Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Camila Xu and Version 1 by Valeria Tiranti.
Mitochondria are cytoplasmic double-membrane organelles defined as eukaryotic cells’ powerhouses due to their involvement in the cellular bioenergetics. In particular, mitochondrial synthesis of adenosine triphosphate (ATP) is associated with the functionality of aerobic oxidative phosphorylation (OXPHOS). In addition to energy production, mitochondria play many critical roles in cellular function and signalling, including fatty acid biosynthesis, calcium homeostasis, reactive oxygen species production, cell survival, proliferation, apoptosis, autophagy, stem cell differentiation, and regulation of the immune response.
- mitochondria
- mitochondrial diseases
- mitochondrial medicine
1. Introduction
Mitochondria are cytoplasmic double-membrane organelles defined as eukaryotic cells’ powerhouses due to their involvement in the cellular bioenergetics. In particular, mitochondrial synthesis of adenosine triphosphate (ATP) is associated with the functionality of aerobic oxidative phosphorylation (OXPHOS) [1]. In addition to energy production, mitochondria play many critical roles in cellular function and signalling, including fatty acid biosynthesis, calcium homeostasis, reactive oxygen species production, cell survival, proliferation, apoptosis, autophagy, stem cell differentiation, and regulation of the immune response [2,3][2][3]. The complex functionality of mitochondria in cell biology is supported by their morphological and ultrastructural plasticity. Under physiological conditions, they form a dynamic and highly interconnected tubular network [4].
Due to their endosymbiotic origin [5], mitochondria have their own circular double-stranded DNA (mtDNA) that is transcribed and replicated following principles that are different from those regulating nuclear DNA (nDNA) [6]. Furthermore, unlike nDNA, mtDNA is characterised by maternal uniparental inheritance [7].
Human mtDNA is 16.6 kb long and comprises 37 genes, of which only 13 code for subunits of complexes I, III, IV, and ATP synthase. The remaining genes code for 2 ribosomal RNAs and 22 transfer RNAs, both of which are necessary for the protein synthesis machinery of mitochondria [8]. Nuclear genes encode the remaining proteins involved in OXPHOS and other enzymes required for mtDNA replication, repair, transcription, and translation [9]. Each individual mammalian cell contains hundreds or thousands of mitochondria, which in turn each contain between 1 and 15 molecules of mtDNA [10]. The ratio of mtDNA molecules/cell varies depending on cell and tissue types [11]. The mtDNA molecules—about 5 μm long—are highly compacted by non-histone proteins in aggregates (or spherical bodies) called nucleoids within the mitochondrial matrix [12]. In addition, mtDNA is characterised by the absence of protective histone proteins and a high exposure to oxygen free radicals (ROS) due to its proximity to OXPHOS sites [13]. The mtDNA repair processes, compared to nDNA, are less diversified, being primarily mediated by the base excision repair pathway [14]. Despite the high degree of conservation in animals, it is estimated that the mitochondrial genome evolves 10–20 times faster than nDNA, exhibiting heterogeneity in the nucleotide sequence [15]. The less efficient damage repair system, combined with continuous and random replication of mtDNA molecules, is probably the reason for the high mutation rate of the mitochondrial genome [16].
2. Treatments for Mitochondrial Diseases
Due to recent developments in understanding the pathogenic mechanisms underlying mitochondrial disorders, various therapeutic approaches of “mitochondrial medicine” aiming to rescue wild-type phenotypes and prevent progression have been developed for MDs [25][17]. However, the development of efficient treatments has been severely hampered by the enormously variegate phenotype–genotype correlation that is typical of MDs. Since most of them lack an approved cure and available therapies are geared toward alleviating symptoms [26][18], there is urgent need to develop novel generalised or personalised approaches. To achieve this, different therapeutic strategies have been developed, which can be classified into two main groups: (1) pharmacological and metabolic approaches and (2) molecular approaches (Figure 1).
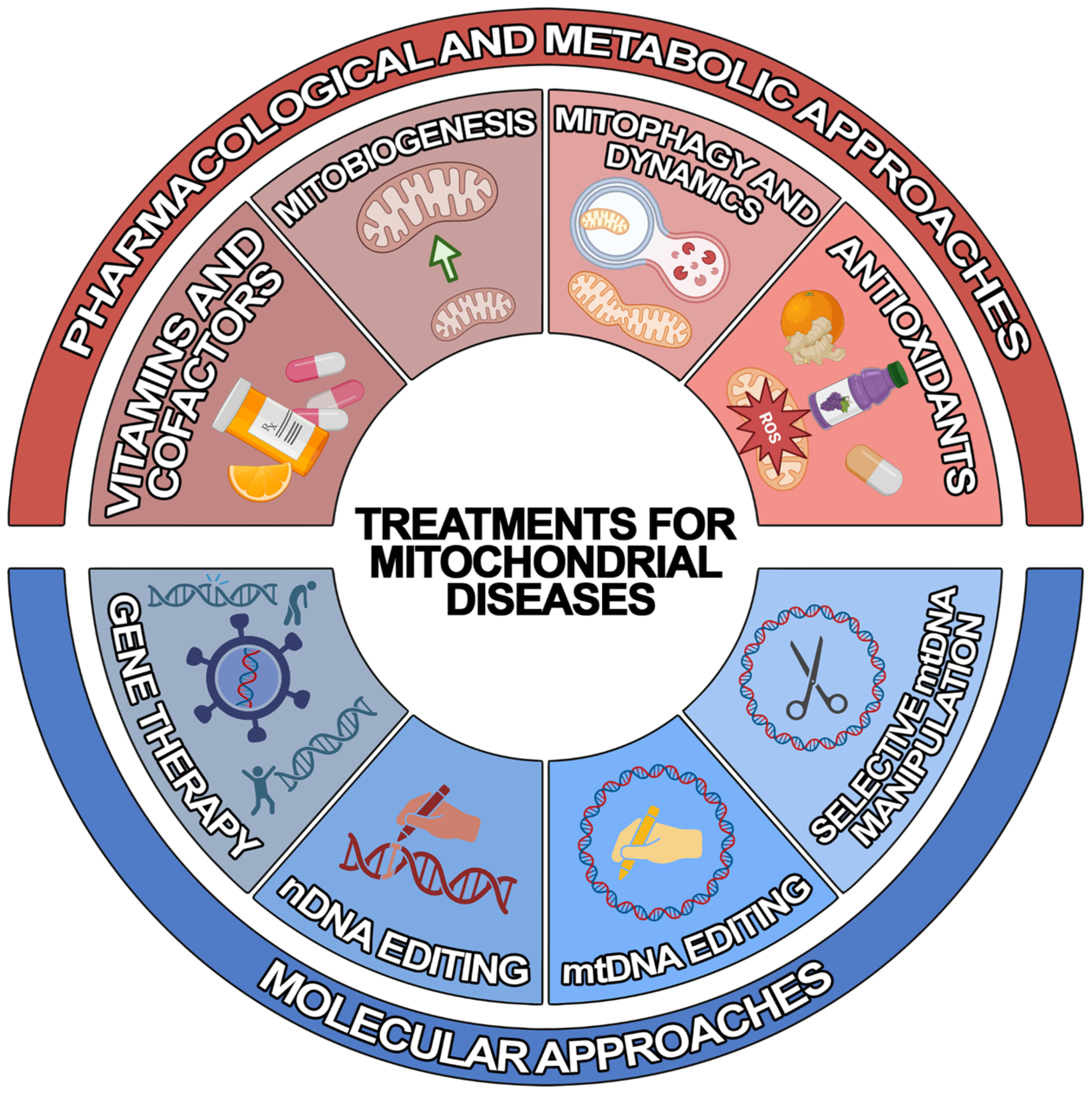
Figure 1. Schematic representation of the treatments for mitochondrial diseases. Illustrated are the current treatment strategies for mitochondrial diseases, which can be classified into pharmacological and metabolic (red) and molecular (blue) approaches.
2.1. Pharmacological and Metabolic Approaches
Most of the pharmacological agents aim to reduce symptoms, particularly exercise intolerance and fatigue, and slow down the progression of the disease. Traditional treatments attempt to improve the function of the electron transport chain (ETC). In particular, several vitamins and cofactors are used to: (1) increase electron flux through the respiratory chain (CoQ10, riboflavin); (2) act as antioxidants (idebenone, alpha-lipoic acid, vitamins C and E), and/or increase ETC substrate availability (dichloroacetate and thiamine); and (3) feed the mitochondria (L-carnitine) [27][19]. However, in recent years, emerging experimental strategies have been proposed which are focused on: (1) stimulating mitochondrial biogenesis, (2) modulating mitophagy and mitochondrial dynamics, (3) bypassing OXPHOS deficits, (4) using mitochondrial replacement therapy (MRT), and (5) treating chronic hypoxia [24][20].
2.2. Molecular Approaches
Molecular strategies aim to override or correct genetic alteration to mitigate the consequent mitochondrial deficits. The nDNA-associated MDs can benefit from genetic approaches based on the targeted re-expression of the wild-type gene and from the genome editing techniques based on clustered regularly interspaced palindromic repeat/Cas9 (CRISPR/Cas9) technology, zinc finger nucleases (ZFNs), or transcription-activator-like effector nucleases (TALENs) [28][21]. However, mtDNA-related MDs must be treated with alternative methods. Indeed, unlike nDNA, not only is mtDNA not easily accessible due to the mitochondrial double membrane structure, but it is also present in multiple copies within the organelle [29][22]. Thus, the delivery of nucleic acids to mitochondria is challenging.
Three main experimental approaches have been developed to overcome this issue: (1) allotopic gene therapy, (2) mtDNA-selective endonuclease-based strategy, and (3) mitochondrial genome editing [28][21]. The first approach aims to re-express the mitochondrial gene at the nuclear level—followed by the import of the gene-encoded protein into the organelle, where it is supposed to function properly—by adding a mitochondrial-targeting sequence (MTS) at the N-terminus. Although clinical trials began in LHON patients, several challenges must be overcome to increase the successful application of this treatment [30][23]. The second approach exploits the observation that a break in the DNA double-strand leads to rapid degradation of the mtDNA molecule. Consequently, the selective elimination of mutated mtDNA via specific endonucleases might promote a heteroplasmic shift towards wild-type mtDNA copies within the organelle [31,32,33][24][25][26]. However, the strategy can only allow the potential treatment of heteroplasmic conditions. The third approach is based on two recent mitochondrial base editor technologies that are potentially able to correct homoplasmic single-nucleotide changes [33][26]. On the one hand, the DddA-derived cytosine base editor (DdCBE), which catalyses the conversions of C-to-T (or G-to-A) in human mtDNA [34][27]. On the other hand, transcription-activator-like effector (TALE)-linked deaminases (TALEDs) introduce targeted A-to-G editing in human mtDNA [35][28]. However, these approaches are still under development, and further studies are needed to verify their efficiency and safety in vivo.
3. Intercellular Transfer of Mitochondria
Cell-to-cell mitochondria transfer is considered a type of intercellular interaction that occurs physiologically in organisms [36][29]. Researchers' knowledge of mitochondrial cell communication was enhanced by two experiments performed in the early 2000s: (1) Rustom et al. discovered cell-to-cell migration of organelles via nanotubular structures [37][30]; (2) Spees et al. demonstrated the natural transfer of wild-type mitochondria from mesenchymal stem cells (MSCs) into parenchymal cells displaying mitochondrial dysfunction [38][31]. Since then, a considerable body of data suggests that mitochondria and their components can be actively released into the extracellular space and transferred between cells under both healthy and pathological conditions [39,40][32][33]. Accordingly, knowledge of mitochondrial function is shifting from a microscale (single cell) to a macroscale cellular perspective, drawing attention to communication between cells through exchange of mitochondria.
The biological role of this interaction is still controversial and depends on (among other things) the metabolic state of the cells. In fact, mitochondria released from a donor cell might be degraded or integrated by a recipient cell [41][34]. According to some studies, transmitophagy processes are responsible for exogenous mitochondria degradation [42][35]: (1) Davis et al. show that the mitopsosis [43][36] of dysfunctional mitochondria from optic nerve neurons occurs in adjacent glial cells [44][37]; (2) Phinney et al. suggest the selective discharge of depolarized mitochondria from MSCs to macrophages. Importantly, the authors unexpectedly noted that the recipient macrophages integrate exogenous organelles in order to enhance their own bioenergetics [45][38].
In line with this, other research papers point out that damaged cells can also take up exogenous functional mitochondria from a donor cell and integrate them into their endogenous mitochondrial networks to improve biological processes [46][39]. This seems to be the case for: (1) astrocytes transferring mitochondria to neurons during focal cerebral ischaemia [47][40], (2) endothelial progenitor cells protecting the brain endothelium during stroke and brain injury [48][41], and (3) bone-marrow-derived mesenchymal stromal cells (BM-MSCs) providing mitochondria to diverse cell types in various conditions [49,50][42][43]. In short, intercellular communication between recipient and donor cells can generate what Liu et al. refer to as the “find me” and “save me” connection signal, a specific mechanism that occurs in damaged tissues [51][44].
In addition to the well-characterized apoptotic processes, other biochemical events have been observed in mitochondria under stress conditions. It could be that mtDNA escapes into the cytosol via the Bak/Bax permeabilization pathway, where it is identified by various pattern recognition receptors (PRRs) as a “foreign” damage-associated molecular pattern (DAMP). This process leads to the activation of the cGAS-STING1-TBK1-IRF3 cascade, which stimulates the expression of type I interferon and proinflammatory cytokine (IL-1, IL,4, IL-10 and TNFα) genes [52][45]. The mtDNA can also bind endosomal toll-like receptor 9 (TLR9) to boost a cellular proinflammatory response [53][46]. Given the cellular connections, one could boldly hypothesize that the proinflammatory response may also trigger a chain reaction involving the neighbouring cells. According to this view, a cell might both help the dysfunctional neighbouring cell to eliminate dysfunctional organelles and restore the pool of healthy mitochondria [54,55][47][48]. In view of the above, cell-to-cell mitochondrial transfer seems to be essential for the maintenance and restoration of the organism’s homeostasis [56][49].
3.1. Mechanisms of Mitochondria Intercellular Transfer
The molecular mechanisms responsible for mitochondrial transfer are far from being clearly and completely understood. The best-known molecular mechanisms for intercellular transfer of mitochondria are: (1) tunnelling nanotubes (TNTs) and (2) extracellular vesicles (EVs) (Figure 2).
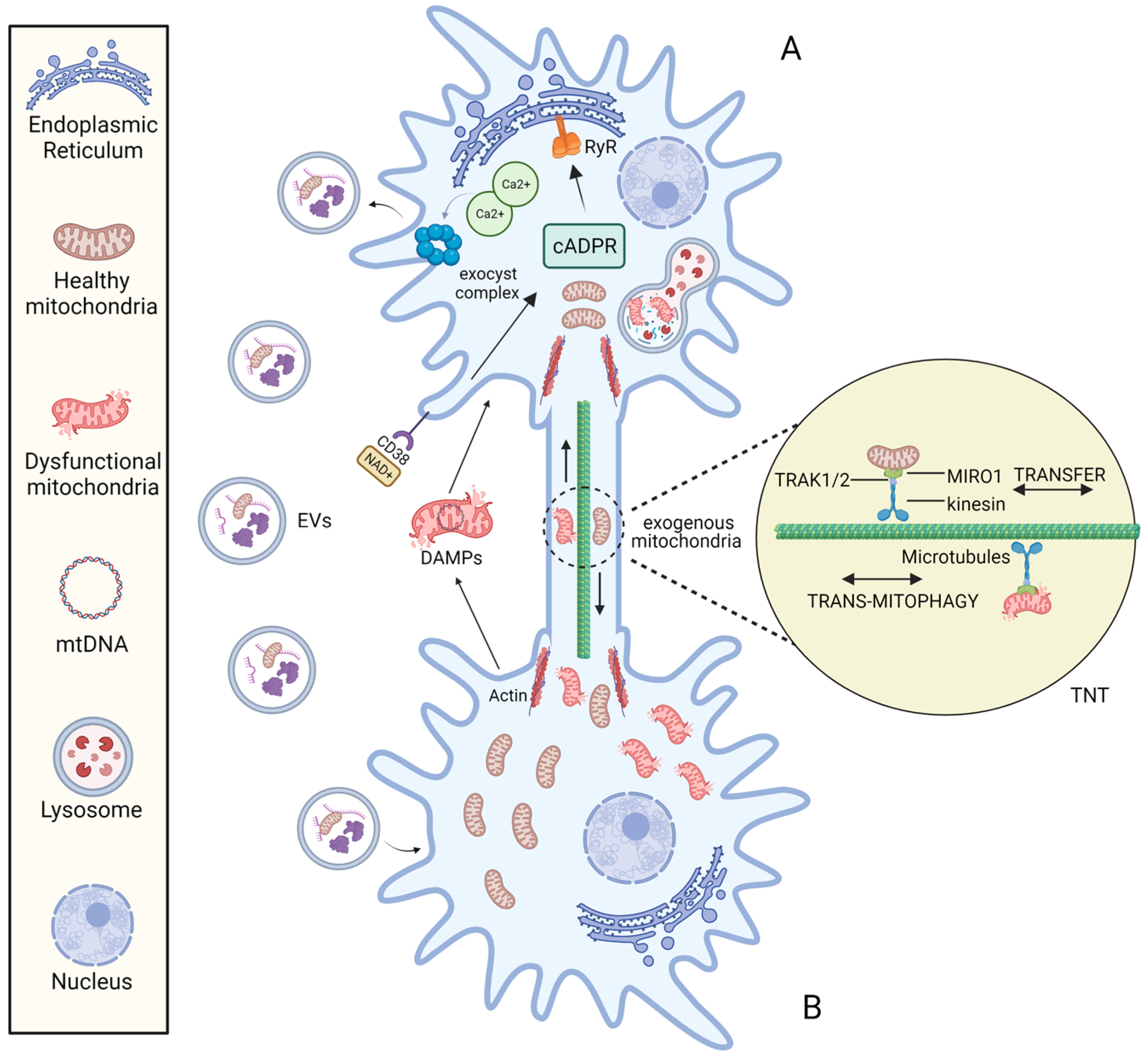
Figure 2. The mechanisms of intercellular mitochondrial transfer. Under the stimulation of stress conditions, mitochondria can be transported bidirectionally between cell A and B via the TNT structure and mitochondria-containing EVs. The formation of TNT is driven by the actin, and mitochondrial transfer between TNT-linked cells is regulated by Miro1. EV endocytosis is mediated by the NAD+/CD38/cADPR/Ca2+ pathway: Under stress, intracellular NAD+ increases and diffuses to the extracellular environment. CD38 then catalyses NAD+ to produce cADPR, a second messenger that acts on Ryanodine Receptor (RyR) on the Endoplasmic reticulum to induce the release of the intracellular Ca2+. Following the increase of cytoplasmic Ca2+ mediates the activation of exocyst complex, leading to the formation and release of vesicles.
3.1.1. Transfer via TNTs
In 2004, Rustom and collaborators identified TNTs as distinct structures for intercellular communication while working on PC12 rat pheochromocytoma cell culture [37][30]. TNTs are plasma membrane-derived tubular cytoplasmic extensions of 50–1500 nm in width and 5–120 in length [57,58][50][51]. These dimensions facilitate unidirectional or reciprocal movement of signalling molecules and cellular components, promoting long-distance cell-to-cell interconnection [59][52]. Different cell types have been reported to form TNTs with each other in both physiological and pathological states [60][53]. TNTs have an F-actin-based structure and rely on adaptor–motor proteins to mediate cargo transfer [61][54].
It is possible to speculate that mitochondrial transfer between TNT-linked cells shares similar mechanisms with Miro1-mediated axonal transport of mitochondria into neurons. Indeed, Miro 1 (the outer mitochondrial membrane (OMM) Rho-GTPase Ca2+-dependent protein) appears to play a central role in regulating mitochondrial mobility along the TNTs [62][55]. In neurons, Miro1 acts as a mitochondria-loaded vehicle that interacts with OMM mitofusins and the molecular motor kinesin-1 via the Milton adapter protein (TRAK1/2). In this way, mitochondria move along the tracks of parallel polarized microtubule array [63][56]. The formation of TNTs is driven by the F-actin cytoskeleton-remodelling-based projection system. Three steps are involved in this mechanism: the development of a membrane protrusion, its expansion, and its fusion with the target cell membrane [64][57]. Currently, many proteins and pathways related to nanotubes formation have been identified [60,65][53][58].
3.1.2. Transfer via EVs
Another system for cell-to-cell transfer of mitochondria is mediated by EVs. EVs were identified in 1971 by Aaronson S. et al., who discovered that the freshwater phytoflagellate Ochromonas danica produces membranous extracellular macromolecules [66][59]. Since then, a large number of reports in the last three decades, have suggested EVs’ involvement in intercellular signalling [67,68][60][61]. EVs are nano-sized circular phospholipid structures that are released into the extracellular space by almost all the cell types under both physiological and pathological conditions [69][62]. EVs are designated as biological cargoes because they carry and deliver a variety of bioactive molecules such as proteins, mRNAs, miRNAs, mtDNA, lipids (including cholesterol and cytokines), mitochondria, and their components [70,71][63][64]. They are classified in three general classes according to their nano-diameter and biogenesis: (1) exosomes (30–150 nm), which are of endocytic origin through inward budding of endosomal membrane; (2) microvesicles (150–1000 nm), which are formed by outward budding of the plasma membrane; and (3) apoptotic bodies (>1 μm), which are released by apoptotic cells [69][62].
Due to exosomes’ limited size, only microvesicles (eMVs) might contain intact mitochondria and contribute to mitochondrial intercellular transfer [72,73][65][66]. Indeed, recent papers have indicated horizontal mitochondrial transfer from donor cells to host cells through eMVs: (1) Islam et al. showed that bone-marrow-derived stromal cells (BMSC) supply alveolar epithelial cells with functional mitochondria via eMVs in a Cx43-dependent process [74][67]; (2) Phinney et al. reported that MSCs deliver eMVs with depolarized mitochondria to macrophages under pathological conditions [45][38]; (3) Hough et al. showed that airway myeloid-derived regulatory cells (MDRCs) secrete eMVs, which transfer mitochondria to T cells in a model of allergic airway disease [75][68]; (4) Davis et al. discovered that retinal ganglion cells can release mitochondria-rich EVs, which are captured by astrocytes in the optic nerve papilla [44][37]; and (5) Hayakawa et al. found that astrocytes release mitochondrial EVs that are captured by damaged neurons to support neuronal survival [76][69].
On the one hand, the eMV-mediated mitochondrial intercellular transfer mechanism is regulated by the NAD+/CD38/cADPR/Ca2+ pathway that activate the exocyst complex, leading to the formation and release of vesicles [36][29]. In addition, the loading of mitochondria into EVs may depend on optic atrophy 1 (OPA1) and sorting nexin 9 (Snx9) proteins, although the precise process is unknown [77][70].
On the other hand, anchorage on recipient cells is mediated by integrins localized on the eMVs surface [78][71]. After attachment to recipient cells, EVs can either immediately fuse with the membrane of the host cell or be internalized by a variety of mechanisms, such as phagocytosis, micropinocytosis, and clathrin-dependent endocytosis [79,80][72][73].
Overall, these finding suggest that cell-to-cell mitochondrial transfer pathway paves the way for a novel approach to mitochondrial transplantation for the treatment of different disorders.
4. Mitochondrial Transplantation (MT)
As further expansion in the field of innovative therapeutic strategies for mitochondrial disorders, two scientific evidences supported the idea of testing mitotherapy based on mitochondria delivery methods [81][74]. On one hand, according to the endosymbiotic theory, α-proteobacteria-derived mitochondria could be integrated into the host cell’s mitochondrial network [82][75]. On the other hand, evidence for cell-to-cell mitochondrial transfer suggested that mitochondria retain their bacterial ability to enter cells [83][76]. Many recent procedures of mitochondria transplantation rely on the transfer of exogenous functional mitochondria to injured cells to finally restore mitochondrial deficiency in several diseases [84][77].
4.1. In Vitro Methods for MT
Mitochondrial transplantation (MT) is an innovative technique based on the possibility of introducing healthy exogenous mitochondria into dysfunctional cells or tissues to modulate mitochondrial function [85][78]. The concept of “mitochondrial transplantation” was developed in 1982 by the pioneering study of Clark and Shay. These researchers purified mitochondria from donor human fibroblasts affected by mtDNA mutations, which induced chloramphenicol (CAP) and efrapeptin (EF) resistance (Figure 3A). Then, they transferred the organelles to antibiotics sensitive dysfunctional human recipient cells via simple coincubation method, and they were able to produce antibiotics-resistant vital mammalian cells, denominated “mitochondrial transformants” [86][79]. They also showed that antibiotics resistance could not be transferred from mouse to human cells. This mitochondrial transformation experiment led to some basic conclusions: (1) isolated mitochondria retain the ability to invade host cells through endocytosis mechanisms; (2) mtDNA from donor cells could be integrated into recipient cells, allowing them to transfer genetic material and induce functional changes; (3) integration of exogenous mitochondria from different species into the recipient mitochondrial network could potentially be limited by species barrier; and (4) cells might have variable mitochondrial uptake capacity according to their physiological state [87][80].
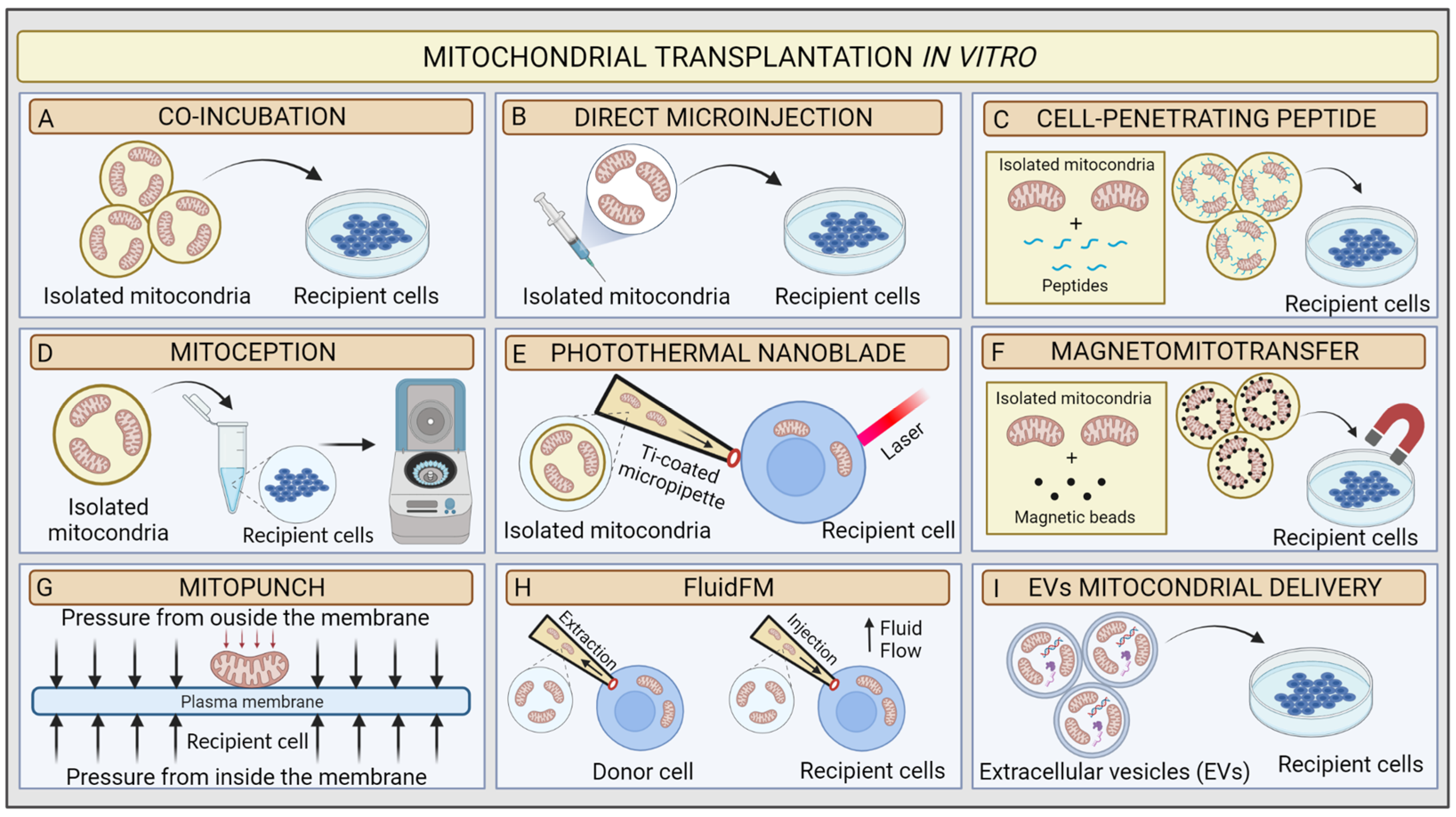
Figure 3. Schematic illustration of mitochondrial transplantation methods in vitro. (A) Coincubation, (B) direct microinjection, (C) cell-penetrating peptide mitochondrial delivery, (D) mitoception, (E) photothermal nanoblade, (F) magnetomitotransfer, (G) Mitopunch, (H) FluidFM, (I) EV mitochondrial delivery.
In 1988, King and Attardi confirmed that mitochondrial transformation is possible. They proposed the first precise MT technology for the transfer of healthy mitochondria in vitro. The researchers demonstrated that antibiotic resistance could be successfully generated in sensitive, partially mtDNA-depleted 143B human osteosarcoma cells through direct microinjection of human mitochondria isolated from CAP-resistant cell lines. They proved the retention of exogenous mtDNA copies in host cells from six to ten weeks after transplantation via the analysis of multiple mtDNA polymorphisms [88][81]. Although very accurate, the injection method was less efficient than Clark and Shay’s coincubation protocol because of the limited number of target cells in each transplantation experiment and the damage to the recipient cell. These preliminary data suggested that the uptake of a few healthy mitochondria could very rapidly repopulate a mtDNA depleted cells. In 1997, Pinkert et al. successfully microinjected mice liver mitochondria into mice zygotes, which incorporated a high rate of foreign mitochondria [89][82]. These results represented an initial step in the development of mitochondrial replacement therapy (MRT) techniques, which in MDs could prevent the transmission of mutant mtDNA to the foetus [90][83] (Figure 3B).
However, only in early 2000s, with the discovery of cell-to-cell transfer of mitochondria, did the real therapeutic potential of MT come to light. Since then, researchers’ interest triggered several MT studies on coincubation methods to investigate mitochondrial uptake in vitro [91,92][84][85]. The development of two strategies proved essential to assess the validity of MT: (1) advances in confocal microscopy technology [93][86] and (2) availability of Rho0 cells, completely deprived of mtDNA by ethidium bromide treatment [94][87], which are suitable models for proving mtDNA transfer [95][88]. Several MT approaches have been developed to improve in vitro transfer using chemical mediators. In 2017, Chang and his research group implemented Clark and Shay’s technique by using a penetrating peptide (Pep-1) conjugated with mitochondria to induce pores in the host cell’s plasma membrane to facilitate mitochondrial transfer. As a result, the conjugation of mitochondria isolated from human osteosarcoma 143B cybrid donor cells with Pep-1 promoted the mitochondrial internalization by a MELAS cybrid model (Figure 3C). The confocal microscopy analysis showed that the exogenous mitochondria colocalized with host cells mitochondria and favoured the recovery of the mitochondrial membrane potential [96][89]. In neonatal rat cardiomyocytes (NRCMs), Maeda et al. used transactivators of transcription dextran complexes (TATdextran) to increase cellular internalization of exogenous mitochondria and improve mitochondrial recipient function [97][90].
Remarkably, Caicedo et al. designed a protocol called MitoCeption based on the application of centripetal force to directly transfer mitochondria, isolated from MSCs, to human breast cancer MDA-MB-231 cells. In this protocol, mitochondria suspension is slowly added to the recipient cells in a Petri dish, which is then centrifuged and placed in the cell incubator facilitating the transplantation. The confocal imaging, fluorescence-activated cell sorting (FACS), and mitochondrial DNA analysis demonstrated the successful dose-dependent transfer of exogenous mitochondria to cancer cells [98][91]. Later, Cabrera et al. proposed a new transplantation protocol to simplify the MitoCeption procedure. Specifically, mitochondria are directly added into a microcentrifuge tube, where the recipient cells are in suspension. Following this, the tubes are centrifuged (Figure 3D). The novel protocol appears to be quicker and successful [99][92].
This procedure allowed for an allogenic transplant in vitro, repairing the metabolic activity, mitochondrial mass, and mtDNA sequence stability in UV-damaged peripheral blood mononuclear cells (PBMCs). In 2018, Kim’s research lab proposed an easy and quick centrifugation method without additional incubation steps. They first isolated mitochondria from human-umbilical-cord-derived MSCs via differential centrifugation, then transferred them into both L6 cells and human umbilical cord Rho0 cells via centrifugation. At the end, in the recipient cells, they observed an increase in ATP production, mitochondrial membrane potential, and oxygen consumption levels and a decrease in ROS [100][93]. According to Kim’s results, the application of an outer physical stimulus in MT experiments, such as centripetal force, appears to be successful regardless of cells’ types and physiological states. However, two critical issues must be considered. On one hand, the possible mechanical damage to the recipient cells’ plasma membrane. On the other hand, such a strategy cannot be performed in vivo. Here, the considerable therapeutic potential of stem cells becomes significant. As a matter of fact, stem cells from patients might be transplanted ex vivo and introduced back in an organism’s injured area to reprogram and/or repair its metabolism [55][48].
Such an attractive approach pushed researchers to explore novel physical-principle-based MT techniques [101][94]. In 2016, Wu et al. utilised a photothermal nanoblade to deliver mitochondria isolated from MDA-MB-453 cells into 143B- Rho0 cells [102][95] (Figure 3E). In detail, the photothermal nanoblade techniques causes the opening of the plasma membrane via laser pulses and the delivery of mitochondria using a titanium-coated borosilicate glass micropipette ~5 mm in length and with a tip ~3 μm in diameter using a fluid pump. Although the respiration of 143B- Rho0 cells seemed restored, only a few cells per hour could be transplanted in each experiment. Thus, the researchers adjusted the protocol with a biophotonic-laser-assisted surgery tool (BLAST) for the rapid massively parallel delivery of micron-sized large cargo into recipient cells [103][96].
In parallel, Macheiner’s groups proposed a novel mitochondrial transfer method, called magneto-mitotransfer, which employs an anti-TOM22 magnetic bead to labelled mitochondria and carries them into host cells with the support of a magnetic plate (Figure 3F). They reported the rapid autologous MT of human fibroblasts MRC-5 and the related functional changes. Moreover, in 2021, Sarcel et al. developed “MitoPunch”, a pressure-driven MT device to transfer mitochondria to recipient cells seeded on a porous polyester membrane (Figure 3G). They show the transfer of mitochondria isolated from HEK293T cells to 143B-Rho0 cells and the host retention of exogenous mtDNA copies [104][97].
Overall, the success of MT seems to be influenced by several factors: (1) the method of isolating mitochondria, which stresses organelles and affects their viability; (2) the selection of the donor cell line, with stem cells being preferred; (3) the amount of mitochondria to be transplanted; and (4) the delivery method [105][98]. The need to deliver intact and functional mitochondria has therefore led researchers to develop new tools.
In this scenario, Gäbelein’s group recently developed a single-cell technology called FluidFM to transplant mitochondria between living cells without compromising mitochondrial and cellular integrity or viability. FluidFM combines atomic force microscopy, optical microscopy, and nanofluidics. This procedure uses specific probes that permit minimally invasive access to cells and fluid flow to extract and inject mitochondria (Figure 3H). However, the technique is still not appropriate for use in vivo [106][99].
For in vitro and in vivo applications, the use of specific physiological vehicles with low immunogenicity and toxicity, such as extracellular vesicles (EVs), improved the preservation of mitochondria integrity when delivered (Table 1). Two main studies explored the novel EV-mediated MT method (Figure 3I). In 2021, Ikeda et al. isolated EVs from human-induced pluripotent stem cell (iPSC)-derived cardiomyocytes (iCMs) and coincubated them with recipient iCMs damaged by hypoxia. The confocal microscopy analysis indicated that exogenous mitochondria were transferred into host cells and fused with their endogenous mitochondrial networks, leading to a significant increase in ATP generation and an improvement in contractility [107][100]. In the same year, Peruzzotti-Jametti et al. investigated the hypothesis that neural stem cells (NSCs) release and transport functioning mitochondria through EVs into host cells to modulate mitochondrial activity. First, they verified whether the EVs were loaded with mitochondria via microscopic and functional analyses. Then, they incubated them with mtDNA-depleted L929 Rho0 cells. Mitochondrial function was then assessed in the recipient cells [71][64].
Table 1.
A comparison of in vitro mitochondrial transplantation methods.
| In Vitro MT Methods | Pros | Cons | Remarks |
|---|
][102]. Next, Cowan et al. performed two in vivo MT procedures: (1) direct injection of mitochondria into rabbit myocardial ischemic zone and (2) vascular perfusion of mitochondria through the coronary artery. Analysis of the results demonstrated that direct mitochondria injection proved to be more efficient in cardiac protection [110][103]. To summarize, direct mitochondrial transplantation appears to be the most promising way to modulate cardiomyocytes metabolism. Since then, several studies in vivo have been performed in different animal tissues, such as liver, lung, and brain [111][104] (Table 2). Worth to mention, two studies demonstrate the feasibility of MT on patients with ischaemia–reperfusion injury and single large-scale mtDNA deletion syndromes (SLSMDs).
Table 2.
In vivo research reports of mitochondrial transplantation.
| Targeted Organs | Species | Disease | Route of Administration | Studies Reference | ||||
|---|---|---|---|---|---|---|---|---|
| Co-incubation | Reduced manipulation | |||||||
| Heart | Broad number of transplanted recipient cells Easy to realise |
Rabbits, pigs, rats, mice, piglets, humansLow accuracy Dependence on physiological state and uptake capacity of recipient cells Mitochondria dose-dependent High risk of mitochondria damage |
mtDNA retention up to 12 passages Moderate transfer Efficiency |
|||||
| Heart regional/global ischaemia; heterotopic heart transplantation; right heart failure. | Local direct injection; | intracoronary injection. | [107,108,109,110,112,119,120,121][100][101]113,[114,115,102][103][105][106][107116,]117,118,[108][109][110][111] | Microinjection | Successful regardless the physiological state and uptake capacity of the target cells | 116][117][118][119][120][121] | ||
| Cell-penetrating peptide | Low manipulation Increased uptake capacity rate of target cells |
Unknown effect of Pep-1 on mitochondrial function High risk of mitochondria damage |
mtDNA is retained 11 days after | |||||
| Lung | Treatment | |||||||
| Rats, mice | Airway hyperresponsiveness; melanoma lung metastasis; acute lung ischaemia–reperfusion; pulmonary hypertension, | experimental sepsis. |
Intratracheally injection; intravenously injection; intracoronary injection; pulmonary artery injection. |
[129,130,131,132,133][122][123][124] | MitoCeption | Time saving Successful regardless the physiological state and uptake capacity of the target cells |
High manipulation Potentially harmful for the target Mitochondria dose-dependent High risk of mitochondria damage |
mtDNA retention not known Moderate transfer Efficiency |
| [ | 112 | ] | [ | 113][114] | Potentially harmful for the target Limited number of cells that can be transplanted per experiment High risk of mitochondria damage |
mtDNA is retained from 6–10 weeks after treatment | ||
| Liver | Rats, mice | Partial liver ischaemia; fatty liver; acetaminophen/carbon-tetrachloride-induced liver injury. | [ | 125][126] | ||||
| Brain | Rats, mice, pigs | Stroke; Parkinson’s; schizophrenia; Alzheimer’s; age-associated cognitive decline, depression; spinal cord injury; optic nerve crush. |
Intracerebral injection; systemic injection; intrathecal injection; intranasal injection; intracerebroventricular injection. |
[71,134,135,136,137,138,139,150,[64][127][128151 | Photothermal nanoblade | Rapid massively delivery Very accurate |
High manipulation Specific equipment High risk of mitochondria damaging Limited number of transplanted cells |
Stable retained 2% transfer efficiency |
| Intrasplenic injection; | intravenously injection. | [ | 122,123,124,125,126,127,128][,]152,153,[129]140,141,142,143,144,154,[130]155,[131]145,][133][134][146,135147,148,][136149,156,[157,158,132159]][137 | Magnetomitotransfer | Rapid massively delivery Very accurate |
Specific supplies High risk of mitochondria damage |
mtDNA retention not known High transfer Efficiency |
|
| 115 | ] | [ | ] | Mitopunch | Rapid massively delivery |
Not suitable for all cell types (only those attaching PET filter) Dependent on nDNA-mtDNA mismatch High risk of mitochondria damage |
Stable retention Moderate transfer Efficiency |
|
| FluidFM | Mitochondria and cellular integrity preservation Minimally invasive |
Specific equipment High cost |
High transfer efficiency mtDNA retention is not known |
|||||
| EVs mitochondrial delivery | Low manipulation Mitochondrial and cellular integrity preservation Easy to realise |
Mitochondria-rich-EV isolation | mtDNA retention not known |
4.2. In Vivo Methods for MT
In addition to the reported in vitro MT studies, mitochondria can also be transplanted directly into animal models. In 2009, McCully et al. [108][101] presented the first in vivo MT study in an ischaemia–reperfusion heart model using an allogenic transplant in a rabbit. They isolated functional mitochondria from the left ventricular tissue of a healthy animal and injected them into the cardiac ischemic area of a different rabbit just before reperfusion. The study highlights the potential of MT to improve functional recovery of the heart and survival of cardiomyocytes after ischaemia. In addition, cardiomyocytes could uptake exogenous mitochondria just 2 h after injection. Further studies demonstrated that MT significantly reduces cardiac ischaemia markers (creatine kinase-MB and cardiac Troponin I) and the apoptosis protein Caspase-3, as well as infarct size [109
In the first case, autologous mitochondria were isolated from a piece of healthy rectus abdominis muscle and direct injected to the myocardium of five paediatric patients who required central extracorporeal membrane oxygenation (ECMO) support for ischaemia–reperfusion dysfunction. The data show improvement in ventricular function within several days after treatment, but future studies are necessary to demonstrate the efficiency of the strategy [112][105].
Recently, in a compassionate use study, a mitochondrial augmentation therapy (MAT) approach has been applied on six patients with SLSMDs [160][153]. MAT is a cell technology platform in which autologous hematopoietic stem and progenitor cells (HSPCs) are augmented ex vivo with mitochondria obtained from donor cells or tissue. HSPCs have been shown to migrate to distal tissues and to abrogate disease-related deterioration of mitochondrial dysfunctions or metabolic disease. In this case, CD34+ cells were collected from a patient with a non-inherited primary mitochondrial disease, and PBMCs were collected from the maternal donor. Using a coincubation method, healthy mitochondria isolated from maternal PBMCs were transplanted into the patients’ CD34+ cells, which in turn were intravenously reinfused into the patients. MAT resulted in decreased heteroplasmy of mtDNA deletion, increased mtDNA levels, and improved ATP content in peripheral blood mononuclear cells in four out of six patients, as well as improved muscle strength and endurance in two individuals. Despite these encouraging results in peripheral tissues, this strategy does not reach the brain, which is also affected in SLSMDs (Table 2). However, this approach is not applicable in the case of homoplasmic mutations or in the presence of high heteroplasmy levels.
Several data show that mitochondrial dysfunction plays a pivotal role in neurodegeneration. Indeed, cells of the nervous system require a considerable amount of energy to carry out synaptic transmission, neurogenesis, and neuronal differentiation [161][154]. Therefore, MT strategy has also been experimented upon in animal models to treat neurodegenerative diseases and to investigate mitochondria supply in the brain [162][155].
For example, Shi et al. reported that systemic injection of mitochondria isolated from human hepatoma cells (HepG2 cells) into the brain of PD mice mitigates the progression of PD by improving ETC function and decreasing ROS production [143][136]. To facilitate mitochondrial transplantation, in another paper, the authors injected PeP-1-conjugated mitochondria into the medial forebrain bundle (MFB) of 6-OHDA PD rats. As a result, neuronal mitochondrial function and dynamics improved in the substantia nigra (SN) [142][135].
Additionally, encouraging data for MT in the CNS came from experimental models of schizophrenia (SZ) and Alzheimer’s disease [162][155]. Dos-Santos et al. demonstrated the therapeutic potential of MT in the crush-based animal model of optic-nerve glaucoma. The group performed an intravitreal injection of liver-isolated mitochondria and showed that MT transiently protects RGCs and reduces oxidative stress [156][149].
It is worth noting that the route of administration is crucial to the effect of MT in vivo. Currently, four routes of administration are used: (1) intracerebral injection, an invasive and painful procedure; (2) intrathecal injection, an alternative to bypassing the blood–brain barrier (BBB); (3) systemic or intracarotid administration, a less invasive and safer route; and (4) intranasal administration, which very recently has been shown as a simple and effective delivery system [140,144,163][133][137][156].
There are two main limitations to consider when performing MT in the CNS. First, the BBB which protects the brain from compounds that are toxic to brain neurons [164][157]. Second, the immune response in the brain is particularly alarming, and mitochondria are strongly immunogenetic organelles [165,166][158][159]. Indeed, some studies have investigated the immune reaction after mitochondrial transplantation in different tissue in vivo, confirming that there is an activation of the immune system following injection of mitochondria [167,168][160][161]. To reduce the immunological risk associated with MT, it would be very useful to untwist the mechanism underlying an immune response during this procedure [111][104]. In summary, future research on in vivo MT should focus on bypassing the BBB and preventing harmful immune responses in the brain. The study by Pluchino’s group mentioned above has provided a strategic method in this direction using NSC-derived EVs [71][64]. Remarkably, the delivery through vesicles not only preserves mitochondrial integrity but also reduces the damage associated with the immune response. Indeed, EVs hiding mitochondria from microglia and macrophages would act as a Trojan Horse, thus reducing immune reactivity.
References
- Rossmann, M.P.; Dubois, S.M.; Agarwal, S.; Zon, L.I. Mitochondrial Function in Development and Disease. Dis. Model. Mech. 2021, 14, dmm048912.
- Vinten-Johansen, J. Commentary: Mitochondria Are More than Just the Cells’ Powerhouse. J. Thorac. Cardiovasc. Surg. 2020, 160, e33–e34.
- Giacomello, M.; Pyakurel, A.; Glytsou, C.; Scorrano, L. The Cell Biology of Mitochondrial Membrane Dynamics. Nat. Rev. Mol. Cell Biol. 2020, 21, 204–224.
- Whitley, B.N.; Engelhart, E.A.; Hoppins, S. Mitochondrial Dynamics and Their Potential as a Therapeutic Target. Mitochondrion 2019, 49, 269–283.
- Roger, A.J.; Muñoz-Gómez, S.A.; Kamikawa, R. The Origin and Diversification of Mitochondria. Curr. Biol. 2017, 27, R1177–R1192.
- Taanman, J.-W. The Mitochondrial Genome: Structure, Transcription, Translation and Replication. Biochim. Biophys. Acta (BBA) Bioenerg. 1999, 1410, 103–123.
- Pagnamenta, A.T.; Wei, W.; Rahman, S.; Chinnery, P.F. Biparental Inheritance of Mitochondrial DNA Revisited. Nat. Rev. Genet. 2021, 22, 477–478.
- Zeviani, M.; Viscomi, C. Mitochondrial Neurodegeneration. Cells 2022, 11, 637.
- Pagliarini, D.J.; Calvo, S.E.; Chang, B.; Sheth, S.A.; Vafai, S.B.; Ong, S.-E.; Walford, G.A.; Sugiana, C.; Boneh, A.; Chen, W.K.; et al. A Mitochondrial Protein Compendium Elucidates Complex I Disease Biology. Cell 2008, 134, 112–123.
- Bogenhagen, D.; Clayton, D.A. The Number of Mitochondrial Deoxyribonucleic Acid Genomes in Mouse L and Human HeLa Cells. Quantitative Isolation of Mitochondrial Deoxyribonucleic Acid. J. Biol. Chem. 1974, 249, 7991–7995.
- Veltri, K.L.; Espiritu, M.; Singh, G. Distinct Genomic Copy Number in Mitochondria of Different Mammalian Organs. J. Cell. Physiol. 1990, 143, 160–164.
- Jevtic, V.; Kindle, P.; Avilov, S.V. SYBR Gold Dye Enables Preferential Labelling of Mitochondrial Nucleoids and Their Time-Lapse Imaging by Structured Illumination Microscopy. PLoS ONE 2018, 13, e0203956.
- Bogenhagen, D.F.; Rousseau, D.; Burke, S. The Layered Structure of Human Mitochondrial DNA Nucleoids. J. Biol. Chem. 2008, 283, 3665–3675.
- Kazak, L.; Reyes, A.; Holt, I.J. Minimizing the Damage: Repair Pathways Keep Mitochondrial DNA Intact. Nat. Rev. Mol. Cell Biol. 2012, 13, 659–671.
- Brown, W.M.; George, M.; Wilson, A.C. Rapid Evolution of Animal Mitochondrial DNA. Proc. Natl. Acad. Sci. USA 1979, 76, 1967–1971.
- Pham, V.H.; Nguyen, V.L.; Jung, H.-E.; Cho, Y.-S.; Shin, J.-G. The Frequency of the Known Mitochondrial Variants Associated with Drug-Induced Toxicity in a Korean Population. BMC Med. Genom. 2022, 15, 3.
- Rahman, J.; Rahman, S. Mitochondrial Medicine in the Omics Era. Lancet 2018, 391, 2560–2574.
- Gorman, G.S.; Chinnery, P.F.; DiMauro, S.; Hirano, M.; Koga, Y.; McFarland, R.; Suomalainen, A.; Thorburn, D.R.; Zeviani, M.; Turnbull, D.M. Mitochondrial Diseases. Nat. Rev. Dis. Prim. 2016, 2, 16080.
- El-Hattab, A.W.; Zarante, A.M.; Almannai, M.; Scaglia, F. Therapies for Mitochondrial Diseases and Current Clinical Trials. Mol. Genet. Metab. 2017, 122, 1–9.
- Bottani, E.; Lamperti, C.; Prigione, A.; Tiranti, V.; Persico, N.; Brunetti, D. Therapeutic Approaches to Treat Mitochondrial Diseases: “One-Size-Fits-All” and “Precision Medicine” Strategies. Pharmaceutics 2020, 12, 1083.
- Di Donfrancesco, A.; Massaro, G.; Di Meo, I.; Tiranti, V.; Bottani, E.; Brunetti, D. Gene Therapy for Mitochondrial Diseases: Current Status and Future Perspective. Pharmaceutics 2022, 14, 1287.
- Slone, J.; Huang, T. The Special Considerations of Gene Therapy for Mitochondrial Diseases. NPJ Genom. Med. 2020, 5, 7.
- Artika, I.M. Allotopic Expression of Mitochondrial Genes: Basic Strategy and Progress. Genes Dis. 2020, 7, 578–584.
- Reddy, P.; Ocampo, A.; Suzuki, K.; Luo, J.; Bacman, S.R.; Williams, S.L.; Sugawara, A.; Okamura, D.; Tsunekawa, Y.; Wu, J.; et al. Selective Elimination of Mitochondrial Mutations in the Germline by Genome Editing. Cell 2015, 161, 459–469.
- Yang, X.; Jiang, J.; Li, Z.; Liang, J.; Xiang, Y. Strategies for Mitochondrial Gene Editing. Comput. Struct. Biotechnol. J. 2021, 19, 3319–3329.
- Falabella, M.; Minczuk, M.; Hanna, M.G.; Viscomi, C.; Pitceathly, R.D.S. Gene Therapy for Primary Mitochondrial Diseases: Experimental Advances and Clinical Challenges. Nat. Rev. Neurol. 2022, 18, 689–698.
- Mok, B.Y.; de Moraes, M.H.; Zeng, J.; Bosch, D.E.; Kotrys, A.V.; Raguram, A.; Hsu, F.; Radey, M.C.; Peterson, S.B.; Mootha, V.K.; et al. A Bacterial Cytidine Deaminase Toxin Enables CRISPR-Free Mitochondrial Base Editing. Nature 2020, 583, 631–637.
- Cho, S.-I.; Lee, S.; Mok, Y.G.; Lim, K.; Lee, J.; Lee, J.M.; Chung, E.; Kim, J.-S. Targeted A-to-G Base Editing in Human Mitochondrial DNA with Programmable Deaminases. Cell 2022, 185, 1764–1776.e12.
- Qin, Y.; Jiang, X.; Yang, Q.; Zhao, J.; Zhou, Q.; Zhou, Y. The Functions, Methods, and Mobility of Mitochondrial Transfer Between Cells. Front. Oncol. 2021, 11, 672781.
- Rustom, A.; Saffrich, R.; Markovic, I.; Walther, P.; Gerdes, H.-H. Nanotubular Highways for Intercellular Organelle Transport. Science 2004, 303, 1007–1010.
- Spees, J.L.; Olson, S.D.; Whitney, M.J.; Prockop, D.J. Mitochondrial Transfer between Cells Can Rescue Aerobic Respiration. Proc. Natl. Acad. Sci. USA 2006, 103, 1283–1288.
- Torralba, D.; Baixauli, F.; Sánchez-Madrid, F. Mitochondria Know No Boundaries: Mechanisms and Functions of Intercellular Mitochondrial Transfer. Front. Cell Dev. Biol. 2016, 4, 107.
- Nakamura, Y.; Park, J.-H.; Hayakawa, K. Therapeutic Use of Extracellular Mitochondria in CNS Injury and Disease. Exp. Neurol. 2020, 324, 113114.
- Miliotis, S.; Nicolalde, B.; Ortega, M.; Yepez, J.; Caicedo, A. Forms of Extracellular Mitochondria and Their Impact in Health. Mitochondrion 2019, 48, 16–30.
- Burdett, T.C.; Freeman, M.R. Astrocytes Eyeball Axonal Mitochondria. Science 2014, 345, 385–386.
- Lyamzaev, K.G.; Nepryakhina, O.K.; Saprunova, V.B.; Bakeeva, L.E.; Pletjushkina, O.Y.; Chernyak, B.V.; Skulachev, V.P. Novel Mechanism of Elimination of Malfunctioning Mitochondria (Mitoptosis): Formation of Mitoptotic Bodies and Extrusion of Mitochondrial Material from the Cell. Biochim. Biophys. Acta (BBA) Bioenerg. 2008, 1777, 817–825.
- Davis, C.O.; Kim, K.-Y.; Bushong, E.A.; Mills, E.A.; Boassa, D.; Shih, T.; Kinebuchi, M.; Phan, S.; Zhou, Y.; Bihlmeyer, N.A.; et al. Transcellular Degradation of Axonal Mitochondria. Proc. Natl. Acad. Sci. USA 2014, 111, 9633–9638.
- Phinney, D.G.; Di Giuseppe, M.; Njah, J.; Sala, E.; Shiva, S.; St Croix, C.M.; Stolz, D.B.; Watkins, S.C.; Di, Y.P.; Leikauf, G.D.; et al. Mesenchymal Stem Cells Use Extracellular Vesicles to Outsource Mitophagy and Shuttle MicroRNAs. Nat. Commun. 2015, 6, 8472.
- Chen, J.; Zhong, J.; Wang, L.; Chen, Y. Mitochondrial Transfer in Cardiovascular Disease: From Mechanisms to Therapeutic Implications. Front. Cardiovasc. Med. 2021, 8, 771298.
- Pluchino, S.; Peruzzotti-Jametti, L.; Frezza, C. Astrocyte Power Fuels Neurons during Stroke. Swiss Med. Wkly. 2016, 146, w14374.
- Hayakawa, K.; Chan, S.J.; Mandeville, E.T.; Park, J.H.; Bruzzese, M.; Montaner, J.; Arai, K.; Rosell, A.; Lo, E.H. Protective Effects of Endothelial Progenitor Cell-Derived Extracellular Mitochondria in Brain Endothelium. Stem Cells 2018, 36, 1404–1410.
- Sinclair, K.A.; Yerkovich, S.T.; Hopkins, P.M.-A.; Chambers, D.C. Characterization of Intercellular Communication and Mitochondrial Donation by Mesenchymal Stromal Cells Derived from the Human Lung. Stem Cell Res. Ther. 2016, 7, 91.
- Rodriguez, A.-M.; Nakhle, J.; Griessinger, E.; Vignais, M.-L. Intercellular Mitochondria Trafficking Highlighting the Dual Role of Mesenchymal Stem Cells as Both Sensors and Rescuers of Tissue Injury. Cell Cycle 2018, 17, 712–721.
- Liu, K.; Ji, K.; Guo, L.; Wu, W.; Lu, H.; Shan, P.; Yan, C. Mesenchymal Stem Cells Rescue Injured Endothelial Cells in an in vitro Ischemia-Reperfusion Model via Tunneling Nanotube like Structure-Mediated Mitochondrial Transfer. Microvasc. Res. 2014, 92, 10–18.
- Kumar, V. A STING to Inflammation and Autoimmunity. J. Leukoc. Biol. 2019, 106, 171–185.
- Rodríguez-Nuevo, A.; Díaz-Ramos, A.; Noguera, E.; Díaz-Sáez, F.; Duran, X.; Muñoz, J.P.; Romero, M.; Plana, N.; Sebastián, D.; Tezze, C.; et al. Mitochondrial DNA and TLR9 Drive Muscle Inflammation upon Opa1 Deficiency. EMBO J. 2018, 37, e96553.
- Liu, K.; Zhou, Z.; Pan, M.; Zhang, L. Stem Cell-derived Mitochondria Transplantation: A Promising Therapy for Mitochondrial Encephalomyopathy. CNS Neurosci. Ther. 2021, 27, 733–742.
- Wang, J.; Li, H.; Yao, Y.; Zhao, T.; Chen, Y.; Shen, Y.; Wang, L.; Zhu, Y. Stem Cell-Derived Mitochondria Transplantation: A Novel Strategy and the Challenges for the Treatment of Tissue Injury. Stem Cell Res. Ther. 2018, 9, 106.
- Domhan, S.; Ma, L.; Tai, A.; Anaya, Z.; Beheshti, A.; Zeier, M.; Hlatky, L.; Abdollahi, A. Intercellular Communication by Exchange of Cytoplasmic Material via Tunneling Nano-Tube Like Structures in Primary Human Renal Epithelial Cells. PLoS ONE 2011, 6, e21283.
- Zhang, J.; Zhang, Y. Membrane Nanotubes: Novel Communication between Distant Cells. Sci. China Life Sci. 2013, 56, 994–999.
- Resnik, N.; Prezelj, T.; De Luca, G.M.R.; Manders, E.; Polishchuk, R.; Veranič, P.; Kreft, M.E. Helical Organization of Microtubules Occurs in a Minority of Tunneling Membrane Nanotubes in Normal and Cancer Urothelial Cells. Sci. Rep. 2018, 8, 17133.
- Dupont, M.; Souriant, S.; Lugo-Villarino, G.; Maridonneau-Parini, I.; Vérollet, C. Tunneling Nanotubes: Intimate Communication between Myeloid Cells. Front. Immunol. 2018, 9, 43.
- Driscoll, J.; Gondaliya, P.; Patel, T. Tunneling Nanotube-Mediated Communication: A Mechanism of Intercellular Nucleic Acid Transfer. Int. J. Mol. Sci. 2022, 23, 5487.
- Shen, J.; Zhang, J.-H.; Xiao, H.; Wu, J.-M.; He, K.-M.; Lv, Z.-Z.; Li, Z.-J.; Xu, M.; Zhang, Y.-Y. Mitochondria Are Transported along Microtubules in Membrane Nanotubes to Rescue Distressed Cardiomyocytes from Apoptosis. Cell Death Dis. 2018, 9, 81.
- Ahmad, T.; Mukherjee, S.; Pattnaik, B.; Kumar, M.; Singh, S.; Kumar, M.; Rehman, R.; Tiwari, B.K.; Jha, K.A.; Barhanpurkar, A.P.; et al. Miro1 Regulates Intercellular Mitochondrial Transport & Enhances Mesenchymal Stem Cell Rescue Efficacy. EMBO J. 2014, 33, 994–1010.
- Tang, B. MIRO GTPases in Mitochondrial Transport, Homeostasis and Pathology. Cells 2015, 5, 1.
- Drab, M.; Stopar, D.; Kralj-Iglič, V.; Iglič, A. Inception Mechanisms of Tunneling Nanotubes. Cells 2019, 8, 626.
- Vignais, M.-L.; Caicedo, A.; Brondello, J.-M.; Jorgensen, C. Cell Connections by Tunneling Nanotubes: Effects of Mitochondrial Trafficking on Target Cell Metabolism, Homeostasis, and Response to Therapy. Stem Cells Int. 2017, 2017, 6917941.
- Aaronson, S. The Synthesis of Extracellular Macromolecules and Membranes by a Population of the Phytoflagellate Ochromonas Danica 1: Extracellular Secretion by Ochromonas. Limnol. Oceanogr. 1971, 16, 1–9.
- Raposo, G.; Nijman, H.W.; Stoorvogel, W.; Liejendekker, R.; Harding, C.V.; Melief, C.J.; Geuze, H.J. B Lymphocytes Secrete Antigen-Presenting Vesicles. J. Exp. Med. 1996, 183, 1161–1172.
- Pitt, J.M.; Kroemer, G.; Zitvogel, L. Extracellular Vesicles: Masters of Intercellular Communication and Potential Clinical Interventions. J. Clin. Investig. 2016, 126, 1139–1143.
- Meng, W.; He, C.; Hao, Y.; Wang, L.; Li, L.; Zhu, G. Prospects and Challenges of Extracellular Vesicle-Based Drug Delivery System: Considering Cell Source. Drug Deliv. 2020, 27, 585–598.
- Mir, B.; Goettsch, C. Extracellular Vesicles as Delivery Vehicles of Specific Cellular Cargo. Cells 2020, 9, 1601.
- Peruzzotti-Jametti, L.; Bernstock, J.D.; Willis, C.M.; Manferrari, G.; Rogall, R.; Fernandez-Vizarra, E.; Williamson, J.C.; Braga, A.; van den Bosch, A.; Leonardi, T.; et al. Neural Stem Cells Traffic Functional Mitochondria via Extracellular Vesicles. PLoS Biol. 2021, 19, e3001166.
- Ratajczak, M.Z.; Ratajczak, J. Horizontal Transfer of RNA and Proteins between Cells by Extracellular Microvesicles: 14 Years Later. Clin. Transl. Med. 2016, 5, 7.
- Mao, J.; Li, C.; Wu, F.; She, Z.; Luo, S.; Chen, X.; Wen, C.; Tian, J. MSC-EVs Transferring Mitochondria and Related Components: A New Hope for the Treatment of Kidney Disease. Front. Immunol. 2022, 13, 978571.
- Islam, M.N.; Das, S.R.; Emin, M.T.; Wei, M.; Sun, L.; Westphalen, K.; Rowlands, D.J.; Quadri, S.K.; Bhattacharya, S.; Bhattacharya, J. Mitochondrial Transfer from Bone-Marrow–Derived Stromal Cells to Pulmonary Alveoli Protects against Acute Lung Injury. Nat. Med. 2012, 18, 759–765.
- Hough, K.P.; Trevor, J.L.; Strenkowski, J.G.; Wang, Y.; Chacko, B.K.; Tousif, S.; Chanda, D.; Steele, C.; Antony, V.B.; Dokland, T.; et al. Exosomal Transfer of Mitochondria from Airway Myeloid-Derived Regulatory Cells to T Cells. Redox Biol. 2018, 18, 54–64.
- Hayakawa, K.; Esposito, E.; Wang, X.; Terasaki, Y.; Liu, Y.; Xing, C.; Ji, X.; Lo, E.H. Transfer of Mitochondria from Astrocytes to Neurons after Stroke. Nature 2016, 535, 551–555.
- Todkar, K.; Chikhi, L.; Desjardins, V.; El-Mortada, F.; Pépin, G.; Germain, M. Selective Packaging of Mitochondrial Proteins into Extracellular Vesicles Prevents the Release of Mitochondrial DAMPs. Nat. Commun. 2021, 12, 1971.
- Fuentes, P.; Sesé, M.; Guijarro, P.J.; Emperador, M.; Sánchez-Redondo, S.; Peinado, H.; Hümmer, S.; Ramón y Cajal, S. ITGB3-Mediated Uptake of Small Extracellular Vesicles Facilitates Intercellular Communication in Breast Cancer Cells. Nat. Commun. 2020, 11, 4261.
- Van Niel, G.; D’Angelo, G.; Raposo, G. Shedding Light on the Cell Biology of Extracellular Vesicles. Nat. Rev. Mol. Cell Biol. 2018, 19, 213–228.
- Zhang, Y.; Tan, J.; Miao, Y.; Zhang, Q. The Effect of Extracellular Vesicles on the Regulation of Mitochondria under Hypoxia. Cell Death Dis. 2021, 12, 358.
- Chang, C.-Y.; Liang, M.-Z.; Chen, L. Current Progress of Mitochondrial Transplantation That Promotes Neuronal Regeneration. Transl. Neurodegener. 2019, 8, 17.
- Tria, F.D.K.; Brueckner, J.; Skejo, J.; Xavier, J.C.; Kapust, N.; Knopp, M.; Wimmer, J.L.E.; Nagies, F.S.P.; Zimorski, V.; Gould, S.B.; et al. Gene Duplications Trace Mitochondria to the Onset of Eukaryote Complexity. Genome Biol. Evol. 2021, 13, evab055.
- Liu, Z.; Sun, Y.; Qi, Z.; Cao, L.; Ding, S. Mitochondrial Transfer/Transplantation: An Emerging Therapeutic Approach for Multiple Diseases. Cell Biosci. 2022, 12, 66.
- Park, A.; Oh, M.; Lee, S.J.; Oh, K.-J.; Lee, E.-W.; Lee, S.C.; Bae, K.-H.; Han, B.S.; Kim, W.K. Mitochondrial Transplantation as a Novel Therapeutic Strategy for Mitochondrial Diseases. Int. J. Mol. Sci. 2021, 22, 4793.
- Lightowlers, R.N.; Chrzanowska-Lightowlers, Z.M.; Russell, O.M. Mitochondrial Transplantation—A Possible Therapeutic for Mitochondrial Dysfunction? EMBO Rep. 2020, 21, e50964.
- Clark, M.A.; Shay, J.W. Mitochondrial Transformation of Mammalian Cells. Nature 1982, 295, 605–607.
- Caicedo, A.; Aponte, P.M.; Cabrera, F.; Hidalgo, C.; Khoury, M. Artificial Mitochondria Transfer: Current Challenges, Advances, and Future Applications. Stem Cells Int. 2017, 2017, 7610414.
- King, M.P.; Attardi, G. Injection of Mitochondria into Human Cells Leads to a Rapid Replacement of the Endogenous Mitochondrial DNA. Cell 1988, 52, 811–819.
- Pinkert, C.A.; Irwin, M.H.; Johnson, L.W.; Moffatt, R.J. Mitochondria Transfer into Mouse Ova by Microinjection. Transgenic Res. 1997, 6, 379–383.
- Reznichenko, A.; Huyser, C.; Pepper, M. Mitochondrial Transfer: Implications for Assisted Reproductive Technologies. Appl. Transl. Genom. 2016, 11, 40–47.
- Ali Pour, P.; Kenney, M.C.; Kheradvar, A. Bioenergetics Consequences of Mitochondrial Transplantation in Cardiomyocytes. J. Am. Heart Assoc. 2020, 9, e014501.
- Maeda, H.; Kami, D.; Maeda, R.; Shikuma, A.; Gojo, S. Generation of Somatic Mitochondrial DNA-Replaced Cells for Mitochondrial Dysfunction Treatment. Sci. Rep. 2021, 11, 10897.
- Reilly, W.M.; Obara, C.J. Advances in Confocal Microscopy and Selected Applications. In Confocal Microscopy; Brzostowski, J., Sohn, H., Eds.; Methods in Molecular Biology; Springer: New York, NY, USA, 2021; Volume 2304, pp. 1–35. ISBN 978-1-07-161401-3.
- King, M.P.; Attardi, G. Human Cells Lacking MtDNA: Repopulation with Exogenous Mitochondria by Complementation. Science 1989, 246, 500–503.
- Gomzikova, M.O.; James, V.; Rizvanov, A.A. Mitochondria Donation by Mesenchymal Stem Cells: Current Understanding and Mitochondria Transplantation Strategies. Front. Cell Dev. Biol. 2021, 9, 653322.
- Chang, J.-C.; Hoel, F.; Liu, K.-H.; Wei, Y.-H.; Cheng, F.-C.; Kuo, S.-J.; Tronstad, K.J.; Liu, C.-S. Peptide-Mediated Delivery of Donor Mitochondria Improves Mitochondrial Function and Cell Viability in Human Cybrid Cells with the MELAS A3243G Mutation. Sci. Rep. 2017, 7, 10710.
- Maeda, H.; Kami, D.; Maeda, R.; Murata, Y.; Jo, J.; Kitani, T.; Tabata, Y.; Matoba, S.; Gojo, S. TAT-dextran–Mediated Mitochondrial Transfer Enhances Recovery from Models of Reperfusion Injury in Cultured Cardiomyocytes. J. Cell. Mol. Med. 2020, 24, 5007–5020.
- Caicedo, A.; Fritz, V.; Brondello, J.-M.; Ayala, M.; Dennemont, I.; Abdellaoui, N.; de Fraipont, F.; Moisan, A.; Prouteau, C.A.; Boukhaddaoui, H.; et al. MitoCeption as a New Tool to Assess the Effects of Mesenchymal Stem/Stromal Cell Mitochondria on Cancer Cell Metabolism and Function. Sci. Rep. 2015, 5, 9073.
- Cabrera, F.; Ortega, M.; Velarde, F.; Parra, E.; Gallardo, S.; Barba, D.; Soto, L.; Peña, G.; Pedroza, L.A.; Jorgensen, C.; et al. Primary Allogeneic Mitochondrial Mix (PAMM) Transfer/Transplant by MitoCeption to Address Damage in PBMCs Caused by Ultraviolet Radiation. BMC Biotechnol. 2019, 19, 42.
- Kim, M.J.; Hwang, J.W.; Yun, C.-K.; Lee, Y.; Choi, Y.-S. Delivery of Exogenous Mitochondria via Centrifugation Enhances Cellular Metabolic Function. Sci. Rep. 2018, 8, 3330.
- Ulger, O.; Kubat, G.B. Therapeutic Applications of Mitochondrial Transplantation. Biochimie 2022, 195, 1–15.
- Wu, T.-H.; Sagullo, E.; Case, D.; Zheng, X.; Li, Y.; Hong, J.S.; TeSlaa, T.; Patananan, A.N.; McCaffery, J.M.; Niazi, K.; et al. Mitochondrial Transfer by Photothermal Nanoblade Restores Metabolite Profile in Mammalian Cells. Cell Metab. 2016, 23, 921–929.
- Wu, Y.-C.; Wu, T.-H.; Clemens, D.L.; Lee, B.-Y.; Wen, X.; Horwitz, M.A.; Teitell, M.A.; Chiou, P.-Y. Massively Parallel Delivery of Large Cargo into Mammalian Cells with Light Pulses. Nat. Methods 2015, 12, 439–444.
- Sercel, A.J.; Patananan, A.N.; Man, T.; Wu, T.-H.; Yu, A.K.; Guyot, G.W.; Rabizadeh, S.; Niazi, K.R.; Chiou, P.-Y.; Teitell, M.A. Stable Transplantation of Human Mitochondrial DNA by High-Throughput, Pressurized Isolated Mitochondrial Delivery. eLife 2021, 10, e63102.
- Kubat, G.B.; Ulger, O.; Akin, S. Requirements for Successful Mitochondrial Transplantation. J. Biochem. Mol. Toxicol. 2021, 35, e22898.
- Gäbelein, C.G.; Feng, Q.; Sarajlic, E.; Zambelli, T.; Guillaume-Gentil, O.; Kornmann, B.; Vorholt, J.A. Mitochondria Transplantation between Living Cells. PLoS Biol. 2022, 20, e3001576.
- Ikeda, G.; Santoso, M.R.; Tada, Y.; Li, A.M.; Vaskova, E.; Jung, J.-H.; O’Brien, C.; Egan, E.; Ye, J.; Yang, P.C. Mitochondria-Rich Extracellular Vesicles From Autologous Stem Cell–Derived Cardiomyocytes Restore Energetics of Ischemic Myocardium. J. Am. Coll. Cardiol. 2021, 77, 1073–1088.
- McCully, J.D.; Cowan, D.B.; Pacak, C.A.; Toumpoulis, I.K.; Dayalan, H.; Levitsky, S. Injection of Isolated Mitochondria during Early Reperfusion for Cardioprotection. Am. J. Physiol.-Heart Circ. Physiol. 2009, 296, H94–H105.
- Masuzawa, A.; Black, K.M.; Pacak, C.A.; Ericsson, M.; Barnett, R.J.; Drumm, C.; Seth, P.; Bloch, D.B.; Levitsky, S.; Cowan, D.B.; et al. Transplantation of Autologously Derived Mitochondria Protects the Heart from Ischemia-Reperfusion Injury. Am. J. Physiol.-Heart Circ. Physiol. 2013, 304, H966–H982.
- Cowan, D.B.; Yao, R.; Akurathi, V.; Snay, E.R.; Thedsanamoorthy, J.K.; Zurakowski, D.; Ericsson, M.; Friehs, I.; Wu, Y.; Levitsky, S.; et al. Intracoronary Delivery of Mitochondria to the Ischemic Heart for Cardioprotection. PLoS ONE 2016, 11, e0160889.
- Yamada, Y.; Ito, M.; Arai, M.; Hibino, M.; Tsujioka, T.; Harashima, H. Challenges in Promoting Mitochondrial Transplantation Therapy. Int. J. Mol. Sci. 2020, 21, 6365.
- Emani, S.M.; Piekarski, B.L.; Harrild, D.; del Nido, P.J.; McCully, J.D. Autologous Mitochondrial Transplantation for Dysfunction after Ischemia-Reperfusion Injury. J. Thorac. Cardiovasc. Surg. 2017, 154, 286–289.
- Kaza, A.K.; Wamala, I.; Friehs, I.; Kuebler, J.D.; Rathod, R.H.; Berra, I.; Ericsson, M.; Yao, R.; Thedsanamoorthy, J.K.; Zurakowski, D.; et al. Myocardial Rescue with Autologous Mitochondrial Transplantation in a Porcine Model of Ischemia/Reperfusion. J. Thorac. Cardiovasc. Surg. 2017, 153, 934–943.
- Moskowitzova, K.; Shin, B.; Liu, K.; Ramirez-Barbieri, G.; Guariento, A.; Blitzer, D.; Thedsanamoorthy, J.K.; Yao, R.; Snay, E.R.; Inkster, J.A.H.; et al. Mitochondrial Transplantation Prolongs Cold Ischemia Time in Murine Heart Transplantation. J. Heart Lung Transplant. 2019, 38, 92–99.
- Shin, B.; Saeed, M.Y.; Esch, J.J.; Guariento, A.; Blitzer, D.; Moskowitzova, K.; Ramirez-Barbieri, G.; Orfany, A.; Thedsanamoorthy, J.K.; Cowan, D.B.; et al. A Novel Biological Strategy for Myocardial Protection by Intracoronary Delivery of Mitochondria: Safety and Efficacy. JACC Basic Transl. Sci. 2019, 4, 871–888.
- Blitzer, D.; Guariento, A.; Doulamis, I.P.; Shin, B.; Moskowitzova, K.; Barbieri, G.R.; Orfany, A.; del Nido, P.J.; McCully, J.D. Delayed Transplantation of Autologous Mitochondria for Cardioprotection in a Porcine Model. Ann. Thorac. Surg. 2020, 109, 711–719.
- Guariento, A.; Blitzer, D.; Doulamis, I.; Shin, B.; Moskowitzova, K.; Orfany, A.; Ramirez-Barbieri, G.; Staffa, S.J.; Zurakowski, D.; del Nido, P.J.; et al. Preischemic Autologous Mitochondrial Transplantation by Intracoronary Injection for Myocardial Protection. J. Thorac. Cardiovasc. Surg. 2020, 160, e15–e29.
- Weixler, V.; Lapusca, R.; Grangl, G.; Guariento, A.; Saeed, M.Y.; Cowan, D.B.; del Nido, P.J.; McCully, J.D.; Friehs, I. Autogenous Mitochondria Transplantation for Treatment of Right Heart Failure. J. Thorac. Cardiovasc. Surg. 2021, 162, e111–e121.
- Doulamis, I.P.; Guariento, A.; Duignan, T.; Orfany, A.; Kido, T.; Zurakowski, D.; del Nido, P.J.; McCully, J.D. Mitochondrial Transplantation for Myocardial Protection in Diabetic Hearts. Eur. J. Cardio-Thorac. Surg. 2020, 57, 836–845.
- Sun, X.; Gao, R.; Li, W.; Zhao, Y.; Yang, H.; Chen, H.; Jiang, H.; Dong, Z.; Hu, J.; Liu, J.; et al. Alda-1 Treatment Promotes the Therapeutic Effect of Mitochondrial Transplantation for Myocardial Ischemia-Reperfusion Injury. Bioact. Mater. 2021, 6, 2058–2069.
- Sun, X.; Chen, H.; Gao, R.; Qu, Y.; Huang, Y.; Zhang, N.; Hu, S.; Fan, F.; Zou, Y.; Hu, K.; et al. Intravenous Transplantation of an Ischemic-Specific Peptide-TPP-Mitochondrial Compound Alleviates Myocardial Ischemic Reperfusion Injury. ACS Nano 2023.
- Lin, H.-C.; Liu, S.-Y.; Lai, H.-S.; Lai, I.-R. Isolated Mitochondria Infusion Mitigates Ischemia-Reperfusion Injury of the Liver in Rats. Shock 2013, 39, 304–310.
- Fu, A.; Shi, X.; Zhang, H.; Fu, B. Mitotherapy for Fatty Liver by Intravenous Administration of Exogenous Mitochondria in Male Mice. Front. Pharmacol. 2017, 8, 241.
- Shi, X.; Bai, H.; Zhao, M.; Li, X.; Sun, X.; Jiang, H.; Fu, A. Treatment of Acetaminophen-Induced Liver Injury with Exogenous Mitochondria in Mice. Transl. Res. 2018, 196, 31–41.
- Liu, X.; Khouri-Farah, N.; Wu, C.H.; Wu, G.Y. Targeted Delivery of Mitochondria to the Liver in Rats. J. Gastroenterol. Hepatol. 2020, 35, 2241–2247.
- Ko, S.; Chen, Y.; Sung, P.; Chiang, J.Y.; Chu, Y.; Huang, C.; Huang, C.; Yip, H. Hepatic 31 P-magnetic Resonance Spectroscopy Identified the Impact of Melatonin-pretreated Mitochondria in Acute Liver Ischaemia-reperfusion Injury. J. Cell. Mol. Med. 2020, 24, 10088–10099.
- Ulger, O.; Kubat, G.B.; Cicek, Z.; Celik, E.; Atalay, O.; Suvay, S.; Ozler, M. The Effects of Mitochondrial Transplantation in Acetaminophen-Induced Liver Toxicity in Rats. Life Sci. 2021, 279, 119669.
- Zhao, Z.; Hou, Y.; Zhou, W.; Keerthiga, R.; Fu, A. Mitochondrial Transplantation Therapy Inhibit Carbon Tetrachloride-induced Liver Injury through Scavenging Free Radicals and Protecting Hepatocytes. Bioeng. Transl. Med. 2021, 6, 69–83.
- Su, Y.; Zhu, L.; Yu, X.; Cai, L.; Lu, Y.; Zhang, J.; Li, T.; Li, J.; Xia, J.; Xu, F.; et al. Mitochondrial Transplantation Attenuates Airway Hyperresponsiveness by Inhibition of Cholinergic Hyperactivity. Theranostics 2016, 6, 1244–1260, Erratum in Theranostics 2019, 9, 1385–1386.
- Fu, A.; Hou, Y.; Yu, Z.; Zhao, Z.; Liu, Z. Healthy Mitochondria Inhibit the Metastatic Melanoma in Lungs. Int. J. Biol. Sci. 2019, 15, 2707–2718.
- Moskowitzova, K.; Orfany, A.; Liu, K.; Ramirez-Barbieri, G.; Thedsanamoorthy, J.K.; Yao, R.; Guariento, A.; Doulamis, I.P.; Blitzer, D.; Shin, B.; et al. Mitochondrial Transplantation Enhances Murine Lung Viability and Recovery after Ischemia-Reperfusion Injury. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2020, 318, L78–L88.
- de Carvalho, L.R.P.; Abreu, S.C.; de Castro, L.L.; Andrade da Silva, L.H.; Silva, P.M.; Vieira, J.B.; Santos, R.T.; Cabral, M.R.; Khoury, M.; Weiss, D.J.; et al. Mitochondria-Rich Fraction Isolated From Mesenchymal Stromal Cells Reduces Lung and Distal Organ Injury in Experimental Sepsis. Crit. Care Med. 2021, 49, e880–e890.
- Hsu, C.-H.; Roan, J.-N.; Fang, S.-Y.; Chiu, M.-H.; Cheng, T.-T.; Huang, C.-C.; Lin, M.-W.; Lam, C.-F. Transplantation of Viable Mitochondria Improves Right Ventricular Performance and Pulmonary Artery Remodeling in Rats with Pulmonary Arterial Hypertension. J. Thorac. Cardiovasc. Surg. 2022, 163, e361–e373.
- Huang, P.-J.; Kuo, C.-C.; Lee, H.-C.; Shen, C.-I.; Cheng, F.-C.; Wu, S.-F.; Chang, J.-C.; Pan, H.-C.; Lin, S.-Z.; Liu, C.-S.; et al. Transferring Xenogenic Mitochondria Provides Neural Protection against Ischemic Stress in Ischemic Rat Brains. Cell Transpl. 2016, 25, 913–927.
- Zhang, Z.; Ma, Z.; Yan, C.; Pu, K.; Wu, M.; Bai, J.; Li, Y.; Wang, Q. Muscle-Derived Autologous Mitochondrial Transplantation: A Novel Strategy for Treating Cerebral Ischemic Injury. Behav. Brain Res. 2019, 356, 322–331.
- Xie, Q.; Zeng, J.; Zheng, Y.; Li, T.; Ren, J.; Chen, K.; Zhang, Q.; Xie, R.; Xu, F.; Zhu, J. Mitochondrial Transplantation Attenuates Cerebral Ischemia-Reperfusion Injury: Possible Involvement of Mitochondrial Component Separation. Oxidative Med. Cell. Longev. 2021, 2021, 1006636.
- Nakamura, Y.; Lo, E.H.; Hayakawa, K. Placental Mitochondria Therapy for Cerebral Ischemia-Reperfusion Injury in Mice. Stroke 2020, 51, 3142–3146.
- Orrego, M.A.; Levy, S.; Kelly, C.; Arroyo, G.; Toribio, L.; García, H.H.; Walker, M. Procedimiento Para La Infusión de Mitocondrias Autólogas Por La Arteria Carótida En El Cerebro Porcino. Rev. Peru. Med. Exp. Salud Pública 2021, 38, 345–351.
- Pourmohammadi-Bejarpasi, Z.; Roushandeh, A.M.; Saberi, A.; Rostami, M.K.; Toosi, S.M.R.; Jahanian-Najafabadi, A.; Tomita, K.; Kuwahara, Y.; Sato, T.; Roudkenar, M.H. Mesenchymal Stem Cells-Derived Mitochondria Transplantation Mitigates I/R-Induced Injury, Abolishes I/R-Induced Apoptosis, and Restores Motor Function in Acute Ischemia Stroke Rat Model. Brain Res. Bull. 2020, 165, 70–80.
- Hosseini, L.; Karimipour, M.; Seyedaghamiri, F.; Abolhasanpour, N.; Sadigh-Eteghad, S.; Mahmoudi, J.; Farhoudi, M. Intranasal Administration of Mitochondria Alleviated Cognitive Impairments and Mitochondrial Dysfunction in the Photothrombotic Model of MPFC Stroke in Mice. J. Stroke Cerebrovasc. Dis. 2022, 31, 106801.
- Zhang, B.; Gao, Y.; Li, Q.; Sun, D.; Dong, X.; Li, X.; Xin, W.; Zhang, J. Effects of Brain-Derived Mitochondria on the Function of Neuron and Vascular Endothelial Cell After Traumatic Brain Injury. World Neurosurg. 2020, 138, e1–e9.
- Chang, J.-C.; Wu, S.-L.; Liu, K.-H.; Chen, Y.-H.; Chuang, C.-S.; Cheng, F.-C.; Su, H.-L.; Wei, Y.-H.; Kuo, S.-J.; Liu, C.-S. Allogeneic/Xenogeneic Transplantation of Peptide-Labeled Mitochondria in Parkinson’s Disease: Restoration of Mitochondria Functions and Attenuation of 6-Hydroxydopamine–Induced Neurotoxicity. Transl. Res. 2016, 170, 40–56.e3.
- Shi, X.; Zhao, M.; Fu, C.; Fu, A. Intravenous Administration of Mitochondria for Treating Experimental Parkinson’s Disease. Mitochondrion 2017, 34, 91–100.
- Chang, J.-C.; Chao, Y.-C.; Chang, H.-S.; Wu, Y.-L.; Chang, H.-J.; Lin, Y.-S.; Cheng, W.-L.; Lin, T.-T.; Liu, C.-S. Intranasal Delivery of Mitochondria for Treatment of Parkinson’s Disease Model Rats Lesioned with 6-Hydroxydopamine. Sci. Rep. 2021, 11, 10597.
- Robicsek, O.; Ene, H.M.; Karry, R.; Ytzhaki, O.; Asor, E.; McPhie, D.; Cohen, B.M.; Ben-Yehuda, R.; Weiner, I.; Ben-Shachar, D. Isolated Mitochondria Transfer Improves Neuronal Differentiation of Schizophrenia-Derived Induced Pluripotent Stem Cells and Rescues Deficits in a Rat Model of the Disorder. Schizophr. Bull. 2018, 44, 432–442.
- Nitzan, K.; Benhamron, S.; Valitsky, M.; Kesner, E.E.; Lichtenstein, M.; Ben-Zvi, A.; Ella, E.; Segalstein, Y.; Saada, A.; Lorberboum-Galski, H.; et al. Mitochondrial Transfer Ameliorates Cognitive Deficits, Neuronal Loss, and Gliosis in Alzheimer’s Disease Mice. J. Alzheimer’s Dis. 2019, 72, 587–604.
- Zhang, Z.; Wei, D.; Li, Z.; Guo, H.; Wu, Y.; Feng, J. Hippocampal Mitochondrial Transplantation Alleviates Age-Associated Cognitive Decline via Enhancing Wnt Signaling and Neurogenesis. Comput. Intell. Neurosci. 2022, 2022, 9325302.
- Bobkova, N.V.; Zhdanova, D.Y.; Belosludtseva, N.V.; Penkov, N.V.; Mironova, G.D. Intranasal Administration of Mitochondria Improves Spatial Memory in Olfactory Bulbectomized Mice. Exp. Biol. Med. 2022, 247, 416–425.
- Ma, H.; Jiang, T.; Tang, W.; Ma, Z.; Pu, K.; Xu, F.; Chang, H.; Zhao, G.; Gao, W.; Li, Y.; et al. Transplantation of Platelet-Derived Mitochondria Alleviates Cognitive Impairment and Mitochondrial Dysfunction in Db/Db Mice. Clin. Sci. 2020, 134, 2161–2175.
- Wang, Y.; Ni, J.; Gao, C.; Xie, L.; Zhai, L.; Cui, G.; Yin, X. Mitochondrial Transplantation Attenuates Lipopolysaccharide- Induced Depression-like Behaviors. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2019, 93, 240–249.
- Javani, G.; Babri, S.; Farajdokht, F.; Ghaffari-Nasab, A.; Mohaddes, G. Mitochondrial Transplantation Improves Anxiety- and Depression-like Behaviors in Aged Stress-Exposed Rats. Mech. Ageing Dev. 2022, 202, 111632.
- Lin, M.-W.; Fang, S.-Y.; Hsu, J.-Y.C.; Huang, C.-Y.; Lee, P.-H.; Huang, C.-C.; Chen, H.-F.; Lam, C.-F.; Lee, J.-S. Mitochondrial Transplantation Attenuates Neural Damage and Improves Locomotor Function After Traumatic Spinal Cord Injury in Rats. Front. Neurosci. 2022, 16, 800883.
- Gollihue, J.L.; Patel, S.P.; Eldahan, K.C.; Cox, D.H.; Donahue, R.R.; Taylor, B.K.; Sullivan, P.G.; Rabchevsky, A.G. Effects of Mitochondrial Transplantation on Bioenergetics, Cellular Incorporation, and Functional Recovery after Spinal Cord Injury. J. Neurotrauma 2018, 35, 1800–1818.
- Fang, S.-Y.; Roan, J.-N.; Lee, J.-S.; Chiu, M.-H.; Lin, M.-W.; Liu, C.-C.; Lam, C.-F. Transplantation of Viable Mitochondria Attenuates Neurologic Injury after Spinal Cord Ischemia. J. Thorac. Cardiovasc. Surg. 2021, 161, e337–e347.
- Zhao, Z.; Yu, Z.; Hou, Y.; Zhang, L.; Fu, A. Improvement of Cognitive and Motor Performance with Mitotherapy in Aged Mice. Int. J. Biol. Sci. 2020, 16, 849–858.
- Nascimento-dos-Santos, G.; de-Souza-Ferreira, E.; Lani, R.; Faria, C.C.; Araújo, V.G.; Teixeira-Pinheiro, L.C.; Vasconcelos, T.; Gonçalo, T.; Santiago, M.F.; Linden, R.; et al. Neuroprotection from Optic Nerve Injury and Modulation of Oxidative Metabolism by Transplantation of Active Mitochondria to the Retina. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2020, 1866, 165686.
- Yan, C.; Ma, Z.; Ma, H.; Li, Q.; Zhai, Q.; Jiang, T.; Zhang, Z.; Wang, Q. Mitochondrial Transplantation Attenuates Brain Dysfunction in Sepsis by Driving Microglial M2 Polarization. Mol. Neurobiol. 2020, 57, 3875–3890.
- Adlimoghaddam, A.; Benson, T.; Albensi, B.C. Mitochondrial Transfusion Improves Mitochondrial Function Through Up-Regulation of Mitochondrial Complex II Protein Subunit SDHB in the Hippocampus of Aged Mice. Mol. Neurobiol. 2022, 59, 6009–6017.
- Norat, P.; Soldozy, S.; Sokolowski, J.D.; Gorick, C.M.; Kumar, J.S.; Chae, Y.; Yağmurlu, K.; Prada, F.; Walker, M.; Levitt, M.R.; et al. Mitochondrial Dysfunction in Neurological Disorders: Exploring Mitochondrial Transplantation. NPJ Regen. Med. 2020, 5, 22.
- Jacoby, E.; Bar-Yosef, O.; Gruber, N.; Lahav, E.; Varda-Bloom, N.; Bolkier, Y.; Bar, D.; Blumkin, M.B.-Y.; Barak, S.; Eisenstein, E.; et al. Mitochondrial Augmentation of Hematopoietic Stem Cells in Children with Single Large-Scale Mitochondrial DNA Deletion Syndromes. Sci. Transl. Med. 2022, 14, eabo3724.
- Rehman, M.U.; Sehar, N.; Dar, N.J.; Khan, A.; Arafah, A.; Rashid, S.; Rashid, S.M.; Ganaie, M.A. Mitochondrial Dysfunctions, Oxidative Stress and Neuroinflammation as Therapeutic Targets for Neurodegenerative Diseases: An Update on Current Advances and Impediments. Neurosci. Biobehav. Rev. 2022, 144, 104961.
- Nascimento-dos-Santos, G.; de-Souza-Ferreira, E.; Linden, R.; Galina, A.; Petrs-Silva, H. Mitotherapy: Unraveling a Promising Treatment for Disorders of the Central Nervous System and Other Systemic Conditions. Cells 2021, 10, 1827.
- Picone, P.; Nuzzo, D. Promising Treatment for Multiple Sclerosis: Mitochondrial Transplantation. Int. J. Mol. Sci. 2022, 23, 2245.
- Kadry, H.; Noorani, B.; Cucullo, L. A Blood–Brain Barrier Overview on Structure, Function, Impairment, and Biomarkers of Integrity. Fluids Barriers CNS 2020, 17, 69.
- Rodríguez-Nuevo, A.; Zorzano, A. The Sensing of Mitochondrial DAMPs by Non-Immune Cells. Cell Stress 2019, 3, 195–207.
- Rickenbach, C.; Gericke, C. Specificity of Adaptive Immune Responses in Central Nervous System Health, Aging and Diseases. Front. Neurosci. 2022, 15, 806260.
- Lin, L.; Xu, H.; Bishawi, M.; Feng, F.; Samy, K.; Truskey, G.; Barbas, A.S.; Kirk, A.D.; Brennan, T.V. Circulating Mitochondria in Organ Donors Promote Allograft Rejection. Am. J. Transplant. 2019, 19, 1917–1929.
- Pollara, J.; Edwards, R.W.; Lin, L.; Bendersky, V.A.; Brennan, T.V. Circulating Mitochondria in Deceased Organ Donors Are Associated with Immune Activation and Early Allograft Dysfunction. JCI Insight 2018, 3, e121622.
More
