Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Leticia Diaz and Version 2 by Amina Yu.
Artificial insemination (AI) is a widely used technique in swine production. Advances in the technique have made it possible to store spermatozoa at temperatures of 15–20 °C for short periods, up to ten days. Unfortunately, it is currently associated with bacterial contamination of semen during collection and dilution. Although the temperature is reduced to induce sperm inactivity during storage, bacterial growth can still occur [4]. Bacterial growth has been associated with deleterious effects on semen quality and shelf life, such as sperm agglutination, decreased sperm motility and viability. In addition, reproductive output after AI can also be affected by bacteriospermia.
- antibiotics
- antibiotic resistance
- bacteria contamination
- bacteriospermia
- boar semen
1. Bacterial Presence in Boar Semen
Healthy boars usually do not contain bacteria in their semen. Nonetheless, the preputial diverticulum, skin, and hair of the boar contain several microorganisms. In addition, the collection environment or human intervention can contribute to contamination [1][2]. Therefore, there are a large number of species of bacteria in boar semen. The percentage of bacteria contaminated semen samples has been described as 32% in 2002 and 2003, 17% in 2005, and 26% in 2006 [1][2][3][2,6,8]. However, this can be even higher (up to 66.7%) in the case of boars [4][1]. The bacterial and fungal load of fresh boar semen is about 82.41 ± 0.149 × 103 CFU/mL and 0.354 ± 0.140 × 103 CFU/mL after dilution of the ejaculates [5][9].
Most of the contaminants in semen cultures have been reported to be Gram-negative bacteria belonging to the family Enterobacteriaceae [2][6][7][5,6,10]. Recent studies have evaluated the seminal microbiome of a group of boars Proteobacteria (39.1–57.3%), Firmicutes (27.5–31.17%), Actinobacteria (3.41–14.9%), and Bacteroidetes (4.24–5.7%) as the most abundant phyla [8][9][11,12]. In addition, the species Bacillus megaterium, Brachybacterium faecium, and B. coagulans were recognized as post-ejaculation contamination from soil, feces, and water sources. On the other hand, a relatively low percentage of Escherichia coli, Clostridium difficile, C. perfringens, C. botulinum, and Mycobacterium tuberculosis were found in semen [8][11]. Other studies indicate that species such as Candida parapsilosis/sakeare and Escherichia coli are found in higher percentages (92% and 81.2%, respectively) [5][9]. This bacterial contamination can be problematic since they could be pathogenic agents for the sows [8][11].
MALDI-TOF mass spectrometry and API 20E (Analytical Profile Index) were also used to determine the Gram-negative bacterial species present in porcine seminal samples, where a majority of contaminating bacteria belong to the family Enterobacteriaceae, including Seratia marcescens (19.56%), Proteus mirabilis (15.21%), E. coli (10.86%); and to the Pseudomonaceae family, including Ralstonia picketii (17.39%), Burkholderia cepacia (10.86%), Pseudomonas aeruginosa (8.69%) and Pseudomonas fluorescens (4.34%), respectively [10][13]. Similar results are reported from Brazil boars’ semen samples, showing 43% contamination with a predominance of Gram-negative bacteria such as P. aeruginosa, P. mirabilis, E. coli, Kerstersia gyiorum, Aerococcus viridans, Brevibacterium casei, Providencia stuartii, Citrobacter koseri, and Staphylococcus pasteuri [11][14]. Leptospira is also present in some samples; however, this is not a pathogen of concern in porcine seminal samples [12][15].
On the other hand, bacterial diversity changes have been observed in response to seasonal conditions. For example, Lactobacillus is highly abundant in samples taken in winter, which are positively associated with sperm quality and reproductive performance. In contrast, Pseudomonas is highly abundant in summer samples and is negatively associated with sperm quality and reproductive potential [9][12]. Metagenomics by high-throughput sequencing results also suggested bacterial community variation from one AI center to another. This is expected due to the different environmental characteristics in which the pigs are kept. In particular, the type of flooring on which the animals are housed might influence bacterial semen contamination [13][16]. Therefore, providing hygienically impeccable housing conditions will prevent bacterial contamination and preserve spermatic functionality and sanitary quality [14][17]. Actions such as adequate safeguards against the entry of pathogens from outside, systematic veterinary controls, vaccination, deworming protocols, and pest control are required.
About 60% of the variability in the bacterial contamination profile in semen samples is explained by the hygienic conditions of the different control points in artificial insemination [15][18]. The process of sample collection has been analyzed step by step to determine the critical points for bacterial contamination. These are the dripping of preputial fluid from the technician’s hand; the collection time over seven minutes; and the presence of long preputial hair [16][19]. At the laboratory level, sinks or drains show a high rate of bacterial contamination; in fact, multiple isolates of multidrug-resistant bacteria have been isolated from these areas [16][19]. Elevated bacterial contamination is also found in heating cabinets, ejaculate transfer, manual operating elements, and laboratory surfaces [4][1]. Moreover, large amounts of resistant bacteria have been reported in the mucosa of the boar’s prepuce [17][20]. Hygiene management during the collection, maintenance, and insemination processes can significantly reduce bacterial contamination and, therefore, diminish the use of conventional antibiotics [15][18]. Although this contamination cannot be avoided entirely, it can be contained by appropriate hygiene measures and process optimization [14][17].
2. Semen Cold Storage and Bacterial Contamination
AI plays an important role in boar reproduction; 93% of sows are inseminated by artificial insemination, and 99% of all sows are inseminated with extended semen [14][18][17,21]. AI is the most widely used reproductive biotechnology in swine. It is almost entirely performed with liquid-preserved semen, as it is used to bridge the time gap between collection and insemination [19][22]. Preserved extended semen allow for national and international trade of semen doses and increase the efficiency of using an ejaculate, as lower concentrations of spermatozoa are used [19][22]. Extended semen consists of seminal doses of 50–100 mL at a concentration of 1–3 billion spermatozoa per dose [20][23]. To obtain a dose, the ejaculate-rich fraction is collected and then diluted, allowing the preparation of multiple required doses [20][23]. The diluent is an aqueous solution that increases the volume of the ejaculate to the required amount. It helps to reduce the metabolic activity of the spermatozoa, preserves their function, and maintains an adequate level of fertility [20][23].
There are short-term and long-term extender media; the difference between the two is that the short-term allows liquid preservation for up to three days, while the long-term allows preservation for up to ten days [20][23]. Usually, the diluted semen is kept at 17 °C for the recommended time. However, microbial growth can occur at this temperature, damaging the quality and functionality of the spermatozoa. Therefore, in recent years, the diluted samples have been preserved at 5 °C [21][22][24,25].
Even though bacterial growth can be diminished, strains on contaminated semen samples can still colonize and damage the extended semen. This is why antibiotics are commonly added to each extended semen sample at the time of dilution in order to control or delay the colonization of microorganisms. A recent study evaluated the use of both types of extenders (short- and long-term) in relation to microbial growth. The authors found that total colony counts are not affected by the storage time (3 vs. 7 days); however, Gram-negative counts appear to be higher on day 3 of storage compared to day 7. In addition, the bacterial counts in the short-term extender were higher than those in the long-term extender on the third day of storage [23][26]. Another study observed that adding an extender supplemented with antibiotic at 17 °C controls bacterial growth for only a few hours. Strains such as Bacillus spp., Pseudomonas spp., Staphylococcus spp., and Streptococcus spp. significantly increased their growth after 24 h, failing to adequately maintain the sample quality for five days of storage. Therefore, there is a current necessity in the preservation of semen samples and in the control of their bacterial growth [24][27].
3. Consequences of Bacterial Contamination on Boar Semen
The degree of bacterial contamination in ejaculates directly influences sperm quality parameters during cold preservation (Figure 1) [25][28].
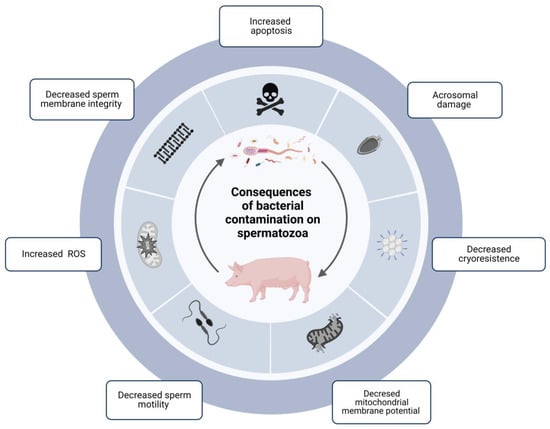
Figure 1. Diagram showing the consequences of the contamination of bacterial communities in boar sperm and how they directly influence sperm quality parameters during cold storage. The presence of bacteria in boar semen has significant consequences on the quality and functionality parameters of spermatozoa: damage to the acrosome, decreased mitochondrial membrane potential, cryoresistance, sperm motility, and integrity of the sperm membrane, and increased ROS and apoptosis. ROS: Reactive oxigen species. Created with BioRender.com.
Among the negative consequences recorded for contaminated boar sperm, seminal samples with bacterial counts over 1.4 × 104 CFU/mL showed decreased motility after 108 and 168 h of storage at 16 °C. In addition, the pH decreases from 7.2 to 6.0 between 24 and 168 h of storage in samples with high contamination levels [6][20][26][27][28][29][30][5,23,29,30,31,32,33]. However, pH changes are species-specific, as E. coli and C. perfringens infections have been reported to lead to an alkalinization of the seminal medium [31][34]. In addition, other consequences associated with integrity and function have been described. Some bacteria affect the integrity of the sperm membrane, reducing their viability, damaging the acrosome, and decreasing its cryoresistance [32][35]. Mitochondrial membrane potential has also been associated with bacterial concentration and storage time [33][36]. Finally, other studies have demonstrated that bacteria cause agglutination of the cells, preventing the correct mobilization of the spermatozoa [28][30][34][35][31,33,37,38]. This agglutination is associated with type 1 fimbriae in some bacteria, which bind to glycoprotein receptors on sperm cell surfaces. These fimbriae mediate specific adhesion to the host cell surface. Tail-to-tail, tail-to-head, or head-to-head adhesion occur between spermatozoa [30][33]. All these consequences have been found to be increased in semen samples stored at 15–17 °C [29][32] and to a much lesser extent in samples stored at 5 °C; this is why the shelf life of semen doses stored at 15–17 °C is reduced. One study described that the presence of 106 or 108 CFU/mL of P. aeruginosa in capacitating media results in low motility of spermatozoa and a decrease in phosphotyrosine—an indicator of sperm capacitation [27][30]. This result is of great concern since fertilization of oocytes with non- or low-capacitated spermatozoa is inefficient.
Additionally, the adhesion of bacteria to spermatozoa can also occur. Different bacteria, such as E. coli and C. perfringens, have different adhesion patterns. The proportions of sperm with adhering E. coli are higher than those with C. perfringens from the fourth day of storage over 5 °C. The adhesion of both bacteria to the spermatozoa surface is non-specific, but it shows a significantly higher adhesion to the middle piece in those infected with E. coli, and higher adhesion to the main piece in those infected with C. perfringens, causing significantly more damage to the affected area [36][39]. Additionally, bacterial adhesion increases with higher infective doses and longer storage times [37][41].
Bacteria, such as P. mirabilis, also produce outer membrane vesicles that influence boar sperm function by inducing sperm membrane reconstruction, autophagy, and apoptosis. They affect motility, increase ROS production, alter oocyte binding, increase LC3 and caspase-3 expression, and decrease Bcl2 anti-apoptosis in spermatozoa [38][40]. In addition, large counts of Gram-negative bacteria produce large amounts of lipopolysaccharides (LPS). LPS can directly bind to the sperm head region, decreasing sperm motility and inducing sperm apoptosis due to its toxicity [39][42]. Thus, sperm damage is not only directly associated with the bacterial presence on the medium but also with their subproducts. Although functionality and quality are affected, no significant changes in sperm morphology associated with microbial contamination have been described in samples stored for 11 days at 15 °C and 96 h at 37 °C [29][32].
4. Antibiotic Use for Preservation of Boar Semen
The addition of antibiotics to the diluent is obligatory by law (EU Directive 90/429/EEC Annex C). Therefore, most commercial sperm media already include antibiotics or those added before semen storage to prevent microbial growth. Several combinations and concentrations have been tested, including 200 μg/mL gentamicin + 200 μg/mL florfenicol, 200 μg/mL gentamicin + 200 μg/mL polymyxin B, 100 μg/mL gentamicin + 100 μg/mL florfenicol and 100 μg/mL gentamicin + and 100 μg/mL polymyxin B. The combination 100 μg/mL gentamicin + 100 μg/mL florfenicol has shown the highest percentage of progressive motility, viability, and mitochondrial membrane potential over ten days. In addition, this combination does not damage DNA integrity, allowing for the desired reproductive performance in terms of the number of pregnant sows and litter size [40][43]. Sulfanilamide has also been tested for porcine semen storage at 17 °C for 6 days, where concentration of 0.02 g/L is optimal for maintaining the quality parameters. This antibiotic controls the growth of Staphylococcus and Pseudomonas and enables an increase in litter size and pregnancy rate [41][44]. In addition, with traditional antibiotics, such as gentamicin, half a dose can reduce the bacterial load to preserve sperm quality and be safe for sow [42][45]. High concentrations of gentamicin did not have optimal results. In this context, Schulze et al. [43][46] proposed using an extender without antibiotics and manually supplementing it with the right amount of gentamicin according to the sample volume to ensure its efficiency.
Regarding commercial sperm media, the intrinsic capacity of Androstar® Premium (Minitube) and Beltsville Thawing Solution, BTS® (Pursel and Johnson) is efficient compared to the use of extrinsic antibiotics; therefore, with their use, no extra dose of antibiotics is necessary [42][45]. On the other hand, using Androstar plus® at 17 °C for 72 h with antibiotics such as gentamicin, aminoglycoside, cephalosporin, lincomycin, and spectinomycin may decrease the total bacterial count. However, gentamicin alone is not sufficiently effective against Gram-positive bacteria. In addition, it was reported that with the combination of three antibiotics, bacterial growth is optimally controlled, showing only a few colonies of Enterococcus hirae, B. subtilis, and Corynebacterium spp. Therefore, the use of antibiotics should be specific and should not be added randomly in commercial media. Specific antibiotics for semen extension and hygiene standards should preserve semen quality and reduce the indiscriminate use of antibiotics and the antibiotic resistance crisis in the pig industry [44][47].
The bacterial exposition to different concentrations of antibiotics leads to selection pressure and increases antibiotic resistance emergence. In boar semen preservation, ejaculates are diluted to adjust sperm concentration; therefore, the final antibiotic concentration should depend on the dilution factor of the ejaculate. In some swine AI centers, this method is already being used with successful results [21][24]. Despite the efforts of some groups to improve and optimize the use of antibiotics, multiple studies have reported antibiotic resistance in semen samples, including E. coli against chloramphenicol, Neisseria meningitides against spectinomycin, and S. aureus against linezolid [45][48]. Worryingly, Costinar et al. [10][13] observed that 56.52% of isolates were resistant to gentamicin and 58.69% to penicillin, demonstrating that the indiscriminate use of antibiotics stimulates antibiotic resistance [10][13].
