The coenzyme Q10 is a naturally occurring benzoquinone derivative widely prescribed as a food supplement for different physical conditions and pathologies. Thanks to its favourable combination of functional activity and safety profile, it is widely prescribed for an ever increasing number of physical conditions. Ageing, myopathy, cardiomyopathy, high blood pressure, dyslipidemia, migraine, diabetes, infertility, Friedreich's ataxia, and neurologic disorders like Parkinson’s and Huntington’s diseases, are but a few examples that today prompt practitioners to prescribe CoQ10. To this already extensive list, fibromyalgia has recently been added, once it has been clear that CoQ10 deficiency and mitochondrial dysfunction are both implicated in its pathophysiology.
- ubiquinone
- ubiquinol
- coenzyme Q
- CoQ10
1. Fibromyalgia: Key Characteristics and Therapeutic Approaches
2. Coenzyme Q10 as a Key Functional Derivative
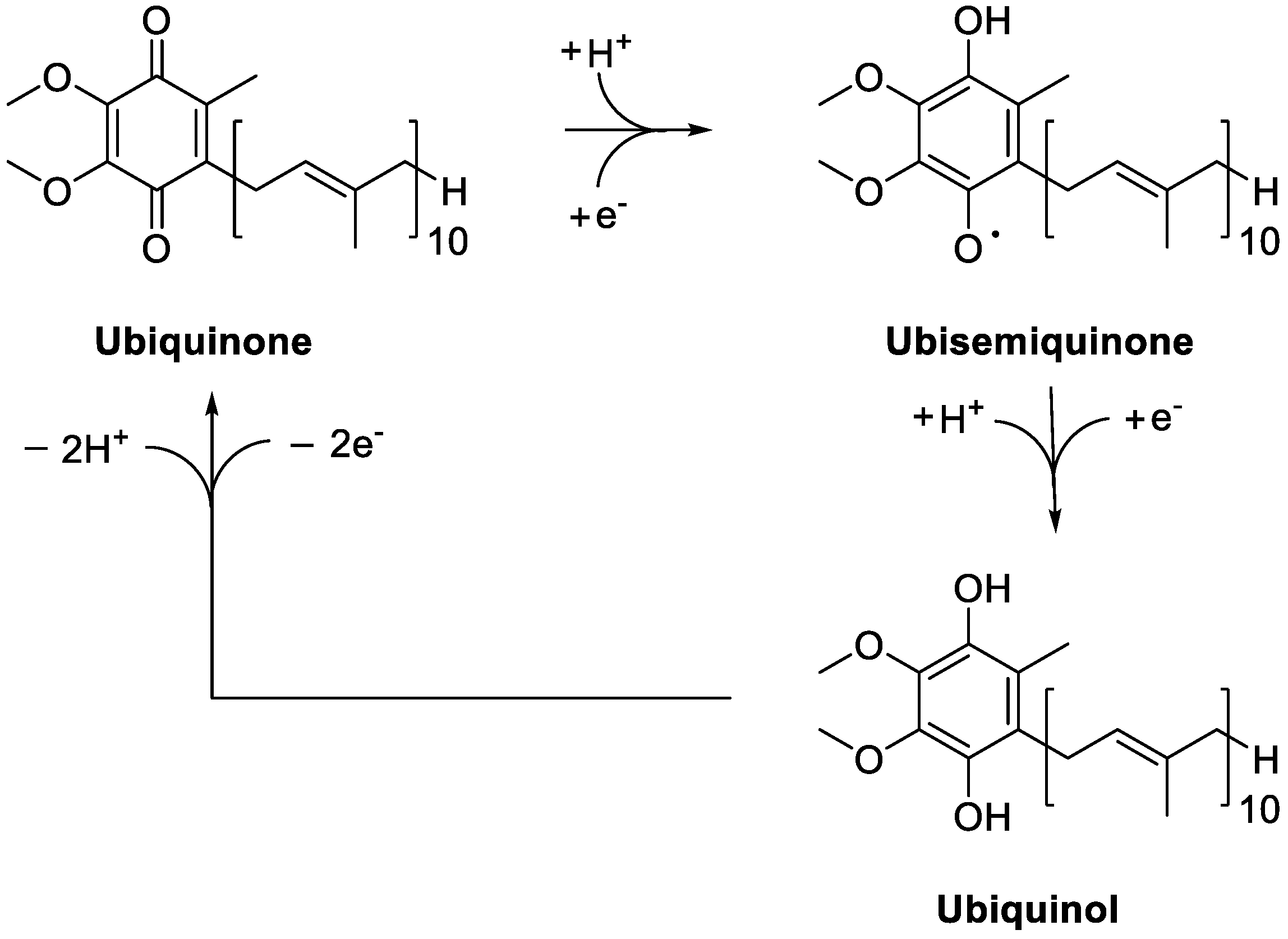
The number of investigations aiming at demonstrate the effectiveness of CoQ10 in the management of FM has been increasing over time, outlining a positive causal relationship between the amount of its supplementation and the relief in FM symptoms, first and foremost the feeling of fatigue. Therefore, according on the outcomes obtained so far, CoQ10 might be reasonably regarded as the gold standard supplement for people affected by FM.
However, a closer look at the available data highlights the lack of a sound and clear scientific basis justifying this trumpeted claim. Actually, most, but not all, the authors highlighted reduced levels of CoQ10 in FM patients, therefore the consensus of the scientific community on this condition is not unanimous. Moreover, scientific investigations claiming the functional efficacy of this supplementation suffers from clear and obvious limits. In particular, besides biased reading of the experimental outcomes, the number of patients enrolled in the studies are often limited, even consisting of a single patient [94], thus inadequate to provide evidences expendable for the whole FM population. Therefore, prospective and randomized trial with hundreds of patients per study group are now required, to corroborate the efficacy of CoQ10 in the management of FM observed in pilot studies.
References
- Häuser, W.; Ablin, J.; Fitzcharles, M.-A.; Littlejohn, G.; Luciano, J.V.; Usui, C.; Walitt, B. Fibromyalgia. Nat. Rev. Dis. Prim. 2015, 13, 15022.
- Marques, A.P.; de Sousa do Espirito Santo, A.; Berssaneti, A.A.; Matsutani, L.A.; Yuan, S.L.K. Prevalence of fibromyalgia: Literature review update. Rev. Bras. Reumatol. Engl. Ed. 2017, 57, 356–363.
- Creed, F. A review of the incidence and risk factors for fibromyalgia and chronic widespread pain in population-based studies. PAIN 2020, 161, 1169–1176.
- Kashikar-Zuck, S.; Ting, T.V. Juvenile fibromyalgia: Current status of research and future developments. Nat. Rev. Rheumatol. 2014, 10, 89–96.
- Jacobson, S.A.; Simpson, R.G.; Lubahn, C.; Hu, C.; Belden, C.M.; Davis, K.J.; Nicholson, L.R.; Long, K.E.; Osredkar, T.; Lorton, D. Characterization of fibromyalgia symptoms in patients 55 to 95 years old: A longitudinal study showing symptom persistence with suboptimal treatment. Aging Clin. Exp. Res. 2015, 27, 75–82.
- Ursini, F.; Naty, S.; Grembiale, R.D. Fibromyalgia and obesity: The hidden link. Rheumatol. Int. 2011, 31, 1403–1408.
- Vincent, A.; Clauw, D.; Oh, T.H.; Whipple, M.O.; Toussaint, L.L. Decreased physical activity attributable to higher body mass index influences fibromyalgia symptoms. PM R 2014, 6, 802–807.
- Salaffi, F.; Di Carlo, M.; Farah, S.; Atzeni, F.; Buskila, D.; Ablin, J.N.; Häuser, W.; Sarzi-Puttini, W. Diagnosis of fibromyalgia: Comparison of the 2011/2016 ACR and AAPT criteria and validation of the modified Fibromyalgia Assessment Status. Rheumatology 2020, 59, 3042–3049.
- Ablin, J.N.; Buskila, D.; Clauw, D.J. Biomarkers in fibromyalgia. Curr. Pain Headache Rep. 2009, 13, 343–349.
- Harris, R.E.; Clauw, D.J.; Scott, D.J.; McLean, S.A.; Gracely, R.H.; Zubieta, J.K. Decreased central mu-opioid receptor availability in fibromyalgia. J. Neurosci. 2007, 27, 10000–10006.
- Watkins, L.R.; Hutchinson, M.R.; Rice, K.C.; Maier, S.F. The “toll” of opioid-induced glial activation: Improving the clinical efficacy of opioids by targeting glia. Trends Pharmacol. Sci. 2009, 30, 581–591.
- Harris, R.E.; Sundgren, P.C.; Craig, A.D.; Kirshenbaum, E.; Sen, A.; Napadow, V.; Clauw, D.J. Elevated insular glutamate (Glu) in fibromyalgia (FM) is associated with experimental pain. Arthritis Rheum. 2009, 60, 3146–3152.
- Foerster, B.R.; Petrou, M.; Edden, R.A.; Sundgren, P.C.; Schmidt-Wilcke, T.; Lowe, S.E.; Harte, S.E.; Clauw, D.J.; Harris, R.E. Reduced insular gamma-aminobutyric acid in fibromyalgia. Arthritis Rheum. 2012, 64, 579–583.
- Petrovic, P.; Kalso, E.; Petersson, K.M.; Ingvar, M. Placebo and opioid analgesia-- imaging a shared neuronal network. Science 2002, 295, 1737–1740.
- Littlejohn, G. Neurogenic neuroinflammation in fibromyalgia and complex regional pain syndrome. Nat. Rev. Rheumatol. 2015, 11, 639–648.
- Guedj, E.; Taieb, D.; Cammilleri, S.; Lussato, D.; de Laforte, C.; Niboyet, J.; Mundler, O. 99mTc-ECD brain perfusion SPECT in hyperalgesic fibromyalgia. Eur. J. Nucl. Med. Mol. Imaging 2007, 34, 130–134.
- Gracely, R.H.; Petzke, F.; Wolf, J.M.; Clauw, D.J. Functional magnetic resonance imaging evidence of augmented pain processing in fibromyalgia. Arthritis Rheum. 2002, 46, 1333–1343.
- Feraco, P.; Bacci, A.; Pedrabissi, F.; Passamonti, L.; Zampogna, G.; Pedrabissi, F.; Malavolta, N.; Leonardi, M. Metabolic abnormalities in pain-processing regions of patients with fibromyalgia: A 3T MR spectroscopy study. Am. J. Neuroradiol. 2011, 32, 1585–1590.
- Üçeyler, N.; Zeller, D.; Kahn, A.-K.; Kewenig, S.; Kittel-Schneider, S.; Schmid, A.; Casanova-Molla, J.; Reiners, K.; Sommer, C. Small fibre pathology in patients with fibromyalgia syndrome. Brain 2013, 136, 1857–1867.
- Castro-Marrero, J.; Cordero, M.D.; Saez-Francas, N.; Jimenez-Gutierrez, C.; Auilar-Montilla, F.J.; Aliste, L.; Alegre-Martin, J. Could mitochondrial dysfunction be a differentiating marker between chronic fatigue syndrome and fibromyalgia? Antioxid. Redox Signal 2013, 19, 1855–1860.
- Sprott, H.; Salemi, S.M.; Gay, R.E.; Bradley, L.A.; Alarcon, G.S.; Oh, S.J.; Michel, B.A.; Gay, S. Increased DNA fragmentation and ultrastructural changes in fibromyalgic muscle fibres. Ann. Rheum. Dis. 2004, 63, 245–251.
- Cordero, M.D.; De Miguel, M.; Moreno Fernández, A.M.; Carmona López, I.M.; Maraver, J.G.; Cotán, D.; Gómez Izquierdo, L.; Bonal, P.; Campa, F.; Bullon, P.; et al. Mitochondrial dysfunction and mitophagy activation in blood mononuclear cells of fibromyalgia patients: Implications in the pathogenesis of the disease. Arthritis Res. Ther. 2010, 12, R17.
- Bullon, P.; Alcocer-Gómez, E.; Carrión, A.M.; Marín-Aguilar, F.; Garrido-Maraver, J.; Román-Malo, L.; Ruiz-Cabello, J.; Culic, O.; Ryffel, B.; Apetoh, L.; et al. AMPK phosphorylation modulates pain by activation of NLRP3 inflammasome. Antioxid. Redox Signal 2016, 24, 157–170.
- Alcocer-Gómez, E.; Sánchez-Alcazar, J.A.; Battino, M.; Bullón, P.; Cordero, M.D. Aging-related changes in inflammatory and LKB1/AMPK gene expression in fibromyalgia patients. CNS Neurosci. Ther. 2014, 20, 476–478.
- Macfarlane, G.J.; Kronisch, C.; Dean, L.E.; Atzeni, F.; Hauser, W.; Flu, E.; Cho, E.; Kosek, E.; Amris, K.; Branco, J.; et al. EULAR revised recommendations for the management of fibromyalgia. Ann. Rheum. Dis. 2017, 76, 318–328.
- Hauser, W.; Klose, P.; Langhorst, J.; Moradi, B.; Steinbach, M.; Schiltenwolf, M.; Busch, A. Efficacy of different types of aerobic exercise in fibromyalgia syndrome: A systematic review and meta-analysis of randomised controlled trials. Arthritis Res. Ther. 2010, 12, R79.
- Cazzola, M.; Atzeni, F.; Salaffi, F.; Stisi, S.; Cassisi, G.; Sarzi-Puttini, P. Which kind of exercise is best in fibromyalgia therapeutic programmes? A practical review. Clin. Exp. Rheumatol. 2010, 28, S117–S124.
- Larsson, A.; Palstam, A.; Löfgren, M.; Ernberg, M.; Bjersing, J.; Bileviciute-Ljungar, I.; Gerdle, B.; Kosek, E.; Mannerkorpi, K. Resistance exercise improves muscle strength, health status and pain intensity in fibromyalgia--a randomized controlled trial. Arthritis Res. Ther. 2015, 17, 161.
- Pagliai, G.; Giangrandi, I.; Dinu, M.; Sofi, F.; Colombini, B. Nutritional interventions in the management of fibromyalgia syndrome. Nutrients 2020, 12, 2525.
- Lowry, E.; Marley, J.; McVeigh, J.G.; Mc Sorley, E.; Allsopp, P.; Kerr, D. Dietary interventions in the management of fibromyalgia: A systematic review and best-evidence synthesis. Nutrients 2020, 12, 2664.
- Kadayifci, F.Z.; Bradley, M.J.; Onat, A.M.; Shi, H.N.; Zheng, S. Review of nutritional approaches to fibromyalgia. Nutr. Rev. 2022, nuac036.
- Fernández-Araque, A.; Verde, Z.; Torres-Ortega, C.; Sainz-Gil, M.; Velasco-Gonzalez, V.; González-Bernal, J.J.; Mielgo-Ayuso, J. Effects of antioxidants on pain perception in patients with fibromyalgia-A systematic review. J. Clin. Med. 2022, 11, 2462.
- Haddad, H.W.; Mallepalli, N.R.; Scheinuk, J.E.; Bhargava, P.; Cornett, E.M.; Urits, I.; Kaye, A.D. The Role of nutrient supplementation in the management of chronic pain in fibromyalgia: A narrative review. Pain Ther. 2021, 10, 827–848.
- Crane, F.L.; Hatefi, Y.; Lester, R.L.; Widmer, C. Isolation of a quinone from beef heart mitochondria. Biochim. Biophys. Acta 1957, 25, 220–221.
- Morton, R.A. Ubiquinone. Nature 1958, 182, 1764–1767.
- Ernster, L.; Dallner, G. Biochemical, physiological and medical aspects of ubiquinone function. Biochim. Biophys. Acta 1995, 1271, 195–204.
- Frederick, L.; Crane, F.L. Biochemical functions of Coenzyme Q10. J. Am. Coll. Nutr. 2001, 20, 591–598.
- Turunen, M.; Sindelar, P.; Dallner, G. Induction of endogenous coenzyme Q biosynthesis by administration of peroxisomal inducers. Biofactors 1999, 9, 131–139.
- Arroyo, A.; Kagan, V.E.; Tyurin, V.A.; Burgess, J.R.; de Cabo, R.; Navas, P.; Villalba, J.M. NADH and NADPH dependent reduction of coenzyme Q at the plasma membrane. Antioxid. Redox Signal 2000, 2, 251–262.
- Thomas, S.R.; Witting, P.K.; Stocker, R. A role for reduced coenzyme Q in atherosclerosis. Biofactors 1999, 9, 207–224.
- Schneider, D.; Elstner, E.F. Coenzyme Q10, vitamin E and dihydrothioctic acid cooperatively prevent diene conjugation in isolated low density lipoprotein. Antioxid. Redox Signal 2000, 2, 327–333.
- Takahashi, T.; Okamoto, T.; Mori, K.; Sayo, H.; Kishi, T. Distribution of ubiquinone and ubiquinol homologues in rat tissues and subcellular fraction. Lipids 1993, 28, 803–809.
- Quinn, P.J.; Fabisiak, J.P.; Kagan, V.E. Expansion of antioxidant function of vitamin E by coenzyme Q. Biofactors 1999, 9, 149–154.
- Crane, F.L. New functions for coenzyme Q. Protoplasma 2000, 213, 127–133.
- Villalba, J.M.; Navas, P. Plasma membrane redox system in the control of stress-induced apoptosis. Antioxid. Redox Signal 2000, 2, 213–230.
- Sun, I.L.; Sun, E.E.; Crane, F.L.; Morré, D.J.; Lindgren, A.; Löw, H. A requirement for coenzyme Q in plasma membrane electron transport. Proc. Nat. Acad. Sci. USA 1992, 89, 11126–11130.
- Takahashi, T.; Okamoto, T.; Kishi, T. Characterization of NADPH dependent ubiquinone reductase activity in rat liver cytosol. J. Biochem. 1996, 119, 256–263.
- Navarro, F.; Arroyo, A.; Martin, S.F.; Bello, R.I.; de Cabo, R.; Burgess, J.R.; Navas, P.; Villalba, J.M. Protective role of ubiquinone in vitamin E and selenium deficient plasma membranes. Biofactors 1999, 9, 163–170.
- Gorelick, C.; Lopez-Jones, M.; Goldberg, G.L.; Romney, S.L.; Khabele, D. Coenzyme Q10 and lipid-related gene induction in HeLa cells. Am. J. Obstet. Gynecol. 2004, 190, 1432–1434.
- Groneberg, D.A.; Kindermann, B.; Althammer, M.; Klapper, M.; Vormann, J.; Littarru, G.P.; Döring, F. Coenzyme Q10 affects expression of genes involved in cell signalling, metabolism and transport in human CaCo-2 cells. Int. J. Biochem. Cell Biol. 2005, 37, 1208–1218.
- Schmelzer, C.; Lindner, I.; Rimbach, G.; Niklowitz, P.; Menke, T.; Döring, F. Functions of coenzyme Q10 in inflammation and gene expression. Biofactors 2008, 32, 179–183.
