Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Dean Liu and Version 1 by Fabrizio Bianchi.
Lung cancer is the leading cause of cancer-related mortality in the world. The development of drug resistance represents a major challenge for the clinical management of patients. In the last years, microRNAs have emerged as critical modulators of anticancer therapy response.
- non-small cell lung cancer
- NSCLC
- microRNAs
1. Introduction
Lung cancer represents the main cause of cancer-related death worldwide. Despite recent advances in lung cancer management, the prognosis of patients remains adverse with five-year survival rates ranging from 6% in the case of metastatic cancer (i.e., regional and distant metastases) up to 33–60% in patients with localized tumours [1]. Non-small-cell lung cancer (NSCLC) is the most common subtype of lung cancer (i.e., 85% of cases; [2]). Therapeutic approaches for the treatment of NSCLC include surgery, radiotherapy, chemotherapy, targeted therapy, and immunotherapy used alone or in combination. Although the number of therapeutic agents for NSCLC treatment has dramatically increased in the last years, a long-term impact in the setting of advanced disease has not been reached due to the acquisition of drug resistance [3]. Thus, a better understanding of the mechanisms underlying the response to therapy is needed to achieve long-term and durable disease remission. In this regard, the investigation of the coding and non-coding transcriptome (e.g., microRNAs [miRNAs]) of NSCLC in the last years has led to important findings on which and how specific molecular mechanisms are involved in drug resistance. Indeed, several studies which focused on miRNA expression profiling in NSCLC have shown their frequent altered expression pattern in lung cancer [4],[5],[6] and their role as master regulators of cancer genes involved in chemotherapy response [7].
MiRNAs are non-coding RNAs (ncRNAs) of ~22 nucleotides, which act mainly as post-transcriptional gene regulators [8]. The impact of miRNAs in the regulation of the transcription of genes can be relevant since one miRNA can potentially target the messenger RNAs (mRNAs) of distinct genes. Yet, the expression of a single gene can be regulated by different miRNAs containing a similar seed sequence (i.e., a conserved heptametrical sequence which is mostly situated at positions 2–7 from the miRNA 5′-end) which, ultimately, targets the relative complementary sequence in the mRNA, i.e., the miRNA recognition elements (MREs). miRNA can exert other non-canonical functions, such as activating Toll-like receptors that are involved in the activation of innate immune response, binding non-argonaute (AGO) proteins (e.g., hnRNPE2) and triggering transcription, binding other ncRNAs which inhibit miRNA functions (i.e., miRNA sponges) [[9]].
2. Annotation of miRNAs with a Functional Role in NSCLC Therapy Resistance
Researchers initially performed a literature search (see Methods) and reviewed a total of 502 publications (from 2009 to June 2022) that report signatures of miRNAs involved in the responses to different drugs used to treat NSCLC patients. Next, researchers analyzed miRNA expression regulation in the drug-resistant condition and the various experimental models/cohort of samples (Figure 1). Furthermore, researchers assigned an ascending score depending on whether the role of miRNAs was investigated in cell lines (1), in animal models (2), human cancer tissues (3), cell lines AND human cancer tissues OR animal models AND human cancer tissues (4), cell lines AND animal models AND human cancer tissues (5). Importantly, this score describes the strength of the evidence supporting the role of particular miRNAs in drug resistance and can be used to select a miRNA based on the level of evidence (Figure 1). When available, researchers also reported genes that were (i) experimentally validated as a direct target of the indicated miRNA, and (ii) functionally involved in miRNA-mediated regulation of drug resistance. Finally, each miRNA entry on the database was associated with the relative citation by reporting the Pubmed ID (PMID) of the publication, thus allowing fast retrieval of the study of interest. In the case of multiple independent reports about a single miRNA, researchers merged the information derived from all the studies to obtain a unique miRNA entry.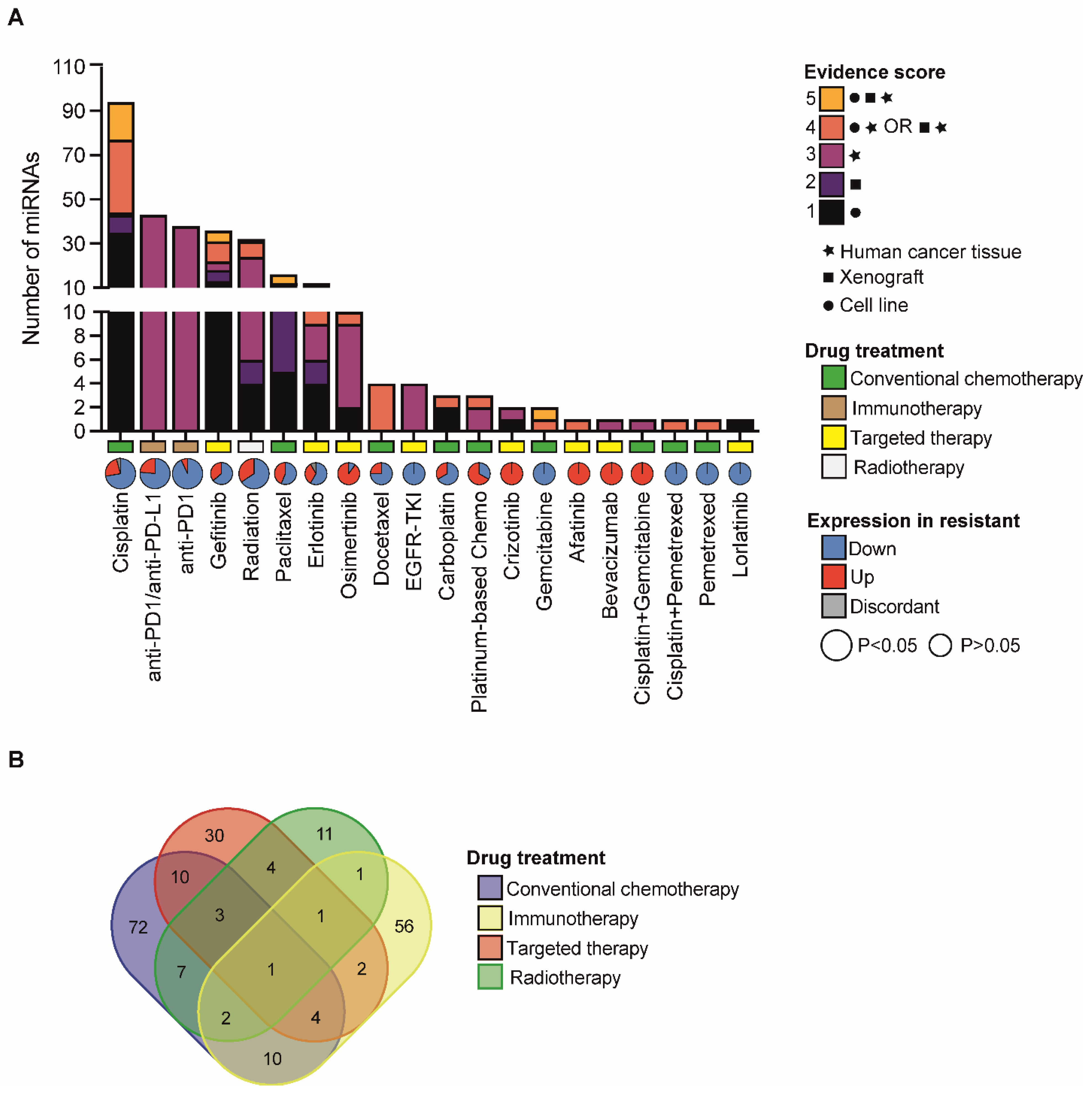
Figure 1. Landscape of miRNAs associated with therapy response in NSCLC. (A) Bar chart reporting the number of miRNAs that are associated with therapy response in NSCLC. Different colours indicate relative evidence scores which were assigned to miRNAs based on the type of experiment conducted (i.e., cell lines, xenograft models and/or human cancer tissue). In X-axes, pie charts indicate the fraction of miRNAs whose expression is upregulated (red) or downregulated (blue) in resistant samples vs. sensitive ones. In grey, fraction of miRNAs with a discordant trend of expression regulation (up or down) in independent studies. Pie sizes are proportional to the statistical significance (binomial test) relative to the unbalance in up- or downregulated miRNAs, as per the legend. Type of drug treatment is also indicated in X-axes, as per the legend. (B) Venn diagram of the overlapping miRNAs associated to the indicated category of drug treatment.
3. The Landscape of miRNAs Which Regulate Cisplatin Resistance in NSCLC
Out of a total of 42 drugs used in the NSCLC clinical setting, found that the expression/activity of a total of 212 unique miRNAs was associated with sensitivity to 22 drugs (Figure 1). Cisplatin emerged as the top drug whose sensitivity can be functionally modulated by miRNAs and showed the highest number of miRNAs with the strongest evidence score (Figure 1). This could be partly explained by an earlier approval (in 1978) of Cisplatin for lung cancer treatment by US food and drug administration (FDA) in comparison with more recently approved drugs [10]. Interestingly, researchers noticed that the resistance to cisplatin was generally and significantly associated with an overall loss of miRNA expression (Figure 1A), in line with the prevalent role of miRNAs as tumor suppressors [11]. Similarly, researchers observed a frequent miRNA downregulation in tumors refractory to radiotherapy and immunotherapy (Figure 1A). Notably, there is a large fraction of miRNAs involved in mechanisms of resistance to conventional chemotherapy, radio, and immunotherapy, which suggests a global transcriptome reprogramming that impacts molecular mechanisms at multiple levels (Figure 1A). Contrariwise, the number of miRNAs involved in mechanisms of resistance to targeted agents is lower and with a more heterogeneous trend of the regulation (Figure 1A), which is reasonable in the case of targeted agents that act by inhibiting/activating specific molecular mechanisms to kill tumor cells [12]. Interestingly, researchers found that a number of miRNAs were commonly altered in response to drugs belonging to different categories (Figure 1B), which suggests common regulatory pathways to be involved in drug resistance. Indeed, as researchers previously discussed, several of these miRNAs have been already reported to regulate common pathways/signaling cascades. Over the last 30 years of research, a variety of molecular mechanisms were described to be involved in the onset of resistance to cisplatin which were extensively reviewed elsewhere [13]. Yet, a recent review reported a large number of genes (~900) to be implicated in platinum response in several human malignancies [14]. Mechanisms of cisplatin resistance in tumor cells can be summarized in those involved in (i) cisplatin import/export, (ii) DNA damage repair, (iii) transcriptome reprogramming by sequestering miRNAs (miRNA sponges), (iv) pro-survival and apoptotic signaling pathways, (v) oxidative stress, (vi) cancer signalling pathways, (vii) epithelial-to-mesenchymal transition (EMT), and (viii) autophagy. Here below, wresearchers provide a schematic report:- −
-
Cisplatin import/export: regulation of cisplatin import/export is an important mechanism of resistance to cisplatin. MiR-369-3p overexpression promoted cisplatin resistance by direct regulation of the expression of the SLC35F5 gene, a nucleotide sugar transporter involved in drug uptake [15]. In addition, miR-495-3p regulates cisplatin resistance by modulating the expression of ATP7A, a copper transporter that regulate cisplatin efflux from the cells [16].
- −
-
DNA damage repair: upon entering the cells, cisplatin becomes activated by the displacement of chloride atoms by water molecules. Active cisplatin exerts its cytotoxic function mainly by generating monoadducts and crosslinks at the level of the DNA. Therefore, genetic and epigenetic alterations of genes involved in DNA damage response represent a major mechanism to cope with cisplatin-induced cell death. In line with this, miR-92a-3p regulates cisplatin resistance by directly binding to the 3′ untranslated region (UTR) of RAD21 mRNA, a member of the cohesin complex that can promote DNA repair at the G2 phase of the cell cycle [17]. Yet, miR-17-5p, which belongs to the same cluster of miR-92a-3p (e.g., miR-17-92 cluster), can protect cancer cells from cisplatin-induced apoptosis by regulating CDKN1A, a cell cycle inhibitor that blocks DNA synthesis by G1 arrest and whose levels are increased upon accumulation of DNA damage due to activation of ATM and ATR and consequent TP53 stabilization [17].
- −
-
miRNA sponges: long non-coding RNAs (lncRNAs) are transcripts longer than 200 nucleotides that do not code for functional proteins. Since lncRNAs can contain MREs in their sequence, they can sequester miRNAs and impair their activity on target genes, therefore, acting as competitive endogenous RNAs or miRNA ‘sponges’. Over the last years, a high number of reports described lncRNAs that regulate cisplatin resistance by sponging specific miRNAs. For example, NORAD is a lncRNA upregulated by DNA damage and was found involved in cisplatin resistance by regulating SOX4 expression by sponging miR-129-1-3p [18]. Similarly, circular RNA (circRNA) Circ-PRMT5 promoted the resistance to cisplatin by competing with the binding of miR-4458 to REV3L [19], a catalytic subunit of DNA polymerase implicated in the tolerance of DNA adduct through translesion synthesis.
- −
-
Pro-survival and apoptotic signaling pathways: when cells are exposed to platinum, both pro-survival and apoptotic signaling pathways are activated and compete for the final fate of the cells. A number of miRNAs have been associated with the direct or indirect regulation of apoptotic proteins or survival signaling pathways. MiR-103a-3p induces ERK signaling in NSCLC cells by targeting NF1 expression, a key negative regulator of the Ras signaling pathway [20]. In an independent study, Wang et al., found that miR-103a-3p could be released in an exosome (nanosized extracellular vesicles actively released by a variety of cells) from cancer-associated fibroblast and can induce resistance of NSCLC cells via direct regulation of BAK1, a pro-apoptotic BCL-2 family member [21]. Yet, miR-29c-3p affects cisplatin resistance by regulating PI3K/AKT signaling pathways due to binding to 3′UTR of AKT2 [22]. Similarly, miR-126-5p overexpression increases cisplatin sensitivity by inhibiting the PTEN/PI3K/AKT signaling pathway, an effect partly induced by the direct regulation of the metalloprotease ADAM9 [23]. In addition, miR-539-5p increases the sensitivity of cisplatin-resistant cells via the inactivation of the P13K/AKT/mTOR signaling pathway by targeting the protein kinase DCLK1 [24]. Interestingly, the increase in DCLK1 expression and cisplatin resistance was found to be also mediated by lncRNA SNHG1-dependent sponging of miR-330-5p [25]. Several independent studies associated also miR-21 upregulation with increased resistance to Cisplatin[26][27],[28],[29],[30],[31],[32], mostly due to a direct regulation of PTEN expression [27],[28],[30],[31]. Furthermore, miR-21 expression was reported to be regulated by KRAS wild-type or mutant [32], a major driver mutation in NSCLC.
- −
-
Reactive oxygen species: besides DNA damage, activated cisplatin is also a potent inducer of reactive oxygen species, which induces cell death. In this scenario, miR-495-3p overexpression was reported to modulate cisplatin resistance through direct inhibition of NRF2 [33], a transcription factor that regulates the expression of important NADPH-generating enzymes and redox proteins crucial for protecting the cells from oxidative stress.
- −
-
Epithelial-to-mesenchymal transition (EMT): EMT contributes to resistance to several therapeutic agents, including cisplatin [34]. In line with this, miR-128-3p upregulation drives chemoresistance and was associated with the overactivation of Wnt/beta-catenin and TGF-beta (TGF-β) pathways and consequent acquisition of mesenchymal and stem-like features [35]. Likewise, miR-181b-5p regulates the TGF-β pathway by direct inhibition of TGFBR1 expression, thus modulating EMT and sensitivity to cisplatin [36]. An independent study found that miR-181b-5p suppresses stem cell properties in tumor cells and enhances sensitivity to cisplatin treatment by directly targeting NOTCH [37].
- −
-
Autophagy: autophagy is a crucial process that allows the recycling of important cellular components in response to stress conditions such as those induced by cisplatin treatment. Indeed, regulation of autophagy has been widely associated with cisplatin resistance phenotype. Rescue of miR-1-3p increases the sensitivity of cisplatin resistance cells by inhibiting ATG3, a key autophagic protein [38]. Furthermore, exosomal transfer of miR-425-3p was found to increase autophagic flux and chemoresistance by inhibiting AKT1 in the targeted NSCLC cells [39].
4. Beyond Targeting NSCLC Cells: The Role of miRNA in Regulating Immune Response to Cisplatin Treatment
Besides targeting tumor cells, platinum compounds can also elicit an immune response against cancer cells through different mechanisms, i.e., by augmenting CD8+ T cells tumor infiltration, inducing maturation of Antigen-presenting cells (APCs), by downmodulating regulatory T cells (Tregs) and decreasing Myeloid-derived suppressor cells (MDSCs) which compose the so-called ‘immune permissive environment’ [40][41]. Moreover, cisplatin was also described to induce PD-L1 expression in vitro and in vivo [42][43][44][45][46], a co-inhibitory factor of the immune response which in turn augments the sensitivity of cancer cells to therapies using immune checkpoint inhibitors (ICIs) against PD-L1 [[47][48]. In keeping with this, Fujita et al., demonstrated that miR-197-3p modulated cisplatin resistance by targeting the cyclin-dependent kinase CKS1B and, indirectly, the transcription factor STAT3, which ultimately leads to increased PD-L1 expression thus sensitizing PD-L1high tumor cells to cisplatin [42]. Alternatively, an independent report showed that LncRNA MALAT1 induces chemoresistance of NSCLC cells through direct regulation of p120 catenin by competing with miR-197-3p [49]. Other miRNAs were found functionally involved in modulating cisplatin resistance though the exact molecular mechanisms were unclear [17][50][51][52][53]5. miRNA Regulation of Platinum-Based Therapy Response
Cisplatin is the standard first-line treatment for advanced-stage NSCLC. It can be administered in combination with third-generation anticancer agents such as Gemcitabine, Docetaxel, Paclitaxel, or Vinorelbine, thus also called platinum-based doublet chemotherapy aka P-doublet [54]. As shown in Figure 1, a lower number of miRNAs was found involved in regulating responses to P-doublet in NSCLC, which is mainly ascribable to a lower number of research studies that analyzed miRNAs roles in P-doublet therapy. However, Lin et al. showed that miR-30-5p expression impairs resistance to cisplatin alone or in combination with pemetrexed [55] while it enhances paclitaxel sensitivity in an independent study [56]. Cai et al. found that miR-128-3p upregulation increased resistance to multiple drugs though used alone (cisplatin, gemcitabine, or paclitaxel; [57]), probably suggesting a similar effect when combined therapy would be otherwise used. Other miRNAs were also identified to regulate sensitivity to both cisplatin and paclitaxel (i.e., miR-186-5p [58][59]; miR-17-5p [17][60][61]; miR-34c-3p [62]) or cisplatin and docetaxel (miR-141-3p [63][64]; and miR-379-5p [65],[66]).6. miRNAs Which Modulate Response to EGFR Inhibitors
Mutations in EGFR are frequent in NSCLC patients (~10–20% of Caucasian, and up to 60% of South-East Asian patients) and cause the constitutive ligand-independent activation of EGFR receptor thus promoting cell growth and survival [67]. EGFR tyrosin kinase inhibitors (TKIs) are small molecules that bind the adenosine triphosphate (ATP) pocket of EGFR and inhibit its autophosphorylation and downstream signal transduction [68], including RAS–MAPK, PI3K–AKT, and JAK–STAT signaling. Initially, first- (gefitinib, erlotinib) and second-generation TKIs (afatinib, dacomitinib) have been developed and showed a better efficacy vs. platinum-based therapy alone in EGFR-mutated patients [69][[70][71]. More recently, third-generation TKIs, such as Osimertinib, have shown a significant improvement in progression-free survival and overall survival [72], thus becoming the front-line therapy for patients with EGFR-mutated NSCLC [73]. Despite the initial response, resistance to EGFR TKIs inevitably occurs through both EGFR-target-dependent and independent mechanisms [74]. Some miRNAs were shown to modulate sensitivity to Gefitinib, Erlotinib, Afatinib, and Osimertinib with a high level of evidence (score ≥ 4 or 5) (Figure 1). In this context, transcriptome rewiring of TKIs resistant cells upon miRNA induction would activate alternative signaling pathways to bypass EGF inhibition [74]. For example, the rescue of miR-19a expression reverses gefitinib resistance in NSCLC by directly targeting 3′UTR of c-MET [75] which is one of the most altered pathways implicated in EGFR-TKIs inhibition [3][74]. Loss of PTEN was associated with both primary and acquired EGFR-TKIs resistance due to an increased PI3K signaling. Accordingly, miR-21 overexpression is associated with acquired resistance to gefitinib via inhibition of PTEN and PDCD4 and induction of PI3K/AKT [28][76]. Notably, increased expression of miR-21 was associated with afatinib resistance in both in vitro experimental models and in patients [77]. Other pathways were implicated in modulating sensitivity to EGFR-TKIs such as the regulation of NF1 which is a GTPase that negatively regulates the Ras/MEK/ERK pathway and is targeted by miR-641 regulation [78]. Yet, miR-326 impairs resistance to Gefitinib by direct inhibition of the type I interferon receptor IFNAR2 [79], which is relevant for JAK-STAT signaling activation [80] and is associated with the response to EGFR-TKIs [81]. Likewise, miR-762 is a potential downstream effector of the IL-6/STAT3 pathway and regulates gefitinib resistance via direct regulation of the target gene ABR [82]. Yet, loss of expression of miR-206 induces the over-activation of STAT3 which leads to Gefitinib resistance [83]. Furthermore, miR-206 have been reported to regulate response to gefinitib via other molecular mechanisms, including the direct regulation of the multidrug resistance protein ABCB1 [84] and by overcoming HGF activity (i.e., the ligand of Met) and promote MAPK and PI3K/AKT downstream pathways [85]. Gefinitib activity can be also modulated by the regulation of miR-146b-5p/nuclear factor kB (NF-kB) axis which impacts the NF-kB-related IL-6 and IL-8 production and ultimately enhances gefitinib-induced apoptosis [86]. Other miRNAs can influence Gefitinib response by regulating phenotypic traits of resistant cells. For example, Let-7 downregulation concomitantly increases in vitro self-renewal capability and Gefitinib resistance via direct regulation of MYC [87]. Furthermore, miR-17-5p increases tumor sphere formation and Gefitinib resistance by targeting CDKN1A (aka p21) [87][88]. This miRNA has also been associated with Erlotinib resistance although the molecular mechanisms have not been yet investigated [89]. Modulation of YAP1 gene expression by miR-7 is another mechanism of Gefitinib resistance via induction of EMT. Indeed, YAP1 was found to be targeted by miR-7 also through exosomal miR-7 transfer [90]. Similarly, miR-200c-3p inhibits downstream signaling pathways of EGFR and regulates both EMT and gefitinib resistance [91][[92]. MiR-124-3p was found to be downregulated in gefitinib-resistant NSCLC patients, and its overexpression reversed EMT transformation and gefitinib resistance, at least in part, by the direct regulation of SNAI2 and STAT3 [93]. Contrariwise, miR-124-3p inhibition was associated with an increased sphere-forming efficiency and gefitinib resistance of EGFR-mutated cells through the unleashed expression of USP14 [94]. Other miRNAs were reported to regulate response to first-generation EGFR-TKIs through direct regulation of newly identified target genes including PELI3 by miR-365a-5p [95]; ATG5 by miR-153-3p [96]; TGFBR2 by miR-942-5p [95] and LHX6 by miR-214 [97]. Lastly, exosomal miR-184 was recently proposed as a biomarker resistance to third-generation TKI such as Osimertinib, which was reported to alter AKT phosphorylation and Osimertinib-induced cell death in cooperation with miR-22-3p [98][[99] Further studies are urgently needed to better understand the role of miRNAs in the regulation of response to third-generation TKIs, with a particular focus on atypical EGFR mutations that showed low response to EGFR-TKIs.7. miRNAs Associated to Resistance to Radiotherapy
Radiotherapy is an important component of the multi-modality treatment for metastatic NSCLC, with ~50% of NSCLC patients undergoing radiotherapy either for a curative or palliative intent [100]. Radiotherapy kills tumor cells either by directly inducing DNA damage, which leads to cell death as well as increases anti-tumor immune response [101] or by indirectly altering tumor vasculature [102]. As said before, a large fraction of miRNAs associated with radiotherapy was downregulated in radioresistant samples and vice versa upregulated in radiosensitive ones, for example:(i) miR-218-5p overexpression restored the sensitivity of radiation-resistant cells through direct regulation of PRKDC, a member of the non-homologous end-joining pathway in the DNA double-strand break repair response [103]; (ii) rescue of miR-126-3p expression increased radiation-induced apoptosis by inactivation of the PI3K/AKT signaling [104]. MiR-449a, a p53-responsive miRNA, was found to regulate LDHA expression, which in turn inhibited glycolysis and increased radiotherapy sensitivity [105]; (iii) miR-375-3p and miR-513a-3p were respectively sponged by circ_0086720 and LINC00473, and their inhibition attenuated the knockdown-mediated radiosensitivity induced by these two ncRNAs [106][[107]; (iv) re-expression of members of the let-7 family was functionally associated to increased radiosensitivity in NSCLC cells, although the molecular mechanisms were not fully investigated [97]; (v) miR-21-5p was found overexpressed in radioresistant samples while its inhibition was able to increase the sensitivity to radiotherapy through several molecular mechanisms including direct regulation of PTEN and PDCD4 genes, inactivation of the PI3K/AKT signaling or through modulation of HIF1-alpha dependent metabolism regulation [108][109][110][111][112]. Similarly, miR-25-3p was upregulated in radiation-resistant patients and was found to affect radiotherapy sensitivity by direct regulation of the newly identified target gene BTG2, a cell cycle modulator that acts as an effector of p53-induced cell cycle arrest [113] .
References
- Miller, K.D.; Nogueira, L.; Devasia, T.; Mariotto, A.B.; Yabroff, K.R.; Jemal, A.; Kramer, J.; Siegel, R.L. Cancer Treatment and Survivorship Statistics, 2022. CA Cancer J. Clin. 2022, 72, 409–436.
- Chen, Z.; Fillmore, C.M.; Hammerman, P.S.; Kim, C.F.; Wong, K.-K. Non-Small-Cell Lung Cancers: A Heterogeneous Set of Diseases. Nat. Rev. Cancer 2014, 14, 535–546.
- Lim, Z.-F.; Ma, P.C. Emerging Insights of Tumor Heterogeneity and Drug Resistance Mechanisms in Lung Cancer Targeted Therapy. J. Hematol. Oncol. 2019, 12, 134.
- Yanaihara, N.; Caplen, N.; Bowman, E.; Seike, M.; Kumamoto, K.; Yi, M.; Stephens, R.M.; Okamoto, A.; Yokota, J.; Tanaka, T.; et al. Unique MicroRNA Molecular Profiles in Lung Cancer Diagnosis and Prognosis. Cancer Cell 2006, 9, 189–198.
- Cancer Genome Atlas Research, N. Comprehensive Molecular Profiling of Lung Adenocarcinoma. Nature 2014, 511, 543–550.
- Dama, E.; Melocchi, V.; Colangelo, T.; Cuttano, R.; Bianchi, F. Deciphering the Molecular Profile of Lung Cancer: New Strategies for the Early Detection and Prognostic Stratification. J. Clin. Med. 2019, 8, 108
- Si, W.; Shen, J.; Zheng, H.; Fan, W. The Role and Mechanisms of Action of MicroRNAs in Cancer Drug Resistance. Clin. Epigenet. 2019, 11, 25.
- Treiber, T.; Treiber, N.; Meister, G. Regulation of MicroRNA Biogenesis and Its Crosstalk with Other Cellular Pathways. Nat. Rev. Mol. Cell Biol. 2019, 20, 5–20
- Dragomir, M.P.; Knutsen, E.; Calin, G.A. Classical and Noncanonical Functions of MiRNAs in Cancers. Trends Genet. 2022, 38, 379–394.
- Kelland, L. The Resurgence of Platinum-Based Cancer Chemotherapy. Nat. Rev. Cancer 2007, 7, 573–584.
- Kumar, M.S.; Lu, J.; Mercer, K.L.; Golub, T.R.; Jacks, T. Impaired MicroRNA Processing Enhances Cellular Transformation and Tumorigenesis. Nat. Genet. 2007, 39, 673–677.
- Yuan, M.; Huang, L.-L.; Chen, J.-H.; Wu, J.; Xu, Q. The Emerging Treatment Landscape of Targeted Therapy in Non-Small-Cell Lung Cancer. Signal Transduct. Target. Ther. 2019, 4, 61.
- Rottenberg, S.; Disler, C.; Perego, P. The Rediscovery of Platinum-Based Cancer Therapy. Nat. Rev. Cancer 2021, 21, 37–50.
- Huang, D.; Savage, S.R.; Calinawan, A.P.; Lin, C.; Zhang, B.; Wang, P.; Starr, T.K.; Birrer, M.J.; Paulovich, A.G. A Highly Annotated Database of Genes Associated with Platinum Resistance in Cancer. Oncogene 2021, 40, 6395–6405
- Hao, G.-J.; Ding, Y.-H.; Wen, H.; Li, X.-F.; Zhang, W.; Su, H.-Y.; Liu, D.-M.; Xie, N.-L. Attenuation of Deregulated MiR-369-3p Expression Sensitizes Non-Small Cell Lung Cancer Cells to Cisplatin via Modulation of the Nucleotide Sugar Transporter SLC35F5. Biochem. Biophys. Res. Commun. 2017, 488, 501–508
- Song, L.; Li, Y.; Li, W.; Wu, S.; Li, Z. MiR-495 Enhances the Sensitivity of Non-Small Cell Lung Cancer Cells to Platinum by Modulation of Copper-Transporting P-Type Adenosine Triphosphatase A (ATP7A). J. Cell. Biochem. 2014, 115, 1234–1242.
- Zhao, J.; Fu, W.; Liao, H.; Dai, L.; Jiang, Z.; Pan, Y.; Huang, H.; Mo, Y.; Li, S.; Yang, G.; et al. The Regulatory and Predictive Functions of MiR-17 and MiR-92 Families on Cisplatin Resistance of Non-Small Cell Lung Cancer. BMC Cancer 2015, 15, 731.
- Huang, Q.; Xing, S.; Peng, A.; Yu, Z. NORAD Accelerates Chemo-Resistance of Non-Small-Cell Lung Cancer via Targeting at MiR-129-1-3p/SOX4 Axis. Biosci. Rep. 2020, 40, BSR20193489.
- Pang, J.; Ye, L.; Zhao, D.; Zhao, D.; Chen, Q. Circular RNA PRMT5 Confers Cisplatin-Resistance via MiR-4458/REV3L Axis in Non-Small-Cell Lung Cancer. Cell Biol. Int. 2020, 44, 2416–2426.
- Zhu, H.; Yang, J.; Yang, S. MicroRNA-103a-3p Potentiates Chemoresistance to Cisplatin in Non-Small Cell Lung Carcinoma by Targeting Neurofibromatosis 1. Exp. Ther. Med. 2020, 19, 1797–1805.
- Wang, H.; Huang, H.; Wang, L.; Liu, Y.; Wang, M.; Zhao, S.; Lu, G.; Kang, X. Cancer-Associated Fibroblasts Secreted MiR-103a-3p Suppresses Apoptosis and Promotes Cisplatin Resistance in Non-Small Cell Lung Cancer. Aging 2021, 13, 14456–14468
- Sun, D.-M.; Tang, B.-F.; Li, Z.-X.; Guo, H.-B.; Cheng, J.-L.; Song, P.-P.; Zhao, X. MiR-29c Reduces the Cisplatin Resistance of Non-Small Cell Lung Cancer Cells by Negatively Regulating the PI3K/Akt Pathway. Sci. Rep. 2018, 8, 8007.
- Liu, B.; Wang, R.; Liu, H. Mir-126-5p Promotes Cisplatin Sensitivity of Non-Small-Cell Lung Cancer by Inhibiting ADAM9. BioMed Res. Int. 2021, 2021, 6622342.
- Deng, H.; Qianqian, G.; Ting, J.; Aimin, Y. MiR-539 Enhances Chemosensitivity to Cisplatin in Non-Small Cell Lung Cancer by Targeting DCLK1. Biomed. Pharmacother. 2018, 106, 1072–1081
- Ge, P.; Cao, L.; Zheng, M.; Yao, Y.; Wang, W.; Chen, X. LncRNA SNHG1 Contributes to the Cisplatin Resistance and Progression of NSCLC via MiR-330-5p/DCLK1 Axis. Exp. Mol. Pathol. 2021, 120, 104633
- Wei, J.; Gao, W.; Zhu, C.-J.; Liu, Y.-Q.; Mei, Z.; Cheng, T.; Shu, Y.-Q. Identification of Plasma MicroRNA-21 as a Biomarker for Early Detection and Chemosensitivity of Non-Small Cell Lung Cancer. Chin. J. Cancer 2011, 30, 407–414.
- Gao, W.; Lu, X.; Liu, L.; Xu, J.; Feng, D.; Shu, Y. MiRNA-21: A Biomarker Predictive for Platinum-Based Adjuvant Chemotherapy Response in Patients with Non-Small Cell Lung Cancer. Cancer Biol. Ther. 2012, 13, 330–340.
- Liu, Z.-L.; Wang, H.; Liu, J.; Wang, Z.-X. MicroRNA-21 (MiR-21) Expression Promotes Growth, Metastasis, and Chemo- or Radioresistance in Non-Small Cell Lung Cancer Cells by Targeting PTEN. Mol. Cell. Biochem. 2013, 372, 35–45.
- Xu, L.; Huang, Y.; Chen, D.; He, J.; Zhu, W.; Zhang, Y.; Liu, X. Downregulation of MiR-21 Increases Cisplatin Sensitivity of Non-Small-Cell Lung Cancer. Cancer Genet. 2014, 207, 214–220.
- Yang, Z.; Fang, S.; Di, Y.; Ying, W.; Tan, Y.; Gu, W. Modulation of NF-ΚB/MiR-21/PTEN Pathway Sensitizes Non-Small Cell Lung Cancer to Cisplatin. PLoS ONE 2015, 10, e0121547.
- Cao, L.; Chen, J.; Ou, B.; Liu, C.; Zou, Y.; Chen, Q. GAS5 Knockdown Reduces the Chemo-Sensitivity of Non-Small Cell Lung Cancer (NSCLC) Cell to Cisplatin (DDP) through Regulating MiR-21/PTEN Axis. Biomed. Pharmacother. 2017, 93, 570–579.
- Shi, L.; Middleton, J.; Jeon, Y.-J.; Magee, P.; Veneziano, D.; Laganà, A.; Leong, H.-S.; Sahoo, S.; Fassan, M.; Booton, R.; et al. KRAS Induces Lung Tumorigenesis through MicroRNAs Modulation. Cell Death Dis. 2018, 9, 219.
- Li, C.; Fan, K.; Qu, Y.; Zhai, W.; Huang, A.; Sun, X.; Xing, S. Deregulation of UCA1 Expression May Be Involved in the Development of Chemoresistance to Cisplatin in the Treatment of Non-Small-Cell Lung Cancer via Regulating the Signaling Pathway of MicroRNA-495/NRF2. J. Cell. Physiol. 2020, 235, 3721–3730.
- Shibue, T.; Weinberg, R.A. EMT, CSCs, and Drug Resistance: The Mechanistic Link and Clinical Implications. Nat. Rev. Clin. Oncol. 2017, 14, 611–629.
- Cai, J.; Guan, H.; Fang, L.; Yang, Y.; Zhu, X.; Yuan, J.; Wu, J.; Li, M. MicroRNA-374a Activates Wnt/Beta-Catenin Signaling to Promote Breast Cancer Metastasis. J. Clin. Investig. 2013, 123, 566–579.
- Wang, X.; Chen, X.; Meng, Q.; Jing, H.; Lu, H.; Yang, Y.; Cai, L.; Zhao, Y. MiR-181b Regulates Cisplatin Chemosensitivity and Metastasis by Targeting TGFβR1/Smad Signaling Pathway in NSCLC. Sci. Rep. 2015, 5, 17618.
- Wang, X.; Meng, Q.; Qiao, W.; Ma, R.; Ju, W.; Hu, J.; Lu, H.; Cui, J.; Jin, Z.; Zhao, Y.; et al. MiR-181b/Notch2 Overcome Chemoresistance by Regulating Cancer Stem Cell-like Properties in NSCLC. Stem Cell Res. Ther. 2018, 9, 327.
- Hua, L.; Zhu, G.; Wei, J. MicroRNA-1 Overexpression Increases Chemosensitivity of Non-Small Cell Lung Cancer Cells by Inhibiting Autophagy Related 3-Mediated Autophagy. Cell Biol. Int. 2018, 42, 1240–1249.
- Ma, Y.; Yuwen, D.; Chen, J.; Zheng, B.; Gao, J.; Fan, M.; Xue, W.; Wang, Y.; Li, W.; Shu, Y.; et al. Exosomal Transfer Of Cisplatin-Induced MiR-425-3p Confers Cisplatin Resistance In NSCLC Through Activating Autophagy. Int. J. Nanomed. 2019, 14, 8121–8132
- Rébé, C.; Demontoux, L.; Pilot, T.; Ghiringhelli, F. Platinum Derivatives Effects on Anticancer Immune Response. Biomolecules 2019, 10, 13.
- Hato, S.V.; Khong, A.; de Vries, I.J.M.; Lesterhuis, W.J. Molecular Pathways: The Immunogenic Effects of Platinum-Based Chemotherapeutics. Clin. Cancer Res. 2014, 20, 2831–2837.
- Fujita, Y.; Yagishita, S.; Hagiwara, K.; Yoshioka, Y.; Kosaka, N.; Takeshita, F.; Fujiwara, T.; Tsuta, K.; Nokihara, H.; Tamura, T.; et al. The Clinical Relevance of the MiR-197/CKS1B/STAT3-Mediated PD-L1 Network in Chemoresistant Non-Small-Cell Lung Cancer. Mol. Ther. 2015, 23, 717–727.
- Parra, E.R.; Villalobos, P.; Behrens, C.; Jiang, M.; Pataer, A.; Swisher, S.G.; William, W.N.; Zhang, J.; Lee, J.; Cascone, T.; et al. Effect of Neoadjuvant Chemotherapy on the Immune Microenvironment in Non–Small Cell Lung Carcinomas as Determined by Multiplex Immunofluorescence and Image Analysis Approaches. J. Immunother. Cancer 2018, 6, 48.
- Zhang, P.; Ma, Y.; Lv, C.; Huang, M.; Li, M.; Dong, B.; Liu, X.; An, G.; Zhang, W.; Zhang, J.; et al. Upregulation of Programmed Cell Death Ligand 1 Promotes Resistance Response in Non-small-cell Lung Cancer Patients Treated with Neo-adjuvant Chemotherapy. Cancer Sci. 2016, 107, 1563–1571.
- hin, J.; Chung, J.-H.; Kim, S.H.; Lee, K.S.; Suh, K.J.; Lee, J.Y.; Kim, J.-W.; Lee, J.-O.; Kim, J.-W.; Kim, Y.-J.; et al. Effect of Platinum-Based Chemotherapy on PD-L1 Expression on Tumor Cells in Non-Small Cell Lung Cancer. Cancer Res. Treat. 2019, 51, 1086–1097.
- Guo, L.; Song, P.; Xue, X.; Guo, C.; Han, L.; Fang, Q.; Ying, J.; Gao, S.; Li, W. Variation of Programmed Death Ligand 1 Expression After Platinum-Based Neoadjuvant Chemotherapy in Lung Cancer. J. Immunother. 2019, 42, 215–220.
- Fournel, L.; Wu, Z.; Stadler, N.; Damotte, D.; Lococo, F.; Boulle, G.; Ségal-Bendirdjian, E.; Bobbio, A.; Icard, P.; Trédaniel, J.; et al. Cisplatin Increases PD-L1 Expression and Optimizes Immune Check-Point Blockade in Non-Small Cell Lung Cancer. Cancer Lett. 2019, 464, 5–14.
- Wakita, D.; Iwai, T.; Harada, S.; Suzuki, M.; Yamamoto, K.; Sugimoto, M. Cisplatin Augments Antitumor T-Cell Responses Leading to a Potent Therapeutic Effect in Combination With PD-L1 Blockade. Anticancer Res. 2019, 39, 1749–1760.
- Yang, T.; Li, H.; Chen, T.; Ren, H.; Shi, P.; Chen, M. LncRNA MALAT1 Depressed Chemo-Sensitivity of NSCLC Cells through Directly Functioning on MiR-197-3p/P120 Catenin Axis. Mol. Cells 2019, 42, 270–283.
- Yin, J.; Zhao, J.; Hu, W.; Yang, G.; Yu, H.; Wang, R.; Wang, L.; Zhang, G.; Fu, W.; Dai, L.; et al. Disturbance of the Let-7/LIN28 Double-Negative Feedback Loop Is Associated with Radio- and Chemo-Resistance in Non-Small Cell Lung Cancer. PLoS ONE 2017, 12, e0172787.
- Zhao, X.; Wang, J.; Zhu, R.; Zhang, J.; Zhang, Y. DLX6-AS1 Activated by H3K4me1 Enhanced Secondary Cisplatin Resistance of Lung Squamous Cell Carcinoma through Modulating MiR-181a-5p/MiR-382-5p/CELF1 Axis. Sci. Rep. 2021, 11, 21014.
- Wang, C.; Wang, S.; Ma, F.; Zhang, W. MiRNA-328 Overexpression Confers Cisplatin Resistance in Non-small Cell Lung Cancer via Targeting of PTEN. Mol. Med. Rep. 2018, 18, 4563–4570.
- Jiang, Z.; Yin, J.; Fu, W.; Mo, Y.; Pan, Y.; Dai, L.; Huang, H.; Li, S.; Zhao, J. MiRNA 17 Family Regulates Cisplatin-Resistant and Metastasis by Targeting TGFbetaR2 in NSCLC. PLoS ONE 2014, 9, e94639.
- Hellmann, M.D.; Li, B.T.; Chaft, J.E.; Kris, M.G. Chemotherapy Remains an Essential Element of Personalized Care for Persons with Lung Cancers. Ann. Oncol. 2016, 27, 1829–1835.
- Lin, X.; Lai, X.; Feng, W.; Yu, X.; Gu, Q.; Zheng, X. MiR-30a Sensitized Lung Cancer against Neoadjuvant Chemotherapy by Depressing Autophagy. Jpn. J. Clin. Oncol. 2021, 51, 675–684.
- Xu, X.; Jin, S.; Ma, Y.; Fan, Z.; Yan, Z.; Li, W.; Song, Q.; You, W.; Lyu, Z.; Song, Y.; et al. MiR-30a-5p Enhances Paclitaxel Sensitivity in Non-Small Cell Lung Cancer through Targeting BCL-2 Expression. J. Mol. Med. 2017, 95, 861–871.
- Cai, J.; Fang, L.; Huang, Y.; Li, R.; Xu, X.; Hu, Z.; Zhang, L.; Yang, Y.; Zhu, X.; Zhang, H.; et al. Simultaneous Overactivation of Wnt/β-Catenin and TGFβ Signalling by MiR-128-3p Confers Chemoresistance-Associated Metastasis in NSCLC. Nat. Commun. 2017, 8, 15870.
- Liu, X.; Zhou, X.; Chen, Y.; Huang, Y.; He, J.; Luo, H. MiR-186-5p Targeting SIX1 Inhibits Cisplatin Resistance in Non-Small-Cell Lung Cancer Cells (NSCLCs). Neoplasma 2020, 67, 147–157.
- Ye, J.; Zhang, Z.; Sun, L.; Fang, Y.; Xu, X.; Zhou, G. MiR-186 Regulates Chemo-Sensitivity to Paclitaxel via Targeting MAPT in Non-Small Cell Lung Cancer (NSCLC). Mol. Biosyst. 2016, 12, 3417–3424.
- Chatterjee, A.; Chattopadhyay, D.; Chakrabarti, G. MiR-16 Targets Bcl-2 in Paclitaxel-Resistant Lung Cancer Cells and Overexpression of MiR-16 along with MiR-17 Causes Unprecedented Sensitivity by Simultaneously Modulating Autophagy and Apoptosis. Cell. Signal. 2015, 27, 189–203.
- Chatterjee, A.; Chattopadhyay, D.; Chakrabarti, G. MiR-17-5p Downregulation Contributes to Paclitaxel Resistance of Lung Cancer Cells through Altering Beclin1 Expression. PLoS ONE 2014, 9, e95716.
- Yang, L.-Z.; Lei, C.-C.; Zhao, Y.-P.; Sun, H.-W.; Yu, Q.-H.; Yang, E.-J.; Zhan, X. MicroRNA-34c-3p Target Inhibiting NOTCH1 Suppresses Chemosensitivity and Metastasis of Non-Small Cell Lung Cancer. J. Int. Med. Res. 2020, 48, 300060520904847.
- Fu, W.-F.; Chen, W.-B.; Dai, L.; Yang, G.-P.; Jiang, Z.-Y.; Pan, L.; Zhao, J.; Chen, G. Inhibition of MiR-141 Reverses Cisplatin Resistance in Non-Small Cell Lung Cancer Cells via Upregulation of Programmed Cell Death Protein 4. Eur. Rev. Med. Pharmacol. Sci 2016, 20, 2565–2572.
- Wang, D.; Ma, J.; Ji, X.; Xu, F.; Wei, Y. MiR-141 Regulation of EIF4E Expression Affects Docetaxel Chemoresistance of Non-Small Cell Lung Cancer. Oncol. Rep. 2017, 37, 608–616.
- Hao, G.-J.; Hao, H.-J.; Ding, Y.-H.; Wen, H.; Li, X.-F.; Wang, Q.-R.; Zhang, B.-B. Suppression of EIF4G2 by MiR-379 Potentiates the Cisplatin Chemosensitivity in Nonsmall Cell Lung Cancer Cells. FEBS Lett. 2017, 591, 636–645.
- Zheng, S.; Wang, C.; Yan, H.; Du, Y. Blocking Hsa_circ_0074027 Suppressed Non-Small Cell Lung Cancer Chemoresistance via the MiR-379-5p/IGF1 Axis. Bioengineered 2021, 12, 8347–8357.
- Hsu, W.-H.; Yang, J.C.-H.; Mok, T.S.; Loong, H.H. Overview of Current Systemic Management of EGFR-Mutant NSCLC. Ann. Oncol. 2018, 29, i3–i9.
- Morin, M.J. From Oncogene to Drug: Development of Small Molecule Tyrosine Kinase Inhibitors as Anti-Tumor and Anti-Angiogenic Agents. Oncogene 2000, 19, 6574–6583.
- Sequist, L.V.; Yang, J.C.-H.; Yamamoto, N.; O’Byrne, K.; Hirsh, V.; Mok, T.; Geater, S.L.; Orlov, S.; Tsai, C.-M.; Boyer, M.; et al. Phase III Study of Afatinib or Cisplatin plus Pemetrexed in Patients with Metastatic Lung Adenocarcinoma with EGFR Mutations. J. Clin. Oncol. 2013, 31, 3327–3334.
- Zhou, C.; Wu, Y.-L.; Chen, G.; Feng, J.; Liu, X.-Q.; Wang, C.; Zhang, S.; Wang, J.; Zhou, S.; Ren, S.; et al. Erlotinib versus Chemotherapy as First-Line Treatment for Patients with Advanced EGFR Mutation-Positive Non-Small-Cell Lung Cancer (OPTIMAL, CTONG-0802): A Multicentre, Open-Label, Randomised, Phase 3 Study. Lancet Oncol. 2011, 12, 735–742.
- Mok, T.S.; Wu, Y.-L.; Thongprasert, S.; Yang, C.-H.; Chu, D.-T.; Saijo, N.; Sunpaweravong, P.; Han, B.; Margono, B.; Ichinose, Y.; et al. Gefitinib or Carboplatin-Paclitaxel in Pulmonary Adenocarcinoma. N. Engl. J. Med. 2009, 361, 947–957.
- Soria, J.-C.; Ohe, Y.; Vansteenkiste, J.; Reungwetwattana, T.; Chewaskulyong, B.; Lee, K.H.; Dechaphunkul, A.; Imamura, F.; Nogami, N.; Kurata, T.; et al. Osimertinib in Untreated EGFR-Mutated Advanced Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2018, 378, 113–125.
- Cooper, A.J.; Sequist, L.V.; Lin, J.J. Third-Generation EGFR and ALK Inhibitors: Mechanisms of Resistance and Management. Nat. Rev. Clin. Oncol. 2022, 19, 499–514.
- Passaro, A.; Jänne, P.A.; Mok, T.; Peters, S. Overcoming Therapy Resistance in EGFR-Mutant Lung Cancer. Nat. Cancer 2021, 2, 377–391.
- Cao, X.; Lai, S.; Hu, F.; Li, G.; Wang, G.; Luo, X.; Fu, X.; Hu, J. MiR-19a Contributes to Gefitinib Resistance and Epithelial Mesenchymal Transition in Non-Small Cell Lung Cancer Cells by Targeting c-Met. Sci. Rep. 2017, 7, 2939.
- Shen, H.; Zhu, F.; Liu, J.; Xu, T.; Pei, D.; Wang, R.; Qian, Y.; Li, Q.; Wang, L.; Shi, Z.; et al. Alteration in Mir-21/PTEN Expression Modulates Gefitinib Resistance in Non-Small Cell Lung Cancer. PLoS ONE 2014, 9, e103305.
- Leonetti, A.; Capula, M.; Minari, R.; Mazzaschi, G.; Gregori, A.; El Hassouni, B.; Papini, F.; Bordi, P.; Verzè, M.; Avan, A.; et al. Dynamic Evaluation of Circulating MiRNA Profile in EGFR-Mutated NSCLC Patients Treated with EGFR-TKIs. Cells 2021, 10, 1520.
- Chen, J.; Cui, J.; Guo, X.-T.; Cao, X.; Li, Q. Increased Expression of MiR-641 Contributes to Erlotinib Resistance in Non-Small-Cell Lung Cancer Cells by Targeting NF1. Cancer Med. 2018, 7, 1394–1403.
- Zheng, Y.; Guo, Z.; Li, Y. Long Non-Coding RNA Prostate Cancer-Associated Transcript 6 Inhibited Gefitinib Sensitivity of Non-Small Cell Lung Cancer by Serving as a Competing Endogenous RNA of MiR-326 to up-Regulate Interferon-Alpha Receptor 2. Bioengineered 2022, 13, 3785–3796.
- Shemesh, M.; Lochte, S.; Piehler, J.; Schreiber, G. IFNAR1 and IFNAR2 Play Distinct Roles in Initiating Type I Interferon–Induced JAK-STAT Signaling and Activating STATs. Sci. Signal. 2021, 14, eabe4627.
- Gong, K.; Guo, G.; Panchani, N.; Bender, M.E.; Gerber, D.E.; Minna, J.D.; Fattah, F.; Gao, B.; Peyton, M.; Kernstine, K.; et al. EGFR Inhibition Triggers an Adaptive Response by Co-Opting Antiviral Signaling Pathways in Lung Cancer. Nat. Cancer 2020, 1, 394–409.
- Ge, P.; Cao, L.; Chen, X.; Jing, R.; Yue, W. MiR-762 Activation Confers Acquired Resistance to Gefitinib in Non-Small Cell Lung Cancer. BMC Cancer 2019, 19, 1203.
- Yang, Y.; Wang, W.; Chang, H.; Han, Z.; Yu, X.; Zhang, T. Reciprocal Regulation of MiR-206 and IL-6/STAT3 Pathway Mediates IL6-Induced Gefitinib Resistance in EGFR-Mutant Lung Cancer Cells. J. Cell. Mol. Med. 2019, 23, 7331–7341.
- Wu, K.; Li, J.; Qi, Y.; Zhang, C.; Zhu, D.; Liu, D.; Zhao, S. SNHG14 Confers Gefitinib Resistance in Non-Small Cell Lung Cancer by up-Regulating ABCB1 via Sponging MiR-206-3p. Biomed. Pharmacother. 2019, 116, 108995.
- Jiao, D.; Jiang, C.; Zhu, L.; Zheng, J.; Liu, X.; Liu, X.; Chen, J.; Tang, X.; Chen, Q. MiR-1/133a and MiR-206/133b Clusters Overcome HGF Induced Gefitinib Resistance in Non-Small Cell Lung Cancers with EGFR Sensitive Mutations. J. Drug Target. 2021, 29, 1111–1117.
- Liu, Y.-N.; Tsai, M.-F.; Wu, S.-G.; Chang, T.-H.; Tsai, T.-H.; Gow, C.-H.; Wang, H.-Y.; Shih, J.-Y. MiR-146b-5p Enhances the Sensitivity of NSCLC to EGFR Tyrosine Kinase Inhibitors by Regulating the IRAK1/NF-ΚB Pathway. Mol. Ther. Nucleic Acids 2020, 22, 471–483.
- Yin, J.; Hu, W.; Pan, L.; Fu, W.; Dai, L.; Jiang, Z.; Zhang, F.; Zhao, J. Let-7 and MiR-17 Promote Self-renewal and Drive Gefitinib Resistance in Non-small Cell Lung Cancer. Oncol. Rep. 2019, 42, 495–508.
- Gong, J.; He, L.; Ma, J.; Zhang, J.; Wang, L.; Wang, J. The Relationship between MiR-17-5p, MiR-92a, and Let-7b Expression with Non-Small Cell Lung Cancer Targeted Drug Resistance. J. BUON 2017, 22, 454–461.
- Zhang, W.; Lin, J.; Wang, P.; Sun, J. MiR-17-5p down-Regulation Contributes to Erlotinib Resistance in Non-Small Cell Lung Cancer Cells. J. Drug Target. 2017, 25, 125–131.
- Chen, R.; Qian, Z.; Xu, X.; Zhang, C.; Niu, Y.; Wang, Z.; Sun, J.; Zhang, X.; Yu, Y. Exosomes-Transmitted MiR-7 Reverses Gefitinib Resistance by Targeting YAP in Non-Small-Cell Lung Cancer. Pharmacol. Res. 2021, 165, 105442.
- Zhou, G.; Zhang, F.; Guo, Y.; Huang, J.; Xie, Y.; Yue, S.; Chen, M.; Jiang, H.; Li, M. MiR-200c Enhances Sensitivity of Drug-Resistant Non-Small Cell Lung Cancer to Gefitinib by Suppression of PI3K/Akt Signaling Pathway and Inhibites Cell Migration via Targeting ZEB1. Biomed. Pharmacother. 2017, 85, 113–119.
- Lin, C.-C.; Wu, C.-Y.; Tseng, J.T.; Hung, C.-H.; Wu, S.-Y.; Huang, Y.-T.; Chang, W.-Y.; Su, P.-L.; Su, W.-C. Extracellular Vesicle MiR-200c Enhances Gefitinib Sensitivity in Heterogeneous EGFR-Mutant NSCLC. Biomedicines 2021, 9, 243.
- Hu, F.-Y.; Cao, X.-N.; Xu, Q.-Z.; Deng, Y.; Lai, S.-Y.; Ma, J.; Hu, J.-B. MiR-124 Modulates Gefitinib Resistance through SNAI2 and STAT3 in Non-Small Cell Lung Cancer. J. Huazhong Univ. Sci. Technolog. Med. Sci. 2016, 36, 839–845.
- Yu, F.; Liu, J.-B.; Wu, Z.-J.; Xie, W.-T.; Zhong, X.-J.; Hou, L.-K.; Wu, W.; Lu, H.-M.; Jiang, X.-H.; Jiang, J.-J.; et al. Tumor Suppressive MicroRNA-124a Inhibits Stemness and Enhances Gefitinib Sensitivity of Non-Small Cell Lung Cancer Cells by Targeting Ubiquitin-Specific Protease 14. Cancer Lett. 2018, 427, 74–84.
- Fan, D.; Yang, Y.; Zhang, W. A Novel Circ_MACF1/MiR-942-5p/TGFBR2 Axis Regulates the Functional Behaviors and Drug Sensitivity in Gefitinib-Resistant Non-Small Cell Lung Cancer Cells. BMC Pulm. Med. 2022, 22, 27.
- Zhang, W.; Dong, Y.-Z.; Du, X.; Peng, X.-N.; Shen, Q.-M. MiRNA-153-3p Promotes Gefitinib-Sensitivity in Non-Small Cell Lung Cancer by Inhibiting ATG5 Expression and Autophagy. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 2444–2452.
- Liao, J.; Lin, J.; Lin, D.; Zou, C.; Kurata, J.; Lin, R.; He, Z.; Su, Y. Down-Regulation of MiR-214 Reverses Erlotinib Resistance in Non-Small-Cell Lung Cancer through up-Regulating LHX6 Expression. Sci. Rep. 2017, 7, 781.
- Vadla, G.P.; Daghat, B.; Patterson, N.; Ahmad, V.; Perez, G.; Garcia, A.; Manjunath, Y.; Kaifi, J.T.; Li, G.; Chabu, C.Y. Combining Plasma Extracellular Vesicle Let-7b-5p, MiR-184 and Circulating MiR-22-3p Levels for NSCLC Diagnosis and Drug Resistance Prediction. Sci. Rep. 2022, 12, 6693.
- Li, X.; Chen, C.; Wang, Z.; Liu, J.; Sun, W.; Shen, K.; Lv, Y.; Zhu, S.; Zhan, P.; Lv, T.; et al. Elevated Exosome-Derived MiRNAs Predict Osimertinib Resistance in Non-Small Cell Lung Cancer. Cancer Cell. Int. 2021, 21, 428.
- Delaney, G.P.; Barton, M.B. Evidence-Based Estimates of the Demand for Radiotherapy. Clin. Oncol. 2015, 27, 70–76.
- Hayman, T.J.; Glazer, P.M. Regulation of the Cell-Intrinsic DNA Damage Response by the Innate Immune Machinery. Int. J. Mol. Sci. 2021, 22, 12761.
- Castle, K.D.; Kirsch, D.G. Establishing the Impact of Vascular Damage on Tumor Response to High-Dose Radiation Therapy. Cancer Res. 2019, 79, 5685–5692.
- Chen, X.; Xu, Y.; Jiang, L.; Tan, Q. MiRNA-218-5p Increases Cell Sensitivity by Inhibiting PRKDC Activity in Radiation-Resistant Lung Carcinoma Cells. Thorac. Cancer 2021, 12, 1549–1557.
- Wang, X.-C.; Du, L.-Q.; Tian, L.-L.; Wu, H.-L.; Jiang, X.-Y.; Zhang, H.; Li, D.-G.; Wang, Y.-Y.; Wu, H.-Y.; She, Y.; et al. Expression and Function of MiRNA in Postoperative Radiotherapy Sensitive and Resistant Patients of Non-Small Cell Lung Cancer. Lung Cancer 2011, 72, 92–99
- Li, L.; Liu, H.; Du, L.; Xi, P.; Wang, Q.; Li, Y.; Liu, D. MiR-449a Suppresses LDHA-Mediated Glycolysis to Enhance the Sensitivity of Non-Small Cell Lung Cancer Cells to Ionizing Radiation. Oncol. Res. 2018, 26, 547–556.
- Jin, Y.; Su, Z.; Sheng, H.; Li, K.; Yang, B.; Li, S. Circ_0086720 Knockdown Strengthens the Radiosensitivity of Non-Small Cell Lung Cancer via Mediating the MiR-375/SPIN1 Axis. Neoplasma 2021, 68, 96–107.
- Qin, P.; Li, Y.; Liu, J.; Wang, N. Knockdown of LINC00473 Promotes Radiosensitivity of Non-Small Cell Lung Cancer Cells via Sponging MiR-513a-3p. Free Radic. Res. 2020, 54, 756–764.
- Chen, L.; Ren, P.; Zhang, Y.; Gong, B.; Yu, D.; Sun, X. Long Non-coding RNA GAS5 Increases the Radiosensitivity of A549 Cells through Interaction with the MiR-21/PTEN/Akt Axis. Oncol. Rep. 2020, 43, 897–907.
- Jiang, L.-P.; He, C.-Y.; Zhu, Z.-T. Role of MicroRNA-21 in Radiosensitivity in Non-Small Cell Lung Cancer Cells by Targeting PDCD4 Gene. Oncotarget 2017, 8, 23675–23689.
- Jiang, S.; Wang, R.; Yan, H.; Jin, L.; Dou, X.; Chen, D. MicroRNA-21 Modulates Radiation Resistance through Upregulation of Hypoxia-Inducible Factor-1α-Promoted Glycolysis in Non-Small Cell Lung Cancer Cells. Mol. Med. Rep. 2016, 13, 4101–4107.
- Ma, Y.; Xia, H.; Liu, Y.; Li, M. Silencing MiR-21 Sensitizes Non-Small Cell Lung Cancer A549 Cells to Ionizing Radiation through Inhibition of PI3K/Akt. Biomed Res. Int. 2014, 2014, 617868.
- Wang, X.; Wang, W.; Zhang, Z.-B.; Zhao, J.; Tan, X.-G.; Luo, J.-C. Overexpression of MiRNA-21 Promotes Radiation-Resistance of Non-Small Cell Lung Cancer. Radiat. Oncol. 2013, 8, 146.
- He, Z.; Liu, Y.; Xiao, B.; Qian, X. MiR-25 Modulates NSCLC Cell Radio-Sensitivity through Directly Inhibiting BTG2 Expression. Biochem. Biophys. Res. Commun. 2015, 457, 235–241.
More
