Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Rita Xu and Version 1 by Andrea Bagno.
Tantalum has known ever wider applications for the production of endosseous implantable devices in the orthopedic and dental fields. Its excellent performances are due to its capacity to stimulate new bone formation, thus improving implant integration and stable fixation. Tantalum’s mechanical features can be mainly adjusted by controlling its porosity thanks to a number of versatile fabrication techniques, which allow obtaining an elastic modulus similar to that of bone tissue, thus limiting the stress-shielding effect.
- tantalum
- trabecular metal
- porous metal
1. Introduction
The worldwide increase in the elderly population is testament to the general improvement of living conditions but necessitates a parallel increase in healthcare actions. With specific regard to the orthopedic field, the Italian Arthroplasty Registry (RIAP) evidences an average growth of orthopedic surgeries by 7.2% from 2018 to 2019, in detail: +5.7% for the hip, +8.8% for the knee, and +20.3% for the shoulder [1]. As documented in [2[2][3],3], the worldwide market of orthopedic devices (both accessories and surgical apparatuses) was valued at 40.9 billion USD in 2021, and it is expected to reach approximately 43.1 billion USD by 2024. This is due to multiple reasons: the growing aging population of course, but also the incidence of orthopedic disorders (e.g., degenerative bone disease) caused primarily by sedentary lifestyle and obesity, and the rising number of road accidents. Consequently, manufacturers are investing significantly in the development of more efficient devices with the aim of reducing the costs and introducing technological innovations at the same time.
The search for innovative materials becomes a crucial item to overcome health-related problems, thus increasing patients’ quality of life [4]. Any material intended for use in permanent contact with bone has to “respect” its complex physiology, which ultimately depends on the interconnected roles of osteoblasts and osteoclasts [5]. While osteoblasts are mainly responsible for bone tissue deposition, osteoclasts degrade and resorb mature bone. The fine balancing of these opposite activities allows regulating the so-called “bone remodeling” [6]; this implies the removal of mineralized bone and the formation of newly deposited bone matrix (where mature osteoblasts are embedded within the lacunae and eventually differentiate into osteocytes). Bone remodeling allows adjusting the bone architecture in response to variable mechanical stimuli; it also permits repairing micro-damages to prevent their dangerous accumulation. Lastly, bone remodeling is fundamental to maintain calcemic levels by releasing calcium ions from the bone matrix during degradation, and accumulate them during mineralization.
Being able to convert a mechanical information (load) into a biological activity (remodeling), bone cells are capable of mechanotransduction [7]. If the bone tissue is not properly solicited, e.g., because of the presence of a prosthetic device altering load distribution, the correct remodeling is hampered. Consequently, atrophic bone is produced where the tissue is no more physiologically loaded, and denser bone grows in the area exposed to stresses higher than physiological [8]. This phenomenon is called the “stress-shielding effect”. The capacity of the healthy bone to adapt to variable loading conditions was discovered and described in the 19th century by the well-known Wolff’s law [9]; indeed, the exact mechanism of bone adaptation to load has not yet been completely understood [10].
Endosseous devices, such as those commonly used in orthopedics, have to assure adequate mechanical features and biocompatibility; moreover, they have to promote biological fixation and possibly prevent the stress-shielding effect. On the one hand, materials traditionally used in orthopedics (e.g., stainless steel, cobalt–chromium, and titanium alloys) provide excellent structural support with very good clinical outcomes; on the other hand, their high stiffness and low porosity represent major unsolved limitations [11]. With regard to orthopedic applications, it is worth mentioning the use of nitinol (e.g., nickel and titanium alloy characterized by the shape memory effect [12]) for the production of arthrodesis implants experimentally evaluated both on a polyurethane foam model and on a cadaveric model [13,14][13][14].
Materials other than metals can be exploited for endosseous devices, as well as for blood-contacting applications: for instance, silicon carbide (SiC), due to its inertness, hardness, stiffness, tribological features, and hemocompatibility, has been considered as an alternative to traditional metallic materials such as CoCrMo and Ti6Al4V alloys [15].
Over time, other metallic materials have been proposed. Due to its high resistance to corrosion and excellent biocompatibility, the potential of tantalum (Ta) has emerged for several biomedical applications since the 1940s, particularly surgical suture threads, bone fixation parts, bone implants, vascular stent coatings, and medical imaging contrast agent [16]. Moreover, tantalum exhibits advantageous mechanical ductility, combined with the potential for osteoconductivity, osteoinductivity, and angioinductivity. While Ta presents a relatively high stiffness (185 GPa) in the solid form, when it is produced as a porous material, its elastic modulus decreases to 3 GPa, which is similar to that of human bone (from 0.4 GPa of trabecular bone to 17.9 GPa of cortical bone) [17,18][17][18].
By definition, porous metals (or trabecular metals) present a cellular structure characterized by a three-dimensional network of interconnected pores; they are of particular interest for orthopedic applications [19]. Porous metals exhibit lower but sufficient stiffness and strength compared to solid metals; therefore, they are mainly used for load-bearing and structural purposes. Indeed, any specific application depends on three parameters: composition, macroscopic shape, and pore structure [20]. The composition determines the capacity of the material to suit physical and chemical requirements. The macroscopic shape is critical for the integration/combination with other materials. The pore structure mainly affects the mechanical stability and, in the special case of orthopedic implants, the accommodation of bone cells. Thus, in addition to other advantageous features, porous Ta is intrinsically able to support attachment, proliferation, differentiation, and mineralization of osteoblasts, thus promoting osteogenesis and osteointegration [21].
2. Tantalum and Its Properties
Tantalus was a Greek mythological figure who was condemned to a famous punishment; he had to stand in a water pool under a tree, but he was not allowed to drink the water or to eat the fruits. The myth illustrates the refractory property of tantalum that is highly unreactive in almost all acids, with the exception of hydrofluoric acid and acids containing fluoride and sulfur trioxide [22]. Tantalum is a chemical element with atomic number 73, and with a molecular mass equal to 180.05, represented by the symbol Ta [18]. It was discovered by Anders Ekeberg in 1802 and isolated by Berzelius Jöns in 1820. It is a transition metal present in the Earth’s crust (1–2 ppm) [23]; it is a rather rare metal, found in the minerals tantalite and euxenite. Tantalum is malleable at room temperature and has a bright blue-gray color. Being a refractory metal, it has a high melting point (3017 °C) and can be used as a thermal and electrical conductor. Tantalum quickly reacts with oxygen to form oxides, which exist in two forms: Ta2O5 and TaO2. When tantalum is exposed to the air or is industrially processed, it spontaneously tends to form a layer of Ta2O5 (passivation), which is not conductive, allowing the material to be highly resistant to acids and bases. For this reason, tantalum is not very soluble for any pH and potential value [18]. Tantalum and its alloys possess relevant physicochemical properties, which make them suitable for the production of prosthetic implants and for many other biomedical applications. In particular, the following aspects are considered herein: mechanical properties, corrosion resistance, radiopacity and MRI compatibility, surface properties, hemocompatibility, and osseointegration potential.2.1. Mechanical Properties
The mechanical properties of tantalum are summarized in Table 1. Tantalum exhibits a remarkable elastic modulus (185 GPa), even higher than commercially pure titanium and close to that of 316 L stainless steel and cobalt–chromium (CoCr) alloy. On the other hand, yield strength and tensile strength are much lower than those of the abovementioned materials.| Metal | Elastic Modulus (GPa) | Yield Strength (MPa) | Tensile Strength (MPa) |
|---|---|---|---|
| Tantalum | 185 | 138 | 207 |
| cp Titanium | 110 | 485 | 760 |
| 316 L stainless steel | 190 | 331 | 586 |
| Co-Cr alloy | 210 | 448–648 | 951–1220 |
2.2. Corrosion Resistance
When any metal is implanted into the human body, it has to face an aggressive environment that usually triggers corrosion. Corrosion is the sum of redox reactions that take place in the presence of oxygen in an electrolytic solution, which ultimately result in the release of metal ions and in the degradation of the metallic device. Metal ions can cause toxic effects to the surrounding biological tissues, even systemically. The degradation of the metallic device is accompanied by a progressive loss of physical, chemical, and mechanical features. Therefore, corrosion resistance is of paramount importance for all metals intended for the production of implantable devices. Passivation is a mechanism to increase the corrosion resistance; it consists of the formation of a very compact oxide layer that firmly adheres to the metallic surface. The oxide layer can protect the underlying metal from the direct contact with the biological fluids; moreover, due to its high electrical resistance, it also avoids the transfer of electrons that sustain the redox reactions. As the removal or degradation of the oxide protective layer is often very difficult, corrosion in passivated metals is effectively stopped. The excellent corrosion resistance of Ta is due to the formation of a stable and dense layer of tantalum oxide (Ta2O5), about 2–3 nm thick, which prevents the release of metal ions. Furthermore, a TaO2 film is also present between the overlying Ta2O5 layer and the underlying metal. Several studies highlight the excellent corrosion resistance of Ta and its alloys in acidic and basic environments, particularly in the presence of HCl, H3PO4, and NaOH [24,25,26,27][24][25][26][27]. All these studies also demonstrated that the corrosion rate increases with temperature and acid concentration in solution, but adding Ta alloyed with other elements improves the corrosion resistance. In the study published by Silva et al. [28], the characteristics of the surface oxide layer of Ta in a 0.15 M NaCl solution (simulated body fluid, SBF) were investigated. Discs of pure Ta (15 mm diameter, 5 mm thickness) were immersed in the solution together with the electrodes. After 1 h, the anodic polarization curve showed an initial current increase with a maximum at 1.65 V followed by a decrease; the current growth corresponds to the local dissolution of Ta associated with the degradation of the passivating film, while the decrease is due to the repair of the film thanks to repassivation.2.3. MRI Compatibility
Ta is characterized by high radiopacity thanks to its atomic number and density. Therefore, implantable prosthetic structures made of this metal are easily monitored with fluoroscopy. For example, this imaging technique allows ensuring proper stent release during angioplasty procedures. Table 2 summarizes the atomic numbers and density values of some metals frequently used as biomaterials.| Metal | Atomic Number | Density (g/cm | 3 | ) |
|---|---|---|---|---|
| Tantalum | 73 | 16.6 | ||
| Titanium | 22 | 4.5 | ||
| Iron | 26 | 7.9 | ||
| Cobalt | 27 | 8.9 |
2.4. Surface Properties
The biological response to any implantable material is closely related to its surface properties, including surface energy, morphology, charge, and chemical composition. Surface energy determines the amount of attractive or repulsive forces that the surface can exert on another material. In general, metals have a high surface energy (500–5000 mN/m) due to the presence of the metallic bond. The amount of surface energy controls protein adsorption and the subsequent phases of cell adhesion and growth. The study by M. M. Gentleman and E. Gentleman [31] stated that materials with high surface energy promote cell adhesion and growth, while materials with low surface energy (e.g., some polymers) do not. Tantalum’s surface energy is 100.59 ergs/cm2; this high value is supposed to be a determining factor for promoting the adhesion and growth of osteoblasts and for the osseointegration potential of this metal. Some manufacturing processes can decrease the surface energy for the use of tantalum in contact with blood (e.g., chemical vapor deposition, diffusion coating, nanotube formation, and fluorination).2.5. Hemocompatibility
Surface chemical composition significantly affects the surface energy of any material. In the case of tantalum, after passivation, an oxide layer composed of Ta5+ and O2− ions covers the surface. When these ions attract water molecules, the surface becomes hydrophilic, and this implies a high surface energy. A smooth oxide layer is enough to prevent thrombotic effects when tantalum is in contact with blood, while a very porous structure is used for the surface of orthopedic implants to promote osseointegration. The isoelectric point of tantalum oxide is at 2.7–3.0; thus, its surface is negatively charged at physiological pH (7.4). Platelets, which are negatively charged at physiological pH, are repelled from the surface and this improves the hemocompatibility of the metal. Moreover, tantalum oxide can prevent the electronic transfer to fibrinogen [32], which is the precursor of fibrin, responsible for stabilizing the platelet plug forming the blood clot. This is the reason why the tantalum surface is deemed non-thrombogenic.2.6. Osseointegration Potential
The term “osseointegration” refers to the intimate connection between any endosseous prosthetic implant and the surrounding bone tissue; it implies both the anatomical congruence of the device with respect to the biological environment and the ability to bear physiological loads. With regard to trabecular scaffolds, effective long-term functionality of the implant can be guaranteed by bone ingrowth within the porous structure and not only on the outer surface. The particular structure of trabecular Ta not only facilitates the adhesion and proliferation of osteoblasts, but it also favors the supply of nutrients and oxygen (as well as the removal of catabolites and CO2) that are necessary for new bone tissue formation. This latter begins with the differentiation of pre-osteoblasts and ends with bone mineralization; these processes involve a large number of genes and proteins related to osteogenesis [33]. Bone tissue regeneration is a complex mechanism involving the activation or inhibition of multiple signaling pathways. Interestingly, it has been acknowledged that Ta itself is associated with a large chain of signaling events typical of osteogenesis. Ta contributes to osteogenesis through the regulation of different cellular signaling pathways: the Wnt/β-catenin signaling pathway [34,35][34][35], transforming growth factor-beta (TGF-β) and bone morphogenetic proteins (BMPs) signaling pathway [36,37][36][37], mitogen-activated protein kinases (MAPKs) signaling pathway [38], and integrin signaling pathway [39]. The superior osteoinductivity of Ta to that of titanium received thorough investigations; for instance, the effects of Ta and Ti surfaces on osteogenesis using rat bone mesenchymal stromal cells (rBMSCs) were assessed by Lu et al. [37]. These authors also elucidated the molecular mechanisms regulating metal–cell interactions, which are basically mediated by the integrin α5β1/ERK1/2 pathway. Hu Qian et al. recently reviewed all the mechanisms induced by Ta in osteogenesis [40]. This preseaperrch also pinpointed that many studies elucidated the role of Ta but with various limitations. Firstly, some investigations were just preliminary, while some mechanisms were not described in detail. Secondly, the involvement of Ta in promoting osteogenesis via other pathways associated with bone remodeling has not yet been fully proven.3. Trabecular Tantalum
Two trabecular metals are commonly used for endosseous prosthetic implants: Ta and Ti (and their alloys). Since trabecular Ti shows some limitations, such as low porosity, low coefficient of friction, and an elastic modulus different from that of bone, trabecular Ta is usually preferred. It is characterized by a three-dimensional structure with high porosity (open cell); cells are repeated in a dodecahedral shape similarly to spongy bone. It is obtained via vapor deposition/infiltration of commercially pure Ta onto a vitreous carbon scaffolding. During manufacturing, the glassy carbon bearing structure can be modified to obtain a variety of configurations for a variety of orthopedic applications [11]. Trabecular Ta possesses a high porosity (75–80%), a high coefficient of friction (−1), and an elastic modulus much lower than that of compact Ta (~185 GPa) and more similar to that of bone (Table 3).| Parameter | Value |
|---|---|
| Elastic modulus | 2.5–3.9 GPa |
| Ultimate strength | 50–110 MPa |
| Yield strength | 35–51 MPa |
| Compressive strength | 50–70 MPa |
| Tensile strength | 63 MPa |
| Bending strength | 110 MPa |
3.1. Manufacturing Techniques
Tantalum has good mechanical properties and excellent biocompatibility, is resistant to corrosion, and can play a beneficial role in osteogenesis. All these advantages have been counterbalanced by its difficult manufacturing in the solid (compact) state due to the high melting point and high affinity for oxygen. Only from the early 1990s, thanks to the development of the porous tantalum trabecular-structured metal (PTTM), did this material begin to be used for several prosthetic applications [21]. Therefore, specific manufacturing techniques have been introduced and optimized to address each clinical purpose [20].3.1.1. Chemical Vapor Deposition (CVD)
Chemical vapor deposition (CVD) allows the deposition on a solid support of a molecular precursor, which is supplied in gaseous form and decomposes on the substrate surface. CVD is one of the most important techniques for coating several kinds of materials, and it is commonly used to produce Ta scaffolds in the clinical setting. CVD brings together a set of techniques aimed at depositing thin protective films on a surface; multiple methods are available depending on the process parameters chosen (i.e., pressure, temperature, and type of deposition) [42]. The first step is the production of a low-density vitreous carbon skeleton with a porosity of 98%; it is obtained by pyrolysis of a polymeric foam. The carbon skeleton shows a matrix where a pattern of dodecahedral interconnected pores is repeated. Since the carbon structure can be built with different shapes and sizes, several geometries for as many clinical applications can be produced. Thereafter, TaCl5 (Ta precursor) reacts with H2 at high temperature; Ta then deposits over the carbon structure to get a porous scaffold (99% Ta and 1% glassy carbon by weight). The thickness of the Ta coating varies from 40 to 60 μm, and this parameter can alter the porosity and the mechanical properties of the implant. Generally, the average pore size for orthopedic applications is between 400 and 600 μm and the porosity ranges from 75% to 85% [19]. Different materials other than carbon can be used as a substrate for Ta deposition; in [43], porous Ta scaffolds were produced coating porous silicon carbide (SiC) substrates through CVD at low temperature. The substrate is ultrasonically washed with an acidic solution (HF and HNO3), and then dried under nitrogen. TaCl5 (99.95% pure) is preheated to 223 °C in a stainless sublimator; H2 (99.999%) is used as both a carrier and a reducing agent, while argon (99.998%) is fluxed as a protective gas. TaCl5 vapor is carried by the hydrogen flow through the heated TaCl5 powders. The reaction between TaCl5 and H2 occurs at 1000 °C under a pressure of 2.5 Torr. The deposition process takes approximately 30 min; then, coated samples are ultrasonically cleaned with methanol, acetone, and distilled water. The characteristics of the pores and, consequently, the mechanical properties of the porous structure can be partly tailored by controlling the thickness of the Ta layer deposited onto the substrate.3.1.2. Powder Metallurgy
Powder metallurgy (PM), also known as the “space-holder method”, is a fabrication technique for porous Ta scaffolds associated with low fabrication costs. It consists of a sequence of steps that lead to the compaction and transformation of a metallic powder into a sintered material. This technique consists of five main steps (Figure 1): obtaining the powders, mixing them with the space-holder, compacting the powders, dissolving the space holder particles, and sintering. The Ta powders and the space-holding particles (particles that are later dissolved to generate pores) are mixed, and the mixture is compacted under appropriate pressure (350–450 MPa). The compact compound (green compact) is immersed in distilled water (at 60 °C) to dissolve the space-holding particles and obtain a porous structure Eventually, the porous structure is dried in an oven for 2 h and sintered at 1300–2000 °C under vacuum to obtain the final porous scaffold. The space-holding particles must possess the following features: low cost, fast dissolution in (hot) water, low melting point, non-cytotoxicity and low corrosive effects toward the metal during dissolution. The most used substances are starch, urea, sodium chloride, sucrose, and ammonium bicarbonate.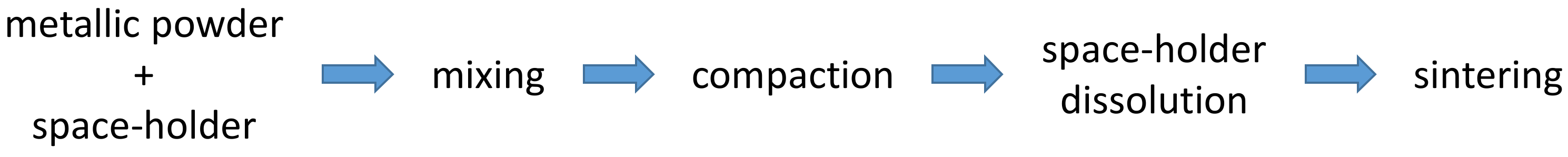
Figure 1. The main steps of the space-holder fabrication technique.
3.1.3. Additive Manufacturing
Medical therapies are moving toward an autologous (personalized) rather than heterologous approach; thus, the need emerges to find manufacturing techniques that easily allow the customization of implantable devices with respect to the specific anatomical characteristics of each patient. In this context, additive manufacturing (AM) appears as a versatile and effective technology to fabricate porous scaffolds intended for tailored orthopedic applications. Additive manufacturing is the process of joining/connecting materials through successive stratification to obtain objects starting from digitalized 3D models [48]. In particular, AM allows obtaining highly customized porous structures with complicated geometries, which accurately correspond to the desired anatomical shape. Furthermore, scaffold porosity can be easily tailored to meet the compressive strength and elastic modulus of bone tissue, avoiding the stress-shielding phenomenon [16]. The ASTM classifies AM techniques into seven groups: vat photopolymerization, material jetting, binder jetting, material extrusion, sheet lamination, direct energy deposition, and powder bed fusion. These classes differ depending on the materials and the process used for layer deposition [49,50][49][50]. Basically, the AM technique requires three main steps [47]: the creation of the 3D model of the object to reproduce, sending the file to the printer, and realization of the physical object layer by layer. Over the years, many AM techniques have been developed, including selective laser melting (SLM), electron beam melting (EBM), direct metal deposition (DMD), direct metal printing (DMP), fused deposition modeling (FDM), direct metal writing (DMW), and binder jetting (BJ). In particular, SLM and EBM are the most widely used AM techniques for the fabrication of metallic porous scaffolds due to their high precision, efficacy, and good stability [51,52][51][52]. In these two techniques, both included in the powder bed fusion category, metal powders are sintered/melted by a different energy source, i.e., an electron beam for EBM and a laser beam for SLM. These systems consist of two platforms, a material delivery platform and a build platform, both enclosed into a chamber. The first platform constantly supplies new material, while a blade gradually removes its excess; the intended object grows onto the build platform by melting metallic powders layer by layer. Once a layer is formed, the platform descends, and a new layer of metal powder is added by the delivery platform. The final step consists of cutting the object formed from the support after obtaining the whole structure.References
- Registro Italiano ArtroProtesi. Annual Report; RIAP: Rome, Italy, 2020.
- Available online: https://www.researchandmarkets.com/reports/4599561/orthopedic-devices-market-size-share-and-trends (accessed on 1 August 2022).
- Available online: https://www.todaysmedicaldevelopments.com/article/medical-device-orthopedic-trends-52218/ (accessed on 1 August 2022).
- Adamovic, D.; Ristic, B.; Zivic, F. Review of Existing Biomaterials—Method of Material Selection for Specific Applications in Orthopedics. In Biomaterials in Clinical Practice; Zivic, F., Affatato, S., Trajanovic, M., Schnabelrauch, M., Grujovic, N., Choy, K., Eds.; Springer: Cham, Switzerland, 2018; pp. 47–99.
- Han, Q.; Wang, C.; Chen, H.; Zhao, X.; Wang, J. Porous Tantalum and Titanium in Orthopedics: A Review. ACS Biomater. Sci. Eng. 2019, 5, 5798–5824.
- Hadjidakis, D.J.; Androulakis, I.I. Bone remodeling. Ann. N. Y. Acad. Sci. 2006, 1092, 385–396.
- Li, M.C.M.; Chow, S.K.H.; Wong, R.M.Y.; Qin, L.; Cheung, W.H. The role of osteocytes-specific molecular mechanism in regulation of mechanotransduction—A systematic review. J. Orthop. Translat. 2021, 29, 1–9.
- Savio, D.; Bagno, A. When the Total Hip Replacement Fails: A Review on the Stress-Shielding Effect. Processes 2022, 10, 612.
- Frost, H.M. Wolff’s Law and bone’s structural adaptations to mechanical usage: An overview for clinicians. Angle Orthod. 1994, 64, 175–188.
- Klein-Nulend, J.; Bakker, A.D.; Bacabac, R.G.; Vatsa, A.; Weinbaum, S. Mechanosensation and transduction in osteocytes. Bone 2013, 54, 182–190.
- Levine, B.R.; Sporer, S.; Poggie, R.A.; Della Valle, C.J.; Jacobs, J.J. Experimental and clinical performance of porous Tantalum in orthopedic surgery. Biomaterials 2006, 27, 4671–4681.
- Eckelmeyer, K.H. The effect of alloying on the shape memory phenomenon in nitinol. Scr. Metall. 1976, 10, 667–672.
- Shen, V.C.; Bumgardner, C.H.; Actis, L.; Ritz, J.; Park, J.; Li, X. 3D digital image correlation evaluation of arthrodesis implants. Clin. Biomech. 2020, 71, 29–36.
- Shen, V.C.; Bumgardner, C.; Actis, L.; Park, J.; Moyer, D.; Nikstaitis, M.D.; Li, X. In Situ Deformation of First Tarsometatarsal Arthrodesis Implants with Digital Image Correlation: A Cadaveric Study. JOM 2022, 74, 3357–3366.
- Li, X.; Wang, X.; Bondokov, R.; Morris, J.; An, Y.H.; Sudarshan, T.S. Micro/nanoscale mechanical and tribological characterization of SiC for orthopedic applications. J. Biomed. Mater. Res. B Appl. Biomater. 2005, 72, 353–361.
- Gao, H.Y.; Yang, J.; Jin, X.; Qu, X.; Zhang, F.; Zhang, D.; Chen, H.; Wei, H.; Zhang, S.; Jia, W.; et al. Porous Tantalum scaffolds: Fabrication, structure, properties, and orthopedic applications. Mater. Des. 2021, 210, 110095.
- Wang, X.; Xu, S.; Zhou, S.; Xu, W.; Leary, M.; Choong, P.; Qian, M.; Brandt, M.; Xie, Y.M. Topological design and additive manufacturing of porous metals for bone scaffolds and orthopaedic implants: A review. Biomaterials 2016, 83, 127–141.
- Wang, X.; Ning, B.; Pei, X. Tantalum and its derivatives in orthopedic and dental implants: Osteogenesis and antibacterial properties. Colloids Surf. B Biointerfaces 2021, 208, 112055.
- Kranzlin, N.; Niederberger, M. Controlled fabrication of porous metals from the nanometer to the macroscopic scale. Mater. Horiz. 2015, 2, 359.
- Wang, Q.; Zhang, H.; Li, Q.; Ye, L.; Gan, H.; Liu, Y.; Wang, H.; Wang, Z. Biocompatibility and osteogenic properties of porous Tantalum. Exp. Ther. Med. 2015, 9, 780–786.
- Bencharit, S.; Byrd, W.C.; Altarawneh, S.; Hosseini, B.; Leong, A.; Reside, G.; Morelli, T.; Offenbacher, S. Development and applications of porous Tantalum trabecular metal-enhanced Titanium dental implants. Clin. Implant. Dent. Relat. Res. 2014, 16, 817–826.
- Mani, G.; Porter, D.; Grove, K.; Collins, S.; Ornberg, A.; Shulfer, R. A comprehensive review of biological and materials properties of Tantalum and its alloys. J. Biomed. Mat. Res. Part A. 2022, 110, 1291–1306.
- George, N.; Nair, A.B. Porous Tantalum: A new biomaterial in orthopedic surgery. In Fundamental Biomaterials: Metals; Balakrishnan, P., Sreekala, M.S., Sabu, T., Eds.; Woodhead Publishing: Sawston, UK, 2018; pp. 243–268.
- Zhou, L.; Niinomi, M.; Akahori, T.; Fukui, H.; Toda, H. Corrosion resistance and biocompatibility of Ti–Ta alloys for biomedical applications. Mater. Sci. Eng. 2005, 398, 28–36.
- Robin, A.; Rosa, J.L. Corrosion behavior of niobium, tantalum and their alloys in hot hydrochloric and phosphoric acid solutions. Int. J. Refract. Met. Hard Met. 2000, 18, 13–21.
- Robin, A. Corrosion behaviour of Tantalum in sodium hydroxide solutions. J. Appl. Electrochem. 2003, 33, 37–42.
- Zhang, Y.; Zheng, Y.; Li, Y.; Wang, L.; Bai, Y.; Zhao, Q.; Xiong, X.; Cheng, Y.; Tang, Z.; Deng, Y.; et al. Tantalum Nitride-Decorated Titanium with Enhanced Resistance to Microbiologically Induced Corrosion and Mechanical Property for Dental Application. PLoS ONE 2015, 10, e0130774.
- Silva, R.A.; Walls, M.; Rondot, B.; Da Cunha Belo, M.; Guidoin, R. Electrochemical and microstructural studies of Tantalum and its oxide films for biomedical applications in endovascular surgery. J. Mater. Sci. Mater. Med. 2002, 13, 495–500.
- Shellock, F.G.; Shellock, V.J. Metallic stents: Evaluation of MR imaging safety. Am. J. Roentgenol. 1999, 173, 543–547.
- Hug, J.; Nagel, E.; Bornstedt, A.; Schnackenburg, B.; Oswald, H.; Fleck, E. Coronary arterial stents: Safety and artifacts during MR imaging. Radiology 2000, 216, 781–787.
- Gentleman, M.M.; Gentleman, E. The role of surface free energy in osteoblast–biomaterial interactions. Int. Mat. Rev. 2014, 59, 417–429.
- Nashed, R.; Hassan, W.M.; Ismail, Y.; Allam, N.K. Unravelling the interplay of crystal structure and electronic band structure of Tantalum oxide (Ta2O5). Phys. Chem. Chem. Phys. 2013, 15, 1352–1357.
- Leng, Y.X.; Chen, J.Y.; Yang, P.; Sun, H.; Wang, J.; Huang, N. The biocompatibility of the Tantalum and Tantalum oxide films synthesized by pulse metal vacuum arc source deposition. Nucl. Instrum. Methods B 2006, 242, 30–32.
- Yang, H.; Guo, Y.; Wang, D.; Yang, X.; Ha, C. Effect of TAK1 on osteogenic differentiation of mesenchymal stem cells by regulating BMP-2 via Wnt/β-catenin and MAPK pathway. Organogenesis 2018, 14, 36–45.
- Yuan, C.; Gou, X.; Deng, J.; Dong, Z.; Ye, P.; Hu, Z. FAK and BMP-9 synergistically trigger osteogenic differentiation and bone formation of adipose derived stem cells through enhancing Wnt-β-catenin signaling. Biomed. Pharmacother. 2018, 105, 753–757.
- Marcellini, S.; Henriquez, J.P.; Bertin, A. Control of osteogenesis by the canonical Wnt and BMP pathways in vivo: Cooperation and antagonism between the canonical Wnt and BMP pathways as cells differentiate from osteochondroprogenitors to osteoblasts and osteocytes. Bioessays 2012, 34, 953–962.
- Lu, M.; Zhuang, X.; Tang, K.; Wu, P.; Guo, X.; Yin, L.; Cao, H.; Zou, D. Intrinsic Surface Effects of Tantalum and Titanium on Integrin α5β1/ERK1/2 Pathway-Mediated Osteogenic Differentiation in Rat Bone Mesenchymal Stromal Cells. Cell. Physiol. Biochem. 2018, 51, 589–609.
- Greenblatt, M.B.; Shim, J.H.; Glimcher, L.H. Mitogen-activated protein kinase pathways in osteoblasts. Annu. Rev. Cell Dev. Biol. 2013, 29, 63–79.
- Wu, M.; Chen, G.; Li, Y.P. TGF-β and BMP signaling in osteoblast, skeletal development, and bone formation, homeostasis and disease. Bone Res. 2016, 4, 16009.
- Qian, H.; Lei, T.; Ye, Z.; Hu, Y.; Lei, P. From the Performance to the Essence: The Biological Mechanisms of How Tantalum Contributes to Osteogenesis. Biomed. Res. Int. 2020, 2020, 5162524.
- Fan, H.; Deng, S.; Tang, W.; Muheremu, A.; Wu, X.; He, P.; Tan, C.; Wang, G.; Tang, J.; Guo, K.; et al. Highly Porous 3D Printed Tantalum Scaffolds Have Better Biomechanical and Microstructural Properties than Titanium Scaffolds. Biomed. Res. Int. 2021, 2021, 2899043.
- Martin, P. Handbook of Deposition Technology for Films and Coatings, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2009.
- Ma, Z.; Xie, H.; Wang, B.; Wei, X.; Zhao, D. A novel Tantalum coating on porous SiC used for bone filling material. Mat. Lett. 2016, 179, 166–169.
- Zhou, Y.; Zhu, Y. Three-dimensional Ta foams produced by replication of NaCl space-holders. Mat Lett. 2013, 99, 8–10.
- Sukumar, V.; Golla, B.R.; Shaik, M.A.; Yadav, A.; Taraka, S.C.D.; Khaple, S. Modeling and Characterization of Porous Tantalum Scaffolds. Trans. Indian Inst. Met. 2019, 72, 935–949.
- Rupérez, E.; Manero, J.; Riccardi, K.; Li, Y.; Aparicio, C.; Gil, F. Development of Tantalum scaffold for orthopedic applications produced by space-holder method. Mater. Des. 2015, 83, 112–119.
- Luo, C.; Wang, C.; Wu, X.; Xie, X.; Wang, C.; Zhao, C.; Zou, C.; Lv, F.; Huang, W.; Liao, J. Influence of porous Tantalum scaffold pore size on osteogenesis and osteointegration: A comprehensive study based on 3D-printing technology. Mater. Sci. Eng. C Mater. Biol. Appl. 2021, 129, 112382.
- ASTM F2792-12; Standard Terminology for Additive Manufacturing Technologies. ASTM: West Conshohocken, PA, USA, 2010.
- Scott, J.; Gupta, N.; Weber, C.; Newsome, S.; Wohlers, T.; Caffrey, T. Additive Manufacturing: Status and Opportunities; Science and Technology Policy Institute: Washington, DC, USA, 2012.
- Bikas, H.; Stavropoulos, P.; Chryssolouris, G. Additive manufacturing methods and modelling approaches: A critical review. Int. J. Adv. Manuf. Technol. 2016, 83, 389–405.
- Huang, S.H.; Liu, P.; Mokasdar, A.; Hou, L. Additive manufacturing and its societal impact: A literature review. Int. J. Adv. Manuf. Technol. 2013, 67, 1191–1203.
- Wauthle, R.; van der Stok, J.; Amin Yavari, S.; Van Humbeeck, J.; Kruth, J.P.; Zadpoor, A.A.; Weinans, H.; Mulier, M.; Schrooten, J. Additively manufactured porous Tantalum implants. Acta Biomater. 2015, 14, 217–225.
More
