Carbon-intensive industries must deem carbon capture, utilization, and storage initiatives to mitigate rising CO2 concentration by 2050. A 45% national reduction in CO2 emissions has been projected by government to realize net zero carbon in 2030. CO2 utilization is the prominent solution to curb not only CO2 but other greenhouse gases, such as methane, on a large scale. Thermocatalytic CO2 conversions into clean fuels and specialty chemicals through catalytic CO2 hydrogenation and CO2 reforming using green hydrogen and pure methane sources have been under scrutiny. However, these processes are still immature for industrial applications because of their thermodynamic and kinetic limitations caused by rapid catalyst deactivation due to fouling, sintering, and poisoning under harsh conditions. Therefore, a key research focus on thermocatalytic CO2 conversion is to develop high-performance and selective catalysts even at low temperatures while suppressing side reactions. Conventional catalysts suffer from a lack of precise structural control, which is detrimental toward selectivity, activity, and stability. Core-shell is a emerged nanomaterial that offers confinement effect to preserve multiple functionalities from sintering in CO2 conversions. Substantial progress has been achieved to implement core-shell in direct or indirect thermocatalytic CO2 reactions, such as methanation, methanol synthesis, Fischer–Tropsch synthesis, and dry reforming methane.
- core-shell nanostructures
- CO2 hydrogenation
- dry reforming methane
1. Introduction
2. CO2 Hydrogenation Reactions
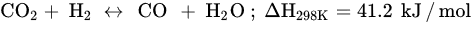
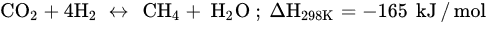
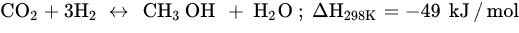
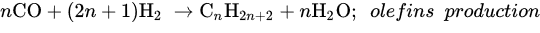
2.1. CO2 Methanation
CO2 methanation, a process of methane production via CO2 hydrogenation, is a pioneering technology that has received extensive recognition for providing solutions toward CO2 emission reduction, renewable energy utilization, and natural gas market reliever [6][79]. Abundant greenhouse gases (GHGs) in the atmosphere can be utilized to yield clean and green fuels via CO2 methanation. Methane (CH4) is the main natural gas formed with higher combustion value, and its combustion products are clean and safe relative to other fossil fuels. CH4 has high energy density and is easy to store, making it an efficient renewable hydrogen energy carrier. The Sabatier reaction (Equation (2)) is a highly exothermic reaction that only occurs completely at low temperatures between 423 K and 573 K and high atmospheric pressure (1 atm) in the presence of a catalyst [7][72] to obtain optimum CO2 conversion and CH4 selectivity. However, the standard heat of CO2 formation is −394.38 kJ/mol, and its high chemical inertness suppresses its activation and molecular dissociation. In addition, at temperatures above 300 °C, RWGS takes over the activity, thereby increasing CO selectivity, decreasing CH4 selectivity, and promoting CO2 conversion.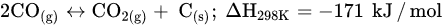 Similar to other CO2 hydrogenation reactions, methanation faces challenges, such as catalyst deactivation due to carbon deposition (fouling, Equation (5)) and decreased activity, resulting in a short catalyst lifetime. In order to prolong the catalyst lifespan and prevent catalyst degradation, CO2 methanation catalysis has received huge attention to develop high-activity catalysts at low temperatures [8][5]. Gao et al. designed an adequate ratio of CO2/H2 and found that a sufficient amount of H2 can significantly influence the production of water vapor (H2O), increase methanation activity rather than CO formation, and inhibit carbon deposition.
Various catalyst preparation methods, including conventional impregnation, sol–gel, and coprecipitation methods, have been developed to discover ways to achieve high activity and selectivity under mild conditions, good stability, and service life of CO2 methanation catalysts. Therefore, suitable active metals, such as Cu, Ni, Fe, Pd, and Co, have been reported for CO2 methanation, and Ni-based catalysts have been selected for industrial commercialization. However, these conventional metal-based catalysts often show low reaction activity at low temperature (180–300 °C) and tend to form particle agglomeration and sintering at high temperatures. For instance, Ni/Al2O3 catalysts prepared by wetness impregnation exhibit low metal dispersion and sinter in the presence of water at high temperatures, thereby increasing CO selectivity and energy consumption. Thus, active Ni metal with low loading is weakly dispersed and becomes less active at low temperatures, whereas the catalyst deactivates at high temperatures due to sintering. Catalyst design has been improved by adding promoters (Fe, Pd, Rh), additives, and support metal oxides (SiO2, ZrO2, CeO2) to enhance activity at low temperatures. However, addition of these components in the conventional method is detrimental toward efficient CO2 adsorption.
Similar to other CO2 hydrogenation reactions, methanation faces challenges, such as catalyst deactivation due to carbon deposition (fouling, Equation (5)) and decreased activity, resulting in a short catalyst lifetime. In order to prolong the catalyst lifespan and prevent catalyst degradation, CO2 methanation catalysis has received huge attention to develop high-activity catalysts at low temperatures [8][5]. Gao et al. designed an adequate ratio of CO2/H2 and found that a sufficient amount of H2 can significantly influence the production of water vapor (H2O), increase methanation activity rather than CO formation, and inhibit carbon deposition.
Various catalyst preparation methods, including conventional impregnation, sol–gel, and coprecipitation methods, have been developed to discover ways to achieve high activity and selectivity under mild conditions, good stability, and service life of CO2 methanation catalysts. Therefore, suitable active metals, such as Cu, Ni, Fe, Pd, and Co, have been reported for CO2 methanation, and Ni-based catalysts have been selected for industrial commercialization. However, these conventional metal-based catalysts often show low reaction activity at low temperature (180–300 °C) and tend to form particle agglomeration and sintering at high temperatures. For instance, Ni/Al2O3 catalysts prepared by wetness impregnation exhibit low metal dispersion and sinter in the presence of water at high temperatures, thereby increasing CO selectivity and energy consumption. Thus, active Ni metal with low loading is weakly dispersed and becomes less active at low temperatures, whereas the catalyst deactivates at high temperatures due to sintering. Catalyst design has been improved by adding promoters (Fe, Pd, Rh), additives, and support metal oxides (SiO2, ZrO2, CeO2) to enhance activity at low temperatures. However, addition of these components in the conventional method is detrimental toward efficient CO2 adsorption.
Core-Shell Nanostructured Catalysts for CO2 Methanation
Core-shell confinement of structure has garnered interest as a potential nanostructure support for CO2 methanation. The activity and selectivity of core-shell structured catalysts are mainly affected by the type of metal used. Among porous shell supports [9][13], metal-organic frameworks (MOFs) are a potential porous shell for Ni confinement because of their porous crystalline materials, high specific surface area, and tunable uniform elemental distribution. Ni precursors were treated solvothermally in a solvent solution at 136 °C to induce crystallization, and the Ni-MOF-74 suspension is calcined and then reduced in 5% hydrogen to form the NixFe@C catalyst. The obtained catalyst achieves a CO2 conversion of 72.3% with 99.3% CH4 selectivity at 350 °C. Encapsulation of Ni-Fe alloy within carbon porous structure facilitates high CO2 adsorption and effectively prevents the aggregation of active metal NPs during the reaction, thereby conferring the core-shell catalyst with superior stability. Moreover, the homogeneity of Ni-Fe NP elemental distribution can be preserved, which improves Ni dispersion. New core-shell nanostructure based on cobalt (Co) catalysts have been successfully fabricated by Cui et al. [5][78] to study catalytic performance of low temperature methanation. MnO-heterostructured NPs injected into porous graphitic carbon (Co/MnO@PGC) were synthesized via a single-step pyrolysis of bimetal CoMn@MOF-74. The resulting nanocomposite features an enriched Co/MnO heterointerface and exhibits excellent catalytic performance for low-temperature CO2 methanation. The synthesized Co/MnO@PGC catalyst allowed CO2 molecules to activate faster at a low heat of 160 °C over 99% selectivity with high STYCH4 of 0.14 μmolCH4⋅s −1 gcat−1. As the temperature reached 240 °C, CO2 conversion and space–time yield (STYCH4) rose to 32.1% and 13.34 μmolCH4⋅gcat−1⋅s−1, respectively. At a high pressure (30 bar), STYCH4 can reach up to 5.60 μmolCH4⋅s−1⋅gcat−1 at 160 °C, which is even comparable to that of the optimal level of Ru-based catalysts. These results indicate that the synergistic interactions between Co and MnO NPs at the Co-MnO heterointerface are responsible for enhancing the catalytic activity toward CH4 production at a low temperature. In addition, the Co/MnO heterostructured NPs encapsulated into PGC play an important role in preventing metal particle aggregation and improving thermal stability. High TOFCH4 suggested that the Co/MnO heterointerface formed inside the PGC of Co/MnO@PGC can significantly boost its activity of low-temperature (160–220 °C) CO2 methanation. Core-shell metal@metal oxide particles can be promising as high thermal-conducting support materials owing to the high thermal conductivity of metal and excellent surface structural properties of the metal oxide itself. Various supports, such as Al2O3 [10][80], CeO2 [11][81], ZrO2, SiO2 [12][60], or zeolites [12][60] have been proposed as metal oxide shell to protect the active metals at the core. In addition, Le et al. [13][82] synthesized a Ni/Al@Al2O3 CSN catalyst for CO and CO2 methanation by using hydrothermal surface oxidation (HTSO). Ni/Al@Al2O3 has selectively yielded carbonate and formate species, which suppress the CO intermediate. The confinement effect helped the CSN catalyst lower the activation energy barriers (74 kJ/mol), which outperforms the activation energy of conventional catalysts, namely, Ni/Al2O3 (80 kJ/mol) and Ni/SiO2 (89 kJ/mol). Apparently, Ni/Al@Al2O3 CSN can successfully enhance the catalytic CO2 adsorption owing to its high Ni dispersion and strong CO2 binding. Meanwhile, Ilsemann et al. [12][60] prepared Co@SiO2 and Co@Silicalite-1 catalysts via a solvothermal method to encapsulate the Co NPs inside two mesoporous structures of silica shells. They found that Co@SiO2 improves the catalytic activity in low-temperature CO2 methanation (230°C–400 °C) by suppressing the side reaction (RWGS), which results in highly selective CO2 hydrogenation to methane. The thermal stability provided by mesoporous silica could preserve the active Co metals at elevated temperatures. However, CO methanation causes slight coking, resulting in a shift of kinetic stability and reduction in methane yield. Similarly, a silicalite-1-confined Ni catalyst was prepared through the selective desilication of the molecular sieve to produce extra voids and pore channels to cage Ni in the crystal [14][83]. The Ni@Silicalite-1 catalyst is characterized by higher CO2 conversion and CH4 selectivity than conventional Ni/Silicalite, which can be attributed to the higher Ni fine dispersion in the void of silicate. The catalyst maintains stable performance over 50 h at 450 °C. CSNs are expected to provide an anti-sintering effect by anchoring the active metal NPs in the porous channel. The formation of metal–metal oxide compounds modulates strong interaction to realize the low deactivation rate of reaction. However, this core-shell design has some shortcomings, such as complex preparation, expensive instrumentation, and lack of exposed defect surface, all of which restrict its industrial applications. Therefore, Yang et al. [11][81] prepared Ni-phyllosilicate@CeO2 CSN by using a hydrothermal method, to create Ni fine dispersion (3.3–6.3 nm). The anchored Ni phyllosilicate could further increase the H2 and CO2 uptake, contributing to high CO2 conversion rate (65%), thus exhibiting high catalytic activity and stability for 100 h lifetime CO2 methanation. Le et al. [15][84] continued their work on the Ni/Al@M-Al2O4 core-shell catalyst by promoting various transition metals (M = Mg, Ni, Co, Zn, or Mn) to develop a synergistic interaction between M/Al and enhance the catalytic activity of the CSN in low-temperature CO2 methanation. Different influences of thermal conductivity on shell NP dispersion were observed as an anti-sintering property. Ni/Al@M-Al2O4 CSNs were prepared using deposition-precipitation (DP) and wet impregnation (WI) methods to facilitate superior heat conductivity and surface properties for highly exothermic and endothermic reactions and control the effect of parameters on metal particle size. Morphological analyses showed that 9 wt% Ni/Al@MnAl2O4 (DP) has a significant BET surface area of 129 m2/g and the highest Ni metal dispersion (9.7%) among other synthesized catalysts. Introduction of Mg into the spinel Ni/Al@MgAl2O4 CSN has provided a larger BET surface area of 171 m2/g with the same dispersion quality to Ni/Al@MnAl2O4. Ni/Al@MnAl2O4 (DP) and Ni/Al@MgAl2O4 (DP) have demonstrated better catalytic performance than the catalyst prepared using the WI method. Both catalysts selectively produce high methane yield by hindering more chain of hydrocarbon while facilitating 90% CO2 conversion under 300 °C, thereby promoting high catalytic activity for CO2 methanation. Bimetallic Ni-Al has facilitated functional heat transfer across Ni/Al@M-Al2O4 CSN catalyst particulates as the Al metal releases high heat conductivity. Interestingly, Ni/Al@MnAl2O4 shows superior catalytic stability because it has a lifetime of 50 h while preventing coke deposition and Ni particle agglomeration during CO2 methanation. Considering the sharp rise in temperature of methanation and rapid catalyst deactivation by Ni particles, Wang et al. [16][85] suggested the interaction of Mg with Ni as a bimetallic core in a Ni/Mg@MCM-41 duo-core@shell catalyst prepared using an in situ hydrothermal method with different Mg contents. Wang et al. also synthesized a conventional Ni/MCM-41 by using WI for comparison of catalytic performance. They found that Ni/MCM-41 features a larger BET surface area of 622.5 m2/g than 0.05 wt% Ni/Mg@MCM-41, and the specific surface area continues to decrease as an additional 0.05 wt% Ni is incorporated. This result can be ascribed to the blockage of Mg particles on the pores when Mg2+ has less tendency to replace Si4+ ions in the SiO2 lattice. However, the Ni/Mg@MCM41 core-shell catalyst produces better atomic composition than the conventional Ni/MCM41 catalyst. Under an optimal temperature of 360 °C, 0.05 wt% Ni/Mg@MCM-41 exhibits the highest CO2 conversion at 88% during CO2 methanation. The selectivity of CH4 gradually decreases with increasing temperature, proving that high temperatures are not advantageous to CH4 production because CO2 methanation is an exothermic reaction. Regardless, the 0.05 wt% Ni/Mg@MCM-41 catalyst shows high catalytic activity at low temperatures (below 360 °C) and a large specific surface area (606.3 m2/g), which are suitable for CO2 methanation. A well-defined nanostructured Ni@SiO2 core-shell catalyst (diameter size of 27.1 nm) was synthesized with distinct metal–metal oxide interfaces in proximity to each other to carry out CO2 hydrogenation [17][86]. Noteworthy, the Ni@SiO2 interface in the catalyst is responsible for RWGS reaction to form CO selectively. The strong interaction between Ni core and SiO2 shell effectively restrains NP growth (agglomeration) and carbon deposition. Thus, the Ni@SiO2 core-shell catalyst, yields 89.8% CH4 and successfully converts 99.0% of CO molecules. Moreover, it retains high catalytic stability in CO methanation under a 100 h lifetime condition, which surpasses the stability of the conventional Ni/SiO2 catalyst, whose CO conversion collapses after 12 h lifetime. Ni@mpCeO2 CSN was synthesized using nanocasting, followed by strong electrostatic adsorption for CO2 methanation [18][74]. The turn of frequency (TOF) for the Ni/mpCeO2 CSN catalyst (0.183 s−1) at 225 °C is threefold higher than that of the Ni catalyst supported on conventional CeO2 prepared using the same method. Compared with the Ni catalyst, the Ni/mpCeO2 CSN provides a rich NiCeO2 interface with more oxygen vacancies, playing a key role in CO2 activation. CO2 activation over the Ni/mpCeO2 CSN catalyst occurs through combined associative and dissociative mechanisms that have been observed through DRIFT mechanism study. Ni NPs are highly dispersed in the channels of mpCeO2, which enhance H2 dissociation, thereby supplying sufficient *H species for the formation of CO and *HCO intermediate species owing to high CH4 selectivity. In addition to enhanced low-temperature activity and selectivity, the Ni/mpCeO2 catalyst maintains its stability 70 h on stream because Ni sintering has been suppressed by the confinement effect of mesoporous CeO2 structure. This study demonstrates the importance of the Ni-CeO2 interface, at which high oxygen vacancy concentration facilitates CO2 adsorption and activation while the adjacent Ni active sites accelerate H2 dissociation.2.2. CO2 Hydrogenation to Methanol
Among all possible products in the CO2 hydrogenation reaction mentioned in the previous section, methanol (MeOH) is the most attractive (Equation (3)). MeOH is a clean, biodegradable, high-energy fuel and is highly versatile as it can easily generate other valuable fuels, such as DME, olefins, hydrocarbons, and long-chain alcohols [19][20][21][27,87,88]. Furthermore, combustion of MeOH generates few carbon side products owing to its freezing point (−96 °C). Thus, MeOH is suitable as a hydrogen carrier without producing a huge number of SOx or NOx. Unlike methanation, MeOH synthesis has thermodynamic and kinetic limitations, such as its high-pressure requirement for complete CO2 activation and low reaction temperature to selectively enhance the methanol yield [22][23][46,89] Nevertheless, this reaction still produces a competing side reaction, RWGS. Therefore, a potential route to suppress CO production must be discovered to maintain high methanol selectivity. CO2 hydrogenation to MeOH is favored at high pressures. Therefore, stable catalysts that are resilient to high temperatures and pressures are required. Active transition metals (Ni, Ru, Ga, Cu, and Co) are usually used for CO2 hydrogenation to MeOH because of their high activity at certain temperatures, abundance, low cost, and different oxidation states/phases to improve catalytic stability and selectivity. However, such active metals can encounter rapid deactivation (sintering, fouling, and poisoning) because methanol synthesis is a naturally structure-sensitive reaction, thus limiting CO2 activation. Cu-based thermal catalysts are those used most often and hold many advantages for effective commercialization at the industrial scale. Cu catalysts are strongly active toward high methanol selectivity because of their three oxidation states (Cu0, Cu1+, and Cu2+) [24][25][26][90,91,92]. Cu provides active sites for H2 dissociation, and metal oxides increase the number of active sites for CO2 activation. Wang et al. reported that methanol selectivity corresponds to the proportion of strong basic sites to the total basic sites. Despite their benefits, Cu NPs agglomerate into large particles at elevated temperatures, which decrease MeOH yield. Therefore, understanding the changes in different surface atom arrangements on Cu NPs is important because the homogeneity of the catalyst structure affects the catalytic activity.Core-Shell Nanostructured Catalyst for CO2 Hydrogenation to Methanol
CSN catalysts are attractive for these conversions because of the impact of the shell materials on reaction selectivity and catalyst stability [27][93] The core-shell nanostructure and surface have many advantages, such as enhancement of the essential properties of conventional catalysts. For example, commercialized Cu/ZnO/Al2O3 catalysts are unstable under high partial pressure at elevated temperatures, accelerating steam in the reaction atmosphere, hence causing rapid agglomeration and undesired crystal growth [19][28][29][30][3,27,94,95] A core-shell arrangement may optimize the interaction between the metal and porous support and minimize Cu sintering by creating unique multifunctionalities of catalytic sites. An et al. [31][96] investigated Cu NPs coated with Zn to boost the surface electronic concentration and surface adsorption from high bimetallic synergy. The Cu/Zn bimetallic particles were anchored within the MOF network, and CuZn@UiO-bpy CSN was suggested to enhance the active metal dispersion and the SMSI. Cu NPs form within a diameter of 0.5–2 nm, depicting that the NPs are homogeneously dispersed, thus spreading more active sites and catalytic activity. Such structural properties result in remarkable CO2 conversion (17.4%) and methanol selectivity (85.6%). In addition, CuZnO@UiO-bpy CSN shows three times higher methanol yield at 250 °C than the conventional catalyst. This report agrees with the findings of Tisseraud et al.’s compilation study [32][33][34][97,98,99] where Cu@ZnOx CSN catalysts exhibit 100% methanol selectivity as the oxygen deficiency formed by Zn migration provides active sites and hinders CO formation by side reactions (RWGS or MeOH decomposition). A recent study has prepared a CuIn@mSiO2 core-shell catalyst using a two-step solvothermal synthesis [35][57]. This catalyst was compared internally with Cu@SiO2, In@SiO2, and conventionally prepared CuIn/SiO2 catalysts. A perfect core-shell shape has been successfully synthesized. The activity results showed that CuIn@SiO2 outperforms the others in terms of CO2 conversion but exhibits the second lowest methanol selectivity (21.8%) owing to CO formation at 250 °C. Nevertheless, the most stable performance over 100 h is dominated by Cu@SiO2 with zero sign of carbon deposits. Considering that mesoporous silica (mSiO2) shells are highly effective in limiting metal agglomeration and preserving the original metal particle sizes by providing a layer of thermally stable surface, Yang et al. [36][100] embedded Cu/ZnO within a layer of mSiO2 and achieved stable CO2 conversion and methanol yield over 160 h lifetime at a low temperature (260 °C). They observed that Cu/ZnO@mSiO2 shows better catalytic performance than the conventional impregnated catalyst Cu/ZnO/SiO2, which is deactivated after only 20 h on stream. Apart from tuning the product selectivity and preventing active sites sintering, the core-shell catalyst can help fix and activate CO2 molecular activation. Hydroxyl species, which can be obtained from transition metal phyllosilicate (TM@SiO2p), enhance CO2 hydrogenation. Jangam et al. [29][94] prepared Cu-SiO2p via a hydrothermal method and compared its performance with conventionally impregnated Cu-SiO2 for CO2 hydrogenation to MeOH at 200–350 °C. Cu-SiO2p reduced at 225 °C produces a stable CO2 conversion and methanol selectivity of 3.5% and 77%, respectively. Recently, hollow Cu@ZrO2 derived from a MOF network has been developed through pyrolysis for selective CO2 hydrogenation to methanol [37][101]. The hollow structure provides easy access of CO2 and H2 to diffuse on active sites. Han et al. found that the basic sites of Cu-ZrO2 interfaces are responsible for the main adsorption and activation sites of CO2. The core-shell confinement structure yields a high methanol selectivity of 85% at 220 °C. Other than Cu-based catalysts, noble metals, such as Pd-based heterogeneous catalysts also received recognition for CO2 hydrogenation. The electronic structure of Pd-based catalysts plays significant roles in the reaction because its metallic sites can be tuned to obtain high-stability catalysts. However, their applications for large-scale plants are limited by sintering and expensive source. Xiao et al. [38][102] have recently designed stale Pd NPs in a confined environment. Pd@Cu core-shell was confined within a layered double hydroxide through modified coprecipitation. They conducted a catalytic test on formate species formation to identify the feasible CO2 hydrogenation pathway without forming CO intermediates. Pd0.4@CuMgAlOx with a CO2/H2 composition of 20:20 successfully yielded 5.68 mmol∙g1∙h-1 of formate, whereas the core-shell catalyst showed no significant loss in formate yield after the fourth cycle, confirming its excellent stability. Kinetically, the Pd metallic sites govern the H2 dissociation by forming active Pd-H.3. CO2-Reforming Reactions
Although renewable H2 production via electrolytic, photoelectrochemical (PEC), and solar thermochemical methods is promising, cost effective, abundant, and sustainable, it is still not viable for industrial commercialization. Thus, thermocatalytic hydrogen production, which is a nonrenewable route that is effective for rich H2 production from biomass, is still relevant in the gas production market. The reforming of fossil fuels, especially natural gasses, via decomposition of hydrocarbon molecules to release H2 is the most common source for H2 production globally. Traditionally, H2 can be produced via several processes, such as steam methane reforming, partial oxidation reforming, methane pyrolysis, coal gasification, and DRM.3.1. CO2 Dry Reforming of Methane
Unlike the abovementioned methods, DRM offers low operating cost, utilizes two hazard greenhouse gases (CO2 and CH4) to produce highly pure gas (CO/H2), and allows easy processing of value-added hydrocarbons and chemicals via the Fischer–Tropsch process [39][104]. DRM is a more economical process relative to other methods because it eases the gas separation of final products. CO2 utilization has a significant influence on DRM performance, considering that the adsorption isotherm and its activation are the main steps to achieve optimal H2 production.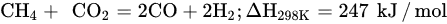
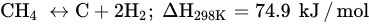 DRM is an endothermic reaction that requires excessive heating (>700 °C) driven by the following main reaction (Equation (6)). Poisoning, catalyst deactivation, and coke deposition are the common issues faced in DRM at high temperature because of methane decomposition (Equation (7)), Boudouard reaction (Equation (5)), and CO2 hydrogenation (Equation (1)), whereas the RGWS reaction usually occurs in DRM at temperatures below 800 °C. To limit the RWGS reaction, DRM must operate at high temperatures, approximately 900 °C, to achieve high yields of H2 and CO. Hence, key factors of efficient and feasible DRM reaction are optimized temperature, pressure, CH4/CO2 ratio, and catalyst design and composition.
An effective DRM reaction mechanism theoretically involves multidisciplinary transitional states, such as methane dissociative adsorption, CO2 dissociative adsorption, hydroxyl group formation, and intermediate oxidation and desorption [40][105]. In detail, CH4 gases must dissociate on the catalyst surface sites to complete their tetravalency, whereby CH3 molecules are adsorbed on top of active metal atoms while another CH2 molecule occurs between two metal atoms, called step sites. Then, another greenhouse gas, CO2, breaks its double bond, where C-O is adsorbed on the surface between the metal and support, leaving one oxygen atom exposed. Later, the H2 molecules migrate from the metal particles to the support atoms to form hydroxyl (−OH) species at temperatures below 800 °C. Finally, the metal-surface oxygen provided by high oxygen mobility support reacts with the S-CHx species to form new S-CHxO intermediates, which potentially form as CO and H2. This kinetic reaction of DRM is influenced by surface electronic properties.
Efforts have been exerted to develop novel catalysts that can increase CO2 and CH4 activities at low temperatures. Nickel is the most prevalent metal-based catalyst because of its abundancy and low cost, making it suitable for industrial catalytic processes; however, Ni is rapidly deactivated because of high carbon formation from side reactions, either methane cracking or CO disproportionation [40][41][105,106] Noble metals, such as Pd [41][106], Ru, and Pt [42][43][44][45][107,108,109,110], are suggested to replace Ni because they are highly resistant to carbon formation. However, their expensiveness restricts their promising properties in the larger market. Hence, catalysts that minimize coke formation and preserve the active sites in DRM need to be developed.
Core-shell catalysts may offer high thermal stability, sintering resistance, and several functionalities, which aid in reducing the rate of carbon deposition [46][47][111,112]. The core-shell structure also promotes good control of dispersion and preservation from metal agglomeration, resulting in enhanced catalyst stability. Porous materials with high thermal resistance and optimum porous size channels that could anchor the active metal core are ideal to stabilize the metal NPs and minimize metal sintering at elevated temperatures [48][113].
DRM is an endothermic reaction that requires excessive heating (>700 °C) driven by the following main reaction (Equation (6)). Poisoning, catalyst deactivation, and coke deposition are the common issues faced in DRM at high temperature because of methane decomposition (Equation (7)), Boudouard reaction (Equation (5)), and CO2 hydrogenation (Equation (1)), whereas the RGWS reaction usually occurs in DRM at temperatures below 800 °C. To limit the RWGS reaction, DRM must operate at high temperatures, approximately 900 °C, to achieve high yields of H2 and CO. Hence, key factors of efficient and feasible DRM reaction are optimized temperature, pressure, CH4/CO2 ratio, and catalyst design and composition.
An effective DRM reaction mechanism theoretically involves multidisciplinary transitional states, such as methane dissociative adsorption, CO2 dissociative adsorption, hydroxyl group formation, and intermediate oxidation and desorption [40][105]. In detail, CH4 gases must dissociate on the catalyst surface sites to complete their tetravalency, whereby CH3 molecules are adsorbed on top of active metal atoms while another CH2 molecule occurs between two metal atoms, called step sites. Then, another greenhouse gas, CO2, breaks its double bond, where C-O is adsorbed on the surface between the metal and support, leaving one oxygen atom exposed. Later, the H2 molecules migrate from the metal particles to the support atoms to form hydroxyl (−OH) species at temperatures below 800 °C. Finally, the metal-surface oxygen provided by high oxygen mobility support reacts with the S-CHx species to form new S-CHxO intermediates, which potentially form as CO and H2. This kinetic reaction of DRM is influenced by surface electronic properties.
Efforts have been exerted to develop novel catalysts that can increase CO2 and CH4 activities at low temperatures. Nickel is the most prevalent metal-based catalyst because of its abundancy and low cost, making it suitable for industrial catalytic processes; however, Ni is rapidly deactivated because of high carbon formation from side reactions, either methane cracking or CO disproportionation [40][41][105,106] Noble metals, such as Pd [41][106], Ru, and Pt [42][43][44][45][107,108,109,110], are suggested to replace Ni because they are highly resistant to carbon formation. However, their expensiveness restricts their promising properties in the larger market. Hence, catalysts that minimize coke formation and preserve the active sites in DRM need to be developed.
Core-shell catalysts may offer high thermal stability, sintering resistance, and several functionalities, which aid in reducing the rate of carbon deposition [46][47][111,112]. The core-shell structure also promotes good control of dispersion and preservation from metal agglomeration, resulting in enhanced catalyst stability. Porous materials with high thermal resistance and optimum porous size channels that could anchor the active metal core are ideal to stabilize the metal NPs and minimize metal sintering at elevated temperatures [48][113].
