impaired vascularisation and the resulting hypoxia, the inability to move on to the healing phase, extended and exacerbated inflammation, and the incapacity of immune cells to manage bacterial infection. Normal wound healing can be hampered by prolonged wound-healing phases or overly aggressive reactions of the organism to the damage [14][9]. The majority of chronic wounds heal through fibrosis, which produces an excessive quantity of connective tissue rather than regeneration. Additionally, persistent inflammation is followed by fibrosis, and fibrosis-healing wounds have been discovered to have higher levels of pro-inflammatory mediators (such as TGF-β) (Figure 1). Unnecessary fibroblast proliferation, neovascularisation, and increased collagen and fibronectin synthesis are all results of poorly controlled growth factor activity. Additionally, a wound contracts too much and for too long, causing fibrotic scar tissue to grow. Keloids and hypertrophic scars are two different types of pathological scars that can result from an injury. In this regard, there is active current study on the change from the inflammatory to the proliferative stage of wound repair. Three to five phases that overlap in both time and location can be artificially created to represent the various stages of wound healing [12,13,14,15,16][7][8][9][10][11].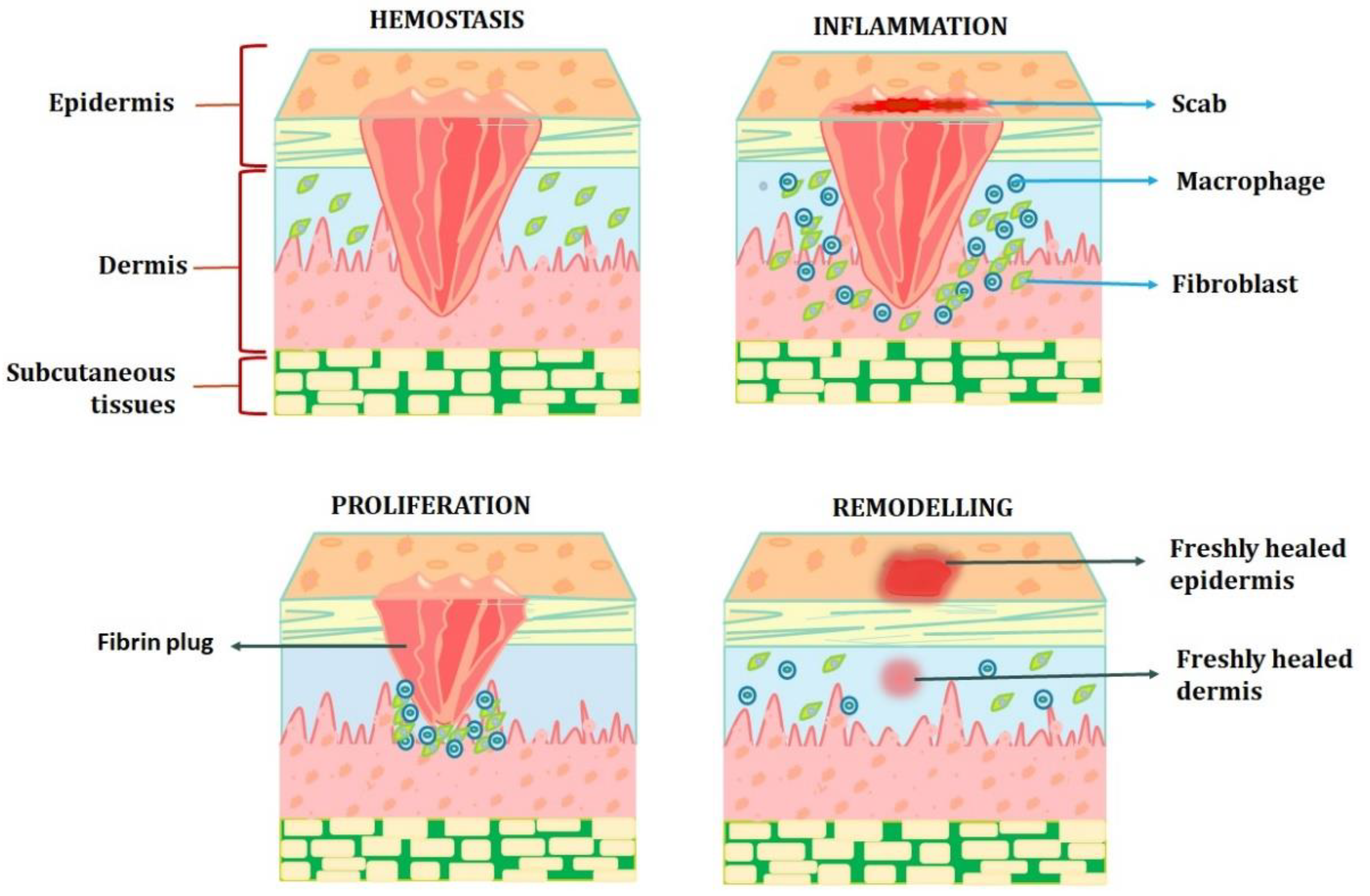
3. Wound Healing Strategies
3.1. Cellular Activity Initiators
DNA synthesis is promoted in fibroblast cells by secretions from healthy wounds. Conversely, the same fibroblasts are inhibited by the secretions from long-lasting, nonhealing wounds, such as leg ulcers. Interestingly, heating the fluid contents of a chronic wound denatures them, removing the inhibitory impact and restoring fibroblast growth. As before, fibroblasts from chronic wounds have the worst response to growth factors than fibroblasts from acute wounds, suggesting that the fibroblasts in chronic wounds are also harmed. The most promising biomarkers are proteases and cytokines. Traditional medicines such as honey, curcumin, and tannin have been studied using modern pharmaceutical practices to learn how they affect cellular activity. One would anticipate that cytokine release, which represents neutrophil and macrophage activity, would increase in an effort to trigger a fibroblast response if fibroblasts stop responding. The pro-inflammatory cytokines IL-1 and TNF-α were found in higher concentrations in non-healing wounds than in healing wounds. When the healing starts, the levels significantly decrease.
Several methods were used to modulate macrophages, including blocking IL-1 or TNF-α, inhibiting the inflammasome pharmacologically, neutralising MCP-1, and chelating iron with desferrioxamine. Sulphated hyaluronic acid is internalised by macrophages after being identified by CD44 and the scavenger receptors CD36 and LOX-1. Most notably, it prevents the phosphorylation of the transcription factors including pNFkB, pSTAT1, and IRF5 that are involved in M1-like activation states and the production of pro-inflammatory genes. Sulphated hyaluronic acid regulates macrophage activation in vivo.
By altering fibroblast activity and proliferation, anti-fibrotic medications such as mitomycin C and 5-FU stop the formation of scars. By disrupting pyrimidine metabolism, the anti-proliferative activity of 5-FU is mediated. By preventing the production of thymidine nucleotides, it prevents DNA synthesis, leading to cell death. It has long-lasting effects on Tenon’s fibroblasts and can effectively limit fibroblast development [34][12]. By reducing to an alkylating agent, mitomycin C is activated and subsequently works by cross-linking DNA. Mitomycin C can impede not just DNA replication but also mitosis and protein synthesis.
3.2. Collagen Synthesis Activators
3.3. Angiogenesis Activators
By acting as a chemoattractant for neutrophils, macrophages, and fibroblasts, TGF-β promotes the development of granulation tissue. As a result, TGF-β is a crucial regulator of angiogenesis during the healing of wounds because it controls cell division, migration, capillary tube formation, and ECM deposition. Through the production of particular proteins, gene augmentation brings about the return of normal cellular function. By delivering DNA or mRNA into the target cells, one can enhance genes. Exciting new choices for treating chronic wounds will be made possible in the following ten years by wound dressings that contain sustained nucleic acid delivery systems for promoting angiogenesis and therapy that targets the underlying morbidities.3.4. Cytokine and Growth Factor Activators
Small, secreted proteins called cytokines influence not only the activity of immune cells but also that of other cells. Interleukins, lymphokines, and several related signalling molecules, such as TNF-α, interferons, and others, are among them. Through the activation of cell surface TGF-β serine/threonine type I and type II receptors and the activation of a Smad3-dependent signal, active TGF-β1 induces the fast chemotaxis of neutrophils and monocytes to the wound site. Leukocytes and fibroblasts that have expressed TGF- β1 are then stimulated to produce additional cytokines, such as TNF-α, IL-1β, and PDGF, as well as chemokines, which are all part of a cytokine cascade. Such factors serve to maintain the inflammatory cell response by influencing neutrophil and monocyte recruitment and activation. TGF-β and other cytokines that activate their corresponding cell surface receptors cause intracellular signalling pathways to be activated, which, in turn, causes target cell populations to respond phenotypically and functionally. NF-κB, early-growth response 1 (EGR1), Smads, and MAPK are some of the upstream signalling cascades involved in acute tissue injury. These cascades activate many cognate target genes, including adhesion molecules, coagulation factors, cytokines, and growth factors. The platform upon which circulating leukocyte-expressing counter-adhesion molecules (integrins, selectins, and Ig superfamily members) tether allows them to sense the microenvironment and react to chemotactic signals at the site of tissue injury. This is accomplished by cytokine-induced enhancement of adhesion molecules (VCAM-1, ELAM-1, and ICAM-1) on the endothelium. In response to various chemotactic cues, transmigration from within to outside the artery wall is made possible by interactions between adhesion molecules on blood leukocytes and endothelium. Numerous chemokines are generated in addition to the chemotactic action of TGF-β1 for neutrophils and monocytes to attract leukocytes to the site of tissue injury. Depending on where the cysteine residues are located, several families of related molecules serve as representations of chemokines [45][15]. Growth factors such as PDGF, TGF-β, and EGF are secreted when platelets degranulate and release alpha granules. PDGF plays a crucial role in luring neutrophils to the wound site to eliminate contaminated germs, coupled with proinflammatory cytokines such as IL-1. A number of pro-inflammatory cytokines (IL-1 and IL-6) and growth factors (FGF, EGF, TGF-β, and PDGF) are released by macrophages to support the development of granulation tissue. One of the first substances to be created in response to skin lesions is a substance known as a proinflammatory cytokine, which controls immune cell actions during epithelialisation. TNF-α, IL-1, IL-6, and IL17 are the main proinflammatory cytokines that play a role in the inflammation phase of wound healing. They also play a role in the epithelialisation phase by promoting cell proliferation and differentiation and mobilising local stem/progenitor cells. Cytokine modulators are a new class of medicinal drugs that prevent fibrogenesis. Scarring and fibrosis of the skin are frequently the results of excessive fibrogenesis [46][16]. As a different strategy to prevent fibrosis, the modulatory effects of natural compounds such as terpenes and honey should be taken into consideration. Mitomycin P modulators such as buckwheat and acacia honey should be taken into account as substances reducing scarring and encouraging re-epithelisation. Terpenoids are frequently present in essential oils and serve as cytokine suppressors, increasing the production of IL-10 and the anti-inflammatory cytokines TNF- and IL-1 [47][17].3.5. Antimicrobials
There is a lot of debate about the application of topical antibiotics to wounds. Topical antimicrobial agents are described as substances that can eliminate, suppress, or lessen the number of bacteria. These substances include disinfectants, antiseptics, and antibiotics. Topical antimicrobial medicines are essential to topical burn care because they are used to prevent and control infection. The ideal topical preventive antimicrobial agent would be able to enter necrotic tissue without being absorbed by the body, have a broad spectrum of activity, a lengthy duration of action, have low toxicity, and have several other qualities [48][18].
A renewed interest in silver-based medications is a result of concerns about bacterial resistance. For thousands of years, silver has been utilised in medicine for its antibacterial properties. A good environment for wound healing can be created by using topical antimicrobials that do not impede epithelial outgrowth and deliver a high concentration of active components to devitalised, devascularised, and, perhaps, necrotic wounds. Topical antibacterial use may reduce the requirement for extensive debridement and subsequent grafting as well as wound deepening. Although microorganisms are present in every wound, the majority do not become infected and heal properly. In these circumstances, the immune system of the host and the bioburden of the wound are in equilibrium [49][19]. NF-κB is nuclear translocated as a result of toll-like receptor (TLR) stimulation through intracellular signalling from adapter proteins, which, in conjunction with mitogen-activated protein kinases, triggers the transcription of a variety of inflammatory cytokines, chemokines, antimicrobial peptides, and costimulatory factors. According to wound specialists, there is a threshold over which antimicrobial intervention is necessary when bacteria loads are more than or equal to 104 CFU/g. Silver compounds are among the antibacterial agents used in burn treatment. In cases when surgery is either not possible or would not be the first option right away, such as in cases of facial burns, silver sulphadiazine is frequently utilised and acts on burn eschar to restrict the area of non-viable tissue [50,51,52,53,54,55][20][21][22][23][24][25]. Since silver is a natural broad-spectrum antibiotic, there has not been any bacterial resistance to its treatments yet. There are numerous types of silver, including silver oxide, silver nitrate, silver sulphate, silver salt, silver zeolite, silver sulfadiazine, and silver nanoparticles. When silver cations come into touch with liquid, they are freed from their carrier dressings. Depending on the dressing employed, there are significant differences in the pace, duration, and peak level of silver released. Once discharged, silver kills germs in a variety of ways. The healing of both acute wounds and chronic wounds is currently aided by various forms of silver. Antiseptic and antibiotic dressings are the two primary categories into which antimicrobial dressings can be divided. Due to their good efficacy and tolerability among the various antimicrobial agents available, iodophor-based formulations such as povidone iodine have remained well-liked after decades of usage for antisepsis and wound healing applications. Povidone iodine has been reported as having a wide range of activity, the capacity to penetrate biofilms, a lack of related resistance, anti-inflammatory qualities, low cytotoxicity, and good tolerability. In clinical practice, no adverse effects on wound healing have been noted. Another antimicrobial agent that penetrates burned tissue is cerium nitrate. It has a wide range of activity against Gram-positive and Gram-negative bacteria, as well as fungal species, and is highly effective when used in conjunction with silver sulphadiazine [56][26]. Povidone iodine is fully hazardous to keratinocytes and fibroblasts at concentrations greater than 0.004 and 0.05%, respectively [57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74][27][28][29][30][31][32][33][34][35][36][37][38][39][40][41][42][43][44].3.6. Stem Cell-Based Therapy
A promising new method in the area of regenerative medicine is stem cell-based therapy. The ability of stem cells to self-renew and specialise into distinct cell types is essential for physiologic tissue renewal and regeneration after injury, and is of great interest to biologists. Adult mesenchymal stem cells, embryonic stem cells, and, the more recently studied, induced pluripotent stem cells are the main sources of stem cells that are used for skin regeneration and wound healing among the several types of stem cells [57][27]. In essence, keratinocytes are produced from stem cells found in the skin’s basal layer, which differentiate for three to six weeks before becoming corneocytes, which in turn create the stratum corneum layer. Keratin, a protein that plays a significant structural role in the stratum corneum, is one of the many proteins that keratinocytes generate. Along with proteins, the stratum corneum’s complex lipid and cell membranes act as a significant barrier against bacteria and dehydration. The migration of fully developed cuboidal basal keratinocytes with large nuclei, phospholipid membranes, and organelles starts from the basal layer after roughly every 28 days [80,81,82,83,84,85,86,87][45][46][47][48][49][50][51][52].3.7. Herbal Alternatives Acting as Activators for Wound Healing Factors
Wound healing may benefit greatly from a variety of plants or chemicals derived from plants that contain high concentrations of antioxidants and have anti-inflammatory, immunomodulatory, and antibacterial characteristics. Antioxidants help tissues recover from injury and can speed up wound healing. The antioxidant activity of flavonoids, anthraquinones, and naphthoquinones is strong. Shikonin, alkanin, lawsone, emodin, epigallocatechin-3-gallate, ellagic acid, and a few herbal extracts have strong antioxidant effects by scavenging ROS, preventing lipid peroxidation, and boosting intracellular antioxidant enzyme activities such as superoxide dismutase, catalase, and glutathione peroxidase [3,8][2][53].4. Localised Delivery Systems for Wound Healing
4.1. Microspheres/Microcarriers
A sort of injectable scaffold is a microsphere, often known as a microcarrier. Microspheres offer enough room for cell development and have good surface area-to-volume ratios. To further improve the transport of cells and bioactive compounds, microspheres with functional architectures (such as hollow or core-shell) can be easily customised and manufactured. As a result, functional microspheres have received a lot of attention recently as a new kind of injectable scaffold. Microspheres are typically referred to as spheres with a diameter between 1 and 1000 m. The diameter of most cells when they adhere and spread on a biomaterial is greater than 20 μm; hence, small-sized microspheres (<20 μm in diameter) are not suited as cellular carriers. Microspheres should therefore be between 20 and 200 μm in size when used as an injectable biomaterial for tissue engineering [103][54].4.2. Inorganic Nanoparticles
Nanomaterials have received a lot of recent attention because of their improved efficacy and broad-spectrum antibacterial potential. Numerous metallic and metal oxide nanoparticles, such as Ag, Fe, Cu, and Au, as well as TiO2, ZnO, and Fe3O4, are being thoroughly researched for the treatment of infectious disorders. Considering that both of their counterparts are typical of low molecular weight, the two most popular systems of metal nanoparticles (NPs), gold (AuNPs) and silver (AgNPs) provide excellent methods for delivering pharmaceuticals. These nanoparticles have advantages over commonly used antibiotics because of their distinctive features, including size, shape, surface charge, dispersion, and chemical composition. Additionally, these synthetic antibiotics frequently only work against a certain type of bacterium or bacterial family, rendering them useless against a diverse range of bacterial species [107][55]. Currently, antibiotic efficiency has decreased mostly as a result of uncontrolled, excessive dosage, and prolonged use, which has favoured the establishment of multi-drug-resistant bacterial strains or “superbugs” such as MRSA-3. AgNPs must be engineered with various compositions to improve their properties and make them suitable for therapeutic application in order to overcome their current constraints. If these nanoparticles pass the required tests by regulatory agencies for better antibacterial efficacy and minimum cytotoxicity for patients, they may prove to be effective antibiotic alternatives or may be used in conjunction with antibiotics [106,107,108][55][56][57]. The antibacterial activity of metallic nanoparticles has been increased by a number of methods, including encasing silver nanoparticles in micelles, covering silver nanoparticles in gold, and capping gold nanoparticles with 5-amindole or sodium borohydride N-heterocyclic molecules. A bimetallic nanoparticle with silver and gold was recently developed. Complex carbohydrates on the caps of these bimetallic nanoparticles improve their stability and other characteristics. These bimetallic NPs can be used for in vivo antimicrobial activities since they have considerably better antibacterial activities and do not harm mammalian cells. The use of silver to improve wound healing dates back to ancient times when it was utilised as an ion. Unlike silver ions (Ag+) or other forms of silver, which are used as nanoparticles, silver metal (Ag) has no known medical applications. They can impede the healing process since many bacteria, viruses, fungi, and even yeast is cytotoxic to them. This ion was used as part of a wound dressing that contained silver ions specifically to treat severe wounds [109][58].4.3. Hydrogel
Insoluble hydrophilic materials known as hydrogels are created using synthetic polymers such as poly (methacrylates) and polyvinyl pyrrolidone. Complex hydrophilic organic cross-linked polymers called hydrogels have a base that is 80–90% water. These hydrogels can provide water to the wound site and aid in keeping it moist, which promotes quicker wound healing. These are created into contact lenses, drug delivery systems, wound dressings, electrodes, and sensors. These gels can be found as fixed flexible sheets or free-flowing amorphous gels. They have a limited capacity for fluid absorption through swelling, but they can also contribute moisture to a dry wound, aiding in autolytic debridement and maintaining a moist, thermally insulated wound environment [113][59]. Hydrogels can be used in conjunction with topical drugs or antibacterial agents, which is one of their advantages. Infected wounds should not be treated with hydrogels in their fixed state. Hydrogels must be coated with additional dressings and left on for up to three days. They transfer oxygen and moisture vapour, but the type of secondary dressing employed affects how permeable they are to bacteria and fluids. Until an equilibrium condition is attained, these systems may swell in water and keep their original shape. The process of hydration, which is related to the presence of chemical groups such as -OH, -COOH, -CONH2, and -CONH-, as well as the existence of capillary regions and variations in osmotic pressure, is one of the interactions that contribute to the water sorption by hydrogels [115][60]. High-intensity radiation, freeze–thaw, or chemical processes can all be used to create hydrogels. Radiation, such as gamma rays, electron beams, X-rays, or ultraviolet light, is thought to be the most suited way for the creation of hydrogels since it allows for simple processing control and eliminates the need for potentially dangerous initiators or cross-linkers. Additionally, sterilisation and possible formation are both possible with irradiation. However, the mechanical strength of the hydrogels produced using this approach is subpar. Today, hydrogels are made using a freeze–thaw process to give them good strength and stability without the need for additional cross-linkers and initiators. The hydrogels’ limited swelling and thermal stability, as well as their opaque appearance, are the principal drawbacks of freeze–thawing. The application of hydrogel appears to considerably stimulate wound healing as compared to the standard gauze therapy. In order to create hydrogel wound dressings, a variety of natural and synthetic polymers with good biocompatibility are used [117][61].4.4. Vesicles Delivery System
Vesicular systems, which can be further divided into liposomes, ultra-deformable liposomes, and ethosomes, are composed of amphiphilic molecules because they have polar or hydrophilic regions and non-polar or lipophilic regions. It has been demonstrated that vesicular systems, including liposomes, niosomes, transferosomes, penetration enhancer-containing vesicles, and ethosomes, can improve the therapeutic activity of medications used to treat wounds. They can lengthen the shelf life of hydrophilic and hydrophobic medications and lessen major side effects including skin irritation, and act as a depot for controlled drug release. They can also improve the penetration of such medications into the skin. The two types of vesicular systems are hard vesicles, such as liposomes and niosomes, and flexible or ultra-flexible vesicles, such as transferosomes. According to reports, rigid vesicles are ineffective for transdermal drug delivery because they stay on the stratum corneum’s outer layer and do not thoroughly penetrate the skin. After topical administration, liposomes can cause a variety of reactions. The majority of efforts have been concentrated on the topical treatments’ antibacterial action, which has fallen short due to the rising rate of antibiotic resistance. They can inhibit systemic absorption, maximise side effects, and provide a localising impact as well as tailored distribution to skin appendages. They can also improve drug deposition within the skin at the site of action. Additionally, these vesicles were crucial in the healing of wounds [121][62]. Antibiotics entrapped in liposomes exhibit reduced toxicity and more target specificity along with increased efficacy in treatment of bacterial infections and thus improve its pharmacokinetics and pharmacodynamics. Increased action against external pathogens that are resistant, as well as increased activity against intracellular pathogens, is also an attractive feature. Due to their occlusive action on the stratum corneum, lipid nanoparticles may be more appropriate in burn wounds and chronic wounds since they can prevent transepidermal water loss and maintain the lesion moisture. Additionally, compared to vesicular systems, nanostructured lipid carriers are offered as superior nano-delivery methods. They have great stability, low toxicity, high drug-loading capacity, and sustained drug release, which helps speed up wound healing and cuts down the number of drugs administered [122][63].4.5. Emulsifying Drug Delivery System
The oral distribution of such medications is frequently linked to inadequate bioavailability, considerable intra- and inter-subject variability, and a lack of dose proportionality. Approximately 40% of novel drug candidates have poor water solubility. The use of surfactants, lipids, permeation enhancers, micronisation, salt formation, cyclodextrins, nanoparticles, and solid dispersions are just a few of the formulation strategies that have been used to tackle these issues. Recently, lipid-based formulations have received a lot of attention, with a focus on self-emulsifying drug delivery systems to increase the oral bioavailability of lipophilic medications. The fact that self-emulsifying drug delivery systems offer a significant interfacial area for the partitioning of the medication between oil and water is another benefit they have over straightforward oily solutions. Therefore, these systems may provide an improvement in the pace and amount of absorption as well as more consistent plasma concentration profiles for lipophilic medicines with dissolution-limited oral absorption [126][64].4.6. Nanofiber/Film/Membrane
A wide range of polymers can be used to create nanofibres. For dressing nanofibres, there are only three types of polymers now available: natural polymers, synthetic polymers, and mixed polymers. Natural polymers are appropriate for use in biomedical applications due to their wide range of benefits, including biocompatibility, non-toxicity, biodegradability, antibacterial properties, and desirable mechanical structure. In the procedure, the solvent evaporates. On the other hand, nanofibres are made of a polymeric base, which makes up most of the fibre’s composition, and a bioactive molecule (such as a protein, hormone, or medication), or another type of polymer, but in a lesser amount than the base polymer. Currently, the three primary techniques for producing nanofibres are electrospinning, the phase-separation method, and the self-assembly method. The method that produces nanofibres most frequently is electrospinning. Depending on the electrospinning technique employed, various types of nanofibres can be produced. Today, the commercialisation of electrospinning equipment is advancing quickly. The most popular electrospinning methods include bubble electrospinning, melt electrospinning, coaxial electrospinning, self-bundling electrospinning, and nano-spider electrospinning [132,133,134,135,136,137,138,139,140,141,142,143,144][65][66][67][68][69][70][71][72][73][74][75][76][77].
Tissue engineering and wound healing are two of the most important and intriguing biomedical uses of nanofibres. Nanofibres have been utilised in the treatment of diabetic ulcers and wounds to aid in wound healing, haemostasis, skin regeneration, and wound dressing. Nanofibre keeps the wound surface moist while healing because it can hold more moisture in its structure. As a result, the nanofibres cannot adhere to the surface of the wound. Additionally, the porous nanofibre network makes it simpler for oxygen to diffuse into the wound area. To remove toxins from the blood of individuals with kidney failure, wearable blood purification systems may integrate the nanofibre membrane. Nanofibre scaffolds hold great promise for wound healing. These scaffolds are used in the treatment of diabetic ulcers, skin rejuvenation, wound dressings to encourage healing, and haemostasis [147,148][78][79].
The polymers used to create scaffolds for wound dressing include collagen, poly-vinyl pyrrolidone, polyacrylic acid, polyvinyl alcohol, gelatin, chitosan, silk fibroin, polyesters, and poly-urethane [149][80]. The goal of a wound dressing is to quickly achieve haemostasis, and it should also have strong antibacterial properties to guard against bacterial infections from the environment. The ability of electrospinning to construct nanofibrous membranes for wound dressings that can provide a moist environment surrounding the wound region to facilitate healing has drawn a great deal of interest. In order to create composite nanofibre membranes that carry a reservoir of biogenic AgNPs for use as a wound dressing, Bardania et al. used T. polium extract as a reducing agent. This method of “green synthesis”, which does not use external stabilisers or reducing agents, produced AgNPs quickly, cheaply, and effectively [76,82,109][47][58][81].4.7. Foam Dressings
Foam dressings are permeable to both gases and water vapour and have a polyurethane foundation. The outer layer’s hydrophobic qualities shield it from liquids while allowing gaseous exchange and the passage of water vapour. Silicone-based rubber foam, or silicone, conforms to the shape of wounds. Depending on the thickness of the wound, foam can absorb varied amounts of wound drainage. There are foam dressings that are both adhesive and non-adhesive. Lower leg ulcers, mild to heavily exuding wounds, and granulating wounds can all benefit from foam dressings. They offer both thermal insulation and excellent absorption thanks to their hydrophilic characteristics [153][82].4.8. Biological Dressings
In order to restore the wound healing process and incorporate active biological agents to assist the wound healing process, advanced biological therapies are currently emerging in ischaemic wound therapies. The use of active biological agents, such as plant-derived active biomolecules with antioxidant, antibacterial, or anti-inflammatory properties, may be used in biological wound-healing therapies, which aim to aid the restoration of the body’s natural repair mechanisms. Biological dressings stop contamination, heat loss, protein and electrolyte loss, and evaporative water loss [15,22][10][83]. Bioactive wound dressings can be created using naturally occurring biomaterials with endogenous activity or materials that release bioactive chemicals. Chitin, chitosan, hydrocolloids, alginate, and derivatives of organic biopolymers are a few examples of these biomaterials. Biological dressings made from animal collagen are known as collagen dressings. The fibroblast-produced protein collagen plays a part in each step of wound healing. The extracellular matrix is primarily made up of collagen, which gives it strength [29][84].4.9. Charcoal Dressings
By absorbing gases produced by bacteria, activated charcoal dressings serve the primary purpose of reducing wound odour. They can absorb odour molecules due to their huge surface area and function as a deodorising agent. Wound odour is very subjective in nature since it is challenging to define and quantify. Leg ulcerations and diverse fungating lesions are the wounds most frequently linked to odour generation. Numerous aerobic bacteria as well as anaerobes including Bacteroides and Clostridium species are among the organisms usually linked to malodorous wounds [51,52,53][21][22][23]. Eliminating the problematic organism is the best strategy for treating wound smells. Antibiotics taken systemically may be successful, but it may be challenging to obtain an adequate concentration of the drug at the infection site. In numerous studies to date, topical treatments such as metronidazole, clindamycin, honey, and sugar have demonstrated promise in this area. Activated charcoal dressings are frequently used in malodorous wounds; however, odour control has not received as much attention in the literature as wound healing has. According to current clinical experiences, it is evident that these dressings can reduce wound odour, but there are not any hard facts on just charcoal ingredients [56,57,58][26][27][28].4.10. Three-Dimensional Skin Substitutes
Recently, several tissue engineering technologies have become available; these technologies take a fundamentally unique and new approach. Among these, three-dimensional free-form fabrication—often referred to as three-dimensional bioprinting—offers several benefits over traditional skin tissue engineering. An innovative method for designing and engineering human organs and tissues is three-dimensional bioprinting, a flexible automated on-demand platform for the free-form production of complex living constructions. In order to simulate the epidermis, dermis, and dermal matrix of the skin, keratinocytes, fibroblasts, and collagen were employed as constituent cells [154][85]. This method has enormous potential for the creation of three-dimensional skin tissue since it can dispense living cells, soluble components, and phase-changing hydrogels in a desired pattern while preserving very high cell viability. The biomimetic mechanical cues that support vascularisation, alignment of fibrous proteins in the ECM, integration of dermal and epidermal components, and adhesion between these layers are absent from the existing skin grafts and their production techniques. When the biomechanics of the repaired tissue at the wound site are compared to those of the healthy tissue nearby wounds, this problem may become more difficult. Therefore, to prevent the separation of the layers during application, a regenerative skin scaffold should take biomimetic mechanical cues into account. By adjusting the physicomechanical characteristics of each layer, the scaffold can offer tailored microenvironments for various cell types [155][86]. The use of autologous epidermal sheets as a kind of skin replacement has progressed into the use of more sophisticated bilayered cutaneous tissue-designed skin substitutes. However, their regular use for restoring normal skin anatomy is constrained by insufficient vascularisation, rigid drug/growth factor loading, and the inability to regenerate skin appendages such as hair follicles [156][87].4.11. Dendrimers
Dendrimers are a class of nanoscale (1–100 nm) three-dimensional globular macromolecules with numerous arms branching out from a central core. They have unique structural characteristics, including high levels of branching, multivalency, globular architecture, and well-defined molecular weight, making them promising drug delivery scaffolds. The distinctive structure of dendrimers makes them an ideal nanomaterial for the administration of medications to target certain tissues or molecules with solubility challenges. This is performed by trapping the drugs inside of their void spaces, branches, or outside functional groups. An expanding field of research involves cross-linking collagen with functionalised nanoparticles to produce scaffolds for use in wound healing. Due to their spherical structure, dendrimers can engage via hydrogen bonding, lipophilicity, and charge interactions with tiny medicines, metals, or imaging moieties that can fit within their branches. In addition to their structure, dendrimers may be the best drug delivery vehicles for many therapeutic treatments because of their size and lipophilicity, which allow them to easily permeate cell membranes. These distinguishing qualities have increased interest in using dendrimers nanoparticles for wound healing in research [158][88].4.12. Carbon Nanotubes
The carbon nanotubes are a class of stiff, stable, hollow nanomaterials with a variety of special physical, chemical, and mechanical properties that have been widely used as catalyst supports, nanowires, electronic components, and more recently in the fields of biomedical engineering and medical chemistry [166][89]. A needle-like structure with a significant surface area, carbon nanotubes are an allotropic form of carbon with nanoscale dimensions and µm lengths. The carbon atoms join to form sheets of graphite, which is made up of six-membered carbon atom rings. Graphite then spirals into tubes. Carbon nanotubes can be divided into single-wall and multiwall varieties based on the number of graphite layers present. CNTs have tensile strengths up to 63 gigapascals, which is around 50 times stronger than steel, and elastic moduli between 1.0 and 1.8 terapascals. With the goal of boosting drug delivery, regulating drug release, and improving therapeutic activity, carbon nanotubes are now being functionalised by various pharmacologically active compounds, with some degree of success [167][90]. The degradation products of carbon nanotubes can be eliminated by the functional tissues of the human body, making them non-toxic and safe for consumption [168][91]. The fibroblasts, which are crucial to the cell renewal system and the healing process of open wounds, may come into contact with the carbon nanotubes employed in the biomedical field and found in the environment as they pass through the skin or open wounds [166][89]. Proteins and receptors found in cell membranes can cling tightly to carbon nanotubes. Carbon nanotubes and cells must adhere to one another [166][89].4.13. Microneedle Drug Delivery Systems
The efficacy of traditional single-drug therapies is subpar, and penetration depth limits the effectiveness of drug delivery [173][92]. In the realm of wound healing, microneedle dressings with transdermal drug delivery capabilities have been crucial [174][93]. Additionally, the microstructure of microneedles allows for efficient medication administration to the target location while preventing overly strong skin and patch adherence. Additionally, temperature-sensitive hydrogel has been used to encapsulate vascular endothelial growth factor (VEGF) in the chitosan microneedle array micropores. As a result, the temperature increase brought on by the inflammatory response at the site of wounds can be used to controllably achieve the smart release of the medications [175][94]. The adaptable approach of the microneedle patch has been presented and has achieved several outstanding successes in the fields of disease therapy, biosensing, skin vaccination, and wound healing. Microneedles can efficiently deliver the desired active pharmaceuticals due to their better loading capacity and well-designed microstructures when compared to those used in conventional drug delivery systems. However, the microneedle that is so frequently used today is typically made from synthetic polymer materials that were created by difficult chemical synthesis using harsh experimental processes and environmentally hazardous organic reagents. This raises the danger of side effects. Additionally, the development of microneedle-based iatrotechnics is constrained by the acquisition of loaded active pharmaceuticals typically through a period of brutal elimination and extremely stringent clinical studies [176][95]. Wang et al. created a three-dimensional origami microneedle patch with extremely tiny needle structures, microfluidic channels, and numerous functionalities that was said to be able to detect biomarkers, distribute medications in a controlled manner, and monitor motions to speed up wound healing [177][96].References
- Kim, H.S.; Sun, X.; Lee, J.H.; Kim, H.W.; Fu, X.; Leong, K.W. Advanced drug delivery systems and artificial skin grafts for skin wound healing. Adv. Drug Deliv. Rev. 2019, 146, 209–239.
- Ryall, C.; Duarah, S.; Chen, S.; Yu, H.; Wen, J. Advancements in Skin Delivery of Natural Bioactive Products for Wound Management: A Brief Review of Two Decades. Pharmaceutics 2022, 14, 1072.
- Martin, P.; Nunan, R. Cellular and molecular mechanisms of repair in acute and chronic wound healing. Br. J. Dermatol. 2015, 173, 370–378.
- Han, G.; Ceilley, R. Chronic Wound Healing: A Review of Current Management and Treatments. Adv. Ther. 2017, 34, 599–610.
- Yuan, Z.; Zhang, K.; Jiao, X.; Cheng, Y.; Zhang, Y.; Zhang, P.; Zhang, X.; Wen, Y. A controllable local drug delivery system based on porous fibers for synergistic treatment of melanoma and promoting wound healing. Biomater. Sci. 2019, 7, 5084–5096.
- Elviri, L.; Bianchera, A.; Bergonzi, C.; Bettini, R. Controlled local drug delivery strategies from chitosan hydrogels for wound healing. Expert Opin. Drug Deliv. 2017, 14, 897–908.
- Jang, M.J.; Bae, S.K.; Jung, Y.S.; Kim, J.C.; Kim, J.S.; Park, S.K.; Suh, J.S.; Yi, S.J.; Ahn, S.H.; Lim, J.O. Enhanced wound healing using a 3D printed VEGF-mimicking peptide incorporated hydrogel patch in a pig model. Biomed. Mater. 2021, 16, 045013.
- Shedoeva, A.; Leavesley, D.; Upton, Z.; Fan, C. Wound Healing and the Use of Medicinal Plants. Evid. Based Complement. Altern. Med. 2019, 2019, 2684108.
- Ibrahim, N.; Wong, S.K.; Mohamed, I.N.; Mohamed, N.; Chin, K.Y.; Ima-Nirwana, S.; Shuid, A.N. Wound Healing Properties of Selected Natural Products. Int. J. Environ. Res. Public Health 2018, 15, 2360.
- Kumari, A.; Raina, N.; Wahi, A.; Goh, K.W.; Sharma, P.; Nagpal, R.; Jain, A.; Ming, L.C.; Gupta, M. Wound-Healing Effects of Curcumin and Its Nanoformulations: A Comprehensive Review. Pharmaceutics 2022, 14, 2288.
- Fatehi, P.; Abbasi, M. Medicinal plants used in wound dressings made of electrospun nanofibers. J. Tissue Eng. Regen. Med. 2020, 14, 1527–1548.
- Cabourne, E.; Clarke, J.C.; Schlottmann, P.G.; Evans, J.R. Mitomycin C versus 5-Fluorouracil for wound healing in glaucoma surgery. Cochrane Database Syst. Rev. 2015, 2015, CD006259.
- Chattopadhyay, S.; Raines, R.T. Review collagen-based biomaterials for wound healing. Biopolymers 2014, 101, 821–833.
- Vivcharenko, V.; Wojcik, M.; Palka, K.; Przekora, A. Highly Porous and Superabsorbent Biomaterial Made of Marine-Derived Polysaccharides and Ascorbic Acid as an Optimal Dressing for Exuding Wound Management. Materials 2021, 14, 1211.
- Ridiandries, A.; Tan, J.T.M.; Bursill, C.A. The Role of Chemokines in Wound Healing. Int. J. Mol. Sci. 2018, 19, 3217.
- Xiao, T.; Yan, Z.; Xiao, S.; Xia, Y. Proinflammatory cytokines regulate epidermal stem cells in wound epithelialization. Stem Cell Res. Ther. 2020, 11, 232.
- Heinrich, P.-C.; Behrmann, I.; Haan, S.; Hermanns, H.-M.; Müller-Newen, G.; Schaper, F. Principles of interleukin (IL)-6-type cytokine signalling and its regulation. Biochem. J. 2003, 15, 374.
- Mi, F.L.; Wu, Y.B.; Shyu, S.S.; Schoung, J.Y.; Huang, Y.B.; Tsai, Y.H.; Hao, J.Y. Control of wound infections using a bilayer chitosan wound dressing with sustainable antibiotic delivery. J. Biomed. Mater. Res. 2002, 59, 438–449.
- Tamahkar, E.; Özkahraman, B.; Süloğlu, A.K.; İdil, N.; Perçin, I. A novel multilayer hydrogel wound dressing for antibiotic release. J. Drug Deliv. Sci. Technol. 2020, 58, 101536.
- Sabitha, M.; Rajiv, S. Preparation and characterization of ampicillin-incorporated electrospun polyurethane scaffolds for wound healing and infection control. Polym. Eng. Sci. 2015, 55, 541–548.
- Ye, S.; Jiang, L.; Wu, J.; Su, C.; Huang, C.; Liu, X.; Shao, W. Flexible amoxicillin-grafted bacterial cellulose sponges for wound dressing: In vitro and in vivo evaluation. ACS Appl. Mater. Interfaces 2018, 10, 5862–5870.
- Basha, M.; AbouSamra, M.M.; Awad, G.A.; Mansy, S.S. A potential antibacterial wound dressing of cefadroxil chitosan nanoparticles in situ gel: Fabrication, in vitro optimization and in vivo evaluation. Int. J. Pharm. 2018, 544, 129–140.
- Nikdel, M.; Rajabinejad, H.; Yaghoubi, H.; Mikaeiliagah, E.; Cella, M.A.; Sadeghianmaryan, A.; Ahmadi, A. Fabrication of cellulosic nonwoven material coated with polyvinyl alcohol and zinc oxide/mesoporous silica nanoparticles for wound dressing purposes with cephalexin delivery. ECS J. Solid State Sci. Technol. 2021, 10, 057003.
- Rădulescu, M.; Holban, A.-M.; Mogoantă, L.; Bălşeanu, T.-A.; Mogos-anu, G.-D.; Savu, D.; Popescu, R.C.; Fufă, O.; Grumezescu, A.M.; Bezirtzoglou, E.; et al. Fabrication, Characterization, and Evaluation of Bionanocomposites Based on Natural Polymers and Antibiotics for Wound Healing Applications. Molecules 2016, 21, 761.
- Bakadia, B.M.; Boni, B.O.O.; Ahmed, A.A.Q.; Zheng, R.; Shi, Z.; Ullah, M.W.; Lamboni, L.; Yang, G. In Situ Synthesized Porous Bacterial Cellulose/Poly (vinyl alcohol)-Based Silk Sericin and Azithromycin Release System for Treating Chronic Wound Biofilm. Macromol. Biosci. 2022, 1, 2200201.
- Ciftci, F.; Ayan, S.; Duygulu, N.; Yilmazer, Y.; Karavelioglu, Z.; Vehapi, M.; ÇakırKoç, R.; Sengor, M.; Yılmazer, H.; Ozcimen, D.; et al. Selenium and clarithromycin loaded PLA-GO composite wound dressings by electrospinning method. Int. J. Polym. Mater. Polym. Biomater. 2022, 13, 71.
- de Souza, R.F.B.; de Souza, F.C.B.; Moraes, Â.M. Polysaccharide-based membranes loaded with erythromycin for application as wound dressings. Appl. Polym. Sci. 2016, 10, 133.
- Alavarse, A.C.; de Oliveira Silva, F.W.; Colque, J.T.; da Silva, V.M.; Prieto, T.; Venancio, E.C.; Bonvent, J.J. Tetracycline hydrochloride-loaded electrospun nanofibers mats based on PVA and chitosan for wound dressing. Mater. Sci. Eng. C 2017, 1, 77.
- Khampieng, T.; Wnek, G.-E.; Supaphol, P. Electrospun DOXY-h loaded-poly(acrylic acid) nanofiber mats:In vitro drug release and antibacterial properties investigation. J. Biomater. Sci. Polym. Ed. 2014, 25, 1292–1305.
- Akota, I.; Alvsaker, B.; Bjørnland, T. The effect of locally applied gauze drain impregnated with chlortetracycline ointment in mandibular third-molar surgery. Acta Odontol. Scand. 1998, 56, 25–29.
- Abbott, P.V.; Hume, W.R.; Pearman, J.W. Antibiotics and endodontics. Aust. Dental. J. 1990, 35, 50–60.
- Michalska-Sionkowska, M.; Kaczmarek, B.; Walczak, M.; Sionkowska, A. Antimicrobial activity of new materials based on the blends of collagen/chitosan/hyaluronic acid with gentamicin sulfate addition. Mater. Sci. Eng. C 2018, 1, 86.
- Anjum, A.; Sim, C.H.; Ng, S.F. Hydrogels containing antibiofilm and antimicrobial agents beneficial for biofilm-associated wound infection: Formulation characterizations and In vitro study. AAPS PharmSciTech 2018, 19, 1219–1230.
- Ahire, J.J.; Robertson, D.D.; van Reenen, A.J.; Dicks, L.M.T. Polyethylene oxide (PEO)-hyaluronic acid (HA) nanofibers with kanamycin inhibits the growth of Listeria monocytogenes. Biomed. Pharmacother. 2017, 86, 143–148.
- Nitanan, T.; Akkaramongkolporn, P.; Rojanarata, T.; Ngawhirunpat, T.; Opanasopit, P. Neomycin-loaded poly (styrene sulfonic acid-co-maleic acid) (PSSA-MA)/polyvinyl alcohol (PVA) ion exchange nanofibers for wound dressing materials. Int. J. Pharm. 2013, 1, 448.
- Denkbaş, E.U.R.B.; Öztürk, E.; Özdem&unknownr, N.; Agalar, C. Norfloxacin-loaded chitosan sponges as wound dressing material. J. Biomater. Appl. 2004, 18, 291–303.
- Contardi, M.; Heredia-Guerrero, J.A.; Perotto, G.; Valentini, P.; Pompa, P.P.; Spanò, R.; Goldonic, L.; Bertorelli, R.; Athanassiou, A.; Bayera, I.S. Transparent ciprofloxacin-povidone antibiotic films and nanofiber mats as potential skin and wound care dressings. Eur. J. Pharm. Sci. 2017, 104, 133–144.
- Li, H.; Williams, G.R.; Wu, J.; Wang, H.; Sun, X.; Zhu, L.M. Poly (N-isopropylacrylamide)/poly (l-lactic acid-co-ɛ-caprolactone) fibers loaded with ciprofloxacin as wound dressing materials. Mater. Sci. Eng. C 2017, 1, 79.
- Pamfil, D.; Vasile, C.; Tarţău, L.; Vereştiuc, L.; Poiată, A. pH-Responsive 2-hydroxyethyl methacrylate/citraconic anhydride–modified collagen hydrogels as ciprofloxacin carriers for wound dressings. J. Bioact. Compat. Polym. 2017, 32, 355–381.
- Pásztor, N.; Rédai, E.; Szabó, Z.I.; Sipos, E. Preparation and Characterization of Levofloxacin-Loaded Nanofibers as Potential Wound Dressings. Acta Med. Marisiensis 2017, 1, 63.
- Singh, B.; Dhiman, A. Designing bio-mimetic moxifloxacin loaded hydrogel wound dressing to improve antioxidant and pharmacology properties. RSC Adv. 2015, 5, 44666–44678.
- Kurczewska, J.; Pecyna, P.; Ratajczak, M.; Gajęcka, M.; Schroeder, G. Halloysite nanotubes as carriers of vancomycin in alginate-based wound dressing. Saudi Pharm. J. 2017, 1, 25.
- Amiri, N.; Ajami, S.; Shahroodi, A.; Jannatabadi, N.; Darban, S.A.; Bazzaz, B.S.F.; Pishavar, E.; Kalalinia, F.; Movaffagh, J. Teicoplanin-loaded chitosan-PEO nanofibers for local antibiotic delivery and wound healing. Int. J. Biol. Macromol. 2020, 162, 645–656.
- Rolston, K.V.I.; Dholakia, N.; Ho, D.H.; LeBlanc, B.; Dvorak, T.; Streeter, H. In-vitro activity of ramoplanin (a novel lipoglycopeptide), vancomycin, and teicoplanin against gram-positive clinical isolates from cancer patients. J. Antimicrob. Chemother. 1996, 38, 265–269.
- Dou, J.L.; Jiang, Y.W.; Xie, J.Q.; Zhang, X.G. New is old, and old is new: Recent advances in antibiotic-based, antibiotic-free and ethnomedical treatments against methicillin-resistant Staphylococcus aureus wound infections. Int. J. Mol. Sci. 2016, 17, 617.
- Fajardo, A.R.; Lopes, L.C.; Caleare, A.O.; Britta, E.A.; Nakamura, C.V.; Rubira, A.F.; Muniz, E.C. Silver sulfadiazine loaded chitosan/chondroitin sulfate films for a potential wound dressing application. Mater. Sci. Eng. C Mater. Biol. Appl. 2013, 33, 588–595.
- Hasselmann, J.; Kühme, T.; Acosta, S. Antibiotic prophylaxis with trimethoprim/sulfamethoxazole instead of cloxacillin fails to improve inguinal surgical site infection rate after vascular surgery. Eur. J. Vasc. Endovasc. Surg. 2015, 49, 129–134.
- Gjorevski, N.; Nikolaev, M.; Brown, T.E.; Mitrofanova, O.; Brandenberg, N.; DelRio, F.W.; Yavitt, F.M.; Liberali, P.; Anseth, K.S.; Lutolf, M.P. Tissue geometry drives deterministic organoid patterning. Science 2022, 375, eaaw9021.
- ValadanTahbaz, S.; Azimi, L.; Asadian, M.; Lari, A.R. Evaluation of synergistic effect of tazobactam with meropenem and ciprofloxacin against multi-drug resistant Acinetobacter baumannii isolated from burn patients in Tehran. GMS Hyg. Infect. Control 2019, 14, Doc08.
- Yang, M.; Hu, Z.; Hu, F. Nosocomial meningitis caused by Acinetobacter baumannii: Risk factors and their impact on patient outcomes and treatments. Future Microbiol. 2012, 7, 787–793.
- NourianDehkordi, A.; MirahmadiBabaheydari, F.; Chehelgerdi, M.; RaeisiDehkordi, S. Skin tissue engineering: Wound healing based on stem-cell-based therapeutic strategies. Stem Cell Res. Ther. 2019, 10, 111.
- Gonzales, K.A.U.; Fuchs, E. Skin and its regenerative powers: An alliance between stem cells and their niche. Dev. Cell 2017, 43, 387–401.
- Gorain, B.; Pandey, M.; Leng, N.H.; Yan, C.W.; Nie, K.W.; Kaur, S.J.; Marshall, V.; Sisinthy, S.P.; Panneerselvam, J.; Molugulu, N.; et al. Advanced drug delivery systems containing herbal components for wound healing. Int. J. Pharm. 2022, 617, 121617.
- Huang, S.; Lu, G.; Wu, Y.; Jirigala, E.; Xu, Y.; Ma, K.; Fu, X. Mesenchymal stem cells delivered in a microsphere-based engineered skin contribute to cutaneous wound healing and sweat gland repair. J. Dermatol. Sci. 2012, 66, 29–36.
- MofazzalJahromi, M.; SahandiZangabad, P.; MoosaviBasri, S.M.; SahandiZangabad, K.; Ghamarypour, A.; Aref, A.; Karimi, M.; Hamblin, M.R. Nanomedicine and advanced technologies for burns: Preventing infection and facilitating wound healing. Adv. Drug Deliv. Rev. 2018, 123, 33–64.
- Negut, I.; Grumezescu, V.; Grumezescu, A.M. Treatment Strategies for Infected Wounds. Molecules 2018, 23, 2392.
- Likus, W.; Bajor, G.; Siemianowicz, K. Nanosilver—Does it have only one face? Acta Biochim. Pol. 2013, 60, 495–501.
- Chen, C.Y.; Yin, H.; Chen, X.; Chen, T.H.; Liu, H.M.; Rao, S.S.; Tan, Y.J.; Qian, Y.X.; Liu, Y.; Hu, X.K. Ångstrom-scale silver particle-embedded carbomer gel promotes wound healing by inhibiting bacterial colonization and inflammation. Sci. Adv. 2020, 6, eaba0942.
- Alven, S.; Aderibigbe, B.A. Chitosan and Cellulose-Based Hydrogels for Wound Management. Int. J. Mol. Sci. 2020, 21, 9656.
- Dhaliwal, K.; Lopez, N. Hydrogel dressings and their application in burn wound care. Br. J. Community Nurs. 2018, 23 (Suppl. 9), S24–S27.
- Tavakoli, S.; Klar, A.S. Advanced Hydrogels as Wound Dressings. Biomolecules 2020, 10, 1169.
- Xu, H.L.; Chen, P.P.; ZhuGe, D.L.; Zhu, Q.Y.; Jin, B.H.; Shen, B.X.; Xiao, J.; Zhao, Y.Z. Liposomes with Silk Fibroin Hydrogel Core to Stabilize bFGF and Promote the Wound Healing of Mice with Deep Second-Degree Scald. Adv. Healthc. Mater. 2017, 6, 1700344.
- Reimer, K.; Vogt, P.M.; Broegmann, B.; Hauser, J.; Rossbach, O.; Kramer, A.; Rudolph, P.; Bosse, B.; Schreier, H.; Fleischer, W. An innovative topical drug formulation for wound healing and infection treatment: In vitro and in vivo investigations of a povidone-iodine liposome hydrogel. Dermatology 2000, 201, 235–241.
- Koshak, A.E.; Algandaby, M.M.; Mujallid, M.I.; Abdel-Naim, A.B.; Alhakamy, N.A.; Fahmy, U.A.; Alfarsi, A.; Badr-Eldin, S.M.; Neamatallah, T.; Nasrullah, M.Z.; et al. Wound Healing Activity of Opuntia ficus-indica Fixed Oil Formulated in a Self-Nanoemulsifying Formulation. Int. J. Nanomed. 2021, 16, 3889–3905.
- Grip, J.; Engstad, R.; Skjæveland, I.; Škalko-Basnet, N.; Isaksson, J.; Basnet, P.; Holsæter, A.M. Beta-glucan-loaded nanofiber dressing improves wound healing in diabetic mice. Eur. J. Pharm. Sci. 2018, 121, 269–280.
- Xu, X.; Wang, X.; Qin, C.; Khan, A.U.R.; Zhang, W.; Mo, X. Silk fibroin/poly-(L-lactide-co-caprolactone) nanofiber scaffolds loaded with Huangbai Liniment to accelerate diabetic wound healing. Colloids Surf. B Biointerfaces 2021, 199, 111557.
- Alzarea, A.I.; Alruwaili, N.K.; Ahmad, M.M.; Munir, M.U.; Butt, A.M.; Alrowaili, Z.A.; Shahari, M.S.B.; Almalki, Z.S.; Alqahtani, S.S.; Dolzhenko, A.V.; et al. Development and Characterization of Gentamicin-Loaded Arabinoxylan-Sodium Alginate Films as Antibacterial Wound Dressing. Int. J. Mol. Sci. 2022, 23, 2899.
- Lv, F.; Wang, J.; Xu, P.; Han, Y.; Ma, H.; Xu, H.; Chen, S.; Chang, J.; Ke, Q.; Liu, M.; et al. A conducive bioceramic/polymer composite biomaterial for diabetic wound healing. Acta Biomater. 2017, 60, 128–143.
- Li, Y.; Zhang, Z.Z. Sustained curcumin release from PLGA microspheres improves bone formation under diabetic conditions by inhibiting the reactive oxygen species production. Drug Des. Dev. Ther. 2018, 12, 1453–1466.
- Elkomy, M.H.; Eid, H.M.; Elmowafy, M.; Shalaby, K.; Zafar, A.; Abdelgawad, M.A.; Rateb, M.E.; Ali, M.R.A.; Alsalahat, I.; Abou-Taleb, H.A. Bilosomes as a promising nanoplatform for oral delivery of an alkaloid nutraceutical: Improved pharmacokinetic profile and snowballed hypoglycemic effect in diabetic rats. Drug Deliv. 2022, 29, 2694–2704.
- Ternullo, S.; Schulte Werning, L.V.; Holsæter, A.M.; Škalko-Basnet, N. Curcumin-in-Deformable Liposomes-in-Chitosan-Hydrogel as a Novel Wound Dressing. Pharmaceutics 2019, 12, 8.
- Cui, M.D.; Pan, Z.H.; Pan, L.Q. DangguiBuxue Extract-Loaded Liposomes in Thermosensitive Gel Enhance In Vivo Dermal Wound Healing via Activation of the VEGF/PI3K/Akt and TGF-β/SmadsSignaling Pathway. Evid. Based Complement. Altern. Med. 2017, 2017, 8407249.
- Kalantari, K.; Mostafavi, E.; Afifi, A.M.; Izadiyan, Z.; Jahangirian, H.; Rafiee-Moghaddam, R.; Webster, T.J. Wound dressings functionalized with silver nanoparticles: Promises and pitfalls. Nanoscale. 2020, 12, 2268–2291.
- Shalaby, M.A.; Anwar, M.M.; Saeed, H. Nanomaterials for application in wound Healing: Current state-of-the-art and future perspectives. J. Polym. Res. 2022, 29, 91.
- Souriyan-Reyhani pour, H.; Khajavi, R.; Yazdanshenas, M.E.; Zahedi, P.; Mirjalili, M. Cellulose acetate/poly(vinyl alcohol) hybrid fibrous mat containing tetracycline hydrochloride and phenytoin sodium: Morphology, drug release, antibacterial, and cell culture studies. J. Bioact. Compat. Polym. 2018, 33, 597–611.
- Kong, Y.; Xu, R.; Darabi, M.A.; Zhong, W.; Luo, G.; Xing, M.M.Q.; Wu, J. Fast and safe fabrication of a free-standing chitosan/alginate nanomembrane to promote stem cell delivery and wound healing. Int. J. Nanomed. 2016, 11, 2543–2555.
- Lohmann, N.; Schirmer, L.; Atallah, P.; Wandel, E.; Ferrer, R.A.; Werner, C.; Simon, J.C.; Franz, S.; Freudenberg, U. Glycosaminoglycan-based hydrogels capture inflammatory chemokines and rescue defective wound healing in mice. Sci. Transl. Med. 2017, 9, eaai9044.
- Cao, L.; Shao, G.; Ren, F.; Yang, M.; Nie, Y.; Peng, Q.; Zhang, P. Cerium oxide nanoparticle-loaded polyvinyl alcohol nanogels delivery for wound healing care systems on surgery. Drug Deliv. 2021, 28, 390–399.
- Ziv-Polat, O.; Topaz, M.; Brosh, T.; Margel, S. Enhancement of incisional wound healing by thrombin conjugated iron oxide nanoparticles. Biomaterials 2010, 31, 741–747.
- Walker, R.M.; Gillespie, B.M.; Thalib, L.; Higgins, N.S.; Whitty, J.A. Foam dressings for treating pressure ulcers. Cochrane Database Syst. Rev. 2017, 10, CD011332.
- Jones, R.N. Critical assessment of the newer non-quinolone oral antimicrobial agents. Antimicrob. Newsl. 1989, 6, 53–60.
- Sillmon, K.; Moran, C.; Shook, L.; Lawson, C.; Burfield, A.H. The Use of Prophylactic Foam Dressings for Prevention of Hospital-Acquired Pressure Injuries: A Systematic Review. J. Wound Ostomy Cont. Nurs. 2021, 48, 211–218.
- Farahani, M.; Shafiee, A. Wound Healing: From Passive to Smart Dressings. Adv. Healthc. Mater. 2021, 10, e2100477.
- Boateng, J.S.; Matthews, K.H.; Stevens, H.N.; Eccleston, G.M. Wound healing dressings and drug delivery systems: A review. J. Pharm. Sci. 2008, 97, 2892–2923.
- Sierra-Sánchez, Á.; Kim, K.H.; Blasco-Morente, G.; Arias-Santiago, S. Cellular human tissue-engineered skin substitutes investigated for deep and difficult to heal injuries. NPJ Regen. Med. 2021, 6, 35.
- Jin, S.; Oh, Y.N.; Son, Y.R.; Kwon, B.; Park, J.H.; Gang, M.J.; Kim, B.W.; Kwon, H.J. Three-Dimensional Skin Tissue Printing with Human Skin Cell Lines and Mouse Skin-Derived Epidermal and Dermal Cells. J Microbiol. Biotechnol. 2022, 32, 238–247.
- Tan, S.H.; Ngo, Z.H.; Sci, D.B.; Leavesley, D.; Liang, K. Recent Advances in the Design of Three-Dimensional and Bioprinted Scaffolds for Full-Thickness Wound Healing. Tissue Eng. Part B Rev. 2022, 28, 160–181.
- Gupta, P.; Sheikh, A.; Abourehab, M.A.S.; Kesharwani, P. Amelioration of Full-Thickness Wound Using Hesperidin Loaded Dendrimer-Based Hydrogel Bandages. Biosensors 2022, 12, 462.
- Zhang, Y.; Wang, B.; Meng, X.; Sun, G.; Gao, C. Influences of acid-treated multiwalled carbon nanotubes on fibroblasts: Proliferation, adhesion, migration, and wound healing. Ann. Biomed. Eng. 2011, 39, 414–426.
- Kittana, N.; Assali, M.; Abu-Rass, H.; Lutz, S.; Hindawi, R.; Ghannam, L.; Zakarneh, M.; Mousa, A. Enhancement of wound healing by single-wall/multi-wall carbon nanotubes complexed with chitosan. Int. J. Nanomed. 2018, 13, 7195–7206.
- Liu, S.; Wu, G.; Chen, X.; Zhang, X.; Yu, J.; Liu, M.; Zhang, Y.; Wang, P. Degradation Behavior In Vitro of Carbon Nanotubes (CNTs)/Poly(lactic acid) (PLA) Composite Suture. Polymers 2019, 11, 1015.
- Yin, M.; Wu, J.; Deng, M.; Wang, P.; Ji, G.; Wang, M.; Zhou, C.; Blum, N.T.; Zhang, W.; Shi, H.; et al. Multifunctional Magnesium Organic Framework-Based Microneedle Patch for Accelerating Diabetic Wound Healing. ACS Nano 2021, 15, 17842–17853.
- Wang, Y.; Lu, H.; Guo, M.; Chu, J.; Gao, B.; He, B. Personalized and Programmable Microneedle Dressing for Promoting Wound Healing. Adv. Healthc. Mater. 2022, 11, e2101659.
- Chi, J.; Zhang, X.; Chen, C.; Shao, C.; Zhao, Y.; Wang, Y. Antibacterial and angiogenic chitosan microneedle array patch for promoting wound healing. Bioact. Mater. 2020, 5, 253–259.
- Chi, J.; Sun, L.; Cai, L.; Fan, L.; Shao, C.; Shang, L.; Zhao, Y. Chinese herb microneedle patch for wound healing. Bioact. Mater. 2021, 6, 3507–3514.
- Wang, Y.; Gao, B.; He, B. Toward Efficient Wound Management: Bioinspired Microfluidic and Microneedle Patch. Small 2022, 19, e2206270.
