Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 3 by Rita Xu and Version 2 by Rita Xu.
The prevalence of osteoarthritis (OA), a degenerative disorder of joints. Its key pathogenic hallmarks include articular cartilage destruction, synovium inflammation, and bone remodeling.
- osteoarthritis
- mesenchymal stem cells
- cartilage regeneration
1. Introduction
Ten percent to fifteen percent of the global population suffers from osteoarthritis (OA), a condition characterized by cartilage deterioration and synovitis. It is a rapidly developing modern disease [1]. Figure 1 summarizes the pathological process in detail [2]. There are many risk factors, such as sex, age, trauma, obesity and genetics. Since the middle of the last century, OA has not only shown its highest worldwide incidence rate [3] but has also forced various developed countries to bear heavy economic burdens because it is a crippling disease [4][5]. Currently, there is no ideal specific drug used for the clinical treatment of OA. To relieve inflammation and discomfort in early stages, nonsteroidal anti-inflammatory medications and analgesics, such as glucosamine, chondroitin supplements, and intra-articular local injections of corticosteroids are primarily employed [6]. Though it is an intrusive procedure that is frequently accompanied by significant uncontrolled complications, joint replacement plays a prominent role in advanced cases [7][8]. However, the service life of the prosthesis and the functional recovery of the damaged limbs are not as positive as researchers originally anticipated and can even be described as restricted [6]. As a result, research into a more secure and efficient treatment for OA is crucial.
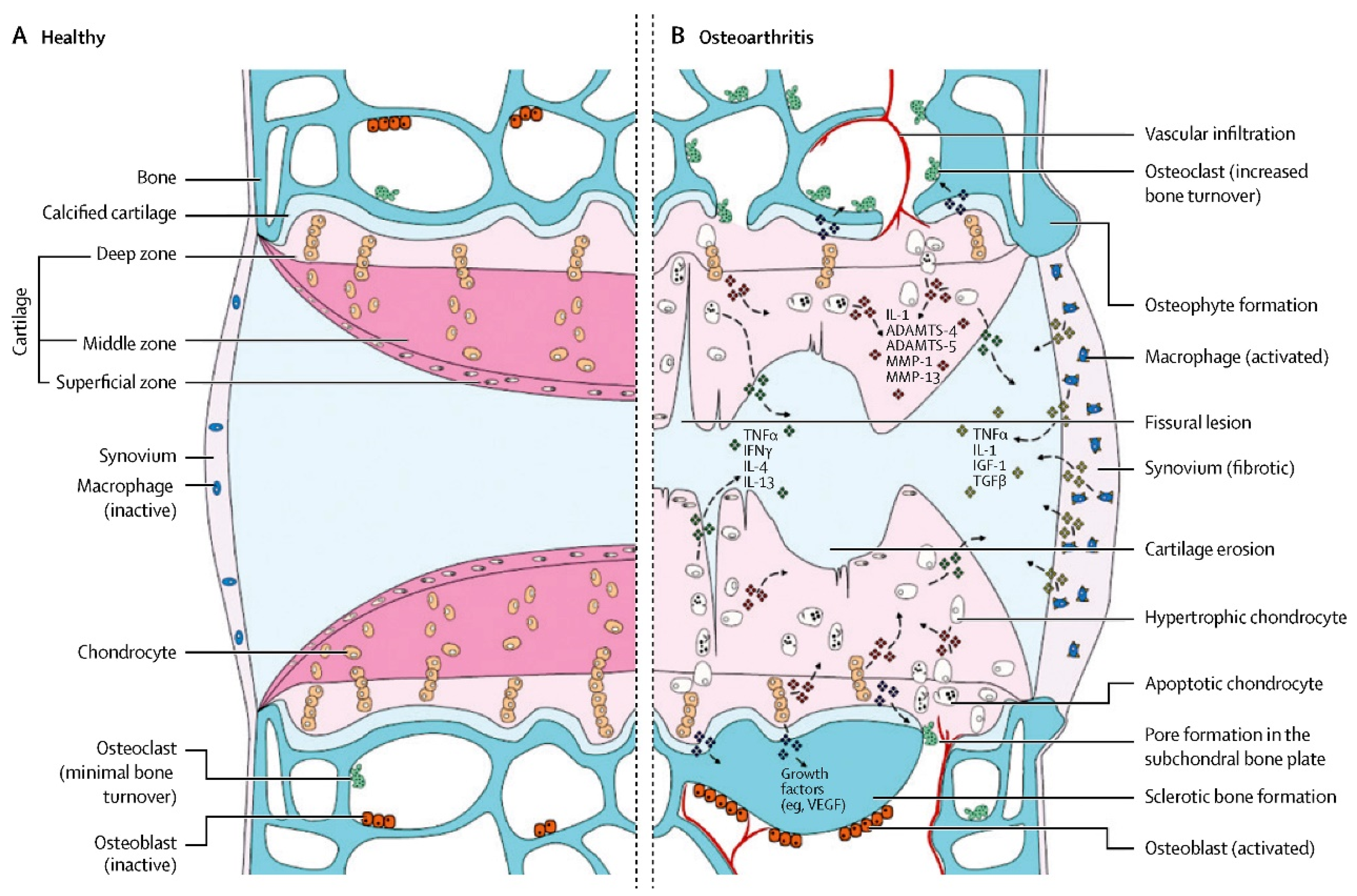
Figure 1. Comparison between normal joints and osteoarthritis. The roles of mechanical, metabolic, and inflammatory factors are identified.
In recent years, several academics have conducted extensive research on MSCs, which are specialized adult stem cells used in many studies [9] due to their capacity for self-renewal and differentiation. MSCs are also widespread throughout the body, suggesting that they might be used as an alternative cell source in OA therapy. Studies have indicated that the surface layer of cartilage sustains the vast majority of damage during OA [10][11]. Importantly, several MSC types have been implicated in the differentiation of chondrocytes. In the process of cartilage remodeling, the surface cartilage is minimally impacted; moreover, following remodeling, bidirectional mitotically active cells can horizontally or vertically replenish chondrocytes [12][13].
In the study of MSCs transplanted into articular cavities in arthritis models, e.g., for mice [14] and horses [15], considerable regeneration and repair in articular cartilage have been observed; moreover, there is evidence that MSCs reduce cartilage lesions by restraining the onset of inflammation. MSCs were found to limit the breakdown of proteoglycan in the cartilage of a rabbit arthritis model by decreasing the production of tumor necrosis factor (TNF)-α and MMP-1 [16]. In addition, by releasing growth factors (such as TGF-beta and IL-6) and antioxidant compounds, MSCs conspicuously suppress apoptosis and fibrosis [17][18].
2. Characteristics of MSCs for the OA Therapy
MSCs, as specific types of adult stem cells, possess great potential in bone tissue engineering and regenerative therapy due to their capacity for self-renewal and differentiation [19][20][21]. The multipotency, wide availability, and low immunogenicity of MSCs have made them a hot topic in the bioremediation field. In 1976, Friedenstein et al. [22] identified and prepared MSCs from different tissues, including fat, placenta, umbilical cord, synovium, bone, and dental pulp [23]. As shown in Figure 2, MSCs, which are mainly extracted from bone marrow, adipose tissue, umbilical cord, and the synovium, possess the ability to differentiate into adipocytes, chondrocytes, and osteoblasts.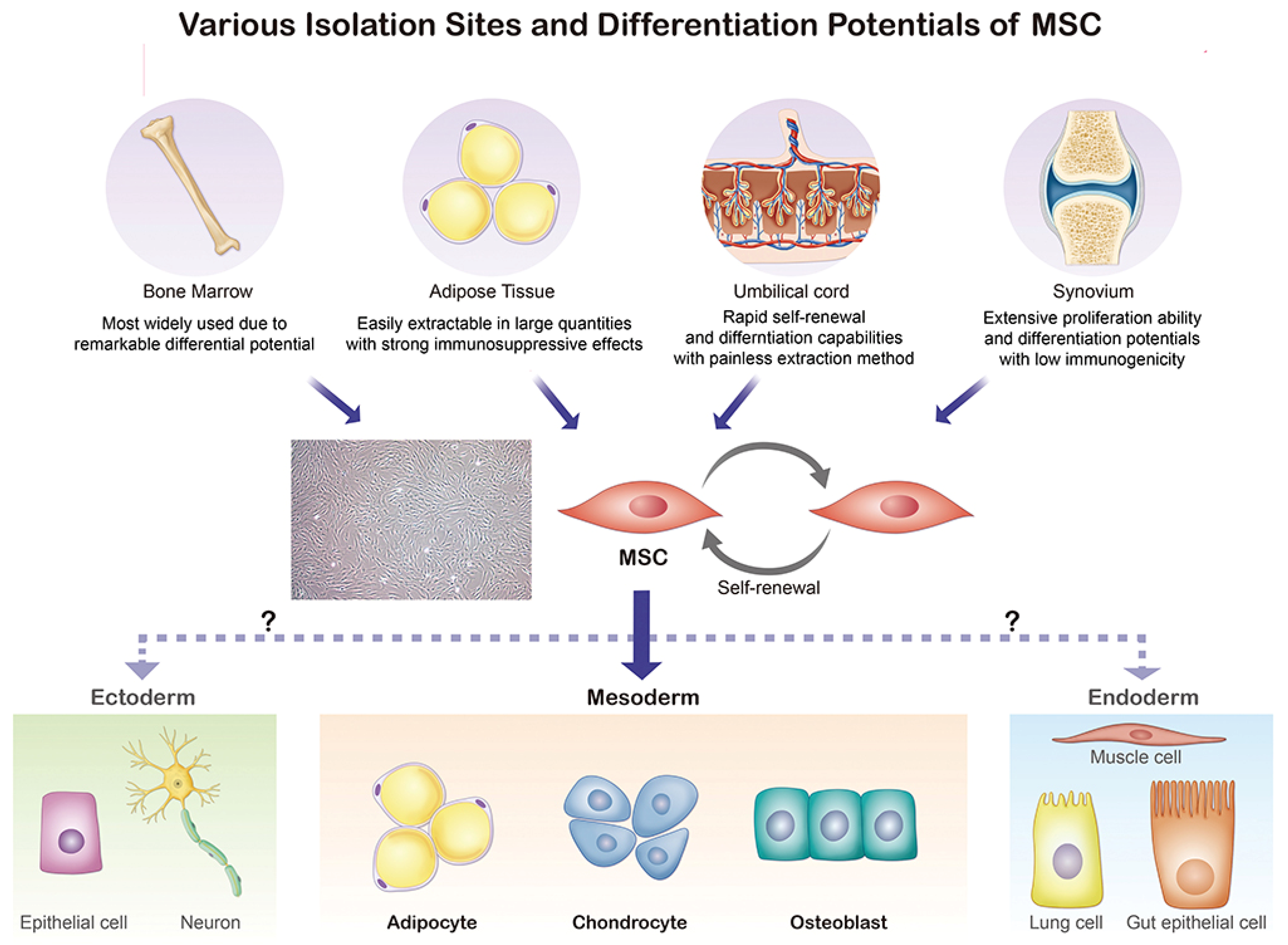
Figure 2. Origin and differentiation directions of MSCs. MSC, mesenchymal stem cell.
3. Underlying Treating Mechanism of MSCs for OA
The therapeutic potential of MSCs in terms of immunological and inflammatory disorders has been explored in several clinical studies [39][40]. BM-MSCs produce substances that are immunoregulatory and anti-inflammatory [41]. Therefore, MSCs are able to efficiently downregulate immune inflammatory processes and boost tissue regeneration because they display particular immunological traits and activities. When tissues are damaged, local tissue progenitor cells that raise the potential to modulate the immune system are called upon and activated [42]. OA is characterized by an influx of immune cells, mostly monocytes/macrophages and then T cells, into the synovium. Furthermore, OA synovia contain mast cells, natural killer (NK) cells, dendritic cells, B cells, and granulocytes. This subject has been more thoroughly covered elsewhere [43]. There might be three ways to accommodate the immune system. Firstly, BM-MSCs may control innate immunity by suppressing the development of mature dendritic cells [44] and lowering the cytotoxicity of NK cells [45]. Secondly, MSCs may modify acquired immunity by preventing cell death (apoptosis) and slowing the development of T and B cells [41][46]. Finally, MSCs may switch macrophages from an inflammatory (M1) phenotype to a restorative (M2) phenotype [47]. It has been demonstrated that BM-MSCs could induce a switch in macrophage phenotype from the pro-inflammatory M1 phenotype, which generates IL-1 and TGF-β, to the anti-inflammatory and chondrogenic phenotype, which produces IL-10, IL-rheumatoid arthritis, and TGF-β [48]. The impact of BM-MSCs on macrophage polarization is mediated by TNF-α, which stimulates gene/protein 6 (TSG-6), prostaglandin E2 (PGE2), and indoleamine 2-dioxygenase 3-dioxygenase (IDO) [48]. The immune switch from M1 to M2 could be beneficial in relieving OA by reducing periarticular inflammation. Moreover, BM-MSCs suppress pathogenic immune responses, remove infections, and improve local cell function [49]. However, the precise mechanism through which BM-MSCs promote joint repair is not yet known. It would be wonderful if MSCs could be implanted and immediately develop into chondrocytes [50][51][52]. Nonetheless, the survival rate of implanted MSCs was found to be low [53], and 50 days after injection, BM-MSCs could not be detected [54]. It has been shown in some studies that implanted MSCs may boost stem/progenitor cell recruitment and cartilage differentiation by secreting substances that encourage the proliferation and anabolism of articular chondrocytes [53][55][56]. Factors produced by MSCs have been shown to affect synovium and articular chondrocytes, which control anabolic and catabolic processes [57][58] and increase the synthesis of molecular mediators of inflammation and chondrogenesis [53][59][60]. Moreover, by downregulating the neuralgia pathway, reduced inflammation may alleviate neuropathic pain.References
- Lim, J.; Kim, J.; Cheon, S. A Deep Neural Network-Based Method for Early Detection of Osteoarthritis Using Statistical Data. Int. J. Environ. Res. Public Health 2019, 16, 1281.
- Glyn-Jones, S.; Palmer, A.; Agricola, R.; Price, A.; Vincent, T.; Weinans, H.; Carr, A. Osteoarthritis. Lancet 2015, 386, 376–387.
- Mandl, L. Osteoarthritis year in review 2018: Clinical. Osteoarthr. Cartil. 2019, 27, 359–364.
- Puig-Junoy, J.; Zamora, A.R. Socio-economic costs of osteoarthritis: A systematic review of cost-of-illness studies. Semin. Arthritis Rheum. 2015, 44, 531–541.
- Iban, M.A.R.; Benavides, J.; Forero, J.P.; Bittelman, S.; Martinez, R.; Mite, M.A.; Heredia, J.D.; Ulloa, S.; Ferrand, M.M.L. Use of strong opioids for chronic pain in osteoarthritis: An insight into the Latin American reality. Expert Rev. Clin. Pharmacol. 2017, 11, 47–59.
- Mahendira, L.; Jones, C.; Papachristos, A.; Waddell, J.; Rubin, L. Comparative clinical and cost analysis between surgical and non-surgical intervention for knee osteoarthritis. Int. Orthop. 2019, 44, 77–83.
- White, R.H.; Romano, P.; Zhou, H.; Rodrigo, J.; Bargar, W. Incidence and Time Course of Thromboembolic Outcomes Following Total Hip or Knee Arthroplasty. Arch. Intern. Med. 1998, 158, 1525–1531.
- Kurtz, S.M.; Lau, E.; Watson, H.; Schmier, J.K.; Parvizi, J. Economic Burden of Periprosthetic Joint Infection in the United States. J. Arthroplast. 2012, 27, 61–65.e1.
- Roemer, F.W.; Guermazi, A.; Javaid, M.K.; Lynch, J.A.; Niu, J.; Zhang, Y.; Felson, D.T.; Lewis, C.E.; Torner, J.; Nevitt, M.C. Change in MRI-detected subchondral bone marrow lesions is associated with cartilage loss: The MOST Study. A longitudinal multicentre study of knee osteoarthritis. Ann. Rheum. Dis. 2008, 68, 1461–1465.
- Hayes, A.J.; MacPherson, S.; Morrison, H.; Dowthwaite, G.; Archer, C.W. The development of articular cartilage: Evidence for an appositional growth mechanism. Anat. Embryol. 2001, 203, 469–479.
- Pritzker, K.P.H.; Gay, S.; Jimenez, S.A.; Ostergaard, K.; Pelletier, J.-P.; Revell, P.A.; Salter, D.; van den Berg, W.B. Osteoarthritis cartilage histopathology: Grading and staging. Osteoarthr. Cartil. 2006, 14, 13–29.
- Dowthwaite, G.P.; Bishop, J.C.; Redman, S.N.; Khan, I.M.; Rooney, P.; Evans, D.J.R.; Haughton, L.; Bayram, Z.; Boyer, S.; Thomson, B.; et al. The surface of articular cartilage contains a progenitor cell population. J. Cell Sci. 2004, 117, 889–897.
- Hunziker, E.; Kapfinger, E.; Geiss, J. The structural architecture of adult mammalian articular cartilage evolves by a synchronized process of tissue resorption and neoformation during postnatal development. Osteoarthr. Cartil. 2007, 15, 403–413.
- Diekman, B.; Wu, C.-L.; Louer, C.; Furman, B.D.; Huebner, J.L.; Kraus, V.B.; Olson, S.A.; Guilak, F. Intra-articular Delivery of Purified Mesenchymal Stem Cells from C57BL/6 or MRL/MpJ Superhealer Mice Prevents Posttraumatic Arthritis. Cell Transplant. 2013, 22, 1395–1408.
- Frisbie, D.D.; Kisiday, J.D.; Kawcak, C.E.; Werpy, N.M.; McIlwraith, C.W. Evaluation of adipose-derived stromal vascular fraction or bone marrow-derived mesenchymal stem cells for treatment of osteoarthritis. J. Orthop. Res. 2009, 27, 1675–1680.
- Huh, J.-E.; Baek, Y.-H.; Ryu, S.-R.; Lee, J.-D.; Choi, D.-Y.; Park, D.-S. Efficacy and mechanism of action of KHBJ-9B, a new herbal medicine, and its major compound triterpenoids in human cartilage culture and in a rabbit model of collagenase-induced osteoarthritis. Int. Immunopharmacol. 2009, 9, 230–240.
- Cosenza, S.; Ruiz, M.; Maumus, M.; Jorgensen, C.; Noël, D. Pathogenic or Therapeutic Extracellular Vesicles in Rheumatic Diseases: Role of Mesenchymal Stem Cell-Derived Vesicles. Int. J. Mol. Sci. 2017, 18, 889.
- Maumus, M.; Manferdini, C.; Toupet, K.; Peyrafitte, J.-A.; Ferreira, R.; Facchini, A.; Gabusi, E.; Bourin, P.; Jorgensen, C.; Lisignoli, G.; et al. Adipose mesenchymal stem cells protect chondrocytes from degeneration associated with osteoarthritis. Stem Cell Res. 2013, 11, 834–844.
- Liu, D.; Kou, X.; Chen, C.; Liu, S.; Liu, Y.; Yu, W.; Yu, T.; Yang, R.; Wang, R.; Zhou, Y.; et al. Circulating apoptotic bodies maintain mesenchymal stem cell homeostasis and ameliorate osteopenia via transferring multiple cellular factors. Cell Res. 2018, 28, 918–933.
- Dalby, M.J.; García, A.J.; Salmeron-Sanchez, M. Receptor control in mesenchymal stem cell engineering. Nat. Rev. Mater. 2018, 3, 17091.
- Battula, V.L.; Cabreira, M.D.G.; Wang, Z.; Ma, W.; Benito, J.; Ruvolo, P.P.; Davis, R.E.; Konopleva, M.; Andreeff, M. Connective Tissue Growth Factor (CTGF) Is Essential for Self Renewal and Proliferation of Mesenchymal Stromal Cells (MSCs) and Affects Leukemia-Stromal Interactions. Blood 2010, 116, 3845.
- Friedenstein, A.; Gorskaja, J.; Kulagina, N. Fibroblast precursors in normal and irradiated mouse hematopoietic organs. Exp. Hematol. 1976, 4, 267–274.
- Tuan, R.S.; Boland, G.; Tuli, R. Adult mesenchymal stem cells and cell-based tissue engineering. Thromb. Haemost. 2003, 5, 32–45.
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.C.; Krause, D.S.; Deans, R.J.; Keating, A.; Prockop, D.J.; Horwitz, E.M. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317.
- Jin, H.J.; Bae, Y.K.; Kim, M.; Kwon, S.-J.; Jeon, H.B.; Choi, S.J.; Kim, S.W.; Yang, Y.S.; Oh, W.; Chang, J.W. Comparative Analysis of Human Mesenchymal Stem Cells from Bone Marrow, Adipose Tissue, and Umbilical Cord Blood as Sources of Cell Therapy. Int. J. Mol. Sci. 2013, 14, 17986–18001.
- Kern, S.; Eichler, H.; Stoeve, J.; Klüter, H.; Bieback, K. Comparative Analysis of Mesenchymal Stem Cells from Bone Marrow, Umbilical Cord Blood, or Adipose Tissue. Stem Cells 2006, 24, 1294–1301.
- Fa, X.; Lixia, W.; Hou, J.; Zhang, R.; Wang, H.; Yang, C. Biological characteristics of human bone marrow mesenchymal stem cell cultured in vitro. J. Huazhong Univ. Sci. Technol. Med. Sci. 2005, 25, 307–309.
- Kim, Y.J.; Kim, H.K.; Cho, H.H.; Bae, Y.C.; Suh, K.T.; Jung, J.S. Direct Comparison of Human Mesenchymal Stem Cells Derived from Adipose Tissues and Bone Marrow in Mediating Neovascularization in Response to Vascular Ischemia. Cell. Physiol. Biochem. 2007, 20, 867–876.
- Panepucci, R.; Siufi, J.; Silva, W., Jr.; Proto-Siquiera, R.; Neder, L.; Orellana, M.; Rocha, V.; Covas, D.; Zago, M. Comparison of gene expression of umbilical cord vein and bone marrow-derived mesenchymal stem cells. Stem Cells 2004, 22, 1263–1278.
- Li, C.Y.; Wu, X.-Y.; Tong, J.-B.; Yang, X.-X.; Zhao, J.-L.; Zheng, Q.-F.; Zhao, G.-B.; Ma, Z.-J. Comparative analysis of human mesenchymal stem cells from bone marrow and adipose tissue under xeno-free conditions for cell therapy. Stem Cell Res. Ther. 2015, 6, 55.
- Winter, A.; Breit, S.; Parsch, D.; Benz, K.; Steck, E.; Hauner, H.; Weber, R.M.; Ewerbeck, V.; Richter, W. Cartilage-like gene expression in differentiated human stem cell spheroids: A comparison of bone marrow-derived and adipose tissue-derived stromal cells. Arthritis Rheum. 2003, 48, 418–429.
- Shao, K.; Koch, C.; Gupta, M.K.; Lin, Q.; Lenz, M.; Laufs, S.; Denecke, B.; Schmidt, M.; Linke, M.; Hennies, H.C.; et al. Induced Pluripotent Mesenchymal Stromal Cell Clones Retain Donor-derived Differences in DNA Methylation Profiles. Mol. Ther. 2013, 21, 240–250.
- Pittenger, M.F.; Mackay, A.M.; Beck, S.C.; Jaiswal, R.K.; Douglas, R.; Mosca, J.D.; Moorman, M.A.; Simonetti, D.W.; Craig, S.; Marshak, D.R. Multilineage Potential of Adult Human Mesenchymal Stem Cells. Science 1999, 284, 143–147.
- Aust, L.; Devlin, B.; Foster, S.; Halvorsen, Y.; Hicok, K.; du Laney, T.; Sen, A.; Willingmyre, G.; Gimble, J. Yield of human adipose-derived adult stem cells from liposuction aspirates. Cytotherapy 2004, 6, 7–14.
- Collins, K.; Lenz, K.; Pollitt, E.; Ferguson, D.; Hutson, I.; Springer, L.; Oestreich, A.; Tang, R.; Choi, Y.; Meyer, G.; et al. Adipose tissue is a critical regulator of osteoarthritis. Proc. Natl. Acad. Sci. USA 2021, 118, e2021096118.
- Sun, Y.-Q.; Deng, M.-X.; He, J.; Zeng, Q.-X.; Wen, W.; Wong, D.S.H.; Tse, H.-F.; Xu, G.; Lian, Q.; Shi, J.; et al. Human Pluripotent Stem Cell-Derived Mesenchymal Stem Cells Prevent Allergic Airway Inflammation in Mice. Stem Cells 2012, 30, 2692–2699.
- Stolzing, A.; Jones, E.; McGonagle, D.; Scutt, A. Age-related changes in human bone marrow-derived mesenchymal stem cells: Consequences for cell therapies. Mech. Ageing Dev. 2008, 129, 163–173.
- Diederichs, S.; Tuan, R.S. Functional Comparison of Human-Induced Pluripotent Stem Cell-Derived Mesenchymal Cells and Bone Marrow-Derived Mesenchymal Stromal Cells from the Same Donor. Stem Cells Dev. 2014, 23, 1594–1610.
- Le Blanc, K.; Frassoni, F.; Ball, L.; Locatelli, F.; Roelofs, H.; Lewis, I.; Lanino, E.; Sundberg, B.; Bernardo, M.; Remberger, M.; et al. Mesenchymal stem cells for treatment of steroid-resistant, severe, acute graft-versus-host disease: A phase II study. Lancet 2008, 371, 1579–1586.
- Connick, P.; Kolappan, M.; Crawley, C.; Webber, D.; Patani, R.; Michell, A.; Du, M.; Luan, S.; Altmann, D.; Thompson, A.; et al. Autologous mesenchymal stem cells for the treatment of secondary progressive multiple sclerosis: An open-label phase 2a proof-of-concept study. Lancet Neurol. 2012, 11, 150–156.
- English, K. Mechanisms of mesenchymal stromal cell immunomodulation. Immunol. Cell Biol. 2012, 91, 19–26.
- Hoogduijn, M.J. Are mesenchymal stromal cells immune cells? Arthritis Res. Ther. 2015, 17, 88.
- Van den Bosch, M.; van Lent, P.; van der Kraan, P. Identifying effector molecules, cells, and cytokines of innate immunity in OA. Osteoarthr. Cartil. 2020, 28, 532–543.
- Spaggiari, G.M.; Abdelrazik, H.; Becchetti, F.; Moretta, L. MSCs inhibit monocyte-derived DC maturation and function by selectively interfering with the generation of immature DCs: Central role of MSC-derived prostaglandin E2. Blood 2009, 113, 6576–6583.
- Spaggiari, G.M.; Capobianco, A.; Becchetti, S.; Mingari, M.C.; Moretta, L. Mesenchymal stem cell-natural killer cell interactions: Evidence that activated NK cells are capable of killing MSCs, whereas MSCs can inhibit IL-2-induced NK-cell proliferation. Blood 2006, 107, 1484–1490.
- Benvenuto, F.; Ferrari, S.; Gerdoni, E.; Gualandi, F.; Frassoni, F.; Pistoia, V.; Mancardi, G.; Uccelli, A. Human Mesenchymal Stem Cells Promote Survival of T Cells in a Quiescent State. Stem Cells 2007, 25, 1753–1760.
- Németh, K.; Leelahavanichkul, A.; Yuen, P.S.T.; Mayer, B.; Parmelee, A.; Doi, K.; Robey, P.G.; Leelahavanichkul, K.; Koller, B.H.; Brown, J.M.; et al. Erratum: Corrigendum: Bone marrow stromal cells attenuate sepsis via prostaglandin E2–dependent reprogramming of host macrophages to increase their interleukin-10 production. Nat. Med. 2009, 15, 462.
- Fernandes, T.L.; Gomoll, A.H.; Lattermann, C.; Hernandez, A.J.; Bueno, D.F.; Amano, M.T. Macrophage: A Potential Target on Cartilage Regeneration. Front. Immunol. 2020, 11, 111.
- Harrell, C.R.; Jankovic, M.G.; Fellabaum, C.; Volarevic, A.; Djonov, V.; Arsenijevic, A.; Volarevic, V. Molecular Mechanisms Responsible for Anti-inflammatory and Immunosuppressive Effects of Mesenchymal Stem Cell-Derived Factors. Adv. Exp. Med. Biol. 2018, 1084, 187–206.
- Mokbel, A.N.; El Tookhy, O.S.; Shamaa, A.A.; Rashed, L.A.; Sabry, D.; El Sayed, A.M. Homing and reparative effect of intra-articular injection of autologus mesenchymal stem cells in osteoarthritic animal model. BMC Musculoskelet. Disord. 2011, 12, 259.
- Sato, M.; Uchida, K.; Nakajima, H.; Miyazaki, T.; Guerrero, A.R.; Watanabe, S.; Roberts, S.; Baba, H. Direct transplantation of mesenchymal stem cells into the knee joints of Hartley strain guinea pigs with spontaneous osteoarthritis. Thromb. Haemost. 2012, 14, R31.
- Mak, J.; Jablonski, C.L.; Leonard, C.A.; Dunn, J.F.; Raharjo, E.; Matyas, J.R.; Biernaskie, J.; Krawetz, R.J. Intra-articular injection of synovial mesenchymal stem cells improves cartilage repair in a mouse injury model. Sci. Rep. 2016, 6, 23076.
- Bárdos, T.; Kamath, R.V.; Mikecz, K.; Glant, T.T. Anti-Inflammatory and Chondroprotective Effect of TSG-6 (Tumor Necrosis Factor-α-Stimulated Gene-6) in Murine Models of Experimental Arthritis. Am. J. Pathol. 2001, 159, 1711–1721.
- Von Bahr, L.; Batsis, I.; Moll, G.; Hägg, M.; Szakos, A.; Sundberg, B.; Uzunel, M.; Ringden, O.; Le Blanc, K. Analysis of Tissues Following Mesenchymal Stromal Cell Therapy in Humans Indicates Limited Long-Term Engraftment and No Ectopic Tissue Formation. Stem Cells 2012, 30, 1575–1578.
- Acharya, C.; Adesida, A.; Zajac, P.; Mumme, M.; Riesle, J.; Martin, I.; Barbero, A. Enhanced chondrocyte proliferation and mesenchymal stromal cells chondrogenesis in coculture pellets mediate improved cartilage formation. J. Cell. Physiol. 2011, 227, 88–97.
- Wu, L.; Prins, H.-J.; Helder, M.N.; van Blitterswijk, C.A.; Karperien, H.B.J. Trophic Effects of Mesenchymal Stem Cells in Chondrocyte Co-Cultures are Independent of Culture Conditions and Cell Sources. Tissue Eng. Part A 2012, 18, 1542–1551.
- Saulnier, N.; Viguier, E.; Perrier-Groult, E.; Chenu, C.; Pillet-Michelland, E.; Roger, T.; Maddens, S.; Boulocher, C. Intra-articular administration of xenogeneic neonatal Mesenchymal Stromal Cells early after meniscal injury down-regulates metalloproteinase gene expression in synovium and prevents cartilage degradation in a rabbit model of osteoarthritis. Osteoarthr. Cartil. 2015, 23, 122–133.
- Desando, G.; Cavallo, C.; Sartoni, F.; Martini, L.; Parrilli, A.; Veronesi, F.; Fini, M.; Giardino, R.; Facchini, A.; Grigolo, B. Intra-articular delivery of adipose derived stromal cells attenuates osteoarthritis progression in an experimental rabbit model. Thromb. Haemost. 2013, 15, R22.
- Lee, C.H.; Cook, J.L.; Mendelson, A.; Moioli, E.K.; Yao, H.; Mao, J.J. Regeneration of the articular surface of the rabbit synovial joint by cell homing: A proof of concept study. Lancet 2010, 376, 440–448.
- Ozeki, N.; Muneta, T.; Koga, H.; Nakagawa, Y.; Mizuno, M.; Tsuji, K.; Mabuchi, Y.; Akazawa, C.; Kobayashi, E.; Matsumoto, K.; et al. Not single but periodic injections of synovial mesenchymal stem cells maintain viable cells in knees and inhibit osteoarthritis progression in rats. Osteoarthr. Cartil. 2016, 24, 1061–1070.
More
