Cancer patients have an increased risk of cardiovascular disease and, notably, a significant prevalence of acute coronary syndrome (ACS). It has been shown that an elevated presence of cardiovascular risk factors in this setting leads to an interaction between these two conditions, influencing their therapeutic strategies and contributing to higher mortality.
- acute coronary syndromes
- atherosclerosis
- cancer
- cardiotoxicity
- thrombosis
1. Introduction
Remarkably, the coexistence of ACS and cancer in the same patient strongly influences prognosis [13]. The treatment in this setting is very challenging, and it should be patient-tailored [14].
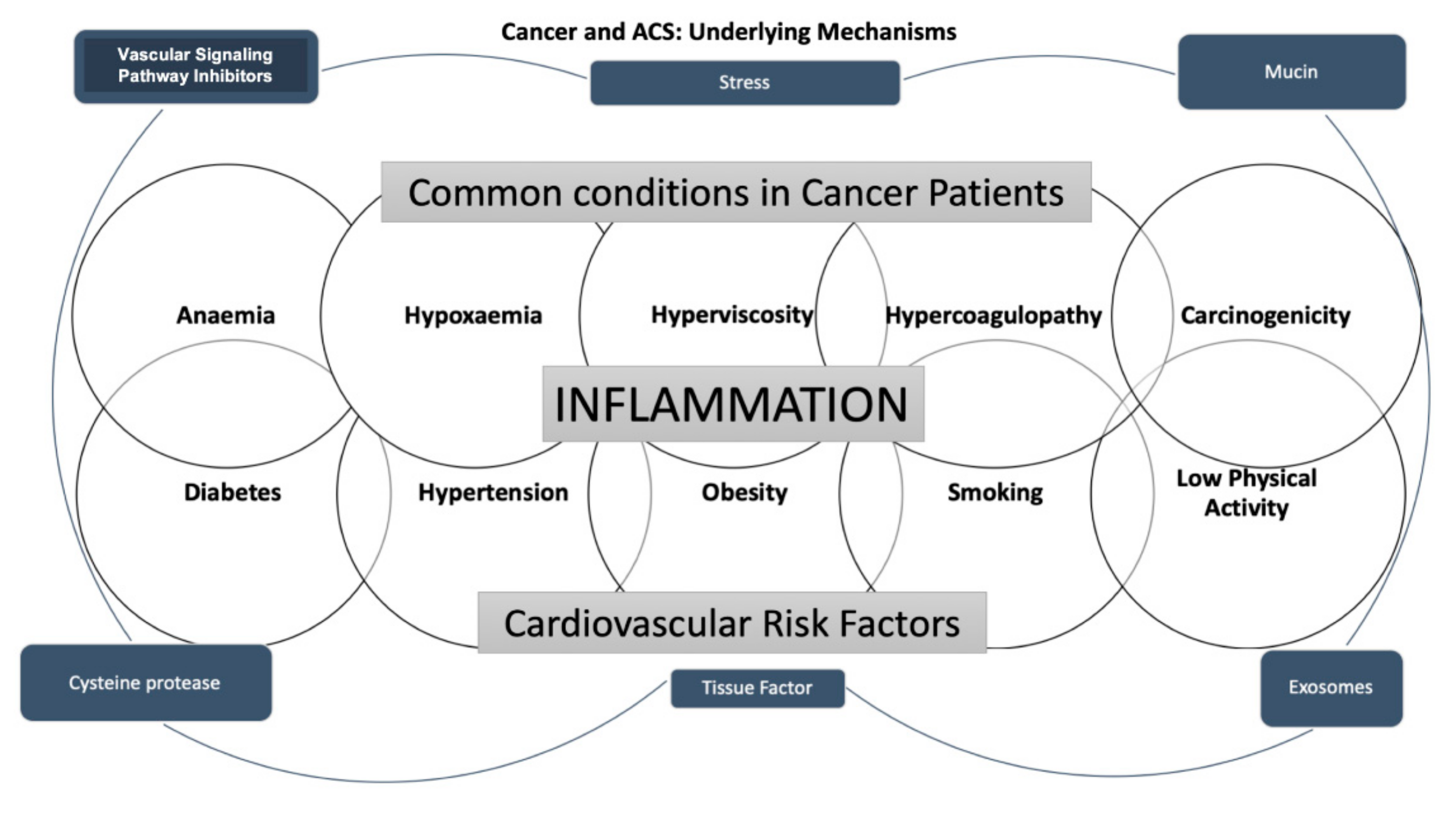
2. The Pathophysiologic Mechanism of Coronary Artery Disease in Cancer Treatment
3. Epidemiology
4. Chemotherapy and ACS Risk
|
Class |
Agents |
Cardiotoxic Effects |
|---|---|---|
|
Immunomodulatory |
Lenalidomide [40] Pomalidomide [41] [39]IPommune Chalidomideckpoint Inhibitors [42,43] [40]Immune Checkpoint Inhibitors [41][42] |
Endothelial Dysfunction → Destabilization of atherosclerotic lesions → Plaque rupture → Cardiovascular events |
|
Anti-microtubule |
Paclitaxel [44] [43] |
Vasoconstriction, Endothelial injury → Cardiovascular events |
|
Proteasome Inhibitor |
Carfilzomib [45] Bortezomib [46] [44]Bortezomib [45] |
Cardiac ubiquitin–proteasome dysfunction → Endothelial injury→ Cardiovascular events |
|
Aromatase Inhibitors |
Anastrozole [47] Letrozole [47] [46]Letrozole [46] |
Vasoconstriction, Endothelial injury → Cardiovascular events |
|
Anti-metabolites |
5-fluorouracil (5-FU) [30] Capecitabine [48] [29]GCapemcitabine [49] Nilotinib [50] [47]Gemcitabine [48]Nilotinib [49] |
Coronary VasospasmThrombus FormationDirect cardiomyocytes and endothelial cells damage → Increase of Von Willebrand Factor’s activity → Cardiovascular events Thrombus Formation Direct cardiomyocytes and endothelial cells damage → Increase of Von Willebrand Factor’s activity → Cardiovascular events |
|
BRC-ABL tyrosine kinase inhibitors |
Nilotinib [50] Ponatinib [51] [49]Ponatinib [50] |
Coronary AtherosclerosisEndothelial Apoptosis → Increase of Factor VII Levels → Prothrombrotic state → Cardiovascular events (cardiac, cerebrovascular, and peripheral events) Endothelial Apoptosis → Increase of Factor VII Levels → Prothrombrotic state → Cardiovascular events (cardiac, cerebrovascular, and peripheral events) |
|
Vascular Endothelial Growth Factor Inhibitors |
Bevacizumab [52] Sorafenib [53] [51]Suorafenitiniib [54] Pazopanib [55] [52]RegorafeSunitinib [56] Axitinib [57] [53]RPazopamucirumanib [58] Aflibercept [59] [54]Regorafenib [55]Axitinib [56]Ramucirumab [57]Aflibercept [58] |
VasospasmInflammationPlatelet Activation → Cardiac ischemia and arterial thrombosis Inflammation Platelet Activation → Cardiac ischemia and arterial thrombosis |
|
Alkilating agents |
[59][60] |
Endothelial dysfunction → platelet aggregation and activation → cardiovascular events |
|
Vinca-alkaloids |
Vincristine [62] [61] |
Thrombus FormationEndothelial Injury → Cardiovascular Events Endothelial Injury → Cardiovascular Events |
|
Platinum |
Cisplatin [63] [62] |
Endothelial dysfunction → Thromboxane Production → Thrombus Formation → Platelet aggregation and activation → Cardiovascular Events |
|
Anti-tumor antibiotics |
Bleomycin [64] [63] |
Endothelial dysfunction → Platelet aggregation and activation → Cardiovascular Events |
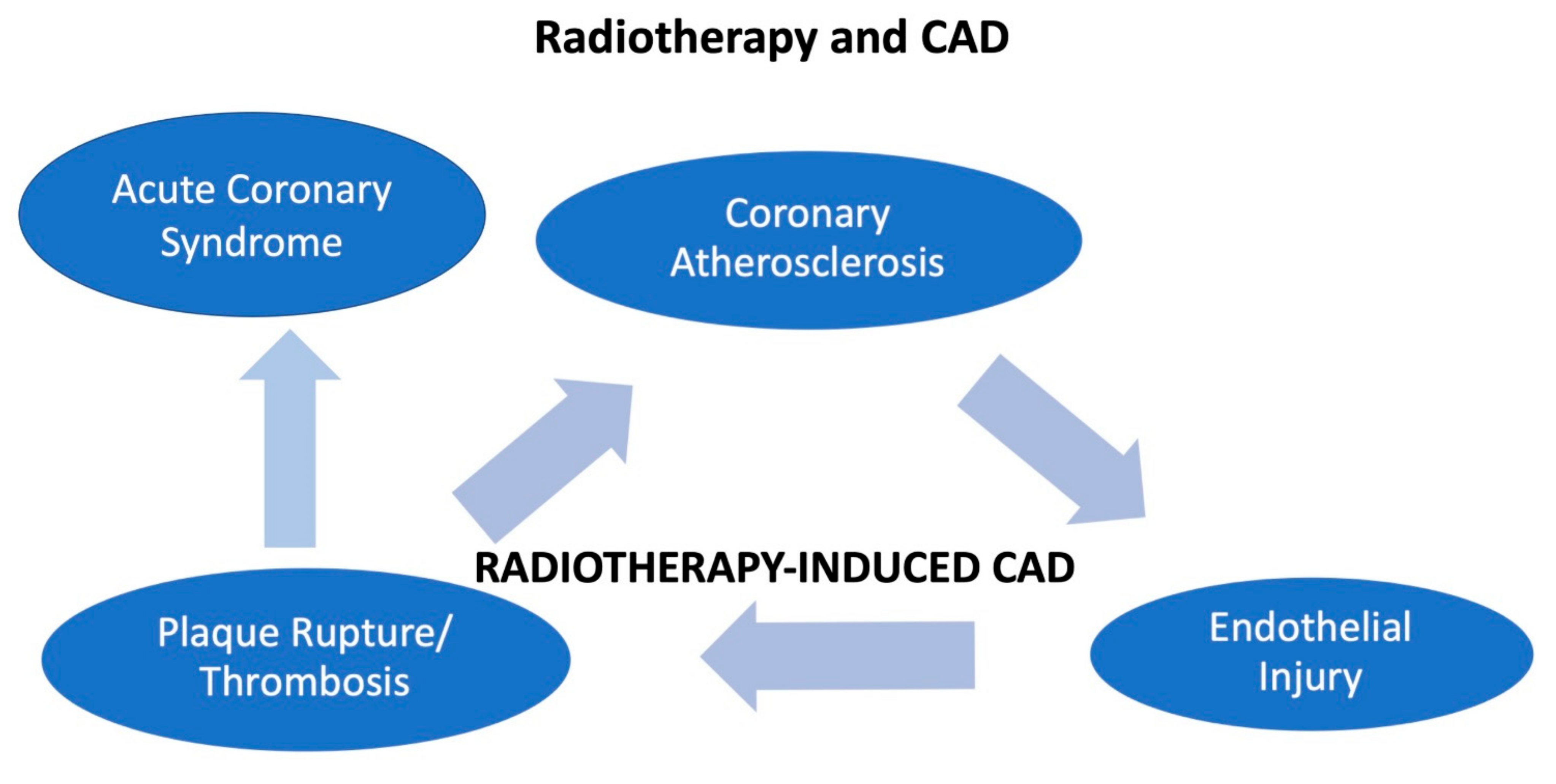
5. Radiotherapy and ACS
6. Clinical Presentation
7. Management
7.1. Cancer and Percutaneous Coronary Intervention
7.1. Cancer and Percutaneous Coronary Intervention
7.2. Pharmacological Treatment of ACS in Cancer Patients
In cancer patients with ACS, there is an increased risk of both thrombotic and hemorrhagic events [93]. Therefore, the clinical decision on antiplatelet drugs and DAPT, aimed at mitigating the risk of stent thrombosis, is challenging for cardiologists and oncologists.7.3. Platelet Concentration Thresholds for Individual Elements of the Therapy
References
- Mertens, A.C.; Liu, Q.; Neglia, J.P.; Wasilewski, K.; Leisenring, W.; Armstrong, G.T.; Robison, L.L.; Yasui, Y. Cause-Specific Late Mortality Among 5-Year Survivors of Childhood Cancer: The Childhood Cancer Survivor Study. J. Natl. Cancer Inst. 2008, 100, 1368–1379.
- Rohrmann, S.; Witassek, F.; Erne, P.; Rickli, H.; Radovanovic, D. Treatment of patients with myocardial infarction depends on history of cancer. Eur. Heart J. Acute Cardiovasc. Care 2017, 7, 639–645.
- Caine, G.J.; Stonelake, P.S.; Lip, G.Y.H.; Kehoe, S.T. The Hypercoagulable State of Malignancy: Pathogenesis and Current Debate. Neoplasia 2002, 4, 465–473.
- Wang, F.; Gulati, R.; Lennon, R.J.; Lewis, B.R.; Park, J.; Sandhu, G.S.; Wright, R.S.; Lerman, A.; Herrmann, J. Cancer History Portends Worse Acute and Long-term Noncardiac (but Not Cardiac) Mortality after Primary Percutaneous Coronary Intervention for Acute ST-Segment Elevation Myocardial Infarction. Mayo Clin. Proc. 2016, 91, 1680–1692.
- Gevaert, S.A.; Halvorsen, S.; Sinnaeve, P.R.; Sambola, A.; Gulati, G.; Lancellotti, P.; Van Der Meer, P.; Lyon, A.R.; Farmakis, D.; Lee, G.; et al. Evaluation and management of cancer patients presenting with acute cardiovascular disease: A Consensus Document of the Acute CardioVascular Care (ACVC) association and the ESC council of Cardio-Oncology—Part 1: Acute coronary syndromes and acute pericardial diseases. Eur. Heart J. Acute Cardiovasc. Care 2021, 10, 947–959.
- Iannaccone, M.; D’Ascenzo, F.; Vadalà, P.; Wilton, S.B.; Noussan, P.; Colombo, F.; Roubín, S.R.; Abu Assi, E.; González-Juanatey, J.R.; Henriques, J.P.S.; et al. Prevalence and outcome of patients with cancer and acute coronary syndrome undergoing percutaneous coronary intervention: A BleeMACS substudy. Eur. Heart J. Acute Cardiovasc. Care 2017, 7, 631–638.
- Edmondson, D.; von Känel, R. Post-traumatic stress disorder and cardiovascular disease. Lancet Psychiatry 2017, 4, 320–329.
- Ariza-Solé, A.; Guerrero, C.; Formiga, F.; Aboal, J.; Abu-Assi, E.; Marín, F.; Bueno, H.; Alegre, O.; López-Palop, R.; Vidán, M.T.; et al. Global Geriatric Assessment and In-Hospital Bleeding Risk in Elderly Patients with Acute Coronary Syndromes: Insights from the LONGEVO-SCA Registry. Thromb. Haemost. 2018, 118, 581–590.
- Manzano-Fernández, S.; Sánchez-Martínez, M.; Flores-Blanco, P.J.; López-Cuenca, Á.; Gómez-Molina, M.; Pastor-Pérez, F.J.; Sánchez-Galian, M.J.; Cambronero-Sanchez, F.; Pérez, E.G.; García-Narbón, A.; et al. Comparison of the Global Registry of Acute Coronary Events Risk Score Versus the Can Rapid Risk Stratification of Unstable Angina Patients Suppress Adverse outcomes with Early Implementation of the ACC/AHA Guidelines Risk Score to Predict In-Hospital Mortality and Major Bleeding in Acute Coronary Syndromes. Am. J. Cardiol. 2016, 117, 1047–1054.
- Guerrero, C.; Garay, A.; Ariza-Solé, A.; Formiga, F.; Raposeiras-Roubín, S.; Abu-Assi, E.; D’Ascenzo, F.; Kinnaird, T.; Manzano-Fernández, S.; Alegre, O.; et al. Anemia in patients with acute coronary syndromes treated with prasugrel or ticagrelor: Insights from the RENAMI registry. Thromb. Res. 2018, 167, 142–148.
- Lancellotti, P.; Marechal, P.; Donis, N.; Oury, C. Inflammation, cardiovascular disease, and cancer: A common link with far-reaching implications. Eur. Heart J. 2019, 40, 3910–3912.
- Das, D.; Asher, A.; Ghosh, A.K. Cancer and Coronary Artery Disease: Common Associations, Diagnosis and Management Challenges. Curr. Treat. Options Oncol. 2019, 20, 46.
- Bisceglia, I.; Canale, M.L.; Lestuzzi, C.; Parrini, I.; Russo, G.; Colivicchi, F.; Gabrielli, D.; Gulizia, M.M.; Iliescu, C.A. Acute coronary syndromes in cancer patients. J. Cardiovasc. Med. 2020, 21, 944–952.
- Radmilovic, J.; Di Vilio, A.; D’Andrea, A.; Pastore, F.; Forni, A.; Desiderio, A.; Ragni, M.; Quaranta, G.; Cimmino, G.; Russo, V.; et al. The Pharmacological Approach to Oncologic Patients with Acute Coronary Syndrome. J. Clin. Med. 2020, 9, 3926.
- Koene, R.J.; Prizment, A.E.; Blaes, A.; Konety, S.H. Shared Risk Factors in Cardiovascular Disease and Cancer. Circulation 2016, 133, 1104–1114.
- Lenneman, C.G.; Sawyer, D.B. Cardio-Oncology: An Update on Cardiotoxicity of Cancer-Related Treatment. Circ. Res. 2016, 118, 1008–1020.
- Canale, M.L.; Bisceglia, I.; Lestuzzi, C.; Parrini, I. Arterial Thrombosis in Cancer: Spotlight on the Neglected Vessels. Anticancer Res. 2019, 39, 4619–4625.
- Libby, P.; Kobold, S. Inflammation: A common contributor to cancer, aging, and cardiovascular diseases—Expanding the concept of cardio-oncology. Cardiovasc. Res. 2019, 115, 824–829.
- Bick, R.L. Cancer-Associated Thrombosis. N. Engl. J. Med. 2003, 349, 109–111.
- Rak, J.; Milsom, C.; May, L.; Klement, P.; Yu, J. Tissue Factor in Cancer and Angiogenesis: The Molecular Link between Genetic Tumor Progression, Tumor Neovascularization, and Cancer Coagulopathy. Semin. Thromb. Hemost. 2006, 32, 54–70.
- Yeh, E.T.H.; Chang, H.-M. Cancer and Clot: Between a Rock and a Hard Place. J. Am. Coll. Cardiol. 2017, 70, 939–941.
- Mitrugno, A.; Tormoen, G.W.; Kuhn, P.; McCarty, O.J. The prothrombotic activity of cancer cells in the circulation. Blood Rev. 2015, 30, 11–19.
- Giza, D.E.; Marmagkiolis, K.; Mouhayar, E.; Durand, J.-B.; Iliescu, C. Management of CAD in Patients with Active Cancer: The Interventional Cardiologists’ Perspective. Curr. Cardiol. Rep. 2017, 19, 56.
- Bharadwaj, A.; Potts, J.; Mohamed, M.O.; Parwani, P.; Swamy, P.; Lopez-Mattei, J.C.; Rashid, M.; Kwok, C.S.; Fischman, D.L.; Vassiliou, V.S.; et al. Acute myocardial infarction treatments and outcomes in 6.5 million patients with a current or historical diagnosis of cancer in the USA. Eur. Heart J. 2020, 41, 2183–2193.
- Navi, B.B.; Reiner, A.S.; Kamel, H.; Iadecola, C.; Okin, P.M.; Elkind, M.S.V.; Panageas, K.S.; DeAngelis, L.M. Risk of Arterial Thromboembolism in Patients with Cancer. J. Am. Coll. Cardiol. 2017, 70, 926–938.
- Navi, B.B.; Reiner, A.S.; Kamel, H.; Iadecola, C.; Okin, P.M.; Tagawa, S.T.; Panageas, K.S.; DeAngelis, L.M. Arterial thromboembolic events preceding the diagnosis of cancer in older persons. Blood 2019, 133, 781–789.
- Park, J.Y.; Guo, W.; Al-Hijji, M.; El Sabbagh, A.; Begna, K.H.; Habermann, T.M.; Witzig, T.E.; Lewis, B.R.; Lerman, A.; Herrmann, J. Acute coronary syndromes in patients with active hematologic malignancies—Incidence, management, and outcomes. Int. J. Cardiol. 2019, 275, 6–12.
- Nanda, A.; Chen, M.-H.; Braccioforte, M.H.; Moran, B.J.; D’Amico, A.V. Hormonal Therapy Use for Prostate Cancer and Mortality in Men with Coronary Artery Disease–Induced Congestive Heart Failure or Myocardial Infarction. JAMA 2009, 302, 866–873.
- Sara, J.D.; Kaur, J.; Khodadadi, R.; Rehman, M.; Lobo, R.; Chakrabarti, S.; Herrmann, J.; Lerman, A.; Grothey, A. 5-fluorouracil and cardiotoxicity: A review. Ther. Adv. Med. Oncol. 2018, 10, 1758835918780140.
- Layoun, M.E.; Wickramasinghe, C.D.; Peralta, M.V.; Yang, E.H. Fluoropyrimidine-Induced Cardiotoxicity: Manifestations, Mechanisms, and Management. Curr. Oncol. Rep. 2016, 18, 35.
- De Forni, M.; Malet-Martino, M.C.; Jaillais, P.; Shubinski, R.E.; Bachaud, J.M.; Lemaire, L.; Canal, P.; Chevreau, C.; Carrie, D.; Soulié, P. Cardiotoxicity of high-dose continuous infusion fluorouracil: A prospective clinical study. J. Clin. Oncol. 1992, 10, 1795–1801.
- Cardinale, D.; Colombo, A.; Colombo, N. Acute coronary syndrome induced by oral capecitabine. Can. J. Cardiol. 2006, 22, 251–253.
- Kanduri, J.; More, L.A.; Godishala, A.; Asnani, A. Fluoropyrimidine-Associated Cardiotoxicity. Cardiol. Clin. 2019, 37, 399–405.
- Moore, R.A.; Adel, N.; Riedel, E.; Bhutani, M.; Feldman, D.R.; Tabbara, N.E.; Soff, G.; Parameswaran, R.; Hassoun, H. High Incidence of Thromboembolic Events in Patients Treated with Cisplatin-Based Chemotherapy: A Large Retrospective Analysis. J. Clin. Oncol. 2011, 29, 3466–3473.
- Touyz, R.M.; Herrmann, J. Cardiotoxicity with vascular endothelial growth factor inhibitor therapy. npj Precis. Oncol. 2018, 2, 13.
- Pouwer, M.G.; Pieterman, E.J.; Verschuren, L.; Caspers, M.P.M.; Kluft, C.; Garcia, R.A.; Aman, J.; Jukema, J.W.; Princen, H.M.G. The BCR-ABL1 Inhibitors Imatinib and Ponatinib Decrease Plasma Cholesterol and Atherosclerosis, and Nilotinib and Ponatinib Activate Coagulation in a Translational Mouse Model. Front. Cardiovasc. Med. 2018, 5, 55.
- Herrmann, J.; Yang, E.H.; Iliescu, C.A.; Cilingiroglu, M.; Charitakis, K.; Hakeem, A.; Toutouzas, K.; Leesar, M.A.; Grines, C.L.; Marmagkiolis, K. Vascular Toxicities of Cancer Therapies: The Old and the New—An Evolving Avenue. Circulation 2016, 133, 1272–1289.
- Lyon, A.R.; Yousaf, N.; Battisti, N.M.L.; Moslehi, J.; Larkin, J. Immune checkpoint inhibitors and cardiovascular toxicity. Lancet Oncol. 2018, 19, e447–e458.
- Li, W.; Cornell, R.F.; Lenihan, D.; Slosky, D.; Jagasia, M.; Piazza, G.; Moslehi, J. Cardiovascular Complications of Novel Multiple Myeloma Treatments. Circulation 2016, 133, 908–912.
- Lee, D.H.; Fradley, M.G. Cardiovascular Complications of Multiple Myeloma Treatment: Evaluation, Management, and Prevention. Curr. Treat. Options Cardiovasc. Med. 2018, 20, 19.
- Drobni, Z.D.; Alvi, R.M.; Taron, J.; Zafar, A.; Murphy, S.P.; Rambarat, P.K.; Mosarla, R.C.; Lee, C.; Zlotoff, D.A.; Raghu, V.K.; et al. Association Between Immune Checkpoint Inhibitors with Cardiovascular Events and Atherosclerotic Plaque. Circulation 2020, 142, 2299–2311.
- Inno, A.; Chiampan, A.; Lanzoni, L.; Verzè, M.; Molon, G.; Gori, S. Immune Checkpoint Inhibitors and Atherosclerotic Vascular Events in Cancer Patients. Front. Cardiovasc. Med. 2021, 8, 652186.
- Pasquier, E.; Honore, S.; Pourroy, B.; Jordan, M.A.; Lehmann, M.; Briand, C.; Braguer, D. Antiangiogenic Concentrations of Paclitaxel Induce an Increase in Microtubule Dynamics in Endothelial Cells but Not in Cancer Cells. Cancer Res. 2005, 65, 2433–2440.
- Waxman, A.J.; Clasen, S.; Hwang, W.-T.; Garfall, A.; Vogl, D.T.; Carver, J.; O’Quinn, R.; Cohen, A.D.; Stadtmauer, E.A.; Ky, B.; et al. Carfilzomib-Associated Cardiovascular Adverse Events: A Systematic Review and Meta-analysis. JAMA Oncol. 2018, 4, e174519.
- Jakubowiak, A.J.; DeCara, J.M.; Mezzi, K. Cardiovascular events during carfilzomib therapy for relapsed myeloma: Practical management aspects from two case studies. Hematology 2017, 22, 585–591.
- Khosrow-Khavar, F.; Filion, K.B.; Bouganim, N.; Suissa, S.; Azoulay, L. Aromatase Inhibitors and the Risk of Cardiovascular Outcomes in Women with Breast Cancer: A Population-Based Cohort Study. Circulation 2020, 141, 549–559.
- Fontanella, C.; Aita, M.; Cinausero, M.; Aprile, G.; Baldin, M.G.; Dusi, V.; Lestuzzi, C.; Fasola, G.; Puglisi, F. Capecitabine-induced cardiotoxicity: More evidence or clinical approaches to protect the patients’ heart? OncoTargets Ther. 2014, 7, 1783–1791.
- Hilmi, M.; Ederhy, S.; Waintraub, X.; Funck-Brentano, C.; Cohen, A.; Vozy, A.; Lebrun-Vignes, B.; Moslehi, J.; Nguyen, L.S.; Salem, J.-E. Cardiotoxicity Associated with Gemcitabine: Literature Review and a Pharmacovigilance Study. Pharmaceuticals 2020, 13, 325.
- Aghel, N.; Lipton, J.H.; Atenafu, E.G.; Kim, D.D.H.; Delgado, D.H. Cardiovascular Events after Exposure to Nilotinib in Chronic Myeloid Leukemia: Long-term Follow-up. Clin. Lymphoma Myeloma Leuk. 2017, 17, 870–878.e871.
- Santoro, M.; Accurso, V.; Mancuso, S.; Contrino, A.D.; Sardo, M.; Novo, G.; Di Piazza, F.; Perez, A.; Russo, A.; Siragusa, S. Management of Ponatinib in Patients with Chronic Myeloid Leukemia with Cardiovascular Risk Factors. Chemotherapy 2019, 64, 205–209.
- Totzeck, M.; Mincu, R.I.; Rassaf, T. Cardiovascular Adverse Events in Patients with Cancer Treated with Bevacizumab: A Meta-Analysis of More Than 20,000 Patients. J. Am. Heart Assoc. 2017, 6, 6.
- Duran, J.M.; Makarewich, C.A.; Trappanese, D.; Gross, P.; Husain, S.; Dunn, J.; Lal, H.; Sharp, T.E.; Starosta, T.; Vagnozzi, R.J.; et al. Sorafenib Cardiotoxicity Increases Mortality after Myocardial Infarction. Circ. Res. 2014, 114, 1700–1712.
- Di Lorenzo, G.; Autorino, R.; Bruni, G.; Cartenì, G.; Ricevuto, E.; Tudini, M.; Ficorella, C.; Romano, C.; Aieta, M.; Giordano, A.; et al. Cardiovascular toxicity following sunitinib therapy in metastatic renal cell carcinoma: A multicenter analysis. Ann. Oncol. 2009, 20, 1535–1542.
- Justice, C.N.; Derbala, M.H.; Baich, T.M.; Kempton, A.N.; Guo, A.S.; Ho, T.H.; Smith, S.A. The Impact of Pazopanib on the Cardiovascular System. J. Cardiovasc. Pharmacol. Ther. 2018, 23, 387–398.
- Chen, J.; Wang, J. Risk of regorafenib-induced cardiovascular events in patients with solid tumors: A systematic review and meta-analysis. Medicine 2018, 97, e12705.
- Tanriverdi, O.; Ates, S.; Sandal, K.K.; Uylas, S.; Bosna, I.C.; Alkan, A. Left ventricular dysfunction associated with axitinib and nivolumab experience in an advanced renal cell carcinoma. J. Oncol. Pharm. Pract. 2020, 26, 1765–1768.
- Grabowski, J.; Glode, A. Ramucirumab: A vascular endothelial growth factor receptor-2 inhibitor with activity in several malignancies. Am. J. Health-Syst. Pharm. 2016, 73, 957–968.
- Sharma, T.; Dhingra, R.; Singh, S.; Sharma, S.; Tomar, P.; Malhotra, M.; Bhardwaj, T.R. Aflibercept: A Novel VEGF Targeted Agent to Explore the Future Perspectives of Anti-Angiogenic Therapy for the Treatment of Multiple Tumors. Mini-Rev. Med. Chem. 2013, 13, 530–540.
- Bikiewicz, A.; Banach, M.; von Haehling, S.; Maciejewski, M.; Bielecka-Dabrowa, A. Adjuvant breast cancer treatments cardiotoxicity and modern methods of detection and prevention of cardiac complications. ESC Heart Fail. 2021, 8, 2397–2418.
- Gür, F.; Cengiz, M.; Kutlu, H.M.; Cengiz, B.P.; Ayhancı, A. Molecular docking analyses of Escin as regards cyclophosphamide-induced cardiotoxicity: In vivo and in Silico studies. Toxicol. Appl. Pharmacol. 2020, 411, 115386.
- Herradón, E.; González, C.; González, A.; Uranga, J.A.; López-Miranda, V. Cardiovascular Toxicity Induced by Chronic Vincristine Treatment. Front. Pharmacol. 2021, 12, 692970.
- Herradón, E.; González, C.; Uranga, J.A.; Abalo, R.; Martín, M.I.; López-Miranda, V. Characterization of Cardiovascular Alterations Induced by Different Chronic Cisplatin Treatments. Front. Pharmacol. 2017, 8, 196.
- Gozhenko, A.; Bestanchuk, O.; Kaschenko, O.; Narbutova, T. Cumulative cardiotoxic effect of bleomycin in experiment. J. Educ. Health Sport 2021, 11, 301–308.
- Desai, M.Y.; Jellis, C.L.; Kotecha, R.; Johnston, D.R.; Griffin, B.P. Radiation-Associated Cardiac Disease: A Practical Approach to Diagnosis and Management. JACC Cardiovasc. Imaging 2018, 11, 1132–1149.
- Belzile-Dugas, E.; Eisenberg, M.J. Radiation-Induced Cardiovascular Disease: Review of an Underrecognized Pathology. J. Am. Heart Assoc. 2021, 10, e021686.
- Darby, S.C.; Cutter, D.J.; Boerma, M.; Constine, L.S.; Fajardo, L.F.; Kodama, K.; Mabuchi, K.; Marks, L.B.; Mettler, F.A.; Pierce, L.J.; et al. Radiation-Related Heart Disease: Current Knowledge and Future Prospects. Int. J. Radiat. Oncol. Biol. Phys. 2010, 76, 656–665.
- Hendry, J.H. Threshold doses and circulatory disease risks. Ann. ICRP 2015, 44, 69–75.
- Stewart, F.A. Mechanisms and dose-response relationships for radiation-induced cardiovascular disease. Ann. ICRP 2012, 41, 72–79.
- Guddati, A.K.; Joy, P.S.; Kumar, G. Analysis of outcomes of percutaneous coronary intervention in metastatic cancer patients with acute coronary syndrome over a 10-year period. J. Cancer Res. Clin. Oncol. 2015, 142, 471–479.
- Munoz, E.; Iliescu, G.; Vejpongsa, P.; Charitakis, K.; Karimzad, K.; Lopez-Mattei, J.; Yusuf, S.W.; Marmagkiolis, K.; Iliescu, C. Takotsubo Stress Cardiomyopathy. J. Am. Coll. Cardiol. 2016, 68, 1143–1144.
- Al-Hawwas, M.; Tsitlakidou, D.; Gupta, N.; Iliescu, C.; Cilingiroglu, M.; Marmagkiolis, K. Acute Coronary Syndrome Management in Cancer Patients: “Good News” in Cancer Patients? Curr. Oncol. Rep. 2018, 20, 78.
- Hindricks, G.; Potpara, T.; Dagres, N.; Arbelo, E.; Bax, J.J.; Blomström-Lundqvist, C.; Boriani, G.; Castella, M.; Dan, G.-A.; Dilaveris, P.E.; et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS): The Task Force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) Developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. Eur. Heart J. 2021, 42, 373–498.
- Collet, J.-P.; Thiele, H.; Barbato, E.; Barthélémy, O.; Bauersachs, J.; Bhatt, D.L.; Dendale, P.; Dorobantu, M.; Edvardsen, T.; Folliguet, T.; et al. 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur. Heart J. 2021, 42, 1289–1367.
- Ibanez, B.; James, S.; Agewall, S.; Antunes, M.J.; Bucciarelli-Ducci, C.; Bueno, H.; Caforio, A.L.P.; Crea, F.; Goudevenos, J.A.; Halvorsen, S.; et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: The Task Force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC). Eur. Heart J. 2018, 39, 119–177.
- Potts, J.E.; Iliescu, C.A.; Lopez-Mattei, J.; Martinez, S.C.; Holmvang, L.; Ludman, P.; De Belder, M.A.; Kwok, C.S.; Rashid, M.; Fischman, D.L.; et al. Percutaneous coronary intervention in cancer patients: A report of the prevalence and outcomes in the United States. Eur. Heart J. 2018, 40, 1790–1800.
- Borovac, J.A.; Kwok, C.S.; Iliescu, C.; Lee, H.J.; Kim, P.Y.; Palaskas, N.L.; Zaman, A.; Butler, R.; Lopez-Mattei, J.C.; Mamas, M.A. Percutaneous Coronary Intervention and Outcomes in Patients with Lymphoma in the United States (Nationwide Inpatient Sample Analysis). Am. J. Cardiol. 2019, 124, 1190–1197.
- Potts, J.; Mohamed, M.O.; Mattei, J.C.L.; Iliescu, C.A.; Konopleva, M.; Rashid, M.; Bagur, R.; Mamas, M.A. Percutaneous coronary intervention and in-hospital outcomes in patients with leukemia: A nationwide analysis. Catheter. Cardiovasc. Interv. 2019, 96, 53–63.
- Mohamed, M.O.; Van Spall, H.G.C.; Kontopantelis, E.; Alkhouli, M.; Barac, A.; Elgendy, I.Y.; Khan, S.U.; Kwok, C.S.; Shoaib, A.; Bhatt, D.L.; et al. Effect of primary percutaneous coronary intervention on in-hospital outcomes among active cancer patients presenting with ST-elevation myocardial infarction: A propensity score matching analysis. Eur. Heart J. Acute Cardiovasc. Care 2021, 10, 829–839.
- Mohamed, M.O.; Lopez-Mattei, J.C.; Parwani, P.; Iliescu, C.A.; Bharadwaj, A.; Kim, P.Y.; Palaskas, N.L.; Rashid, M.; Potts, J.; Kwok, C.S.; et al. Management strategies and clinical outcomes of acute myocardial infarction in leukaemia patients: Nationwide insights from United States hospitalisations. Int. J. Clin. Pract. 2020, 74, e13476.
- Styczkiewicz, K.; Styczkiewicz, M.; Myćka, M.; Mędrek, S.; Kondraciuk, T.; Czerkies-Bieleń, A.; Wiśniewski, A.; Szmit, S.; Jankowski, P. Clinical presentation and treatment of acute coronary syndrome as well as 1-year survival of patients hospitalized due to cancer: A 7-year experience of a nonacademic center. Medicine 2020, 99, e18972.
- Nabiałek-Trojanowska, I.; Dąbrowska-Kugacka, A.; Lewicka-Potocka, Z.; Abdulaziz, Y.; Szerszyńska, A.; Raczak, G.; Lewicka, E. Acute coronary syndrome in patients undergoing anticancer therapies: A single-center, controlled case study. Adv. Clin. Exp. Med. 2019, 28, 1667–1673.
- Iliescu, C.A.; Grines, C.L.; Herrmann, J.; Yang, E.H.; Cilingiroglu, M.; Charitakis, K.; Hakeem, A.; Toutouzas, K.P.; Leesar, M.A.; Marmagkiolis, K. SCAI Expert consensus statement: Evaluation, management, and special considerations of cardio-oncology patients in the cardiac catheterization laboratory (endorsed by the cardiological society of india, and sociedad Latino Americana de Cardiologıa intervencionista). Catheter. Cardiovasc. Interv. 2016, 87, E202–E223.
- Ueki, Y.; Vögeli, B.; Karagiannis, A.; Zanchin, T.; Zanchin, C.; Rhyner, D.; Otsuka, T.; Praz, F.; Siontis, G.C.M.; Moro, C.; et al. Ischemia and Bleeding in Cancer Patients Undergoing Percutaneous Coronary Intervention. JACC CardioOncol. 2019, 1, 145–155.
- Velders, M.A.; Hagström, E.; James, S.K. Temporal Trends in the Prevalence of Cancer and Its Impact on Outcome in Patients with First Myocardial Infarction: A Nationwide Study. J. Am. Heart Assoc. 2020, 9, e014383.
- Heller, S.J.; Tokar, J.L.; Nguyen, M.T.; Haluszka, O.; Weinberg, D.S. Management of bleeding GI tumors. Gastrointest. Endosc. 2010, 72, 817–824.
- Kwok, C.S.; Wong, C.W.; Kontopantelis, E.; Barac, A.; Brown, S.-A.; Velagapudi, P.; Hilliard, A.A.; Bharadwaj, A.S.; Alraies, M.C.; Mohamed, M.; et al. Percutaneous coronary intervention in patients with cancer and readmissions within 90 days for acute myocardial infarction and bleeding in the USA. Eur. Heart J. 2021, 42, 1019–1034.
- Nishikawa, T.; Morishima, T.; Fujii, Y.; Okawa, S.; Otsuka, T.; Kamada, R.; Yasui, T.; Shioyama, W.; Oka, T.; Tabuchi, T.; et al. Prognostic Impact of Cancer Activity on Clinically Relevant Bleeding Events after Percutaneous Coronary Intervention. J. Med. Investig. 2021, 68, 29–37.
- Bharadwaj, A.S.; Swamy, P.M.; Mamas, M.A. Outcomes of percutaneous coronary interventions in cancer patients. Expert Rev. Cardiovasc. Ther. 2020, 18, 25–32.
- Zaleska, M.; Mozenska, O.; Bil, J. Statins use and cancer: An update. Futur. Oncol. 2018, 14, 1497–1509.
- Sarkiss, M.G.; Yusuf, S.W.; Warneke, C.L.; Botz, G.; Lakkis, N.; Hirch-Ginsburg, C.; Champion, J.C.; Swafford, J.; Shaw, A.D.S.; Lenihan, D.J.; et al. Impact of aspirin therapy in cancer patients with thrombocytopenia and acute coronary syndromes. Cancer 2006, 109, 621–627.
- Iliescu, C.; Balanescu, D.V.; Donisan, T.; Giza, D.E.; Gonzalez, E.D.M.; Cilingiroglu, M.; Song, J.; Mukerji, S.S.; Lopez-Mattei, J.C.; Kim, P.Y.; et al. Safety of Diagnostic and Therapeutic Cardiac Catheterization in Cancer Patients with Acute Coronary Syndrome and Chronic Thrombocytopenia. Am. J. Cardiol. 2018, 122, 1465–1470.
