Currently, algae arouse a growing interest in the pharmaceutical and cosmetic area due to the fact that they have a great diversity of bioactive compounds with the potential for pharmacological and nutraceutical applications. Due to lifestyle modifications brought on by rapid urbanization, diabetes mellitus, a metabolic illness, is the third largest cause of death globally. The hunt for an efficient natural-based antidiabetic therapy is crucial to battling diabetes and the associated consequences due to the unfavorable side effects of currently available antidiabetic medications. Finding the possible advantages of algae for the control of diabetes is crucial for the creation of natural drugs.
Many of algae’s metabolic processes produce bioactive secondary metabolites, which give algae their diverse chemical and biological features. Numerous studies have demonstrated the antioxidant and antidiabetic benefits of algae, mostly by blocking carbohydrate hydrolyzing enzyme activity, such as -amylase and -glucosidase. Additionally, bioactive components from algae can lessen diabetic symptoms in vivo. Therefore, the current review concentrates on the role of various secondary bioactive substances found naturally in algae and their potential as antioxidants and antidiabetic materials, as well as the urgent need to apply these substances in the pharmaceutical industry
- diabetes
- antioxidant
- antihyperglycemi
1. Introduction
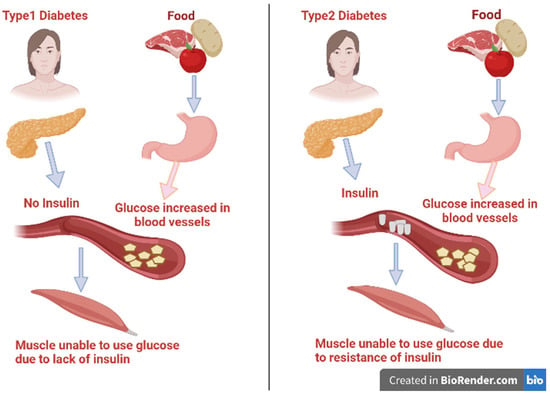
2. Factors That Contribute to Diabetes and Its Complications
The majority of diabetes types across the world may be correlated to modern diets, sedentary lifestyles, and obesity. The mortality associated with diabetes is mainly a result of the augmented danger of several complications of this disease. Even though diabetes is primarily defined by chronic hyperglycemia, many diabetic patients, particularly those with type 2, have elevated blood pressure (hypertension), chronic high levels of insulin (hyperinsulinemia), and abnormal levels of cholesterol, triglycerides, and/or other blood lipids (hyperlipidemia). In addition, lipoprotein abnormalities are some of the most prevalent problems associated with type 2 diabetes [11]. These complications are strictly related to the disease disorders, as well as to the procedures used to diagnose and treat them (Figure 2).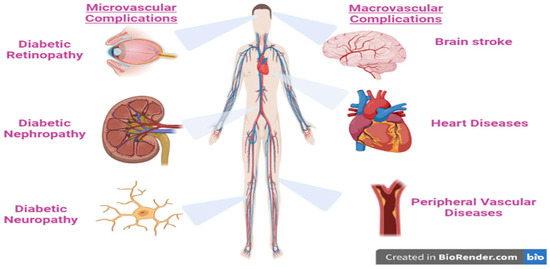
3. Oxidative Stress Linked to Diabetes
Biochemical processes in the body may produce intermediate products called reactive oxygen species (ROS). Among ROS, harmful free radicals have one or more unpaired electrons that make them very reactive with other molecules. Excess ROS may lead to an imbalance in its metabolism and in the body’s ability to detoxify or counteract the harmful oxidant effects of the free radicals [12,13][12][13]. This condition is known as oxidative stress, which depends on the balance between ROS production and antioxidant defenses [14]. Oxidative stress is thus the result of the imbalance between the formation and neutralization of reactive oxygen and nitrogen species [15,16][15][16]. Electron transfers to O2 are catalyzed by oxidase enzymes to produce chemical energy or oxidation of substrates. These enzymes are potential sources of reduced Cu2+ derivatives in biological settings; they also produce O2•− during catalysis [17,18,19][17][18][19]. The mitochondrial electron transport chain reduces O2 to O2•− [20,21][20][21]. Dismutase enzymes reduce O2•− radicals to form hydrogen peroxide (H2O2) and/or further react to form the hydroxyl secondary radical (•−OH) as another type of the ROS [17,19][17][19]. Although the cause-effect relationship remains unsure, there appears to be a strong correlation between mitochondrial dysfunction and chronic metabolic diseases, such as obesity [17] and diabetes mellitus (Figure 3) [22]. This relationship results in oxidative damage to cellular components in the form of lipid peroxidation, protein denaturation or DNA conjugation, and finally, cell death [23]. As a result of the aforementioned reasons, oxidative stress has been related to many diseases, such as cancer, post-ischemic and neural degradation, Parkinson’s and Alzheimer’s diseases, acquired immune deficiency syndrome (AIDS), aging, and cardiovascular diseases [24].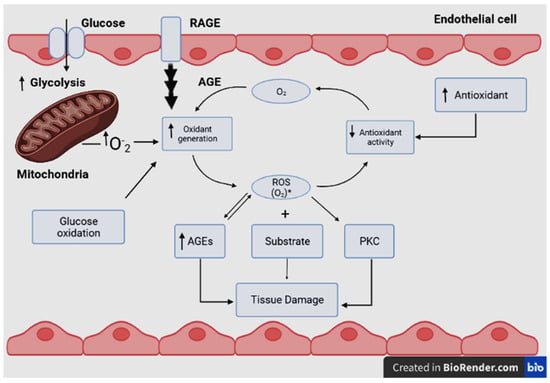
References
- International Diabetes Federation (IDF). IDF Diabetes Atlas. 2022. Available online: https://www.diabetesatlas.org/ (accessed on 31 January 2023).
- WHO. W.H.O. Diabetes. 2022. Available online: https://www.who.int/news-room/fact-sheets/detail/diabetes (accessed on 31 January 2023).
- Ritu, M.; Nandini, J. Nutritional composition of Stevia rebaudiana, a sweet herb, and its hypoglycaemic and hypolipidaemic effect on patients with non-insulin dependent diabetes mellitus. J. Sci. Food Agric. 2016, 96, 4231–4234.
- Akpaso, M.I.; Igiri, A.O.; Odey, P.A. A comparative study on the effect of combined methanolic leaf extracts of Vernonia amygdalina and Gongronema latifolium and metformin on the pancreatic beta cells of streptozocin induced diabetic wistar rats. Asian J. Pharm. Nurs. Med. Sci. 2017, 5, 42–48.
- American Diabetes Association. Classification and diagnosis of diabetes: Standards of medical care in diabetes. Diabetes Care 2021, 44, S15–S33.
- Armstrong, C. Standards of Medical Care for Patients with Diabetes; American Family Physician: Leawood, KS, USA, 2006; Volume 74.
- Fox, I.S. Human Physiology, 8th ed.; McGraw-Hill: New York, NY, USA, 2004.
- Vaidyanathan, K.; Vasudevan, D.M.; Sreekumari, S. Regulation of blood glucose, insulin and diabetes mellitus. In Textbook of Biochemistry for Medical Students; Jaypee Brothers Medical Publishers: New Delhi, India, 2011; pp. 285–290.
- Xu, L.; Li, Y.; Dai, Y.; Peng, J. Natural products for the treatment of type 2 diabetes mellitus: Pharmacology and mechanisms. Pharmacol. Res. 2018, 130, 451–465.
- Franks, P.W.; McCarthy, M.I. Exposing the exposures responsible for type 2 diabetes and obesity. Science 2016, 354, 69–73.
- Sørensen, L.E.; Jeppesen, P.B.; Christiansen, C.B.; Hermansen, K.; Gregersen, S. Nordic seaweed and diabetes prevention: Exploratory studies in KK-Ay mice. Nutrients 2019, 11, 1435.
- Finkel, T.; Holbrook, N.J. Oxidants, oxidative stress and the biology of ageing. Nature 2000, 408, 239–247.
- Tiwari, A. Imbalance in antioxidant defense and human disease: Multiple approach of natural antioxidant therapy. J. Curr. Sci. 2001, 81, 1179–1187.
- Pirian, K.; Moein, S.; Sohrabipour, J.; Rabiei, R.; Blomster, J. Antidiabetic and antioxidant activities of brown and red macroalgae from the Persian Gulf. J. Appl. Phycol. 2017, 29, 3151–3159.
- Poljsak, B.; Fink, R. The protective role of antioxidants in the defence against ROS/RNS-mediated environmental pollution. Oxidative Med. Cell. Longev. 2014, 2014, 22–28.
- Shin, C.S.; Moon, B.S.; Park, K.S.; Kim, S.Y.; Park, S.J.; Chung, M.H.; Lee, H.K. Serum 8-hydroxy-guanine levels are increased in diabetic patients. Diabetes Care 2001, 24, 733–737.
- Alfadda, A.A.; Sallam, R.M. Reactive oxygen species in health and disease. J. Biomed. Biotechnol. 2012, 2012, 14–19.
- Lai, M.Y.; Fung, P.L.; Xiaoqiang, Y.; Zhen-Yu, C.; Yu, H. Reactive oxygen species in vascular wall. Cardiovasc. Hematol. Disord. -Drug Targets 2006, 6, 1–19.
- Styskal, J.; Van Remmen, H.; Richardson, A.; Salmon, A.B. Oxidative stress and diabetes: What can we learn about insulin resistance from antioxidant mutant mouse models? Free. Radic. Biol. Med. 2012, 52, 46–58.
- Bajaj, S.; Khan, A. Antioxidants and diabetes. Indian J. Endocrinol. Metab. 2012, 16, S267–S271.
- Desco, M.-C.; Asensi, M.; Márquez, R.; Martínez-Valls, J.; Vento, M.; Pallardó, F.V.; Sastre, J.; Viña, J. Xanthine oxidase is involved in free radical production in type 1 diabetes. Diabetes 2002, 51, 1118–1124.
- Maiese, K. New insights for oxidative stress and diabetes mellitus. Oxidative Med. Cell. Longev. 2015, 2015, 875961.
- Xin, Y.; Yuan, B.; Yu, B.; Wang, Y.; Wu, J.; Zhou, W.; Qiu, Z. Tet1-mediated DNA demethylation regulates neuronal cell death induced by oxidative stress. Sci. Rep. 2015, 5, 7645–7650.
- Maiese, K. MTOR: Driving apoptosis and autophagy for neurocardiac complications of diabetes mellitus. World J. Diabetes 2015, 6, 217–224.
