Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Rita Xu and Version 1 by Fawad Ali.
Due to their commercial availability, superior processability, and biocompatibility, polymers are frequently used to build three-dimensional (3D) porous scaffolds. The main issues limiting the widespread clinical use of monophasic polymer scaffolds in the bone healing process are their inadequate mechanical strength and inappropriate biodegradation. Due to their mechanical strength and biocompatibility, metal-based scaffolds have been used for various bone regenerative applications.
- Tissue engineering
- Polymer-Mg composite
- Biomedical application
1. Introduction
Biopolymers made from biological sources and with adjustable biodegradability are gaining popularity. Depending on where they came from, biopolymers are divided into natural and synthetic classes [1]. Biopolymers convey their payloads to human bodies, which undergo regulated degradation to form nontoxic byproducts [2]. This distinct characteristic makes tissue engineering viable for treating patients with damaged or missing organs and tissues [3]. Three-dimensional structures for repairing soft and hard tissues are typically created using natural and synthetic biopolymers. However, the poor mechanical properties and the slow degradation rate of biopolymer limit the use of biopolymer in tissue engineering [4].
The search for newer biodegradable and biocompatible materials for tissue engineering has recently increased due to the challenges driven mainly by secondary surgeries associated with the issues of the existing metal implants. Accidents, injuries, and the natural aging process cause bone degeneration, which requires an implant to restore its function [5,6][5][6]. According to a 2019 Australian Orthopaedic Association (AOA) report, the need for hip, knee, and shoulder surgical procedures has exponentially increased, reaching a total number of 459,265 hip, 658,596 knee, and 40,130 shoulder surgeries in 2018 [7]. Similarly, bone fracture costs only around USD 32 billion annually in the US. Over 3 million bone surgeries are conducted worldwide every year [8]. That is the main reason for the importance of highly efficient and cost-effective bone fracture treatment. The orthopedic implant costs USD 45.9 billion in 2017 and is expected to reach USD 66.6 billion by 2025, showing a compound annual growth of 4.7% [9]. The current demand for temporary implants is also soaring for scaffolds, cardiac stimulators, and cardiovascular stents [10,11,12,13,14,15][10][11][12][13][14][15]. Such a steep increase in orthopedic implants has necessitated the acceleration in the research and development for finding new materials and fabrication methods to satisfy the medical requirements of biodegradability, biocompatibility, and on-demand personalized design and manufacturing at a low cost.
Implants, acting as a replacement for natural bones, should imitate the bone in its physical and biological properties as much as possible, that is, strength, hardness, chemical properties, degradation rate, biocompatibility in the physiological environment, and so on [16,17][16][17]. There are mainly two types of implants: (1) permanent implants, which are required to perform the function of the bone for life with no immature failure and additional replacement needs, and (2) temporary implants, which are required to fulfill the functions of a bone until the tissues are healed [18,19][18][19]. Temporary implants stay inside the body, fixed to the bones, until the body part is fully recovered, and then are removed [20,21,22][20][21][22]. Currently, commercially available implants are mostly made of metals and metallic alloys, including titanium, stainless steel, and Co-Cr alloys, due to their high mechanical strength, biocompatibility, and good corrosion resistance properties [23,24,25,26,27,28,29,30,31][23][24][25][26][27][28][29][30][31]. However, due to the differences between the elastic modulus of these implant materials and natural bones, they impart a stress shielding effect [32,33,34,35][32][33][34][35]. Another problem with these materials is the requirement of a second surgery to remove these implants after the healing of the fracture, causing pain to the patient. Such a process not only adds pain, discomfort, and risks to the patient but also costs about 30% of healthcare expenditures [36,37][36][37]. Studies have found that failing to remove these implants can cause severe allergic problems due to ion accumulation, causing osteolysis [36,37][36][37].
Therefore, using biodegradable materials that the body may dissolve is significant for these implants. Such biodegradable implants will replace the natural bone until its recovery without any complications. Owing to its low density (1.8 g/cm3), high strength-to-weight ratio, compressive yield strength, and comparable elastic modulus to natural bone, magnesium (Mg) and its alloys (with some bioactive materials) can be a perfect choice over the existing metal implants [38,39,40][38][39][40]. Mg, a crucial component of natural bone formation and the body’s fourth-most principal cation, has good biocompatibility and is among the ideal materials for temporary implants. On average, a healthy adult weighing 70 kg is known to have 21 to 35 g of stored magnesium [41,42][41][42]. Twenty percent of the available amount of Mg is kept in bones, followed by 35–40% in tissues and ligaments and 1% in bodily fluid [43]. In addition, over 300 enzyme-related processes rely on the availability of Mg, either directly or indirectly [44,45][44][45]. Mg has the second-highest daily permissible intake (420 mg) of all the nutritionally necessary substances [43]. Additionally, the byproducts of the breakdown of magnesium and its alloys can be safely taken by macrophages and eliminated through the urine without endangering physiological function [44].
Despite having several valuable qualities, the unpredictable deterioration rate of Mg-based implants in the physiological environment prevents their commercialization as temporary implants. Mg-based implants deteriorate significantly more quickly in the body fluid than the conventional aqueous solution due to high chloride ion concentration (96–106 mEq/L) and a pH of roughly 7.4–7.6 [46]. Additionally, the accelerated rate of gas bubble creation harms the patient’s health. The implants become particularly susceptible to unexpected failure and lose the necessary mechanical integrity to support the load during this deterioration phase. As a result, creating Mg-based implants with a controlled degradation rate to prolong the bone-required repair duration (between 24 and 32 weeks) is essential.
Tissue engineering scaffolds frequently use polymeric materials, such as polylactic acid (PLA), poly(lactide-co-glycolic acid) (PLGA), and polycaprolactone (PCL), due to their simple processing by 3D printing [47[47][48],48], biocompatibility, osteoinductivity, and negligible inflammatory response. Despite such appealing properties of the polymers that could be utilized as a 3D porous scaffold, inadequate mechanical strength and an improper degradation rate call for additional study in this area [49]. Metal particles as a reinforcement to a polymer matrix is an effective strategy for improving polymers’ biological behavior and mechanical properties. Due to its magnificent properties in biomedical applications, Mg can be an excellent addition to polymer materials, further enhancing biodegradable polymers’ mechanical and biological properties.
Polymer/Mg composite scaffolds must be designed with high dimensional accuracy and reproducible. Furthermore, these scaffolds should be designed according to the patient-specific anatomic requirements. 3D printing technology, as one of the advanced manufacturing techniques, offers high dimensional accuracy and excellent reproducibility with low-cost manufacturing. It can produce customized or personalized structures based on patient-specific anatomic data. Three-dimensional printing controls porosity and surface topology, determining the final implant’s mechanical, chemical, and biodegradation properties. Three-dimensional printing is known to be an easy, fast, and on-site manufacturing process, which can fabricate implants closely resembling native bone properties.
2. Suitability of Polymer/Mg Composites for Biomedical Applications
2.1. Biocompatibility
Biocompatibility and nontoxicity are the two most crucial requirements for every implant material. Soon after a foreign object is inserted into a human body, interactions between the body tissues and implanted material start to happen. These reactions determine whether the body will accept the implant [50]. Biocompatibility for permanent implants depends on how well the newly formed tissue fuses with the implant surface. Contrastingly, temporary implants are intended to stabilize broken bone while it heals before degrading naturally in the body over time [8,51][8][51]. It is pretty concerning whenever a physiological component starts by negatively impacting the breakdown product. Consequently, nontoxic materials should be used for temporary implants. The 3D porous bone scaffolds’ cellular activity is significantly affected by adding Mg particles into polymer matrices, such as PLA, PCL, and PLGA, during 3D printing. The presence of Mg is advantageous for cell growth, proliferation, and differentiation because composite scaffolds have more hydrophilicity, more bioactive area on their surface, and a moderately alkaline pH (pH 10), which is a favorable microenvironment for cell growth [52]. The amount of Mg in the polymer is a crucial element that needs to be tuned since an increase in the pH beyond 10 (pH > 10) harms cellular activity. Excess Mg (greater than 10 wt% (8.9 vol%) in PLGA and five wt% in PCL) has been reported to hurt cytocompatibility [53,54][53][54]. Figure 1 shows suitable cell attachment and proliferation on PCL/Mg composite scaffolds created by the FDM process. Cell viability (green = viable cells, red = dead cells) is markedly increased by adding five wt% Mg particles and decreased by further augmentation, as revealed in CLSM images (Figure 1a–f). Similarly, SEM photos demonstrate that cells in the PCL/Mg scaffold stretch, freely expand, and initiate filopodia, establishing a better cellular adherence to composite scaffolds. However, cells barely attached to the surface on the monophasic PCL scaffold with inappropriate conditions are unacceptable (Figure 1a–f) [53].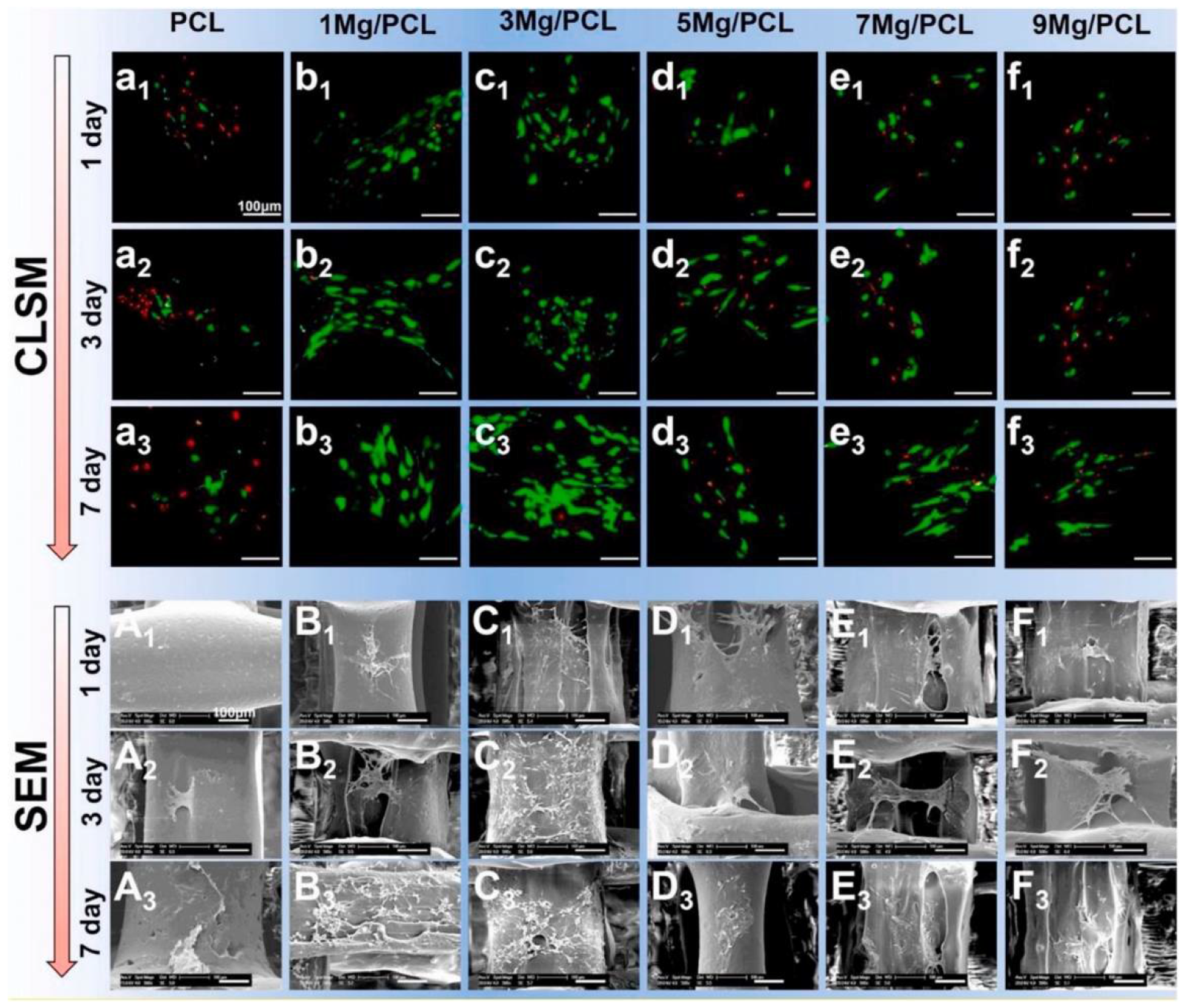
Figure 1. Cell adhesion and proliferation on 3D-printed PCL/Mg composite scaffolds (a–f) and SEM images of PLC/MG composite with different amounts of Mg (Mg = 0, 1, 3, 5, 7, and 9 wt %) (A–F) [53].
2.2. Mechanical Integrity
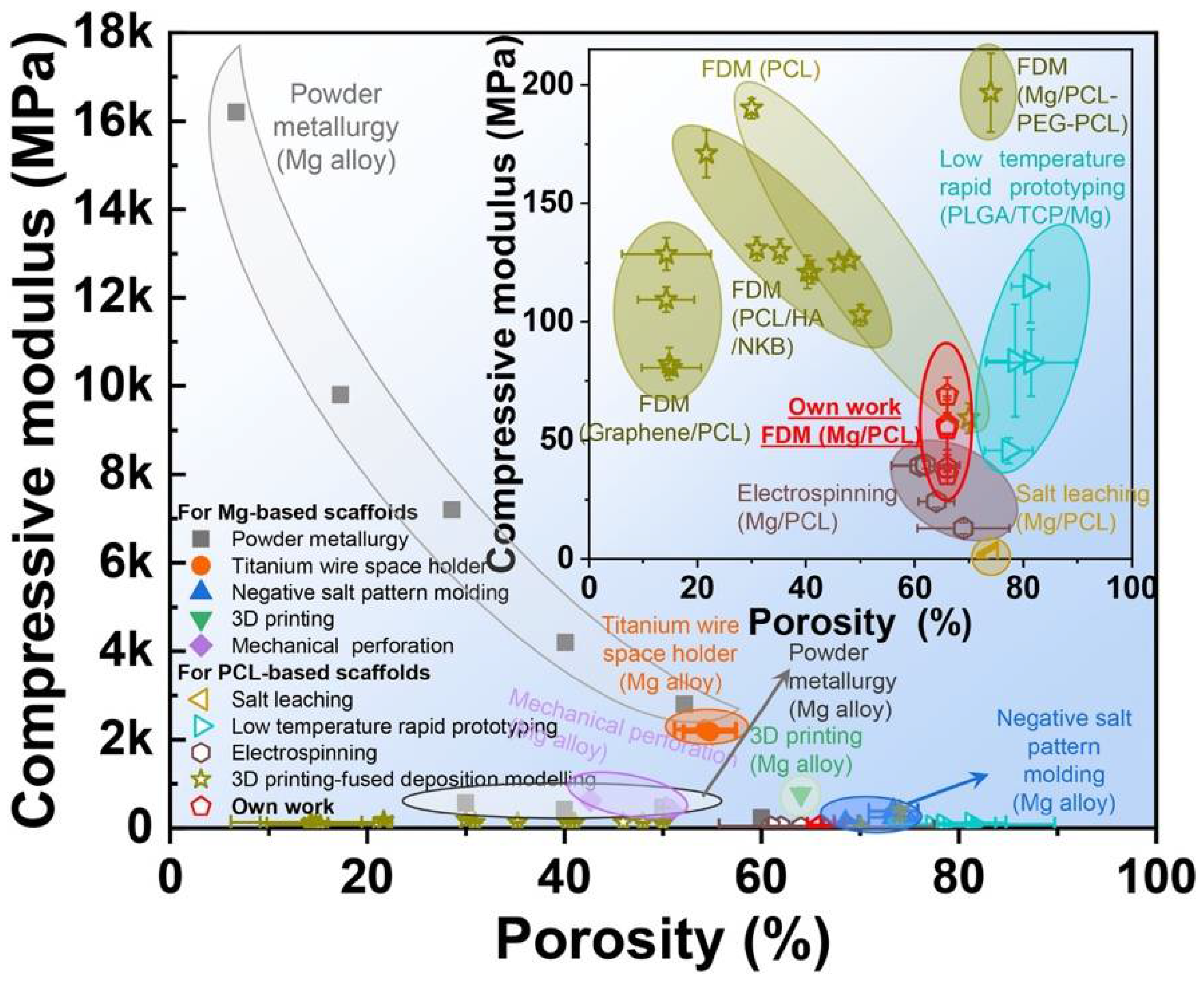
The material suitability to be employed as an orthopedic implant for a particular purpose is highly dependent on its mechanical qualities. Mechanical properties, such as elastic modulus, tensile strength, fatigue strength, hardness, and elongation, are among the rates given top priority. To prevent the “stress shielding effect”, the elastic modulus of biomedical implants should ideally have a similar value to the natural bone [32,33][32][33]. Biopolymers, owing to their excellent biocompatibility and biodegradability, have a strong possibility for scaffolds used in bone tissue engineering. However, they have poor mechanical qualities. This point of view claims that measures have been taken, such as customizing the pore structure and shape to overcome the substandard attribute qualities of the polymer 3D-printed scaffolds [55], modifying printing parameters [56], and incorporating other phases [57,58][57][58]. Among these efforts, the use of metallic powders as fillers is the most promising [59,60][59][60]. Similarly, adding Mg particles into the PLLA matrix demonstrated good mechanical properties (compressive strength and modulus of 5 wt% (3.6 vol%) Mg particles to the PLLA matrix considerably enhanced the compressive strength and modulus of 3D porous scaffolds by 114.5% and 85.7%, respectively). Adding more magnesium can negatively affect the mechanical characteristics of the porous composite scaffold made using the SLS process because of the particle agglomeration [61]. Alizadeh et al. reported a magnesium-reinforced PCL matrix [12]. The correct quantity of magnesium reinforcement strengthens mechanical capabilities, but too much magnesium weakens support due to the regional enhancement of magnesium microparticles in the PCL matrix. The compressive modulus of PCL/Mg scaffolds is summarized in Figure 2 [62,63,64,65,66,67,68,69,70,71,72,73,74,75,76][62][63][64][65][66][67][68][69][70][71][72][73][74][75][76]. Comparatively, PCL-based scaffolds have a lower compressive modulus than Mg-based scaffolds, which helps to limit the stress shielding effect. Additionally, Mg/PCL scaffolds created using 3DP had a greater compressive modulus than those made through electrospinning and salt leaching at a comparable porosity [63,64][63][64]. This scaffold’s mechanical attributes were like those of human cancellous bone, reducing the stress shielding effect. Ma et al. [77] observed an increase in compressive modulus by adding 20 wt% (18.1 vol%) Mg in the 3D porous PLGA scaffold. This enhancement was primarily ascribed to the PLGA/Mg composite scaffold’s increased load-bearing capability because Mg fillers have better strength and modulus.2.3. Biodegradation
The relationship between the synthetic tissue scaffolds’ biodegradability or degradation rate and the rate at which bones mend is crucial. Because the hydrolytic breakdown pathway causes the polymeric chains to separate into monomers and water-solvable oligomers, biopolymers, including PLA, PCL, and PLGA, disintegrate in the physiological environment [49]. The total breakdown of the mentioned polymers in the body would take several years, however [78]. One of the significant problems with biodegradable polymer-based scaffolds is their sluggish disintegration rate, which needs more research. On the other hand, Mg, a very suitable metal with good biocompatibility, has a very high deposition rate. Therefore, incorporating Mg particles into these polymers in a specific ratio can have a controlled degradation rate matching that of the natural bone growth rate and having a good match of the mechanical properties similar to the natural bone. The incorporation of metallic powders into polymers is an effective strategy among the various methods used to increase the degradation of polymers without sacrificing biocompatibility, such as copolymerization [79], blending [80], and surface modification [81]. This is due to the outstanding ability to increase the strength and degradation rate simultaneously [61].2.4. Osteogenic and Angiogenic Characteristics
The process of osteogenesis involves creating new tissues to mend a broken bone. It is a crucial necessity for temporary implants since the damaged bone needs to heal with the help of newly formed tissues and cells before implant disintegration [82,83][82][83]. The osteogenic activity of the synthetic polymers used in the 3D printing of 3D porous scaffolds needs to be improved because they are nonbioactive [84]. A promising strategy in this area has been the fabrication of polymer/metal composite scaffolds and introducing metallic fillers. Due to its inherent ability to promote osteogenesis, Mg is highly preferable compared with other bioactive materials. This suggests that Mg positively affects the osteogenic differentiation of 3D composite porous scaffolds and bone mineralization. According to reports, adding Mg to monophasic PLGA [54] and PCL [53] bone scaffolds remarkably increases the ability of genes relevant to osteoblast development and mineralization to express. The enhanced expression of neuronal calcitonin gene-related polypeptide (CGRP) by an Mg filler is suggested to be the cause of the polymer/Mg composite scaffolds’ robust bone formation. Jing Bai et al. [53] have shown the general research methodology for Mg/PCL scaffolds for bone repair, as shown in Figure 3. The homogeneous distribution of Mg particles in PCL scaffolds is made possible by combining mixing, blending, and 3D printing. Scaffolds made of a Mg/PCL composite show good overall qualities and in vitro and in vivo responses. The Mg particles slowly degrade with a local pH increase in in-vitro degradation studies.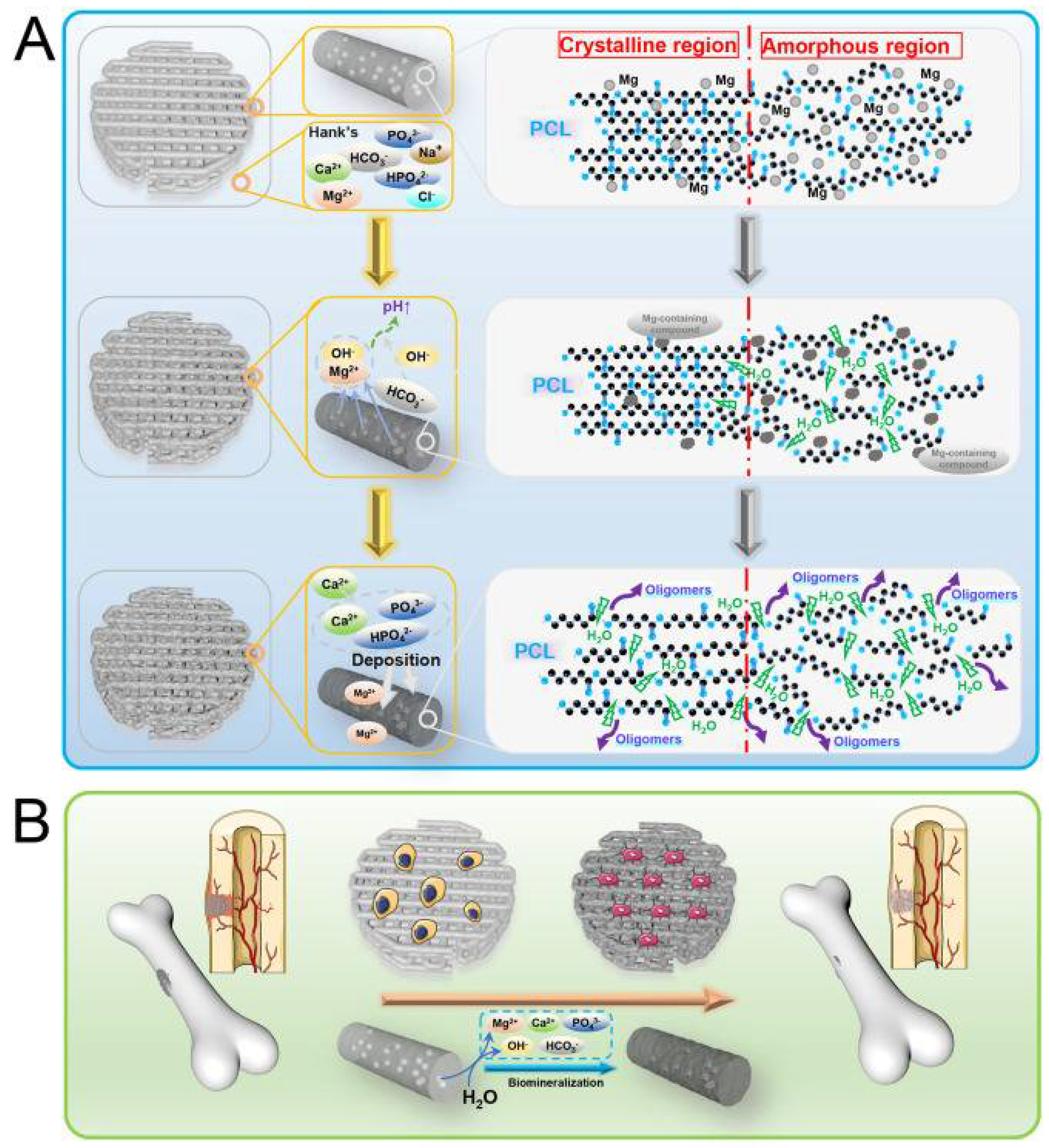
Figure 3.
Reactions of Mg/PCL scaffolds for bone healing in vitro and in vivo, showing (
A
) degradation behavior and (
B
3. Conventional Techniques for Biopolymer and Polymer/Mg Composites
References
- Hassan, M.; Dave, K.; Chandrawati, R.; Dehghani, F.; Gomes, V.G. 3D printing of biopolymer nanocomposites for tissue engineering: Nanomaterials, processing and structure-function relation. Eur. Polym. J. 2019, 121, 109340.
- Tian, H.; Tang, Z.; Zhuang, X.; Chen, X.; Jing, X. Biodegradable synthetic polymers: Preparation, functionalization and biomedical application. Prog. Polym. Sci. 2012, 37, 237–280.
- Freed, L.E.; Novkovic, G.V.; Biron, R.J.; Eagles, B.D.; Lesnoy, D.C.; Barlow, S.K.; Langer, R. Biodegradable Polymer Scaffolds for Tissue Engineering. Nat. Biotechnol. 1994, 12, 689–693.
- van de Velde, K.; Kiekens, P. Biopolymers: Overview of several properties and consequences on their applications. Polym. Test. 2002, 21, 433–442.
- Arif, Z.U.; Khalid, M.Y.; Ahmed, W.; Arshad, H. A review on four-dimensional (4D) bioprinting in pursuit of advanced tissue engineering applications. Bioprinting 2022, 27, e00203.
- Arif, Z.U.; Khalid, M.Y.; Sheikh, M.F.; Zolfagharian, A.; Bodaghi, M. Biopolymeric sustainable materials and their emerging applications. J. Environ. Chem. Eng. 2022, 10, 108159.
- Australian Orthopaedic Association. 20th Australian Orthopaedic Association National Joint Replacement Registry: Hip, Knee & Shoulder Arthroplasty Annual Report. 2019. Available online: https://aoanjrr.sahmri.com/annual-reports-2019 (accessed on 3 August 2022).
- Tsakiris, V.; Tardei, C.; Clicinschi, F.M. Biodegradable Mg alloys for orthopedic implants–A review. J. Magnes. Alloy. 2021, 9, 1884–1905.
- Shinde, O.S.S.; Srivastava, A. Medical Implant Market Statistics. Available online: https://www.alliedmarketresearch.com/medical-implants-market (accessed on 7 August 2022).
- Lee, J.H.; Kim, E.D.; Jun, E.J.; Yoo, H.S.; Lee, J.W. Analysis of trends and prospects regarding stents for human blood vessels. Biomater. Res. 2018, 22, 8.
- Scot, G.; Patrick, W. Coronary Stents. J. Am. Coll. Cardiol. 2010, 56, S1–S42.
- Borhani, S.; Hassanajili, S.; Tafti, S.H.A.; Rabbani, S. Cardiovascular stents: Overview, evolution, and next generation. Prog. Biomater. 2018, 7, 175–205.
- Rigatelli, G.; Carraro, U.; Barbiero, M.; Zanchetta, M.; Pedon, L.; Dimopoulos, K.; Rigatelli, G.; Maiolino, P.; Cobelli, F.; Riccardi, R.; et al. New Advances in Dynamic Cardiomyoplasty: Doppler Flow Wire Shows Improved Cardiac Assistance in Demand Protocol. ASAIO J. 2002, 48, 119–123.
- O’Brien, F.J. Biomaterials & scaffolds for tissue engineering. Mater. Today 2011, 14, 88–95.
- Karunakaran, R.; Ortgies, S.; Tamayol, A.; Bobaru, F.; Sealy, M.P. Additive manufacturing of magnesium alloys. In Bioactive Materials; KeAi Communications Co.: Beijing, China, 2020; pp. 44–54.
- Razavi, M.; Huang, Y. Assessment of magnesium-based biomaterials: From bench to clinic. Biomater. Sci. 2019, 7, 2241–2263.
- Moghaddam, N.S.; Andani, M.T.; Amerinatanzi, A.; Haberland, C.; Huff, S.; Miller, M.; Elahinia, M.; Dean, D. Metals for bone implants: Safety, design, and efficacy. Biomanuf. Rev. 2016, 1, 1.
- Minkowitz, R.B.; Bhadsavle, S.; Walsh, M.; Egol, K.A. Removal of Painful Orthopaedic Implants After Fracture Union. JBJS 2007, 89, 1906–1912.
- Geetha, M.; Singh, A.K.; Asokamani, R.; Gogia, A.K. Ti based biomaterials, the ultimate choice for orthopaedic implants–A review. Prog. Mater. Sci. 2009, 54, 397–425.
- Tian, L.; Tang, N.; Ngai, T.; Wu, C.; Ruan, Y.; Huang, L.; Qin, L. Hybrid fracture fixation systems developed for orthopaedic applications: A general review. J. Orthop. Translat. 2019, 16, 1–13.
- Farraro, K.F.; Kim, K.E.; Woo, S.L.-Y.; Flowers, J.R.; McCullough, M.B. Revolutionizing orthopaedic biomaterials: The potential of biodegradable and bioresorbable magnesium-based materials for functional tissue engineering. J. Biomech. 2014, 47, 1979–1986.
- Chandra, G.; Pandey, A. Biodegradable bone implants in orthopedic applications: A review. Biocybern. Biomed. Eng. 2020, 40, 596–610.
- González, J.E.G.; Mirza-Rosca, J.C. Study of the corrosion behavior of titanium and some of its alloys for biomedical and dental implant applications. J. Electroanal. Chem. 1999, 471, 109–115.
- Elias, C.N.; Lima, J.H.C.; Valiev, R.; Meyers, M.A. Biomedical applications of titanium and its alloys. JOM 2008, 60, 46–49.
- Khoo, L.K.; Kiattavorncharoen, S.; Pairuchvej, V.; Lakkhanachatpan, N.; Wongsirichat, N.; Seriwatanachai, D. The Affinity of Human Fetal Osteoblast to Laser-Modified Titanium Implant Fixtures. Open Dent. J. 2020, 14, 52–58.
- Gepreel, M.A.; Niinomi, M. Biocompatibility of Ti-alloys for long-term implantation. J. Mech. Behav. Biomed. Mater. 2013, 20, 407–415.
- Singh, D.; Singh, R.; Boparai, K.S.; Farina, I.; Feo, L.; Verma, A.K. In-vitro studies of SS 316L biomedical implants prepared by FDM, vapor smoothing and investment casting. Compos. B Eng. 2018, 132, 107–114.
- Chen, R.; Ni, H.; Zhang, H.; Yue, G.; Zhan, W.; Xiong, P. A preliminary study on antibacterial mechanisms of silver ions implanted stainless steel. Vacuum 2013, 89, 249–253.
- Muley, S.V.; Vidvans, A.N.; Chaudhari, G.P.; Udainiya, S. An assessment of ultra fine grained 316L stainless steel for implant applications. Acta Biomater. 2016, 30, 408–419.
- Hunt, J.A.; Callaghan, J.T.; Sutcliffe, C.J.; Morgan, R.H.; Halford, B.; Black, R.A. The design and production of Co–Cr alloy implants with controlled surface topography by CAD–CAM method and their effects on osseointegration. Biomaterials 2005, 26, 5890–5897.
- Xiang, D.D.; Wang, P.; Tan, X.P.; Chandra, S.; Wang, C.; Nai, M.L.S.; Tor, S.B.; Liu, W.Q.; Liu, E. Anisotropic microstructure and mechanical properties of additively manufactured Co–Cr–Mo alloy using selective electron beam melting for orthopedic implants. Mater. Sci. Eng. A 2019, 765, 138270.
- Seal, C.K.; Vince, K.; Hodgson, M.A. Biodegradable surgical implants based on magnesium alloys–A review of current research. IOP Conf. Ser. Mater. Sci. Eng. 2009, 4, 12011.
- Sumner, D.R. Long-term implant fixation and stress-shielding in total hip replacement. J. Biomech. 2015, 48, 797–800.
- Mani, G.; Feldman, M.D.; Patel, D.; Agrawal, C.M. Coronary stents: A materials perspective. Biomaterials 2007, 28, 1689–1710.
- Hermawan, H.; Dubé, D.; Mantovani, D. Degradable metallic biomaterials: Design and development of Fe–Mn alloys for stents. J. Biomed. Mater. Res. A 2010, 93A, 1–11.
- Zielinski, S.M.; Heetveld, M.J.; Bhandari, M.; Patka, P.; van Lieshout, E.M.M.; FAITH Trial Investigators. Implant Removal After Internal Fixation of a Femoral Neck Fracture: Effects on Physical Functioning. J. Orthop. Trauma 2015, 29, e285–e292.
- Backes, M.; Dingemans, S.A.; Dijkgraaf, M.G.W.; Rogier van den Berg, R.; Dijkman, B.V.; Hoogendoorn, J.M.; Joosse, P.; Ritchie, E.D.; Roerdink, W.H.; Schots, J.P.M.; et al. Effect of Antibiotic Prophylaxis on Surgical Site Infections Following Removal of Orthopedic Implants Used for Treatment of Foot, Ankle, and Lower Leg Fractures: A Randomized Clinical Trial. JAMA 2017, 318, 2438–2445.
- Sezer, N.; Evis, Z.; Koç, M. Additive manufacturing of biodegradable magnesium implants and scaffolds: Review of the recent advances and research trends. J. Magnes. Alloy. 2021, 9, 392–415.
- Poinern, G.E.J.; Brundavanam, S.; Fawcett, D. Biomedical Magnesium Alloys: A Review of Material Properties, Surface Modifications and Potential as a Biodegradable Orthopaedic Implant. Am. J. Biomed. Eng. 2013, 2, 218–240.
- Witte, F.; Kaese, V.; Haferkamp, H.; Switzer, E.; Lindenberg, A.M.; Wirth, C.J.; Windhagen, H. In vivo corrosion of four magnesium alloys and the associated bone response. Biomaterials 2005, 26, 3557–3563.
- Witte, F.; Hort, N.; Vogt, C.; Cohen, S.; Kainer, K.U.; Willumeit, R.; Feyerabend, F. Degradable biomaterials based on magnesium corrosion. Curr. Opin. Solid State. Mater. Sci. 2008, 12, 63–72.
- Staiger, M.P.; Pietak, A.M.; Huadmai, J.; Dias, G. Magnesium and its alloys as orthopedic biomaterials: A review. Biomaterials 2006, 27, 1728–1734.
- Vormann, J. Magnesium: Nutrition and metabolism. Mol. Aspects Med. 2003, 24, 27–37.
- Zhao, D.; Witte, F.; Lu, F.; Wang, J.; Li, J.; Qin, L. Current status on clinical applications of magnesium-based orthopaedic implants: A review from clinical translational perspective. Biomaterials 2017, 112, 287–302.
- Saris, N.-E.L.; Mervaala, E.; Karppanen, H.; Khawaja, J.A.; Lewenstam, A. Magnesium: An update on physiological, clinical and analytical aspects. Clinica. Chimica. Acta 2000, 294, 1–26.
- Kannan, M.B.; Raman, R.K.S. In vitro degradation and mechanical integrity of calcium-containing magnesium alloys in modified-simulated body fluid. Biomaterials 2008, 29, 2306–2314.
- Arif, Z.U.; Khalid, M.Y.; Noroozi, R.; Sadeghianmaryan, A.; Jalalvand, M.; Hossain, M. Recent advances in 3D-printed polylactide and polycaprolactone-based biomaterials for tissue engineering applications. Int. J. Biol. Macromol. 2022, 218, 930–968.
- Khalid, M.Y.; Arif, Z.U. Novel biopolymer-based sustainable composites for food packaging applications: A narrative review. Food Packag. Shelf Life 2022, 33, 100892.
- Feng, P.; Jia, J.; Liu, M.; Peng, S.; Zhao, Z.; Shuai, C. Degradation mechanisms and acceleration strategies of poly (lactic acid) scaffold for bone regeneration. Mater. Des. 2021, 210, 110066.
- Shu-Rong, Z.; Huai-Mei, Z.; Snao-Zhen, Q.; Guo-Wei, S.; Kaper, R.F. A long-term study of the efficacy and acceptability of a single-rod hormonal contraceptive implant (Implanon®) in healthy women in China. Eur. J. Contracept. Reprod. Health Care 1999, 4, 85–93.
- Yang, Y.; He, C.; Dianyu, E.; Yang, W.; Qi, F.; Xie, D.; Shen, L.; Peng, S.; Cijun Shuai, C. Mg bone implant: Features, developments and perspectives. Mater. Des. 2020, 185, 108259.
- Zhao, S.; Xie, K.; Guo, Y.; Tan, J.; Wu, J.; Yang, Y.; Fu, P.; Wang, L.; Jiang, W.; Hao, Y. Fabrication and Biological Activity of 3D-Printed Polycaprolactone/Magnesium Porous Scaffolds for Critical Size Bone Defect Repair. ACS Biomater. Sci. Eng. 2020, 6, 5120–5131.
- Dong, Q.; Zhang, M.; Zhou, X.; Shao, Y.; Li, J.; Wang, L.; Chu, C.; Xue, F.; Yao, Q.; Bai, J. 3D-printed Mg-incorporated PCL-based scaffolds: A promising approach for bone healing. Mater. Sci. Eng. C 2021, 129, 112372.
- Long, J.; Zhang, W.; Chen, Y.; Teng, B.; Liu, B.; Li, H.; Yao, Z.; Wang, D.; Li, L.; Yu, X.F.; et al. Multifunctional magnesium incorporated scaffolds by 3D-Printing for comprehensive postsurgical management of osteosarcoma. Biomaterials 2021, 275, 120950.
- Zhou, X.; Zhou, X.; Zhou, G.; Junka, R.; Chang, N.; Anwar, A.; Wang, H.; Yu, X. Fabrication of polylactic acid (PLA)-based porous scaffold through the combination of traditional bio-fabrication and 3D printing technology for bone regeneration. Colloids Surf. B Biointerfaces 2021, 197, 111420.
- Baptista, R.; Guedes, M.; Pereira, M.F.C.; Maurício, A.; Carrelo, H.; Cidade, T. On the effect of design and fabrication parameters on mechanical performance of 3D printed PLA scaffolds. Bioprinting 2020, 20, e00096.
- Singh, S.; Singh, G.; Prakash, C.; Ramakrishna, S.; Lamberti, L.; Pruncu, C.I. 3D printed biodegradable composites: An insight into mechanical properties of PLA/chitosan scaffold. Polym. Test. 2020, 89, 106722.
- Esmaeili, S.; Akbari, A.H.; Motififard, M.; Saber-Samandari, S.; Montazeran, A.H.; Bigonah, M.; Sheikhbahaei, E.; Khandan, A. bA porous polymeric–hydroxyapatite scaffold used for femur fractures treatment: Fabrication, analysis, and simulation. Eur. J. Orthop. Surg. Traumatol. 2020, 30, 123–131.
- Aveen, K.P.; Bhajathari, F.V.; Jambagi, S.C. 3D Printing & Mechanical Characteristion of Polylactic Acid and Bronze Filled Polylactic Acid Components. IOP Conf. Ser. Mater. Sci. Eng. 2018, 376, 12042.
- Mehboob, A.; Mehboob, H.; Chang, S.-H. Evaluation of unidirectional BGF/PLA and Mg/PLA biodegradable composites bone plates-scaffolds assembly for critical segmental fractures healing. Compos. Part A Appl. Sci. Manuf. 2020, 135, 105929.
- Shuai, C.; Li, Y.; Feng, P.; Guo, W.; Yang, W.; Peng, S. Positive feedback effects of Mg on the hydrolysis of poly-l-lactic acid (PLLA): Promoted degradation of PLLA scaffolds. Polym. Test. 2018, 68, 27–33.
- Trachtenberg, J.E.; Placone, J.K.; Smith, B.T.; Fisher, J.P.; Mikos, A.G. Extrusion-based 3D printing of poly(propylene fumarate) scaffolds with hydroxyapatite gradients. J. Biomater. Sci. Polym. Ed. 2017, 28, 532–554.
- Wong, H.M.; Chu, P.K.; Leung, F.K.L.; Cheung, K.M.C.; Luk, K.D.K.; Yeung, K.W.K. Engineered polycaprolactone–magnesium hybrid biodegradable porous scaffold for bone tissue engineering. Prog. Nat. Sci. Mater. Int. 2014, 24, 561–567.
- Adhikari, U.; An, X.; Rijal, N.; Hopkins, T.; Khanal, S.; Chavez, T.; Tatu, R.; Sankar, J.; Little, K.J.; Hom, D.B.; et al. Embedding magnesium metallic particles in polycaprolactone nanofiber mesh improves applicability for biomedical applications. Acta Biomater. 2019, 98, 215–234.
- Dong, Q.; Li, Y.; Jiang, H.; Zhou, X.; Liu, H.; Lu, M.; Chu, C.; Xue, F.; Bai, J. 3D-cubic interconnected porous Mg-based scaffolds for bone repair. J. Magnes. Alloy. 2021, 9, 1329–1338.
- Jiao, Z.; Luo, B.; Xiang, S.; Ma, H.; Yu, Y.; Yang, W. 3D printing of HA/PCL composite tissue engineering scaffolds. Adv. Ind. Eng. Polym. Res. 2019, 2, 196–202.
- Liu, W.; Wang, D.; Huang, J.; Wei, Y.; Xiong, J.; Zhu, W.; Duan, L.; Chen, J.; Sun, R.; Wang, D. Low-temperature deposition manufacturing: A novel and promising rapid prototyping technology for the fabrication of tissue-engineered scaffold. Mater. Sci. Eng. C 2017, 70, 976–982.
- Li, Y.; Zhou, J.; Pavanram, P.; Leeflang, M.A.; Fockaert, L.I.; Pouran, B.; Tümer, N.; Schröder, K.-U.; Mol, J.M.C.; Weinans, H.; et al. Additively manufactured biodegradable porous magnesium. Acta Biomater. 2018, 67, 378–392.
- Zhang, X.; Li, X.-W.; Li, J.-G.; Sun, X.-D. Preparation and mechanical property of a novel 3D porous magnesium scaffold for bone tissue engineering. Mater. Sci. Eng. C 2014, 42, 362–367.
- Jiang, G.; He, G. A new approach to the fabrication of porous magnesium with well-controlled 3D pore structure for orthopedic applications. Mater. Sci. Eng. C 2014, 43, 317–320.
- Cheng, M.; Wahafu, T.; Jiang, G.; Liu, W.; Qiao, Y.; Peng, X.; Cheng, T.; Zhang, X.; He, G.; Liu, X. A novel open-porous magnesium scaffold with controllable microstructures and properties for bone regeneration. Sci Rep. 2016, 6, 24134.
- Liao, Y.; Chen, D.; Niu, J.; Zhang, J.; Wang, Y.; Zhu, Z.; Yuan, G.; He, Y.; Jiang, Y. In vitro degradation and mechanical properties of polyporous CaHPO4-coated Mg–Nd–Zn–Zr alloy as potential tissue engineering scaffold. Mater. Lett. 2013, 100, 306–308.
- Zhuang, H.; Han, Y.; Feng, A. Preparation, mechanical properties and in vitro biodegradation of porous magnesium scaffolds. Mater. Sci. Eng. C 2008, 28, 1462–1466.
- Parai, R.; Bandyopadhyay-Ghosh, S. Engineered bio-nanocomposite magnesium scaffold for bone tissue regeneration. J. Mech. Behav. Biomed. Mater. 2019, 96, 45–52.
- Lee, J.; Farag, M.M.; Park, E.K.; Lim, J.; Yun, H. A simultaneous process of 3D magnesium phosphate scaffold fabrication and bioactive substance loading for hard tissue regeneration. Mater. Sci. Eng. C 2014, 36, 252–260.
- Malikmammadov, E.; Tanir, T.E.; Kiziltay, A.; Hasirci, N. Preparation and characterization of poly(ε-caprolactone) scaffolds modified with cell-loaded fibrin gel. Int. J. Biol. Macromol. 2019, 125, 683–689.
- Ma, R.; Wang, W.; Yang, P.; Wang, C.; Guo, D.; Wang, K. In vitro antibacterial activity and cytocompatibility of magnesium-incorporated poly(lactide-co-glycolic acid) scaffolds. Biomed. Eng. Online 2020, 19, 12.
- Shasteen, C.; Choy, Y.B. Controlling degradation rate of poly(lactic acid) for its biomedical applications. Biomed. Eng. Lett. 2011, 1, 163.
- Zerankeshi, M.M.; Bakhshi, R.; Alizadeh, R. Polymer/metal composite 3D porous bone tissue engineering scaffolds fabricated by additive manufacturing techniques: A review. Bioprinting 2022, 25, e00191.
- Wang, Y.; Wei, X.; Duan, J.; Yang, J.; Zhang, N.; Huang, T.; Wang, Y. Greatly enhanced hydrolytic degradation ability of poly(L-lactide) achieved by adding poly(ethylene glycol). Chin. J. Polym. Sci. 2017, 35, 386–399.
- Höglund, A.; Hakkarainen, M.; Edlund, U.; Albertsson, A.-C. Surface Modification Changes the Degradation Process and Degradation Product Pattern of Polylactide. Langmuir 2010, 26, 378–383.
- Cheng, S.; Zhang, D.; Li, M.; Liu, X.; Zhang, Y.; Qian, S.; Peng, F. Osteogenesis, angiogenesis and immune response of Mg-Al layered double hydroxide coating on pure Mg. Bioact. Mater. 2021, 6, 91–105.
- Chen, W.; Xu, K.; Tao, B.; Dai, L.; Yu, Y.; Mu, C.; Shen, X.; Hu, Y.; He, Y.; Cai, K. Multilayered coating of titanium implants promotes coupled osteogenesis and angiogenesis in vitro and in vivo. Acta Biomater. 2018, 74, 489–504.
- Safari, B.; Aghanejad, A.; Roshangar, L.; Davaran, S. Osteogenic effects of the bioactive small molecules and minerals in the scaffold-based bone tissue engineering. Colloids Surf. B Biointerfaces 2021, 198, 111462.
- Mabrouk, M.; Beherei, H.H.; Das, D.B. Recent progress in the fabrication techniques of 3D scaffolds for tissue engineering. Mater. Sci. Eng. C 2020, 110, 110716.
- Gelinsky, M.; Welzel, P.B.; Simon, P.; Bernhardt, A.; König, U. Porous three-dimensional scaffolds made of mineralised collagen: Preparation and properties of a biomimetic nanocomposite material for tissue engineering of bone. Chem. Eng. J. 2008, 137, 84–96.
- Hartgerink, J.D.; Beniash, E.; Stupp, S.I. Peptide-amphiphile nanofibers: A versatile scaffold for the preparation of self-assembling materials. Proc. Natl. Acad. Sci. USA 2002, 99, 5133–5138.
- Matson, J.B.; Stupp, S.I. Self-assembling peptide scaffolds for regenerative medicine. Chem. Commun. 2012, 48, 26–33.
- Tampieri, A.; Sandri, M.; Landi, E.; Pressato, D.; Francioli, S.; Quarto, R.; Martin, I. Design of graded biomimetic osteochondral composite scaffolds. Biomaterials 2008, 29, 3539–3546.
- Lu, T.; Li, Y.; Chen, T. Techniques for fabrication and construction of three-dimensional scaffolds for tissue engineering. Int. J. Nanomed. 2013, 8, 337–350.
- Barnes, C.P.; Sell, S.A.; Boland, E.D.; Simpson, D.G.; Bowlin, G.L. Nanofiber technology: Designing the next generation of tissue engineering scaffolds. Adv. Drug Deliv. Rev. 2007, 59, 1413–1433.
- Adhikari, U.; Rijal, N.P.; Khanal, S.; Pai, D.; Sankar, J.; Bhattarai, N. Magnesium incorporated chitosan based scaffolds for tissue engineering applications. Bioact. Mater. 2016, 1, 132–139.
- Allaf, R.M. Melt-molding technologies for 3D scaffold engineering. In Functional 3D Tissue Engineering Scaffolds: Materials, Technologies, and Applications; Elsevier BV: Amsterdam, The Netherlands, 2018.
- Dutta, S.; Devi, K.B.; Roy, M. Processing and degradation behavior of porous magnesium scaffold for biomedical applications. Adv. Powder Technol. 2017, 28, 3204–3212.
- Brown, A.; Zaky, S.; Ray, H.; Sfeir, C. Porous magnesium/PLGA composite scaffolds for enhanced bone regeneration following tooth extraction. Acta Biomater. 2015, 11, 543–553.
More