Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Jingan Li and Version 3 by Conner Chen.
There are many studies on the different synthesis methods of carbon dots. Each process aims to improve the synthesis strategy and optimize the reaction conditions so that the carbon dots are not only more cost-effective and eco-friendly, but also provide more excellent performanceOK.
- carbon dots
- composite materials
- synthesis
- bioapplication
1. Introduction
Carbon is the most basic element in organic matter and exists in the form of different compounds. Carbon dots (CDs) are a new carbon based zero-dimensional nanomaterial [1]. During the process of separation and purification of single-walled carbon nanotubes (SWCNTs) by gel electrophoresis, Sun et al. used laser ablation to make a series of surface treatments on the obtained carbon nanoparticles and named them carbon dots [2]. CDs are a large number of different, quasi spherical carbon atom aggregation regions with a size of less than 10 nm. CDs are usually composed of sp2 and sp3 hybrid carbon nuclei and rich active functional groups (such as hydroxyl, carboxyl, amino, etc.), which makes them have excellent water solubility. Through a series of chemical reactions, small molecules, organic polymers, or biomolecules can be adsorbed on the surface of CDs for surface passivation or functionalization, but the quantum yield of CDs is low, which greatly limits their practical application in the development of biological imaging and therapeutic diagnostics. In addition, because there is no specific analyte recognition group on the surface of CDs, the detection process of CDs in biosensors is often affected by potential disruptors. Most importantly, the interaction between CDs and biological systems is usually poor and lacks specificity, which greatly limits their potential clinical application. Generally, surface passivation can enhance the luminescence properties of CDs, and surface functionalization can change the physicochemical properties of CDs [3]. The preparation method and application classification of carbon dots and carbon dot composites are shown in Figure 1 [4][5][6][7][8][9][10][4,5,6,7,8,9,10]. Carbon dot matrix composites have excellent luminescence properties and good biocompatibility, and they have attracted extensive attention in the fields of biological imaging [11][12][13][11,12,13], sensing [14][15][16][14,15,16], detection, and biochemical analysis.
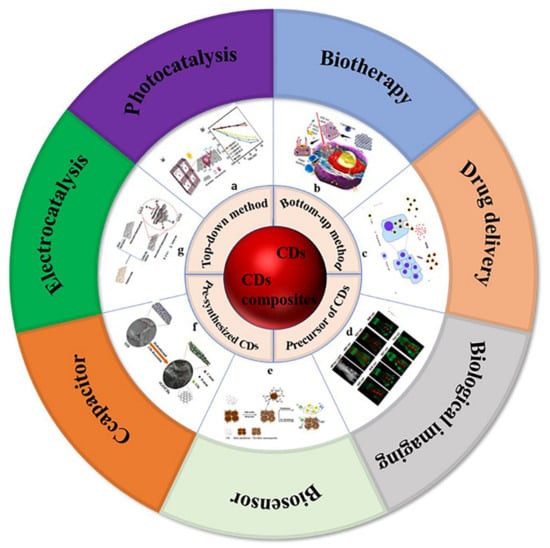
Figure 1. Preparation methods and application classification of carbon dots and carbon dots composites. (a) Photocatalysis: Photocatalytic mechanism of 3D colloidal quantum dots (CQDs)/GA composites, photocatalytic reduction of Cr (VI) with different proportions of CQDs/GA and original CQD [4]. (b) Biotherapy: Structure of PCCN and schematic diagram of 630 nm light-driven water splitting enhanced PDT [5]. (c) Drug delivery: Image of fluorescent carbon nanoparticles in medical cancer treatment [6]. (d) Biological imaging: In vivo imaging and biodistribution of the carboxylated Graphene Quantum Dots [7]. (e) Biosensor: Schematic diagram of GSH detection of CD-MnO2 nanocomposites [8]. (f) Capacitor: Schematic diagram of synthesis process of three-dimensional interconnected CD decorative reduced graphene oxide nanosheets (rGO/CDs) [9]. (g) Electrocatalysis: Schematic preparation process of NS-CD@gf [10].
2. Synthesis of Carbon Dots
There are many studies on the different synthesis methods of carbon dots. Each process aims to improve the synthesis strategy and optimize the reaction conditions so that the carbon dots are not only more cost-effective and eco-friendly, but also provide more excellent performance. According to the carbon source and the used process, the synthesis methods are mainly divided into the “top-down” method and “bottom-up” method (as shown in Figure 2).
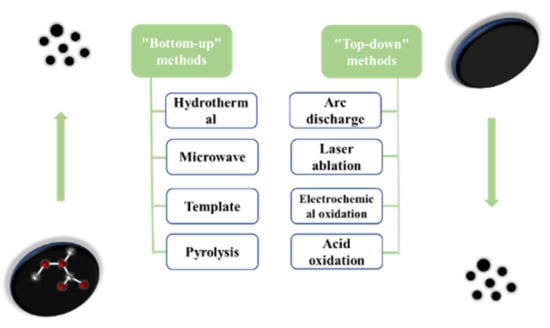
Figure 2.
The two preparation methods of carbon dots.
2.1. “Top-Down” Method
The “top-down” method is a method of peeling or breaking large carbon materials to form small carbon nanoparticles, and then modifying their surfaces to improve their luminous efficiency, mainly including arc discharge method [17], laser ablation [18], electrochemical oxidation [19], and acid oxidation.
2.1.1. Arc Discharge Method
The earliest method to prepare carbon dots is the arc discharge method, which means that under a certain voltage condition, gaseous charged particles are used as conductors, and the reaction is accelerated by generating a strong current and high temperature. The advantage is that the prepared carbon dots have a small particle size and high oxygen content; the disadvantage is that the components of arc discharge are complex and there are many impurities, so the prepared carbon dots are difficult to separate and purify, the yield is very low, and the fluorescence quantum efficiency is also very low.
Research evolution: Xu et al. prepared single wall carbon nanotubes by the arc discharge method and separated the mixed products by electrophoresis. In this process, carbon dots were found [17]. In 2006, Bottini et al. [20] removed the fluorescent nanoparticles from the original carbon nanotubes and the fluorescence emission wavelength of the fluorescence nanoparticles gradually redshifted with the increase of the molecular weight of the nanoparticles, and the fluorescence emission from blue to yellow-green carbon points were obtained.
2.1.2. Laser Ablation
Laser ablation refers to the rapid laser passivation of carbon nanoparticles in organic solvents. The advantage is that the prepared carbon dots have bright, tunable, and stable photoluminescence properties; the disadvantage is that the carbon dots prepared by laser ablation usually need irradiation, oxidation, and passivation processes. The preparation method is complex, the output of carbon dots is low, the particle size distribution is uneven, and the purity is low.
Research evolution: Sun et al. prepared fluorescent carbon dots with good luminescence properties by laser ablation with graphite powder as the carbon source [21]. Firstly, graphite powder and cement were dried and solidified to make the graphite target, which was then annealed in a high-purity Ar atmosphere. Then, the graphite target was bombarded with Nd:YAG solid-state laser at 900 °C and 75 kPa, and then refluxed with concentrated nitric acid for 12 h. Finally, the surface of the previously prepared carbon nanoparticles was passivated using some simple organic substances to obtain fluorescent carbon dots. The fluorescence properties of the carbon dots prepared by this method depend on the surface post modified groups, and the emission wavelength is excitation dependent. By adjusting the excitation wavelength, the full band emission from red to blue light can be realized. Hu et al. reported an effective method to synthesize carbon dots by laser irradiation of carbon materials suspended in organic solution [22]. Firstly, a certain amount of graphite powder is dispersed in polyethylene glycol to form a black suspension, and then a Nd:YAG pulse laser with a wavelength of 1.064 mm is used. Finally, the suspension after laser irradiation was further separated and purified to obtain carbon dots.
2.1.3. Electrochemical Oxidation
Electrochemical oxidation is a method of preparing carbon quantum dots by electrolyzing some carbon materials, such as carbon nanotubes and graphite. The advantage is that the size and luminescence properties of the carbon dots can be adjusted by changing the current intensity, the preparation cost is low, and the yield is high; The disadvantage is that the fluorescence quantum efficiency of the product is low.
Research evolution: Zhou et al. prepared carbon dots by electrochemical synthesis for the first time. Firstly, the composite of multi walled carbon nanotubes and carbon film was prepared as the working electrode, Ag/AgClO4 as the reference electrode, and Pt wire as the counter electrode to form a three-electrode system [23]. An appropriate amount of tetrabutylammonium perchlorate was dissolved in an acetonitrile solution and used as an electrolyte after complete dissolution. The appropriate test conditions for a cyclic voltammetry test of the whole system are selected. In this process, the acetonitrile solution of tetrabutylammonium perchlorate changes from colorless to yellowish brown. After irradiation by UV lamp, it can emit blue fluorescence. Finally, it is separated and purified to obtain carbon dots with uniform size. Bao et al. proposed a new strategy to controllably prepare luminescent carbon dots by etching carbon fibers using the electrochemical method [24].
In this study, the target carbon dots could be prepared controllably only by adjusting the applied voltage.
