Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Rita Xu and Version 1 by Nirmal Mazumder.
Cancer is one of the dreaded diseases to which a sizeable proportion of the population succumbs every year. Circulating tumour cells (CTCs) have grabbed the attention of researchers in the detection of metastasis and there has been a huge surge in the surrounding research activities. Acting as a biomarker, CTCs prove beneficial in a variety of aspects. Nanomaterial-based strategies have been devised to have a tremendous impact on the early and rapid examination of tumor cells.
- circulating tumour cells
- metastasis
- nanotechnology
- nanomaterials
1. Introduction
Cancer is a broad term that often refers to a set of diseases characterized by uncontrolled cell growth with the ability to spread throughout the animal body [1]. It is one of the major causes of mortality globally, affecting patients from all socioeconomic backgrounds and their families. According to World Health Organization (WHO) statistics, 18.1 million new cases and 9.6 million deaths occurred in 2018 due to cancer, with secondary cancer or metastasis accounting for more than 90% of them [2,3][2][3]. The growing prevalence of cancer has encouraged the development of rapid and sensitive techniques for early detection. Currently, several methods, such as detecting the levels of metabolites or tumour markers in the blood, biopsy followed by histological evaluation, ultrasonography, magnetic resonance imaging, computerised tomography, positron emission tomography, and endoscopic-based examination are available [4]. The majority of the techniques are routinely used for cancer diagnosis and disease monitoring at a later stage only after the appearance of initial symptoms. As cellular and molecular sciences advance, various indicators and markers for early cancer detection are being investigated. Biomarkers based on gene and RNA expression are regularly used for the expeditious evaluation of cancers [5,6,7][5][6][7]. However, the requirements of extensive facilities and expensive instruments have limited its diagnostic application. On the other hand, dissemination of circulating tumour cells (CTCs) from primary tumours due to metastasis—which then spreads into the lymphatic nodes and blood, has been seen as an important marker for early diagnosis of cancer [8]. They stay either as a single cell or in a group, which can move from one site to another causing tumour [9] The early invasion of CTCs along with fast progression is vital for accurate monitoring, and detection of the tumour cells and an important part of liquid biopsy [10,11][10][11]. Detection and genomic characterization of CTCs can also help to investigate the mechanism of metastasis [12,13][12][13]. Liquid biopsy, often referred to as fluid biopsy or liquid phase biopsy, is a non-invasive diagnostic method for the analysis of non-solid biological samples such as blood. It overcomes the limitation of the inability to obtain heterogeneous information with traditional methods and helps in the identification of both genetic and epigenetic causes related to any diseases [14]. Liquid biopsy is not a routine test in clinical practice; however, over the past few years, it has evolved as a promising tool for the effective and early detection, monitoring of recurrence and evaluation treatment efficacy of cancer. The early description of circulating free DNA (cf DNA) and RNA in 1948, without knowing in detail about it, is said to be the first step towards the biopsy [15]. Ever since, various other biomarkers have also been looked into, for effective disease management (Figure 1). Micro-RNA, cf DNA, circulating tumour DNA, metabolites, CTCs, exosomes and other extracellular vesicles are some of the markers which have been significantly worked upon. Researches isolate these markers from blood and analyze them by different biochemical and analytical methods such as next-generation sequencing, polymerase chain reactions and mass spectrometry-based genotyping assay [7,16][7][16]. With the advancement of cancer treatment towards a shift to personalized medicine, this developing technology has the potential to change and/or complement functional cancer care. The advancement of liquid biopsy procedures has resulted in the incorporation of extremely effective approaches for the rapid detection and separation of CTCs [17], with nanotechnology at the forefront. Due to the unique physicochemical properties of nanomaterials emerging from their high surface area, shape, size, and optical nature, nanotechnology has proven to be the most reliable strategy in cancer diagnosis [18]. Higher surface-to-volume ratio property allows strong binding of the ligands which helps in recognizing critical biomarker molecules. As such, with the advent of this strategy, CTCs isolation and detection with higher efficiency and specificity, even at extremely low numbers in the circulatory system, has recently been achievable, allowing for early cancer diagnosis [19].
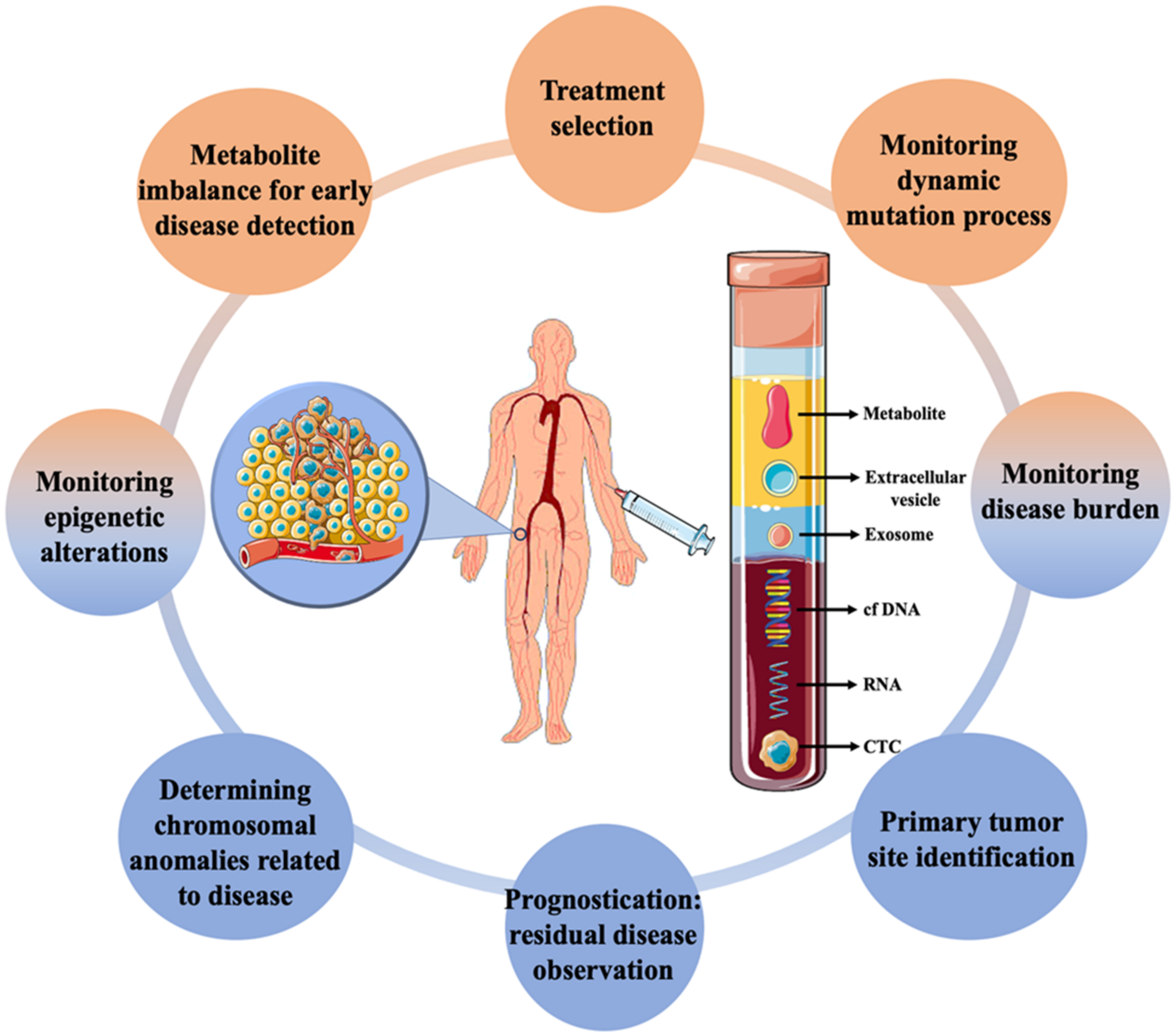
Figure 1. Applications of liquid biopsy in cancer diagnosis along with the different biomarkers commonly targeted using the technique.
2. Biogenesis of CTCs
CTCs are a rare subset of cells that are released from primary tumours and act as the precursor for the growth of additional cancerous tissue in a different location in the body [20]. The hallmarks of cancer such as invasiveness and motility give rise to the ability of malignant tumours to be metastatic. This is devised to be the main factor for the formation and release of CTCs in the circulatory system [21]. Though the formation and progression of cancer is a complex and little understood process, with the development of modern tools, this can be explored. The metastasis and carcinogenesis of tumour cells are commonly described to be a multi-step process, as shown in Figure 2 [22]. Initially, the cell loses its normal cell cycle ability, whether being triggered by internal factors or due to extremal stimulus. This causes the tumour cells to accumulate numerous mutations, prompting them to become malignant [23]. In most cases, it is not the original tumour that causes fatalities, but the spread of malignant cells to other organs. Once the primary tumour grows to a certain size, the available nutrients and oxygen become limited for the continuously dividing cells. This triggers the formation of hypoxia-inducing factors (HIFs). HIFs, with other pathways in the tumours microenvironment, trigger the expression of angiogenesis-promoting factors such as VEGF and angiopoietin-1 and 2, promoting neovascularization around the tumour [24,25][24][25]. Further, the expression of HIFs, angiogenesis, and other factors such as laminin have been shown to reduce the expression of E-cadherin through the pleiotropic effect [26]. The suppressed expression of E-cadherin in the tumour microenvironment reduces cell-to-cell adhesion and increases the mobility of the tumor cells. This begins the process of epithelial-to-mesenchymal transition (EMT). It is observed that changes in the expression of makers and molecules on its surface, such as altered expression of extracellular matrix (ECM)-cell adhesion molecules, suppress expression of epithelial markers, and increase expression of mesenchymal markers [27,28][27][28]. EMT is often considered to be the most accepted hypothesis regulating the formation of CTCs. However, this process is highly complex and still unknown. After the EMT process, the tumour cells adopt a motile, invasive phenotype leading to their detachment from the primary tumour and migration into the neighboring tissue, also known as local invasion. The process is regulated by complex mechanistic changes in cytoskeleton structure, proteolysis of surrounding tissue and alteration in cell–ECM dynamics [29]. Thereafter, some of the cancer cells evade into the network of blood vessels formed around the primary tumor body by intravasation. This process allows cancer cells to initiate metastases at a distant, different locations within the animal body. The cancer cells in the circulation (CTCs) have the ability to evade anoikis due to their mesenchymal properties and also due to the expression of anoikis inhibitors (such as XIAP) in the sub-populations of the cells, thereby resulting in the cells being resistant to apoptosis [30]. The CTCs in circulation are believed to be non-proliferative in nature and travel around the body until they adhere to the capillary bed of a different organ due to their molecular properties or size. Therefore, it is observed that secondary tumours can be formed at distant organs from the original site of the primary neoplasm causing the most fatal malignancies [31]. In addition, it is worth mentioning that these cancer cells also take part in local metastasis, thereby often reaching the lymph nodes that make the disease more severe. Once the cancer cells are attached to the capillary surface, they penetrate the endothelial layer of the blood vessel, thereby moving into the host organ. Then the cells undergo another round of phenotypic alteration almost similar to EMT which is known as mesenchymal to epithelial transition [32,33][32][33]. Though this process is less understood and is still being studied, it is observed that the cancer cells regain their epithelial characteristics, which helps them to acclimatize to new surroundings. Apart from the EMT-mediated motility of cancer cells in the circulation, the non-EMT-associated scenarios have also been known to give rise to CTCs. Centrosome amplification is one of them, in which the E-cadherin expression is maintained throughout the process of CTC formation. The centrosome is sought to play the role of organizing flagella and cilla, apart from also facilitating the separation of chromosomes. Thus, the centrosome has been shown to induce the propagation and motility of cancer cells [34]. Another method is by the non-EMT-mediated dissemination to form CTCs clusters, in which 2–50 cells are clustered together by intracellular adhesion as they move across the vessels. These cells have a higher probability of surviving anoikis than individual circulating cancer cells [35]. This contributes to a better sustainable metastasis and increases the likelihood of extravasation in secondary sites. As not all CTCs lose their epithelial properties, it is also necessary to consider these properties for the effective detection of cells in the blood. It should also be noted that cancer cells might enter the blood following the surgical or microscopic procedures to remove the tumour (such as in case of minimum residual disease) in addition to the naturally occurring CTCs from primary or secondary neoplasms [36,37][36][37].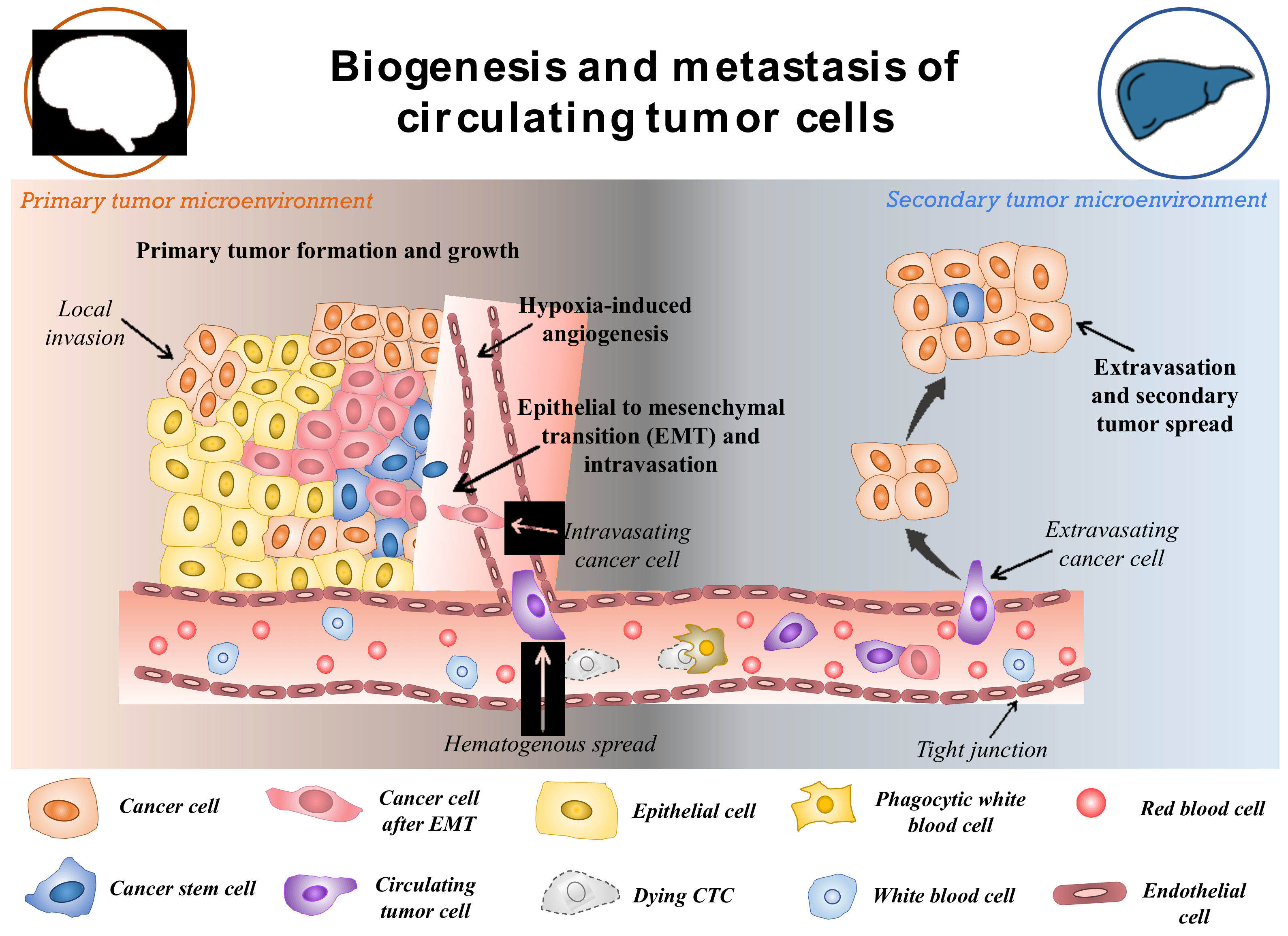
Figure 2. Schematic representation of potential biogenesis and the transport mechanism of circulating tumor cells. Reproduced from [38].
3. Nanotechnology
Nanotechnology has been utilized in various biological research to carry out with high-throughput, sensitivity, selectivity, and specificity and it possesses a high capacity to measure multiplexed systems. Due to their smaller size, they possess a high surface-to-volume ratio, which increases their efficiency towards cellular binding capacity; they are utilized in cancer research which has led to the advancement in significant detection, isolation and enrichment of CTCs from the blood sample. The nanoparticles that are implemented in the analysis of CTCs are classified into various types depending upon their type, shape, and structure. Some of them include quantum dots/fluorescent nanoparticles, surface-enhanced Raman scattering nanoparticles and many more. Each of them has its unique advantages and disadvantages, as discussed in Table 1, which make them suitable for cancer research.Table 1. Different types of nanomaterials used for the detection of CTCs.
| Types of Nanoparticles | Properties | Advantages | Utilization in CTC Analysis | References | |||||
|---|---|---|---|---|---|---|---|---|---|
| Quantum Dots | Tunable narrow fluorescence emission Large absorption coefficient High brightness High photostability High quantum yield Longer fluorescence lifetime |
High specificity High sensitivity |
Detection of CTCs with high metastatic potential Quantified detection of CTCs in the blood vessel. |
[53,54,55,56] | [53][54][55][56] | ||||
| SERS Nanoparticle | Requires a single source of excitation Display minimized photobleaching |
Helps in efficient detection of even a single cell Development of ultrasensitive probes with high specificity | Enrichment, detection, multicolour imaging and enumeration of CTCs | [55,57, | 55][57] | 58, | [58] | 59] | [[59] |
| Magnetic Nanoparticle | High cellular binding capability Outstanding stability in blood Capability to attach large number of MNPs to a single cell without any aggregation. |
Low cost High stability |
Immunomagnetic Separation, enrichment, and detection of CTCs | [55,60,61, | [60][61 | 62] | [55]][62] | ||
| Conductive Nanoparticles/CNTs | Extraordinary mechanical strength High electrical conductivity Conductivity varies with change in the chemical binding |
Extraordinary electrical conductivity Outstanding heat conductivity |
Electronic detection of CTCs with low expression of protein without any enrichment process by real-time electrical impedance sensing method | [54,55,63,64] | [54][55][63][64] | ||||
| Upconversion Nanoparticles | Strong and sharp emission spectra High penetration depth Large Stokes shifts Low background signal High resistance to photobleaching |
High photo stability High thermal stability |
High sensitivity bio-detection and imaging of CTCs | [54,55,65, | 54][55] | 66, | [65] | 67] | [[66][67] |
| Metallic nanoparticles/Plasmonic nanoparticles | Collective coherent oscillation of the electrons at resonance High amount of absorption and scattering of light at resonance |
Biocompatibility High chemical stability Minimum toxicity |
Colometric based detection of CTCs in blood | [54, | [ | 68, | 54 | 69,70] | ][68][69][70] |
3.1. Quantum Dots (QDs)/Fluorescent Nanoparticles
QDs are zero-dimensional fluorescent nanoparticles, where all the dimensions of the particles are less than 100 nm. They possess a semiconductor core covered by a shell as shown in Figure 3a. They are generally made up of elements from groups II to VI or III to V of the periodic table. They pose discrete energy levels with varying bandgap depending upon their size, which ranges between 2–10 nm, which is less than the size of the exciton Bohr radius. They display unique electronic and optical properties such as higher quantum yield, longer fluorescence lifetime, large absorption coefficient, high-quality brightness, excellent photostability, and narrow and tunable fluorescence emission which lies between visible to infrared wavelengths. Due to these properties, QDs have led to the development of modern-day probes which have high specificity and sensitivity [53,54,55,56,71][53][54][55][56][71].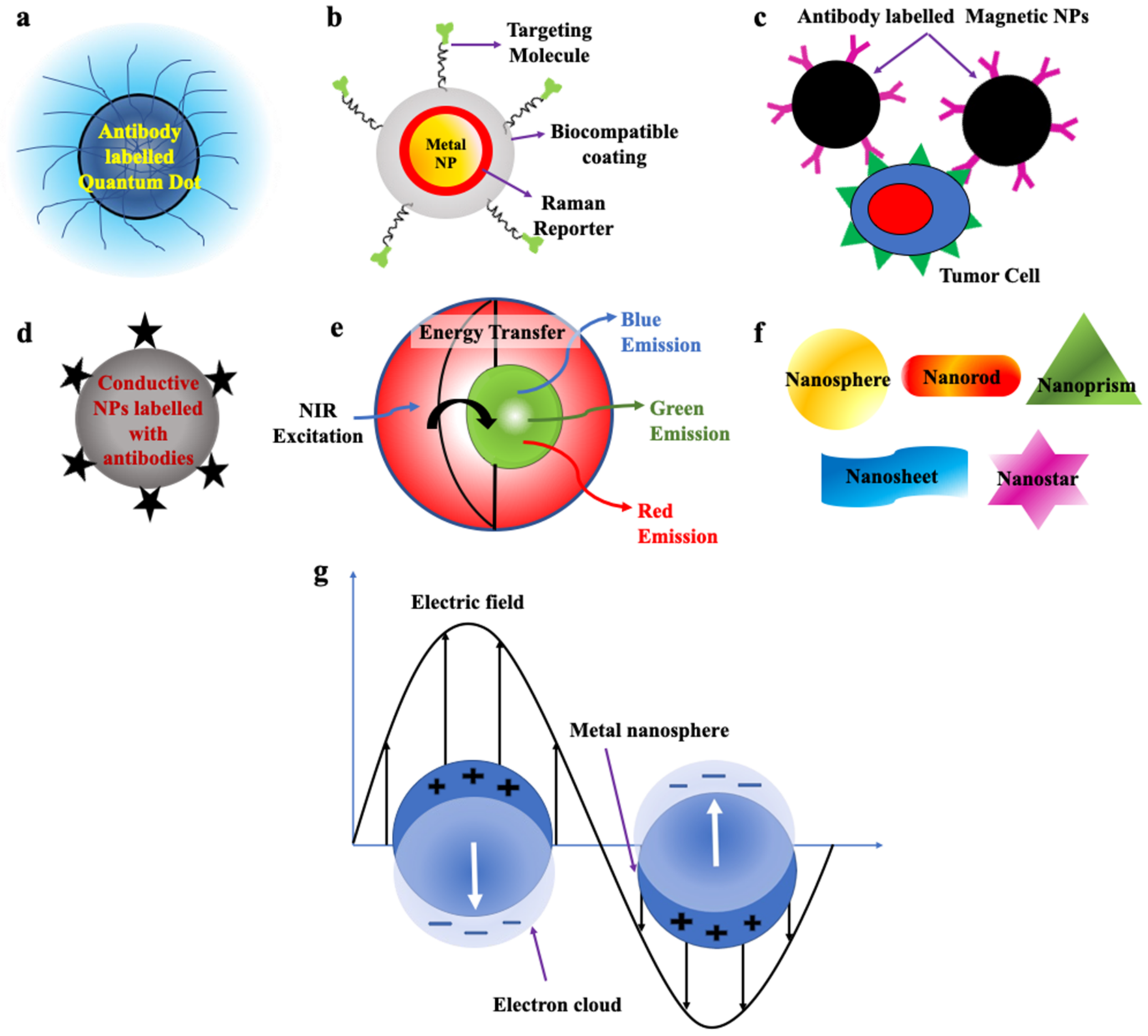
Figure 3. Schematic representation of (a) Quantum dot labelled with antibodies (Redrawn after [72]). (b) SERS nanoparticles (Redrawn after [73]). (c) Magnetic Nanoparticles labelled with antibodies (Redrawn after [62]). (d) Conductive nanoparticles. (e) Upconversion nanoparticles (Redrawn after [74]). (f) Nanoparticles of various morphologies used to detect CTCs. (g) Metallic nanoparticle displaying surface plasmon resonance (Redrawn after [75]).
3.2. Surface-Enhanced Raman Scattering Nanoparticles (SERS NPs)
SERS nanoparticles are formed as a result of absorbance of the analyte, e.g., organic dyes on the surface of plasmonic metal nanoparticles which results in significant enhancement of their Raman signals due to the amplification of electromagnetic field upon excitation by an external light source, which helps in detection of even single molecule (Figure 3b). Its development has led to the foundation of new-generation ultrasensitive probes with high specificity and minimized photobleaching. Probes fabricated by these NPs can be used in biomedical imaging, disease detection and diagnosis [55,85][55][85]. By conjugating it with antibodies, it can be used in enrichment, multicolour imaging and enumeration of even a single cell of CTCs [57,58,59,85,86][57][58][59][85][86].3.3. Magnetic Nanoparticles (MNPs)
Magnetic nanoparticles (MNPs) are shown in Figure 3c. They commonly pose a core–shell structure with a magnetic core that is covered by a magnetically disordered shell. They are generally made up of elements such as cobalt (Co), iron (Fe), nickel (Ni) and ferrite oxide (Fe3O4) possessing magnetic properties. When the external magnetic field is passed through them, under saturation conditions, all the magnetic moments of the particles align along the direction of the applied field. MNPs can display ferromagnetic or superparamagnetic behavior which can be changed by altering their morphology, structure and composition, which helps in their separation. If the size of the NPs is less than 20 nm, then after removal of the external field, due to strong thermal fluctuations magnetic nanoparticles display superparamagnetic behaviour, which possesses no residual magnetization in absence of a magnetic field. Whereas, if the size of the NPs is higher than this limit then some amount of residual magnetization is present in them and they are classified as ferromagnetic NPs [55,61,62][55][61][62]. Their unique properties such as high cellular binding capability, outstanding stability in blood and their capacity to attach a large number of MNPs to a single cell without any aggregation make them very useful for immunomagnetic separation, enrichment and detection of CTCs from blood. These NPs bind easily to the cells and, hence, can be used for in vitro separation under the presence of an externally applied magnetic field [53,60,61,62,87,88][53][60][61][62][87][88].3.4. Conductive Nanoparticles/Carbon Nanotubes (CNTs)
Carbon nanotubes (CNTs) are one-dimensional nanostructures, due to the propagation of electrons only along the axis of the nanotube. They possess a hollow cylindrical core composed of single or multiple sheets of graphite (Figure 3d). Due to the strong carbon bonds in them and their cylindrical symmetry, they possess extraordinary mechanical, unique structural, and outstanding electrical properties. Their conductivity can be varied with a change in chemical binding and structure which makes them an excellent choice for the fabrication of biological sensors. If the CNTs are made up of multi-walled graphite, their surface area and electrical conductivity increased and, thus, can be utilized in the electronic detection of CTCs by the method of real-time electrical impedance sensing. When the CNTs bind with CTCs their conductivities are decreased by around 60%. They are mainly utilized to detect CTCs which have low protein expression without going through any enrichment process [54,55,63,64,89][54][55][63][64][89].3.5. Up Conversion Nanoparticles (UCNPs)
UCNPs made up of lanthanide ions resulted in the breakthrough discovery of next-generation fluorophores, bearing the capability to convert two or more low-energy incident photons in the near-infrared region to a single photon emission with relatively higher energy in the visible region resulting in fluorescence emission. This conversion of energy occurs via a nonlinear optical process as shown in Figure 3e. The extraordinary properties displayed by them such as strong and sharp emission spectra not only prevent damage the normal tissues but also facilitate deep penetration [55,66][55][66]. Their unique luminescent properties, large Stokes shifts, low background signals, and less photobleaching, are used for high-sensitivity detection and imaging of CTCs [54,55,65,66,67][54][55][65][66][67].3.6. Nanostructures of Various Shapes
The shape and size of the nanostructural probes greatly influence their properties and thus, their sensitivity. The most popularly utilised shapes of the nanostructure substrates are nanosphere, nanorod, nanosheet, nanoprism and nanostars (Figure 3f) [54,67,90][54][67][90]. Some properties associated with these nanostructures are enhanced cell capture and binding affinity which enables them to functionalise targeting ligands to be utilised for capturing and enrichment of CTCs by ligand–antigen binding. The shape and size of the NPs also affect the enhanced permeability and retention effect (EPR) [54,67,90,91,92,93,94,95][54][67][90][91][92][93][94][95].3.7. Metallic Nanoparticles/Surface Plasmonic Nanoparticles
Metallic NPs display unique optical properties, due to the presence of free conduction band electrons which oscillate collectively when the frequency of the external electromagnetic radiation matches the frequency of oscillation of the conduction band electrons. Under this condition, localized surface plasmon resonance (LSPR) occurs, which results in significant enhancement of absorbance in smaller-sized metallic nanoparticles. (Figure 3g). The resonance condition in each metallic nanoparticle is dependent on its size, morphology, composition, and geometry. Plasmonic NPs such as gold and silver display biocompatibility and high chemical stability with minimum toxicity and hence, can be used for biological purposes [54,70,96][54][70][96]. Colorimetric assays are based on the use of metallic nanoparticles can quantifiably detect the presence of CTCs in blood. The sensitivity of functionalized metallic nanoparticles in the detection of CTCs extends up to several CTCs per mL of blood [54,68,69,97,98][54][68][69][97][98].References
- Weinberg, R.A. The Biology of Cancer, 2nd ed.; Garland Science: New York, NY, USA, 2013.
- World Health Organization. WHO Report on Cancer: Setting Priorities, Investing Wisely and Providing Care for All; World Health Organization: Geneva, Switzerland, 2020.
- Seyfried, T.N.; Huysentruyt, L.C. On the origin of cancer metastasis. Crit. Rev. Oncog. 2013, 18, 43–73.
- National Cancer Institute. How Cancer Is Diagnosed. 17 July 2019. Available online: https://www.cancer.gov/about-cancer/diagnosis-staging/diagnosis (accessed on 7 August 2022).
- Dai, X.; Xiang, L.; Li, T.; Bai, Z. Cancer hallmarks, biomarkers and breast cancer molecular subtypes. J. Cancer 2016, 7, 1281.
- Inamura, K.; Ishikawa, Y. MicroRNA in lung cancer: Novel biomarkers and potential tools for treatment. J. Clin. Med. 2016, 5, 36.
- Shukla, V.; Varghese, V.K.; Kabekkodu, S.P.; Mallya, S.; Chakrabarty, S.; Jayaram, P.; Pandey, D.; Banerjee, S.; Sharan, K.; Satyamoorthy, K. Enumeration of deregulated miRNAs in liquid and tissue biopsies of cervical cancer. Gynecol. Oncol. 2019, 155, 135–143.
- Gerges, N.; Rak, J.; Jabado, N. New technologies for the detection of circulating tumour cells. Br. Med. Bull. 2010, 94, 49–64.
- Micalizzi, D.S.; Maheswaran, S.; Haber, D.A. A conduit to metastasis: Circulating tumor cell biology. Genes Dev. 2017, 31, 1827–1840.
- Xu, C.M.; Tang, M.; Feng, J.; Xia, H.F.; Wu, L.L.; Pang, D.W.; Chen, G.; Zhang, Z.L. A liquid biopsy-guided drug release system for cancer theranostics: Integrating rapid circulating tumour cell detection and precision tumour therapy. Lab Chip 2020, 20, 1418–1425.
- Yang, C.; Xia, B.R.; Jin, W.L.; Lou, G. Circulating tumor cells in precision oncology: Clinical applications in liquid biopsy and 3D organoid model. Cancer Cell Int. 2019, 19, 341.
- Karthick, S.; Pradeep, P.N.; Kanchana, P.; Sen, A.K. Acoustic impedance-based size-independent isolation of circulating tumour cells from blood using acoustophoresis. Lab Chip 2018, 18, 3802–3813.
- Lu, S.; Chang, C.-J.; Guan, Y.; Szafer-Glusman, E.; Punnoose, E.; Do, A.; Suttmann, B.; Gagnon, R.; Rodriguez, A.; Landers, M.; et al. Genomic analysis of circulating tumor cells at the single-cell level. J. Mol. Diagn. 2020, 22, 770–781.
- Alix-Panabières, C.; Pantel, K. Circulating tumor cells: Liquid biopsy of cancer. Clin. Chem. 2013, 59, 110–118.
- Crowley, E.; Di Nicolantonio, F.; Loupakis, F.; Bardelli, A. Liquid biopsy: Monitoring cancer-genetics in the blood. Nat. Rev. Clin. Oncol. 2013, 10, 472–484.
- Palmirotta, R.; Lovero, D.; Cafforio, P.; Felici, C.; Mannavola, F.; Pellè, E.; Quaresmini, D.; Tucci, M.; Silvestris, F. Liquid biopsy of cancer: A multimodal diagnostic tool in clinical oncology. Ther. Adv. Med. Oncol. 2018, 10, 1758835918794630.
- Cai, L.L.; Ye, H.M.; Zheng, L.M.; Ruan, R.S.; Tzeng, C.M. Circulating tumor cells (CTCs) as a liquid biopsy material and drug target. Curr. Drug Targets 2014, 15, 965–972.
- Choi, Y.E.; Kwak, J.W.; Park, J.W. Nanotechnology for early cancer detection. Sensors 2010, 10, 428–455.
- Kowalik, A.; Kowalewska, M.; Góźdź, S. Current approaches for avoiding the limitations of circulating tumor cells detection methods—Implications for diagnosis and treatment of patients with solid tumors. Transl. Res. 2017, 185, 58–84.
- Potdar, P.D.; Lotey, N.K. Role of circulating tumor cells in future diagnosis and therapy of cancer. J. Cancer Metastasis Treat. 2015, 1, 44–56.
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674.
- Petri, B.J.; Klinge, C.M. Regulation of breast cancer metastasis signalling by miRNAs. Cancer Metastasis Rev. 2020, 39, 837–886.
- Weigelt, B.; Peterse, J.L.; Van’t Veer, L.J. Breast cancer metastasis: Markers and models. Nat. Rev. Cancer 2005, 5, 591–602.
- Lodish, H.; Berk, A.; Zipursky, S.L.; Matsudaira, P.; Baltimore, D.; Darnell, J. Molecular Cell Biology, 4th ed.; W. H. Freeman and Company: New York, NY, USA, 2000.
- Nishida, N.; Yano, H.; Nishida, T.; Kamura, T.; Kojiro, M. Angiogenesis in cancer. Vasc. Health Risk Manag. 2006, 2, 213.
- Carmeliet, P.; Jain, R.K. Angiogenesis in cancer and other diseases. Nature 2000, 407, 249–257.
- Kalluri, R.; Weinberg, R.A. The basics of epithelial-mesenchymal transition. J. Clin. Investig. 2009, 119, 1420–1428.
- Jie, X.X.; Zhang, X.Y.; Xu, C.J. Epithelial-to-mesenchymal transition, circulating tumor cells and cancer metastasis: Mechanisms and clinical applications. Oncotarget 2017, 8, 81558.
- Tinhofer, I.; Saki, M.; Niehr, F.; Keilholz, U.; Budach, V. Cancer stem cell characteristics of circulating tumor cells. Int. J. Radiat. Biol. 2014, 90, 622–627.
- Liu, H.; Zhang, X.; Li, J.; Sun, B.; Qian, H.; Yin, Z. The biological and clinical importance of epithelial–mesenchymal transition in circulating tumor cells. J. Cancer Res. Clin. Oncol. 2015, 141, 189–201.
- Leber, M.F.; Efferth, T. Molecular principles of cancer invasion and metastasis. Int. J. Oncol. 2009, 34, 881–895.
- Hamilton, G.; Rath, B. Mesenchymal-epithelial transition and circulating tumor cells in small cell lung cancer. Isol. Mol. Charact. Circ. Tumor Cells 2017, 994, 229–245.
- Martin, T.A.; Ye, L.; Sanders, A.J.; Lane, J.; Jiang, W.G. Cancer invasion and metastasis: Molecular and cellular perspective. In Madame Curie Bioscience Database; Landes Bioscience: Austin, TX, USA, 2013.
- Godinho, S.A.; Picone, R.; Burute, M.; Dagher, R.; Su, Y.; Leung, C.T.; Polyak, K.; Brugge, J.S.; Théry, M.; Pellman, D. Oncogene-like induction of cellular invasion from centrosome amplification. Nature 2014, 510, 167–171.
- Aceto, N.; Bardia, A.; Miyamoto, D.T.; Donaldson, M.C.; Wittner, B.S.; Spencer, J.A.; Yu, M.; Pely, A.; Engstrom, A.; Zhu, H.; et al. Circulating tumor cell clusters are oligoclonal precursors of breast cancer metastasis. Cell 2014, 158, 1110–1122.
- Cristofanilli, M.; Budd, G.T.; Ellis, M.J.; Stopeck, A.; Matera, J.; Miller, M.C.; Reuben, J.M.; Doyle, G.V.; Allard, W.J.; Terstappen, L.W.M.M.; et al. Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N. Engl. J. Med. 2004, 351, 781–791.
- Kasimir-Bauer, S.; Bittner, A.K.; König, L.; Reiter, K.; Keller, T.; Kimmig, R.; Hoffmann, O. Does primary neoadjuvant systemic therapy eradicate minimal residual disease? Analysis of disseminated and circulating tumor cells before and after therapy. Breast Cancer Res. 2016, 18, 20.
- Chen, L.; Bode, A.M.; Dong, Z. Circulating Tumor Cells: Moving Biological Insights into Detection. Theranostics 2017, 7, 2606–2619.
- Cristofanilli, M.; Hayes, D.F.; Budd, G.T.; Ellis, M.J.; Stopeck, A.; Reuben, J.M.; Doyle, G.V.; Matera, J.; Allard, W.J.; Miller, M.C.; et al. Circulating Tumor Cells: A Novel Prognostic Factor for Newly Diagnosed Metastatic Breast Cancer. J. Clin. Oncol. 2005, 23, 1420–1430.
- Thorsteinsson, M.; Jess, P. The clinical significance of circulating tumor cells in non-metastatic colorectal cancer—A review. Eur. J. Surg. Oncol. 2011, 37, 459–465.
- Alix-Panabières, C.; Pantel, K. Challenges in circulating tumour cell research. Nat. Rev. Cancer 2014, 14, 623–631.
- Krog, B.L.; Henry, M.D. Biomechanics of the Circulating Tumor Cell Microenvironment. Adv. Exp. Med. Biol. 2018, 1092, 209–233.
- Hou, H.W.; Warkiani, M.E.; Khoo, B.L.; Li, Z.R.; Soo, R.A.; Tan, D.S.-W.; Lim, W.-T.; Han, J.; Bhagat, A.A.S.; Lim, C.T. Isolation and retrieval of circulating tumor cells using centrifugal forces. Sci. Rep. 2013, 3, 1259.
- Bagnall, S.; Byun, S.; Begum, S.; Miyamoto, J.D.T.; Hecht, V.C.; Maheswaran, S.; Stott, S.L.; Toner, M.; Hynes, R.O.; Manalis, S.R. Deformability of Tumor Cells versus Blood Cells. Sci. Rep. 2015, 5, 18542.
- Munz, M.; Baeuerle, P.A.; Gires, O. The emerging role of EpCAM in cancer and stem cell signaling. Cancer Res. 2009, 69, 5627–5629.
- Aktas, B.; Tewes, M.; Fehm, T.; Hauch, S.; Kimmig, R.; Kasimir-Bauer, S. Stem cell and epithelial-mesenchymal transition markers are frequently overexpressed in circulating tumor cells of metastatic breast cancer patients. Breast Cancer Res. 2009, 11, R46.
- Hayashi, N.; Nakamura, S.; Tokuda, Y.; Shimoda, Y.; Yagata, H.; Yoshida, A.; Ota, H.; Hortobagyi, G.N.; Cristofanilli, M.; Ueno, N.T. Prognostic value of HER2-positive circulating tumor cells in patients with metastatic breast cancer. Int. J. Clin. Oncol. 2012, 17, 96–104.
- Miyamoto, D.T.; Lee, R.J.; Stott, S.L.; Ting, D.T.; Wittner, B.S.; Ulman, M.; Smas, M.E.; Lord, J.B.; Brannigan, B.W.; Trautwein, J.; et al. Androgen receptor signaling in circulating tumor cells as a marker of hormonally responsive prostate cancer. Cancer Discov. 2012, 2, 995–1003.
- Allard, W.J.; Matera, J.; Miller, M.C.; Repollet, M.; Connelly, M.C.; Rao, C.; Tibbe, A.G.; Uhr, J.W.; Terstappen, L.W. Tumor cells circulate in the peripheral blood of all major carcinomas but not in healthy subjects or patients with nonmalignant diseases. Clin. Cancer Res. 2004, 10, 6897–6904.
- Haber, D.A.; Velculescu, V.E. Blood-based analyses of cancer: Circulating tumor cells and circulating tumor DNA. Cancer Discov. 2014, 4, 650–661.
- Chang, Y.S.; di Tomaso, E.; McDonald, D.M.; Jones, R.; Jain, R.K.; Munn, L.L. Mosaic blood vessels in tumors: Frequency of cancer cells in contact with flowing blood. Proc. Natl. Acad. Sci. USA 2000, 97, 14608–14613.
- Hanssen, A.; Riebensahm, C.; Mohme, M.; Joosse, S.A.; Velthaus, J.L.; Berger, L.A.; Bernreuther, C.; Glatzel, M.; Loges, S.; Lamszus, K.; et al. Frequency of Circulating Tumor Cells (CTC) in Patients with Brain Metastases: Implications as a Risk Assessment Marker in Oligo-Metastatic Disease. Cancers 2018, 10, 527.
- Valizadeh, A.; Mikaeili, H.; Samiei, M.; Farkhani, S.; Zarghami, N.; Kouh, M.; Akbarzadeh, A.; Davaran, S. Quantum dots: Synthesis, bioapplications, and toxicity. Nanoscale Res. Lett. 2012, 7, 480.
- Huang, Q.; Wang, Y.; Chen, X.; Wang, Y.; Li, Z.; Du, S.; Wang, L.; Chen, S. Nanotechnology-Based Strategies for Early Cancer Diagnosis Using Circulating Tumor Cells as a Liquid Biopsy. Nanotheranostics 2018, 2, 21–41.
- Bhana, S.; Wang, Y.; Huang, X. Nanotechnology for enrichment and detection of circulating tumour cells. Nanomedicine 2015, 10, 1973–1990.
- Michalet, X.; Pinaud, F.; Lacoste, T.D.; Dahan, M.; Bruchez, M.P.; Alivisatos, A.P.; Weiss, S. Properties of fluorescent semiconductor nanocrystals and their application to biological labeling. Single Mol. 2001, 2, 261–276.
- Sha, M.Y.; Xu, H.X.; Natan, M.J.; Cromer, R. Surface-enhanced Raman scattering tags for rapid and homogeneous detection of circulating tumor cells in the presence of human whole blood. J. Am. Chem. Soc. 2008, 130, 17214–17215.
- Zhang, P.; Zhang, R.; Gao, M.; Zhang, X. Novel nitrocellulose membrane substrate for efficient analysis of circulating tumour cells coupled with surface-enhanced Raman scattering imaging. ACS Appl. Mater. Interfaces 2014, 6, 370–376.
- Nima, Z.A.; Mahmood, M.; Xu, Y.; Mustafa, T.; Watanabe, F.; Nedosekin, D.A.; Juratli, M.A.; Fahmi, T.; Galanzha, E.I.; Nolan, J.P.; et al. Circulating tumour cell identification by functionalized silver-gold nanorods with multicolor, super-enhanced SERS and photothermal resonances. Sci. Rep. 2014, 4, 4752.
- Habli, Z.; AlChamaa, W.; Saab, R.; Kadara, H.; Khraiche, M. Circulating Tumour Cell Detection Technologies and Clinical Utility: Challenges and Opportunities. Cancers 2020, 12, 1930.
- Akbarzadeh, A.; Samiei, M.; Davaran, S. Magnetic nanoparticles: Preparation, physical properties, and applications in biomedicine. Nanoscale Res. Lett. 2012, 7, 144.
- Liu, P.; Jonkheijm, P.; Terstappen, L.W.M.M.; Stevens, M. Magnetic Particles for CTC Enrichment. Cancers 2020, 12, 3525.
- Bernholc, J.; Brenner, D.; Nardelli, M.B.; Meunier, V.; Roland, C. Mechanical and electric properties of nanotubes. Annu. Rev. Mater. Res. 2002, 32, 347–375.
- Yue, M.; Yuanyuan, L.; Haiyan, X.; Minghe, L.; Ziwei, L.; Jianhong, C.; Jingxin, M.; Sanjun, S. Circulating Tumor Cells: From Theory to Nanotechnology-Based Detection. Front. Pharmacol. 2017, 8, 35.
- DaCosta, M.; Doughan, S.; Han, Y.; Krul, U. Lanthanide upconversion nanoparticles and applications in bioassays and bioimaging: A review. Anal. Chim. Acta 2014, 832, 1–33.
- Wang, M.; Abbineni, G.; Clevenger, A.; Mao, C.; Xu, S. Upconversion nanoparticles: Synthesis, surface modification and biological applications. Nanomedicine 2011, 7, 710–729.
- Zhang, Y.; Li, M.; Gao, X.; Chen, Y.; Liu, T. Nanotechnology in cancer diagnosis: Progress, challenges and opportunities. J. Hematol. Oncol. 2019, 12, 137.
- Pallares, R.M.; Thanh, N.T.K.; Su, X. Sensing of circulating cancer biomarkers with metal nanoparticles. Nanoscale 2019, 11, 22152–22171.
- Blanco-Formoso, M.; Alvarez-Puebla, R.A. Cancer Diagnosis through SERS and Other Related Techniques. Int. J. Mol. Sci. 2020, 21, 2253.
- Das, U.; Mazumder, N.; Biswas, R. An Appraisal on Plasmonic Heating of Nanostructures. In Recent Advances in Plasmonic Probes; Springer: Cham, Switzerland, 2022; pp. 341–354.
- Kairdolf, B.A.; Smith, A.M.; Stokes, T.H.; Wang, M.D.; Young, A.N.; Nie, S. Semiconductor quantum dots for bioimaging and biodiagnostic applications. Annu. Rev. Anal. Chem. 2013, 6, 143–162.
- Wang, Y.; Hu, A. Carbon quantum Dots: Synthesis, properties and applications. J. Mater. Chem. C. 2014, 2, 6921.
- Wang, Y.; Kang, S.; Doerksen, J.D.; Glaser, A.K.; Liu, J.T. Surgical guidance via multiplexed molecular imaging of fresh tissues labeled with SERS-coded nanoparticles. IEEE J. Sel. Top. Quantum Electron. 2015, 22, 154–164.
- Vetrone, F.; Naccache, R.; Mahalingam, V.; Morgan, C.G.; Capobianco, J.A. The active-core/active-shell approach: A strategy to enhance the upconversion luminescence in lanthanide-doped nanoparticles. Adv. Funct. Mater. 2009, 19, 2924–2929.
- Szunerits, S.; Spadavecchia, J.; Boukherroub, R. Surface plasmon resonance: Signal amplification using colloidal gold nanoparticles for enhanced sensitivity. Rev. Anal. Chem. 2014, 33, 153–164.
- Bruchez, M.; Moronne, M.; Gin, P.; Weiss, S.; Alivisatos, A.P. Semiconductor nanocrystals as fluorescent biological labels. Science 1998, 281, 2013–2016.
- Hsieh, Y.H.; Liu, S.J.; Chen, H.W.; Lin, Y.K.; Liang, K.S.; Lai, L.J. Highly sensitive rare cell detection based on quantum dot probe fluorescence analysis. Anal. Bioanal. Chem. 2010, 396, 1135–1141.
- Winnik, F.M.; Maysinger, D. Quantum dot cytotoxicity and ways to reduce it. Acc. Chem. Res. 2013, 46, 672–680.
- Zhang, H.; Fu, X.; Hu, J.; Zhu, Z. Microfluidic bead-based multienzyme-nanoparticle amplification for detection of circulating tumour cells in the blood using quantum dots labels. Anal. Chim. Acta 2013, 779, 64–71.
- Maeda, Y.; Yoshino, T.; Matsunaga, T. Novel nanocomposites consisting of in vivo-biotinylated bacterial magnetic particles and quantum dots for magnetic separation and fluorescent labelling of cancer cells. J. Mater. Chem. 2009, 19, 6361–6366.
- Gazouli, M.; Lyberopoulou, A.; Pericleous, P.; Rizos, S.; Aravantinos, G.; Nikiteas, N.; Anagnou, N.P.; Efstathopoulos, E.P. Development of a quantum-dot-labelled magnetic immunoassay method for circulating colorectal cancer cell detection. World J. Gastroenterol. 2012, 18, 4419–4426.
- Kuo, C.W.; Chueh, D.Y.; Chen, P. Real-time in vivo imaging of subpopulations of circulating tumor cells using antibody conjugated quantum dots. J. Nanobiotechnology 2019, 17, 26.
- Myung, J.H.; Tam, K.A.; Park, S.J.; Cha, A.; Hong, S. Recent advances in nanotechnology-based detection and separation of circulating tumour cells. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2016, 8, 223–239.
- Mazumder, N.; Lyn, R.K.; Singaravelu, R.; Ridsdale, A.; Moffatt, D.J.; Hu, C.-W.; Tsai, H.-R.; Mclauchlan, J.; Stolow, A.; Kao, F.-J.; et al. Fluorescence lifetime imaging of alterations to cellular metabolism by domain 2 of the hepatitis C virus core protein. PLoS ONE 2013, 8, e66738.
- Quester, K.; Avalos-Borja, M.; Vilchis-Nestor, A.; Camacho-López, M.; Castro-Longoria, E. SERS Properties of Different Sized and Shaped Gold Nanoparticles Biosynthesized under Different Environmental Conditions by Neurospora crassa Extract. PLoS ONE 2013, 8, e77486.
- Zavaleta, C.L.; Smith, B.R.; Walton, I.; Doering, W.; Davis, G.; Shojaei, B.; Natan, M.J.; Gambhir, S.S. Multiplexed imaging of surface enhanced Raman scattering nanotags in living mice using noninvasive Raman spectroscopy. Proc. Natl. Acad. Sci. USA 2009, 106, 13511–13566.
- Jha, D.K.; Shameem, M.; Patel, A.B.; Kostka, A.; Schneider, P.; Erbe, A.; Deb, P. Simple synthesis of superparamagnetic magnetite nanoparticles as highly efficient contrast agent. Mater. Lett. 2013, 95, 186–189.
- Deb, P.; Basumallick, A.; Chatterjee, P.; Sengupta, S.P. Preparation of α-Fe2O3 nanoparticles from a nonaqueous precursor medium. Scr. Mater. 2001, 45, 341–346.
- Bora, M.; Deb, P. Magnetic proximity effect in two-dimensional van der Waals heterostructure. J. Phys. Mater. 2021, 4, 034014.
- Wang, S.; Wang, H.; Jiao, J.; Chen, K.J.; Owens, G.; Kamei, K.; Sun, J.; Sherman, D.; Behrenbruch, C.; Wu, H.; et al. Three-dimensional nanostructured substrates toward efficient capture of circulating tumour cells. Angew. Chem. Int. Ed. 2009, 48, 8970–8973.
- Zhang, N.; Deng, Y.; Tai, Q.; Cheng, B.; Zhao, L.; Shen, Q.; He, R.; Hong, L.; Liu, W.; Guo, S.; et al. Electrospun TiO2 nanofiber-based cell capture assay for detecting circulating tumour cells from colorectal and gastric cancer patients. Adv. Mater. 2012, 24, 2756–2760.
- Yoon, H.J.; Kim, T.H.; Zhang, Z.; Azizi, E.; Pham, T.; Paoletti, C.; Lin, J.; Ramnath, N.; Wicha, M.; Hayes, D.; et al. Sensitive capture of circulating tumour cells by functionalized graphene oxide nanosheets. Nat. Nanotechnol. 2013, 8, 735–881.
- Chen, G.D.; Fachin, F.; Fernandez-Suarez, M.; Wardle, B.L.; Toner, M. Nanoporous elements in microfluidics for multiscale manipulation of bioparticles. Small 2011, 7, 1061–1067.
- Wu, X.; Xia, Y.; Huang, Y.; Li, J.; Ruan, H.; Chen, T.; Luo, L.; Shen, Z.; Wu, A. Improved SERS-Active Nanoparticles with Various Shapes for CTC Detection without Enrichment Process with Supersensitivity and High Specificity. ACS Appl. Mater. Interfaces 2016, 8, 19928–19938.
- Gogoi, A.; Mazumder, N.; Konwer, S.; Ranawat, H.; Chen, N.T.; Zhuo, G.Y. Enantiomeric recognition and separation by chiral nanoparticles. Molecules 2019, 24, 1007.
- Das, U.; Biswas, R.; Mazumder, N. Elucidating thermal effects in plasmonic metal nanostructures: A tutorial review. Eur. Phys. J. Plus 2022, 137, 1248.
- Paul, D.; Biswas, R. Facile Fabrication of Novel Sensing System for Size Detection of Nanoparticles. IEEE Trans. Nanotechnol. 2018, 17, 596–602.
- Boruah, B.S.; Biswas, R. Selective detection of arsenic (III) based on colorimetric approach in aqueous medium using functionalized gold nanoparticles unit. Mater. Res. Express 2018, 5, 015059.
More
