Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Beatrix Zheng and Version 3 by Beatrix Zheng.
Redox equilibria and the modulation of redox signalling play crucial roles in physiological processes. Overproduction of reactive oxygen species (ROS) disrupts the body’s antioxidant defence, compromising redox homeostasis and increasing oxidative stress, leading to the development of several diseases. Manganese superoxide dismutase (MnSOD) is a principal antioxidant enzyme that protects cells from oxidative damage by converting superoxide anion radicals to hydrogen peroxide and oxygen in mitochondria. Systematic studies have demonstrated that MnSOD plays an indispensable role in multiple diseases.
- MnSOD
- oxidative stress
- ROS
- human diseases
- MnSOD mimetics
1. Introduction
Reactive oxygen species (ROS) are a series of molecular oxygen derivatives that regulate numerous physiological and pathological processes [1]. As a group of chemical species, ROS can be categorized into non-radical and free radical species. Non-radical ROS include hydrogen peroxide (H2O2), organic hydroperoxides (ROOH), singlet molecular oxygen (1O2), ozone (O3), hypochlorous acid (HOCl), and hypobromous acid (HOBr), whereas free radical ROS include superoxide anion radical (O2•−), hydroxyl radical (·OH), peroxyl radical (ROO·) and alkoxyl radical (RO·) [2]. Cellular ROS are generated endogenously through mitochondrial oxidative phosphorylation, or exogenously stimulated by xenobiotics, cytokines, and bacterial infection [3][4]. O2•− and H2O2 are acknowledged as the most physiologically relevant ROS. O2•− is mainly generated by nicotinamide adenine dinucleotide phosphate oxidases (NOXs), or the complexes I and III of the mitochondrial electron transport chain (ETC). H2O2 is generated by superoxide dismutases (SOD), NOX4, monoamine oxidases, and xanthine oxidases within a few different organelles (endoplasmic reticulum, mitochondria, and peroxisomes). More importantly, ROS are required for numerous cellular processes including cell growth, differentiation, and death by acting as signalling molecules [5]. Multiple signalling pathways are involved in mediating ROS, including nuclear factor kappa-B (NF-κB), mitogen-activated protein kinase (MAPK) cascade, Kelch-like ECH-associated protein 1-nuclear factor erythroid 2-related factor 2-antioxidant response elements (Keap1-Nrf2-ARE) signalling, adenosine 5′-monophosphate-activated protein kinase (AMPK), phosphoinositide-3 kinase-(PI3K-) Akt pathway, etc. [6][7].
The dynamic balance between ROS production and antioxidant capacity responsibly maintains the cellular redox homeostasis [8]. However, oxidative stress occurs when elevated ROS overwhelms the cellular antioxidant defence, damaging nucleic acids, proteins, and lipids [4][5][9]. To protect cells from oxidative damage as well as to maintain physiological ROS levels, organisms have developed antioxidant defence systems that comprise small-molecular-weight antioxidants and antioxidant enzymes. Small-molecular-weight antioxidants include glutathione (GSH), cysteine, ascorbic acid, and α-tocopherol, while antioxidant enzymes include SOD, catalase (CAT), glutathione peroxidase (GPX), glutaredoxin (GRX), thioredoxin (TXN), and peroxiredoxin (PRX) [10][11][12]. From a pharmacokinetic point of view in scavenging intracellular ROS, targeting antioxidant enzymes is more effective than small-molecule antioxidants, and ultimately maintains the antioxidant defence [13]. A series of antioxidant enzymes such as CAT, GPX, and PRX eliminate H2O2 by converting H2O2 into H2O [14][15][16]. On the other hand, SOD is the most powerful O2•− scavenger, which catalyses the dismutation of O2•− into H2O2 and O2 [17]. Currently, three isoforms of SOD have been discovered in mammalian cells, namely, copper–zinc superoxide dismutase (Cu/ZnSOD, SOD1), manganese superoxide dismutase (MnSOD, SOD2), and the extracellular superoxide dismutase (EcSOD, SOD3) [5]. Of these isoforms, MnSOD garners widespread interest as it potently scavenges mitochondrial O2•−, which is the major source of cellular ROS [18]. The indispensable role of MnSOD is further highlighted by the evidence of significant neonatal mortality in MnSOD-deficient mice, compared to the considerable neonatal survival in the SOD1- or SOD3-deficient mice [19][20][21][22].
MnSOD is a nuclear-encoded enzyme that translocates into the mitochondrial matrix [12]. It is constituted by a homotetramer containing an active site with manganese as a cofactor. Despite its enzymatic antioxidant function, MnSOD readily binds with iron, and the produced iron-substituted enzyme attains peroxidase activity that generates highly reactive hydroxyl radicals [23][24]. Therefore, in protecting against oxidative stress, manganese seems like the perfect metal, while the quaternary structure of MnSOD responsibly maintains the catalytic and dismutase activity [25]. The expression and activity of MnSOD is regulatable at multiple levels, from transcription and translation to posttranslational modifications [26]. NF-κB, specificity protein 1 (Sp1), activating protein-1 (Ap1), p53, and CCAAT binding protein (C/EBP) are the major transcription factors that regulate MnSOD gene expression by directly binding to specific DNA elements or interacting with its partners [27][28]. Furthermore, posttranslational modifications regulate MnSOD protein expression and activity through nitration, phosphorylation, and acetylation. Peroxynitrite (ONOO-) inactivates MnSOD by nitrating the tyrosine-34 amino acid residue of MnSOD protein. Acetylation at lysine-68, lysine-122, lysine-53, and lysine-89 amino acid residues, also inactivate MnSOD. In addition, sirtuin 3 (SIRT3) reactivates MnSOD by deacetylating the above lysine residues in response to irradiation, nutritional deprivation, and oxidative stress [25][27]. Moreover, MnSOD activity and stability are enhanced through phosphorylation at Ser106 by the mitochondrial cell-cycle kinase 1 (Cdk1) [29].
Considering that MnSOD engages in detoxifying ROS through O2•− dismutation, which possibly becomes a contributing factor or consequence in multiple diseases, it is vital to understand the physiological and pathological role of MnSOD. Recent evidence has revealed the pathological involvement of MnSOD in a number of diseases (Figure 1). For example, the enzymatic activity or expression of MnSOD is frequently downregulated in numerous diseases, such as diabetes and neurodegenerative diseases [30][31][32][33], whereas overexpression of MnSOD protects against pro-oxidant insults resulting from inflammatory cytokines, irradiation, hyperoxic injury, and ischaemia/reperfusion [34][35][36][37][38][39][40]. In addition, selective MnSOD mimetics have therapeutic potential in treating rheumatoid arthritis (RA), ischaemic stroke, and kidney diseases by mimicking the activity of MnSOD [41][42][43][44]. Nevertheless, a comprehensive summary of MnSOD regulatory roles in a range of diseases is lacking. In this research, the researchers focus on the mechanisms of MnSOD in regulating various diseases that are associated with imbalanced redox status, including inflammation, fibrotic diseases, diabetes, neurodegenerative diseases, vascular diseases, and cancer (Figure 2). Advances in the therapeutic application of MnSOD activators and MnSOD mimetics are also discussed (Table 1). Moreover, limitations and critical issues in these MnSOD-related studies are addressed. Further understanding of how MnSOD can be regulated in the clinic may provide strategies and prospects for antioxidant therapy. Additionally, the comprehensive information and scientific advances presented in this research may facilitate the understanding of the physiological and pathological roles of MnSOD.
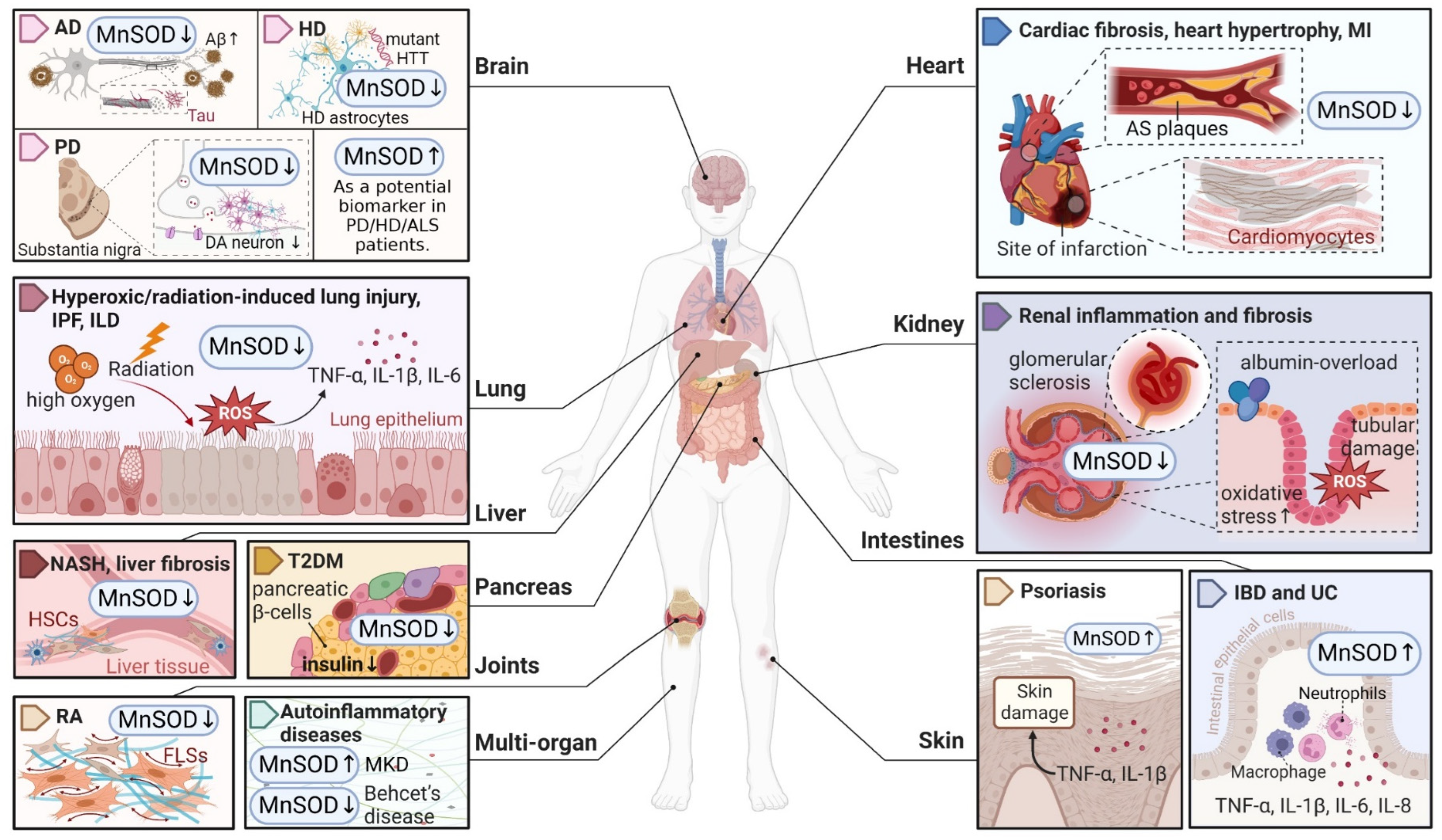
Figure 1. An overview of MnSOD expression in various pathological states of human diseases. Abnormal expression levels of MnSOD have been detected in multiple diseases and have a wide impact on different organs, followed by various pathological alterations such as inflammation, fibrosis, sclerosis, and neuron degeneration. MnSOD, manganese superoxide dismutase; ROS, reactive oxygen species; AD, Alzheimer’s disease; Aβ, amyloid β-protein; HD, Huntington’s disease; HTT, huntingtin; PD, Parkinson’s disease; DA, dopaminergic; ALS, amyotrophic lateral sclerosis; IPF, idiopathic pulmonary fibrosis; ILD, interstitial lung disease; TNF, tumour necrosis factor; IL, interleukin; NASH, non-alcoholic steatohepatitis; HSCs, hepatic stellate cells; T2DM, type 2 diabetes mellitus; RA, rheumatoid arthritis; FLSs, fibroblast-like synoviocytes; MKD, mevalonate kinase deficiency; MI, myocardial infarction; AS, atherosclerosis; IBD, inflammatory bowel disease; UC, ulcerative colitis. ‘↑’ represents increased or upregulated, while ‘↓’ represents decreased or downregulated (created with BioRender.com (accessed on 4 November 2022)).
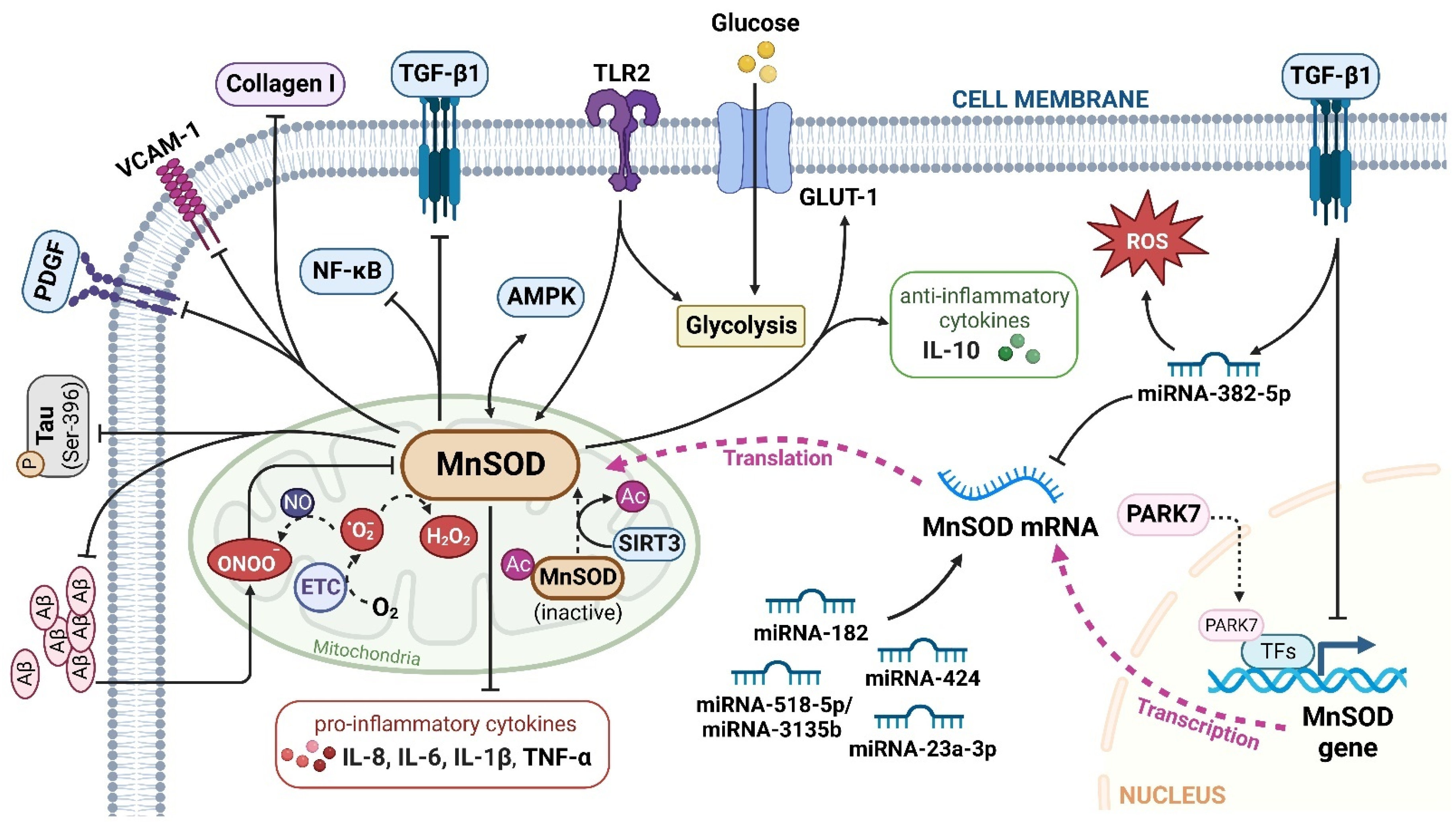
Figure 2. The major signalling pathways involved in MnSOD regulation. MnSOD is localized in the mitochondrial matrix to catalyse the dismutation of O2•− to H2O2, and regulate cellular redox homeostasis. Multiple factors, cytokines, proteins, and miRNAs have been involved in the transcriptional and translational modulation of MnSOD. TGF-β1, transforming growth factor-β1; TLR2, toll-like receptor 2; Aβ, amyloid, β-protein; GLUT-1, glucose transporter member 1; IL, interleukin; VCAM-1, vascular cell adhesion molecule-1; AMPK, adenosine 5′-monophosphate-activated protein kinase; P, phospho; PDGF, platelet-derived growth factor; NF-κB, nuclear factor kappa-B; ETC, electron transport chain; PARK7, Parkinson disease protein 7; TFs, transcriptional factors; Ac, acetyl; SIRT3, sirtuin 3. ‘↑’ represents increased or upregulated, while ‘⊥’ represents suppressed or downregulated (created with BioRender.com (accessed on 4 November 2022)).
Table 1. MnSOD-based therapeutic strategies.
| Type | Treatment | Chemical Structures | Disease | Mechanism | Therapeutic Effect | Ref |
|---|---|---|---|---|---|---|
| Clinical agents | Rebamipide | 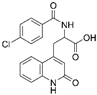 |
Bowel inflammation | ↑MnSOD protein levels ↓superoxide anion leakage |
↓NSAID-induced mucosal injury | [45] |
| Melatonin | 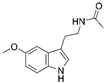 |
Cardiac fibrosis | ↑MnSOD activity ↑SIRT3-mediated MnSOD deacetylation |
↓cardiac dysfunction and fibrosis | [46] | |
| Vitamin C | 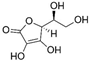 |
Diabetes | ↑MnSOD mRNA levels | protected diabetic tissues | [47] | |
| NAC | 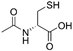 |
HD | ↑MnSOD activity ↓mitochondrial dysfunction |
↓neurobehavioural deficits | [48] | |
| Idebenone | 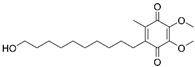 |
AS | ↑SIRT3-MnSOD-mtROS pathway | stabilized atherosclerotic plaques | [49] | |
| Asprosin | N/A | Ischaemic heart disease | ↑ERK1/2-MnSOD signalling ↓mesenchymal stromal cell apoptosis |
↑function of cardiac ejection ↓MI-induced myocardial remodelling |
[50] | |
| Nestorone | 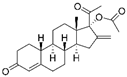 |
Ischaemic stroke | ↑MnSOD protein levels | ↓behavioural and histological stroke outcomes | [51] | |
| Sevoflurane | 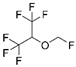 |
Myocardial I/R injury | ↑MnSOD activity and protein levels | ↓myocardial I/R injury | [36][37][38] | |
| Trans sodium crocetinate | 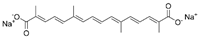 |
|||||
| Irisin | N/A | |||||
| MnSOD mimetics |
ZMTP | N/A | RA | ↑transition of M1 macrophages to M2 phenotype ↓IL-6, IL-1β and TNF-α ↑arginase-1 and IL-10 |
↑antiarthritic efficacy | [41] |
| MnTBAP | 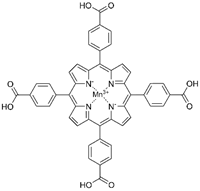 |
Kidney disease | ↓angiotensin converting enzyme/angiotensin II signalling ↓mitochondria-derived oxidative stress |
↓albumin-overload-induced abnormalities | [42][44] | |
| AS | reversed the proatherosclerotic macrophage phenotype | ↓AS progression | [52] | |||
| MnTMPyP | 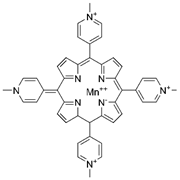 |
AD | ↓ROS during neurodegeneration | protected hippocampal neurons | [53] | |
| PD | ↑MnSOD protein levels ↓DA neurons loss |
↓nigrostriatal DA neurodegeneration | [54] | |||
| Ischaemic stroke | ↓intracellular superoxide radical levels | ↓cerebral infarct volume ↑neurological function |
[43] | |||
| mitoTEMPO | 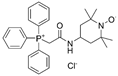 |
Hypertension | ↑reendothelialization capability of endothelial progenitor cells | ↓blood pressure ↑vasorelaxation |
[55][56] | |
| AS | ↓intimal smooth muscle cell apoptosis and matrix degradation | ↓features of atherosclerotic plaque vulnerability | [57] | |||
| Natural compounds | Curcumin | 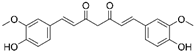 |
Hepatic fibrosis | ↑MnSOD activity and mRNA levels ↓HSC activation and ROS |
↓fibrogenesis | [58][59] |
| Alpha mangostin | 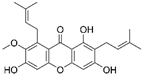 |
|||||
| Hawthorn fruit extract | N/A | AS | ↑MnSOD mRNA levels | ↓atherogenesis | [49] | |
| Proteins | rhMnSOD | N/A | RA | ↑plasma concentration of MnSOD | ↓paw swelling and bone destruction | [60] |
| Hepatic fibrosis | ↓HSC activation | ↓hepatic fibrosis | [61] | |||
| rhMnSOD-Hirudin fusion protein | N/A | IPF | ↓fibroblasts proliferation ↓profibrotic genes (Ctgf, Fn, Col1a1) |
↓lung inflammation and fibrosis | [62] | |
| Oncolytic vaccinia virus | OVV-MnSOD | N/A | Lymphoma | ↑lymphocyte infiltration ↑tumour sensitivity to anti-PD-L1 treatment |
↓tumour progression | [63] |
Note: mtROS, mitochondrial reactive oxygen species; NSAID, nonsteroidal anti-inflammatory drug; SIRT3, sirtuin 3; NAC, N-acetyl-L-cysteine; HD, Huntington’s disease; PD, Parkinson disease; AD, Alzheimer’s disease; PD-L1, programmed death ligand 1; ERK1/2, extracellular signal-regulated kinase 1/2; MI, myocardial infarction; I/R, ischaemia/reperfusion; IL, interleukin; TNF, tumour necrosis factor; DA, dopaminergic; HSC, hepatic stellate cell; OVV, oncolytic vaccinia virus; AS, atherosclerosis; RA, rheumatoid arthritis; IPF, idiopathic pulmonary fibrosis; ZMTP, Zn-MnIII meso-tetrakis (4-carboxyphenyl) porphyrin-polyvinylpyrrolidone; MnTBAP, MnIII tetrakis (4-benzoic acid) porphyrin chloride; MnTMPyP, MnIII tetrakis (1-methyl-4-pyridyl) porphyrin. ‘↑’ represents increased, upregulated, induced, enhanced, or activated, while ‘↓’ represents decreased, downregulated, or inhibited, and ‘N/A’ represents not applicable.
2. MnSOD in Diseases
2.1. Inflammatory Diseases
Inflammation acts as a defensive mechanism to confront harmful stimuli such as infection and tissue damage [64][65]. Oxidative stress in activated infiltrating immune cells and tissue-resident immune cells, such as macrophages and T cells, is responsible for inflammation-related diseases [66][67]. Numerous studies have demonstrated that MnSOD plays a vital role in protecting organismal integrity and homeostasis by regulating inflammatory chemokines and cytokines [68][69] (Figure 2).
2.2. Fibrotic Diseases
Fibrosis is defined by the excessive accumulation of extracellular matrix (ECM) components such as collagen and fibronectin in damaged tissues, which ultimately induce tissue sclerosis and organ failure [70]. This process is initiated by the continuous activation and transdifferentiation of local fibroblasts in response to chronic injury [71]. To date, the regulatory role of MnSOD in the fibrosis of multiple organs has been widely reported and summarized (Figure 1).
2.3. Diabetes
Diabetes mellitus occurs whereby an abnormally high blood glucose level (hyperglycaemia) results from insulin insufficiency, and/or impaired tissue response to insulin (i.e., insulin resistance) [72]. It is a heterogeneous disorder with two major forms, type 1 diabetes mellitus (T1DM) and type 2 diabetes mellitus (T2DM), both of which are exacerbated by free radicals produced via glucose autoxidation [73][74][75]. Emerging evidence has indicated that the CT genotype of the MnSOD 47C/T gene is a significant risk factor for T1DM susceptibility (OR = 2.37; CI 95% = 1.03 to 5.46; p = 0.040) [76]. Here, the researchers summarize the different roles of MnSOD in regulating diabetes and its complications.
2.4. Neurodegenerative Diseases
Neurodegenerative diseases are indicated by progressive degeneration and the selective loss of neuronal systems, leading to cognitive impairment, dementia, motor dysfunction, and even death [77]. The majority of neurodegenerative diseases are characterized by the accumulation of misfolded proteins in the central nerve system, e.g., amyloid-β (Aβ) and tau aggregates in Alzheimer disease (AD), SOD1 pathology in amyotrophic lateral sclerosis (ALS), and mutated huntingtin (HTT) in Huntington disease (HD), and accompanied by a progressive loss of neurons, e.g., the loss of dopaminergic (DA) neurons in PD (Parkinson’s disease) in the affected regions [78]. A number of studies have highlighted the potential therapeutic role of MnSOD in detoxifying cerebral ROS to inhibit the aggregation of misfolded proteins and protect against neurodegenerative disorders (Figure 1).
2.5. Vascular Diseases
Redox homeostasis plays a vital role in vascular function. Dysregulated redox signalling can induce endothelial dysfunction and vascular abnormalities, contributing to different types of vascular diseases, such as hypertension and atherosclerosis (AS) and the occurrence of infarction [79][80][81]. Herein, the researchers summarize the mechanisms of MnSOD-mediated protective effects through vascular remodelling in vascular diseases.
2.6. Cancer
Various aspects of the extensive role of MnSOD in cancer have been explored. MnSOD can directly mediate multiple cancer cell death signalling pathways, including apoptosis [82], pyroptosis [83], and autophagy [84]. High MnSOD expression contributes to chemoresistance [85][86][87] and radioresistance [88][89] in different cancer types. Recently, posttranslational modification of MnSOD, with particular emphasis on acetylation at lysine residue 68, was proposed and explored, which addressed its vital roles in promoting cancer progression [90][91][92]. These functional roles of MnSOD in cancer have been well-reviewed elsewhere [12][27][93][94]. Herein, the researchers summarize some emerging research hotspots in MnSOD-regulated cancer progression, with an emphasis on metabolic reprogramming and the tumour immune microenvironment.
Metabolic reprogramming has been regarded as a hallmark of cancer for centuries. The Warburg effect primarily demonstrates that malignant cells preferentially rely on glycolysis rather than oxidative phosphorylation (OXPHOS) for energy supply [27][95][96]. Moreover, pathways involved in redox control are commonly reprogrammed due to tumorigenic mutations [97]. As MnSOD is one of the major redox metabolism regulators, the effect of MnSOD on cancer metabolic reprogramming has attracted broad attention. For instance, Liu et al. reported that toll-like receptor 2 (TLR 2) induced glycolysis and metabolic shift by markedly upregulating MnSOD expression in human gastric cancer cells. Furthermore, patient-derived tissue microarrays demonstrated a significant correlation between TLR2 and MnSOD expression in gastric tumours, indicating that the TLR2-MnSOD axis could serve as a potential biomarker for therapy decisions and prognosis determination in gastric cancer [98]. Zhou et al. observed that enhanced MnSOD expression activated AMPK signalling and shifted cell energy metabolism to glycolysis, thus facilitating tumorigenesis and progression in colorectal cancer cells [99]. Similarly, upregulation of MnSOD in cancer cells engaged AMPK to perform and sustain the Warburg effect, therefore supporting cancer cell survival [100]. Cells with a high level of MnSOD were more resistant to glucose-deprivation-induced cell death because MnSOD increased glucose uptake, glucose transporter member 1 (GLUT-1) availability, and OXPHOS electron transfer [101]. These results indicate a vital role of MnSOD in cancer glycolytic metabolism reprogramming, which deserves more in-depth mechanistic studies.
As the role of the immune system in cancer development has attracted increasing attention recently, the involvement of MnSOD in the tumour immune microenvironment has also been highlighted. Accumulating evidence has suggested that tumour-infiltrating immune cells participate in cancer progression, which is highly associated with MnSOD expression in different cancer types. For example, Su et al. analysed public datasets and found that MnSOD expression is positively correlated with CXCL8 and neutrophil infiltration, indicating an involvement of the ‘MnSOD-CXCL8-neutrophil recruitment’ axis in cancer progression [102]. MnSOD was also found to be positively correlated with CD68+ macrophage infiltration and may indicate poor outcomes in inflammation-driven lung adenocarcinoma [103]. High MnSOD expression was observed in aggressive triple-negative breast cancer (TNBC) patients and was regarded as a poor prognostic marker. MnSOD promotes the immunosuppressive tumour microenvironment by promoting M2 macrophage invasiveness and infiltration, supporting TNBC progression [104]. In addition, Lou et al. demonstrated that MnSOD-overexpressing oncolytic vaccinia virus (OVV-MnSOD) enhanced lymphocyte infiltration and tumour sensitivity to anti-PD-L1 treatment in lymphomatous mice, suggesting a promising therapeutic potential of MnSOD in cancer immunotherapy [63].
In addition, the researchers recently explored whether MnSOD overexpression could prevent cancer relapse by inhibiting reactivation of quiescent cancer cells (data not published). This novel function of MnSOD further increases the potential benefits of targeting MnSOD in cancer treatment. Although the pleiotropic role of MnSOD in cancer progression has aroused extensive interest, the underlying mechanisms remain controversial and unclear. Breakthroughs in finding key players in MnSOD-cancer regulation will be a promising research area, and will provide positive insights into the development of new drugs based on MnSOD.
References
- Yang, B.; Chen, Y.; Shi, J. Reactive Oxygen Species (ROS)-Based Nanomedicine. Chem. Rev. 2019, 119, 4881–4985.
- Sies, H.; Jones, D.P. Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nat. Rev. Mol. Cell Biol. 2020, 21, 363–383.
- Bae, Y.S.; Oh, H.; Rhee, S.G.; Yoo, Y.D. Regulation of reactive oxygen species generation in cell signaling. Mol. Cells 2011, 32, 491–509.
- Ray, P.D.; Huang, B.W.; Tsuji, Y. Reactive oxygen species (ROS) homeostasis and redox regulation in cellular signaling. Cell. Signal. 2012, 24, 981–990.
- Schieber, M.; Chandel, N.S. ROS function in redox signaling and oxidative stress. Curr. Biol. 2014, 24, R453–R462.
- Zhang, J.; Wang, X.; Vikash, V.; Ye, Q.; Wu, D.; Liu, Y.; Dong, W. ROS and ROS-Mediated Cellular Signaling. Oxid. Med. Cell. Longev. 2016, 2016, 4350965.
- Zhao, Y.; Hu, X.; Liu, Y.; Dong, S.; Wen, Z.; He, W.; Zhang, S.; Huang, Q.; Shi, M. ROS signaling under metabolic stress: Cross-talk between AMPK and AKT pathway. Mol. Cancer 2017, 16, 79.
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84.
- Demir, Y.; Balcı, N.; Gürbüz, M. Differential effects of selective serotonin reuptake inhibitors on paraoxonase-1 enzyme activity: An in vitro study. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2019, 226, 108608.
- Sarsour, E.; Kalen, A.; Goswami, P. Manganese Superoxide Dismutase Regulates a Redox Cycle Within the Cell Cycle. Antioxid. Redox Signal. 2013, 20, 1618–1627.
- Sena, L.A.; Chandel, N.S. Physiological roles of mitochondrial reactive oxygen species. Mol. Cell 2012, 48, 158–167.
- Kim, Y.S.; Gupta Vallur, P.; Phaëton, R.; Mythreye, K.; Hempel, N. Insights into the Dichotomous Regulation of SOD2 in Cancer. Antioxidants 2017, 6, 86.
- Forman, H.J.; Zhang, H. Targeting oxidative stress in disease: Promise and limitations of antioxidant therapy. Nat. Rev. Drug Discov. 2021, 20, 689–709.
- Lennicke, C.; Cochemé, H.M. Redox metabolism: ROS as specific molecular regulators of cell signaling and function. Mol. Cell 2021, 81, 3691–3707.
- Holley, A.K.; Bakthavatchalu, V.; Velez-Roman, J.M.; St Clair, D.K. Manganese superoxide dismutase: Guardian of the powerhouse. Int. J. Mol. Sci. 2011, 12, 7114–7162.
- Demir, Y. Naphthoquinones, benzoquinones, and anthraquinones: Molecular docking, ADME and inhibition studies on human serum paraoxonase-1 associated with cardiovascular diseases. Drug Dev. Res. 2020, 81, 628–636.
- Angelova, P.R.; Abramov, A.Y. Functional role of mitochondrial reactive oxygen species in physiology. Free Radic. Biol. Med. 2016, 100, 81–85.
- Bresciani, G.; da Cruz, I.B.; Gonzalez-Gallego, J. Manganese superoxide dismutase and oxidative stress modulation. Adv. Clin. Chem. 2015, 68, 87–130.
- Li, Y.; Huang, T.T.; Carlson, E.J.; Melov, S.; Ursell, P.C.; Olson, J.L.; Noble, L.J.; Yoshimura, M.P.; Berger, C.; Chan, P.H.; et al. Dilated cardiomyopathy and neonatal lethality in mutant mice lacking manganese superoxide dismutase. Nat. Genet. 1995, 11, 376–381.
- Lebovitz, R.M.; Zhang, H.; Vogel, H.; Cartwright, J., Jr.; Dionne, L.; Lu, N.; Huang, S.; Matzuk, M.M. Neurodegeneration, myocardial injury, and perinatal death in mitochondrial superoxide dismutase-deficient mice. Proc. Natl. Acad. Sci. USA 1996, 93, 9782–9787.
- Reaume, A.G.; Elliott, J.L.; Hoffman, E.K.; Kowall, N.W.; Ferrante, R.J.; Siwek, D.F.; Wilcox, H.M.; Flood, D.G.; Beal, M.F.; Brown, R.H., Jr.; et al. Motor neurons in Cu/Zn superoxide dismutase-deficient mice develop normally but exhibit enhanced cell death after axonal injury. Nat. Genet. 1996, 13, 43–47.
- Carlsson, L.M.; Jonsson, J.; Edlund, T.; Marklund, S.L. Mice lacking extracellular superoxide dismutase are more sensitive to hyperoxia. Proc. Natl. Acad. Sci. USA 1995, 92, 6264–6268.
- Aguirre, J.D.; Culotta, V.C. Battles with iron: Manganese in oxidative stress protection. J. Biol. Chem. 2012, 287, 13541–13548.
- Yamakura, F.; Kobayashi, K.; Furukawa, S.; Suzuki, Y. In vitro preparation of iron-substituted human manganese superoxide dismutase: Possible toxic properties for mitochondria. Free Radic. Biol. Med. 2007, 43, 423–430.
- Bonetta Valentino, R. The structure-function relationships and physiological roles of MnSOD mutants. Biosci. Rep. 2022, 42, 1618–1627.
- Zelko, I.N.; Mariani, T.J.; Folz, R.J. Superoxide dismutase multigene family: A comparison of the CuZn-SOD (SOD1), Mn-SOD (SOD2), and EC-SOD (SOD3) gene structures, evolution, and expression. Free Radic. Biol. Med. 2002, 33, 337–349.
- Dhar, S.K.; St Clair, D.K. Manganese superoxide dismutase regulation and cancer. Free Radic. Biol. Med. 2012, 52, 2209–2222.
- Miao, L.; St Clair, D.K. Regulation of superoxide dismutase genes: Implications in disease. Free Radic. Biol. Med. 2009, 47, 344–356.
- Candas, D.; Fan, M.; Nantajit, D.; Vaughan, A.T.; Murley, J.S.; Woloschak, G.E.; Grdina, D.J.; Li, J.J. CyclinB1/Cdk1 phosphorylates mitochondrial antioxidant MnSOD in cell adaptive response to radiation stress. J. Mol. Cell Biol. 2013, 5, 166–175.
- Belluzzi, E.; Bisaglia, M.; Lazzarini, E.; Tabares, L.C.; Beltramini, M.; Bubacco, L. Human SOD2 modification by dopamine quinones affects enzymatic activity by promoting its aggregation: Possible implications for Parkinson’s disease. PLoS ONE 2012, 7, e38026.
- Suri, S.; Mitra, P.; Bankul, A.; Saxena, I.; Garg, M.K.; Bohra, G.K.; Sharma, P. Altered expression of specific antioxidant (SOD1 and SOD2) and DNA repair (XRCC1 and OGG1) genes in patients with newly diagnosed type-2 diabetes mellitus. Minerva Endocrinol. 2021, 46.
- Cho, I.K.; Yang, B.; Forest, C.; Qian, L.; Chan, A.W.S. Amelioration of Huntington’s disease phenotype in astrocytes derived from iPSC-derived neural progenitor cells of Huntington’s disease monkeys. PLoS ONE 2019, 14, e0214156.
- Diaz-Castro, B.; Gangwani, M.R.; Yu, X.; Coppola, G.; Khakh, B.S. Astrocyte molecular signatures in Huntington’s disease. Sci. Transl. Med. 2019, 11, eaaw8546.
- Joseph, A.; Li, Y.; Koo, H.C.; Davis, J.M.; Pollack, S.; Kazzaz, J.A. Superoxide dismutase attenuates hyperoxia-induced interleukin-8 induction via AP-1. Free Radic. Biol. Med. 2008, 45, 1143–1149.
- Chen, H.X.; Xiang, H.; Xu, W.H.; Li, M.; Yuan, J.; Liu, J.; Sun, W.J.; Zhang, R.; Li, J.; Ren, Z.Q.; et al. Manganese Superoxide Dismutase Gene-Modified Mesenchymal Stem Cells Attenuate Acute Radiation-Induced Lung Injury. Hum. Gene Ther. 2017, 28, 523–532.
- Dong, J.W.; Xu, M.J.; Zhang, W.Y.; Che, X.M. Effects of Sevoflurane Pretreatment on Myocardial Ischemia-Reperfusion Injury Through the Akt/Hypoxia-Inducible Factor 1-alpha (HIF-1 alpha)/Vascular Endothelial Growth Factor (VEGF) Signaling Pathway. Med. Sci. Monit. 2019, 25, 3100–3107.
- Wang, Z.; Chen, K.; Han, Y.; Zhu, H.; Zhou, X.Y.; Tan, T.; Zeng, J.; Zhang, J.; Liu, Y.K.; Li, Y.; et al. Irisin Protects Heart Against Ischemia-Reperfusion Injury Through a SOD2-Dependent Mitochondria Mechanism. J. Cardiovasc. Pharmacol. 2018, 72, 259–269.
- Chang, G.D.; Chen, Y.W.; Zhang, H.W.; Zhou, W. Trans sodium crocetinate alleviates ischemia/reperfusion-induced myocardial oxidative stress and apoptosis via the SIRT3/FOXO3a/SOD2 signaling pathway. Int. Immunopharmacol. 2019, 71, 361–371.
- Koo, H.C.; Davis, J.M.; Li, Y.; Hatzis, D.; Opsimos, H.; Pollack, S.; Strayer, M.S.; Ballard, P.L.; Kazzaz, J.A. Effects of transgene expression of superoxide dismutase and glutathione peroxidase on pulmonary epithelial cell growth in hyperoxia. Am. J. Physiol. Lung Cell. Mol. Physiol. 2005, 288, L718–L726.
- Khan, A.A.; Paget, J.T.; McLaughlin, M.; Kyula, J.N.; Wilkinson, M.J.; Pencavel, T.; Mansfield, D.; Roulstone, V.; Seth, R.; Halle, M.; et al. Genetically modified lentiviruses that preserve microvascular function protect against late radiation damage in normal tissues. Sci. Transl. Med. 2018, 10, eaar2041.
- Yang, B.; Yao, H.; Yang, J.; Chen, C.; Shi, J. Construction of a two-dimensional artificial antioxidase for nanocatalytic rheumatoid arthritis treatment. Nat. Commun. 2022, 13, 1988.
- Jia, Z.; Zhuang, Y.; Hu, C.; Zhang, X.; Ding, G.; Zhang, Y.; Rohatgi, R.; Hua, H.; Huang, S.; He, J.C.; et al. Albuminuria enhances NHE3 and NCC via stimulation of mitochondrial oxidative stress/angiotensin II axis. Oncotarget 2016, 7, 47134–47144.
- Huang, H.-F.; Guo, F.; Cao, Y.-Z.; Shi, W.; Xia, Q. Neuroprotection by manganese superoxide dismutase (MnSOD) mimics: Antioxidant effect and oxidative stress regulation in acute experimental stroke. CNS Neurosci. Ther. 2012, 18, 811–818.
- Zhuang, Y.; Yasinta, M.; Hu, C.; Zhao, M.; Ding, G.; Bai, M.; Yang, L.; Ni, J.; Wang, R.; Jia, Z.; et al. Mitochondrial dysfunction confers albumin-induced NLRP3 inflammasome activation and renal tubular injury. Am. J. Physiol. Ren. Physiol. 2015, 308, F857–F866.
- Nagano, Y.; Matsui, H.; Shimokawa, O.; Hirayama, A.; Tamura, M.; Nakamura, Y.; Kaneko, T.; Rai, K.; Indo, H.P.; Majima, H.J.; et al. Rebamipide attenuates nonsteroidal anti-inflammatory drugs (NSAID) induced lipid peroxidation by the manganese superoxide dismutase (MnSOD) overexpression in gastrointestinal epithelial cells. J. Physiol. Pharm. 2012, 63, 137–142.
- Jiang, J.; Liang, S.; Zhang, J.; Du, Z.; Xu, Q.; Duan, J.; Sun, Z. Melatonin ameliorates PM(2.5)-induced cardiac perivascular fibrosis through regulating mitochondrial redox homeostasis. J. Pineal Res. 2021, 70, e12686.
- Sadi, G.; Güray, T. Gene expressions of Mn-SOD and GPx-1 in streptozotocin-induced diabetes: Effect of antioxidants. Mol. Cell. Biochem. 2009, 327, 127–134.
- Sandhir, R.; Sood, A.; Mehrotra, A.; Kamboj, S.S. N-Acetylcysteine reverses mitochondrial dysfunctions and behavioral abnormalities in 3-nitropropionic acid-induced Huntington’s disease. Neurodegener. Dis. 2012, 9, 145–157.
- Jiang, W.; Geng, H.; Lv, X.; Ma, J.; Liu, F.; Lin, P.; Yan, C. Idebenone Protects against Atherosclerosis in Apolipoprotein E-Deficient Mice Via Activation of the SIRT3-SOD2-mtROS Pathway. Cardiovasc. Drugs Ther. 2021, 35, 1129–1145.
- Zhang, Z.B.; Tan, Y.Z.; Zhu, L.W.; Zhang, B.; Feng, P.; Gao, E.H.; Xu, C.N.; Wang, X.M.; Yi, W.; Sun, Y. Asprosin improves the survival of mesenchymal stromal cells in myocardial infarction by inhibiting apoptosis via the activated ERK1/2-SOD2 pathway. Life Sci. 2019, 231, 116554.
- Lee, J.Y.; Castelli, V.; Kumar, N.; Sitruk-Ware, R.; Borlongan, C.V. Contraceptive drug, Nestorone, enhances stem cell-mediated remodeling of the stroke brain by dampening inflammation and rescuing mitochondria. Free Radic. Biol. Med. 2022, 183, 138–145.
- Baumer, Y.; Ng, Q.M.; Sanda, G.E.; Dey, A.K.; Teague, H.L.; Sorokin, A.V.; Dagur, P.K.; Silverman, J.I.; Harrington, C.L.; Rodante, J.A.; et al. Chronic skin inflammation accelerates macrophage cholesterol crystal formation and atherosclerosis. JCI Insight 2018, 3, e97179.
- Sen, A.; Nelson, T.J.; Alkon, D.L.; Hongpaisan, J. Loss in PKC Epsilon Causes Downregulation of MnSOD and BDNF Expression in Neurons of Alzheimer’s Disease Hippocampus. J. Alzheimer’s Dis. 2018, 63, 1173–1189.
- Dixit, A.; Srivastava, G.; Verma, D.; Mishra, M.; Singh, P.K.; Prakash, O.; Singh, M.P. Minocycline, levodopa and MnTMPyP induced changes in the mitochondrial proteome profile of MPTP and maneb and paraquat mice models of Parkinson’s disease. Biochim. Biophys. Acta 2013, 1832, 1227–1240.
- Dikalova, A.E.; Itani, H.A.; Nazarewicz, R.R.; McMaster, W.G.; Flynn, C.R.; Uzhachenko, R.; Fessel, J.P.; Gamboa, J.L.; Harrison, D.G.; Dikalov, S.I. Sirt3 Impairment and SOD2 Hyperacetylation in Vascular Oxidative Stress and Hypertension. Circ. Res. 2017, 121, 564–574.
- He, J.; Liu, X.; Su, C.; Wu, F.; Sun, J.; Zhang, J.; Yang, X.; Zhang, C.; Zhou, Z.; Zhang, X.; et al. Inhibition of Mitochondrial Oxidative Damage Improves Reendothelialization Capacity of Endothelial Progenitor Cells via SIRT3 (Sirtuin 3)-Enhanced SOD2 (Superoxide Dismutase 2) Deacetylation in Hypertension. Arter. Thromb. Vasc. Biol. 2019, 39, 1682–1698.
- Vendrov, A.E.; Stevenson, M.D.; Alahari, S.; Pan, H.; Wickline, S.A.; Madamanchi, N.R.; Runge, M.S. Attenuated Superoxide Dismutase 2 Activity Induces Atherosclerotic Plaque Instability During Aging in Hyperlipidemic Mice. J. Am. Heart Assoc. 2017, 6, e006775.
- Zhai, X.G.; Qiao, H.W.; Guan, W.; Li, Z.Q.; Cheng, Y.Y.; Jia, X.; Zhou, Y.J. Curcumin regulates peroxisome proliferator-activated receptor-gamma coactivator-1 alpha expression by AMPK pathway in hepatic stellate cells in vitro. Eur. J. Pharmacol. 2015, 746, 56–62.
- Lestari, N.; Louisa, M.; Soetikno, V.; Suwana, A.G.; Ramadhan, P.A.; Akmal, T.; Arozal, W. Alpha Mangostin Inhibits the Proliferation and Activation of Acetaldehyde Induced Hepatic Stellate Cells through TGF-beta and ERK 1/2 Pathways. J. Toxicol. 2018, 2018, 5360496.
- Shingu, M.; Takahashi, S.; Ito, M.; Hamamatu, N.; Suenaga, Y.; Ichibangase, Y.; Nobunaga, M. Anti-inflammatory effects of recombinant human manganese superoxide dismutase on adjuvant arthritis in rats. Rheumatol. Int. 1994, 14, 77–81.
- Guillaume, M.; Rodriguez-Vilarrupla, A.; Gracia-Sancho, J.; Rosado, E.; Mancini, A.; Bosch, J.; Garcia-Pagán, J.C. Recombinant human manganese superoxide dismutase reduces liver fibrosis and portal pressure in CCl4-cirrhotic rats. J. Hepatol. 2013, 58, 240–246.
- Shen, L.H.; Lei, S.J.; Huang, L.Y.; Li, S.G.; Yi, S.Z.; Breitzig, M.; Huang, M.Y.; Mo, X.M.; Sun, H.X.; Zheng, Q.; et al. Therapeutic effects of the rhSOD2-Hirudin fusion protein on bleomycin-induced pulmonary fibrosis in mice. Eur. J. Pharmacol. 2019, 852, 77–89.
- Lou, J.; Dong, J.; Xu, R.; Zeng, H.; Fang, L.; Wu, Y.; Liu, Y.; Wang, S. Remodeling of the tumor microenvironment using an engineered oncolytic vaccinia virus improves PD-L1 inhibition outcomes. Biosci. Rep. 2021, 41, BSR20204186.
- Nathan, C. Points of control in inflammation. Nature 2002, 420, 846–852.
- Medzhitov, R. Origin and physiological roles of inflammation. Nature 2008, 454, 428–435.
- Gray, J.I.; Farber, D.L. Tissue-Resident Immune Cells in Humans. Annu. Rev. Immunol. 2022, 40, 195–220.
- Caiado, F.; Pietras, E.M.; Manz, M.G. Inflammation as a regulator of hematopoietic stem cell function in disease, aging, and clonal selection. J. Exp. Med. 2021, 218, e20201541.
- Biswas, S.K. Does the Interdependence between Oxidative Stress and Inflammation Explain the Antioxidant Paradox? Oxid. Med. Cell. Longev. 2016, 2016, 5698931.
- McGarry, T.; Biniecka, M.; Veale, D.J.; Fearon, U. Hypoxia, oxidative stress and inflammation. Free Radic. Biol. Med. 2018, 125, 15–24.
- Wynn, T.A.; Ramalingam, T.R. Mechanisms of fibrosis: Therapeutic translation for fibrotic disease. Nat. Med. 2012, 18, 1028–1040.
- Henderson, N.C.; Rieder, F.; Wynn, T.A. Fibrosis: From mechanisms to medicines. Nature 2020, 587, 555–566.
- Sever, B.; Altıntop, M.D.; Demir, Y.; Türkeş, C.; Özbaş, K.; Çiftçi, G.A.; Beydemir, Ş.; Özdemir, A. A new series of 2,4-thiazolidinediones endowed with potent aldose reductase inhibitory activity. Open Chem. 2021, 19, 347–357.
- Ashcroft, F.M.; Rorsman, P. Diabetes mellitus and the β cell: The last ten years. Cell 2012, 148, 1160–1171.
- Rains, J.L.; Jain, S.K. Oxidative stress, insulin signaling, and diabetes. Free Radic. Biol. Med. 2011, 50, 567–575.
- Wu, J.; Zhou, Y.; Hu, H.; Yang, D.; Yang, F. Effects of β-carotene on glucose metabolism dysfunction in humans and type 2 diabetic rats. Acta Mater. Med. 2022, 1, 138–153.
- Eddaikra, A.; Amroun, H.; Raache, R.; Galleze, A.; Abdallah-Elhadj, N.; Azzouz, M.; Meçabih, F.; Mechti, B.; Abbadi, M.C.; Touil-Boukoffa, C.; et al. Clinical variables and ethnicity may influenced by polymorphism of CAT-262C/T and MnSOD 47C/T antioxidant enzymes in Algerian type1 diabetes without complications. Gene 2018, 670, 182–192.
- Lin, M.T.; Beal, M.F. Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature 2006, 443, 787–795.
- Peng, C.; Trojanowski, J.Q.; Lee, V.M. Protein transmission in neurodegenerative disease. Nat. Rev. Neurol. 2020, 16, 199–212.
- Fukai, T.; Ushio-Fukai, M. Superoxide dismutases: Role in redox signaling, vascular function, and diseases. Antioxid. Redox Signal. 2011, 15, 1583–1606.
- Siti, H.N.; Kamisah, Y.; Kamsiah, J. The role of oxidative stress, antioxidants and vascular inflammation in cardiovascular disease (a review). Vasc. Pharm. 2015, 71, 40–56.
- Perumareddi, P. Prevention of Hypertension Related to Cardiovascular Disease. Prim. Care 2019, 46, 27–39.
- Li, M.; Wu, C.; Muhammad, J.S.; Yan, D.; Tsuneyama, K.; Hatta, H.; Cui, Z.G.; Inadera, H. Melatonin sensitises shikonin-induced cancer cell death mediated by oxidative stress via inhibition of the SIRT3/SOD2-AKT pathway. Redox Biol. 2020, 36, 101632.
- Liu, J.; Yao, L.; Zhang, M.; Jiang, J.; Yang, M.; Wang, Y. Downregulation of LncRNA-XIST inhibited development of non-small cell lung cancer by activating miR-335/SOD2/ROS signal pathway mediated pyroptotic cell death. Aging 2019, 11, 7830–7846.
- Jeong, S.; Kim, B.G.; Kim, D.Y.; Kim, B.R.; Kim, J.L.; Park, S.H.; Na, Y.J.; Jo, M.J.; Yun, H.K.; Jeong, Y.A.; et al. Cannabidiol Overcomes Oxaliplatin Resistance by Enhancing NOS3- and SOD2-Induced Autophagy in Human Colorectal Cancer Cells. Cancers 2019, 11, 781.
- Ma, C.S.; Lv, Q.M.; Zhang, K.R.; Tang, Y.B.; Zhang, Y.F.; Shen, Y.; Lei, H.M.; Zhu, L. NRF2-GPX4/SOD2 axis imparts resistance to EGFR-tyrosine kinase inhibitors in non-small-cell lung cancer cells. Acta Pharmacol. Sin. 2021, 42, 613–623.
- Paku, M.; Haraguchi, N.; Takeda, M.; Fujino, S.; Ogino, T.; Takahashi, H.; Miyoshi, N.; Uemura, M.; Mizushima, T.; Yamamoto, H.; et al. SIRT3-Mediated SOD2 and PGC-1alpha Contribute to Chemoresistance in Colorectal Cancer Cells. Ann. Surg. Oncol. 2021, 28, 4720–4732.
- Hsueh, W.T.; Chen, S.H.; Chien, C.H.; Chou, S.W.; Chi, P.I.; Chu, J.M.; Chang, K.Y. SOD2 Enhancement by Long-Term Inhibition of the PI3K Pathway Confers Multi-Drug Resistance and Enhanced Tumor-Initiating Features in Head and Neck Cancer. Int. J. Mol. Sci. 2021, 22, 11260.
- Zhang, Y.; Xu, Z.; Ding, J.; Tan, C.; Hu, W.; Li, Y.; Huang, W.; Xu, Y. HZ08 suppresses RelB-activated MnSOD expression and enhances Radiosensitivity of prostate Cancer cells. J. Exp. Clin. Cancer Res. CR 2018, 37, 174.
- Noh, J.K.; Woo, S.R.; Yun, M.; Lee, M.K.; Kong, M.; Min, S.; Kim, S.I.; Lee, Y.C.; Eun, Y.G.; Ko, S.G. SOD2- and NRF2-associated Gene Signature to Predict Radioresistance in Head and Neck Cancer. Cancer Genom. Proteom. 2021, 18, 675–684.
- He, C.; Danes, J.M.; Hart, P.C.; Zhu, Y.; Huang, Y.; de Abreu, A.L.; O’Brien, J.; Mathison, A.J.; Tang, B.; Frasor, J.M.; et al. SOD2 acetylation on lysine 68 promotes stem cell reprogramming in breast cancer. Proc. Natl. Acad. Sci. USA 2019, 116, 23534–23541.
- Hjelmeland, A.B.; Patel, R.P. SOD2 acetylation and deacetylation: Another tale of Jekyll and Hyde in cancer. Proc. Natl. Acad. Sci. USA 2019, 116, 23376–23378.
- Zhu, Y.; Zou, X.; Dean, A.E.; Brien, J.O.; Gao, Y.; Tran, E.L.; Park, S.-H.; Liu, G.; Kieffer, M.B.; Jiang, H.; et al. Lysine 68 acetylation directs MnSOD as a tetrameric detoxification complex versus a monomeric tumor promoter. Nat. Commun. 2019, 10, 2399.
- Kim, A. Modulation of MnSOD in Cancer:Epidemiological and Experimental Evidence. Toxicol. Res. 2010, 26, 83–93.
- Konzack, A.; Kietzmann, T. Manganese superoxide dismutase in carcinogenesis: Friend or foe? Biochem. Soc. Trans. 2014, 42, 1012–1016.
- Gatenby, R.A.; Gillies, R.J. Why do cancers have high aerobic glycolysis? Nat. Rev. Cancer 2004, 4, 891–899.
- Hanahan, D. Hallmarks of Cancer: New Dimensions. Cancer Discov. 2022, 12, 31–46.
- Faubert, B.; Solmonson, A.; DeBerardinis, R.J. Metabolic reprogramming and cancer progression. Science 2020, 368, eaaw5473.
- Liu, Y.D.; Yu, L.; Ying, L.; Balic, J.; Gao, H.; Deng, N.T.; West, A.; Yan, F.; Ji, C.B.; Gough, D.; et al. Toll-like receptor 2 regulates metabolic reprogramming in gastric cancer via superoxide dismutase 2. Int. J. Cancer 2019, 144, 3056–3069.
- Zhou, C.; Lyu, L.-H.; Miao, H.-K.; Bahr, T.; Zhang, Q.-Y.; Liang, T.; Zhou, H.-B.; Chen, G.-R.; Bai, Y. Redox regulation by SOD2 modulates colorectal cancer tumorigenesis through AMPK-mediated energy metabolism. Mol. Carcinog. 2020, 59, 545–556.
- Hart, P.C.; Mao, M.; de Abreu, A.L.P.; Ansenberger-Fricano, K.; Ekoue, D.N.; Ganini, D.; Kajdacsy-Balla, A.; Diamond, A.M.; Minshall, R.D.; Consolaro, M.E.L.; et al. MnSOD upregulation sustains the Warburg effect via mitochondrial ROS and AMPK-dependent signalling in cancer. Nat. Commun. 2015, 6, 6053.
- Quiros-Gonzalez, I.; Gonzalez-Menendez, P.; Mayo, J.C.; Hevia, D.; Artime-Naveda, F.; Fernandez-Vega, S.; Fernandez-Fernandez, M.; Rodriguez-Gonzalez, P.; Garcia-Alonso, J.I.; Sainz, R.M. Androgen-Dependent Prostate Cancer Cells Reprogram Their Metabolic Signature upon GLUT1 Upregulation by Manganese Superoxide Dismutase. Antioxidants 2022, 11, 313.
- Su, H.; Cai, T.; Zhang, S.; Yan, X.; Zhou, L.; He, Z.; Xue, P.; Li, J.; Zheng, M.; Yang, X.; et al. Identification of hub genes associated with neutrophils infiltration in colorectal cancer. J. Cell. Mol. Med. 2021, 25, 3371–3380.
- Han, X.; Liu, X.; Wang, X.; Guo, W.; Wen, Y.; Meng, W.; Peng, D.; Lv, P.; Zhang, X.; Shen, H. TNF-alpha-dependent lung inflammation upregulates superoxide dismutase-2 to promote tumor cell proliferation in lung adenocarcinoma. Mol. Carcinog. 2020, 59, 1088–1099.
- Al Haq, A.T.; Tseng, H.-Y.; Chen, L.-M.; Wang, C.-C.; Hsu, H.-L. Targeting prooxidant MnSOD effect inhibits triple-negative breast cancer (TNBC) progression and M2 macrophage functions under the oncogenic stress. Cell Death Dis. 2022, 13, 49.
More
