Photocatalysis is a more recently applied concept and is proven to be able to completely remove and degrade pollutants into simpler organic compounds. Titanium dioxide (TiO2) is a fine example of a photocatalyst owing to its cost-effectiveness and superb efficiency. However, issues such as the high recombination rate of photogenerated electrons along with positive holes while being only limited to UV irradiation need to be addressed. Carbonaceous materials such as graphene oxide (GO) can overcome such issues by reducing the recombination rate and providing a platform for adsorption accompanied by photocatalytic degradation of TiO2.
- graphene oxide
- titanium dioxide
- photocatalysis
1. Conventional Wastewater Treatment Methods for Waste Removal
With the rapid development of industry in recent years, and the manufacturing industry in particular, the amount of untreated organic waste being released into the environment has gradually increased, and it also poses potential health problems to both aquatic life and humans. Many methods have been developed and implemented over the years to control pollution. Conventional wastewater treatment methods can be split into three main branches which are chemical, physical, and biological methods.
2. Photocatalysis
2.1. TiO
2.1. TiO
2
2.2. GO
2.3. Production of GO
There are two mainstream approaches to synthesize GO: bottom-up methods whereby simple carbon molecules are used to generate pristine graphene, and top-down methods whereby layers of graphite are used to extract graphene sheets [28][100]. The drawbacks of bottom-up techniques are that they are time-consuming and are not suitable for large-scale production yet. Bottom-up techniques include chemical vapor deposition, epitaxial growth on silicon carbide, and so on. Therefore, the top-down method would be a more favorable and preferred option in the production of graphene derivatives. The products that are first obtained are graphene oxide or even reduced graphene oxide, which are well established in the application of nanocomposite materials.
2.4. Production of TiO
2
2.4.1. Electrophoretic Deposition
2.4.2. Spray Pyrolysis
2.4.3. Sol Gel Method
Generally speaking, the sol-gel process entails transforming a system from a liquid “sol” phase, which is mostly in colloidal form, into a solid “gel” phase. Metal organic compounds, such as metal alkoxide and inorganic metal salts, are common precursors in the synthesis of “sol.” A sol or colloidal suspension is created by a succession of hydrolysis and polymerization processes. A wet “gel” will form when a sol is cast into a mold. The gel can be further dried and heated to create solid products.2.4.4. Sonochemical and Microwave-Assisted Methods
Efficient photoactive TiO2 nanoparticles can also be synthesized via the sonochemical method while using ultrasonic irradiation for the hydrolysis of titanium tetraisopropoxide (TTIP) in pure water or in an ethanol/water mixture. The concept of acoustic cavitation causes the formation, growth, and collapse of bubbles in the solution while temperature at about 5000 K and pressures at about 1000 atm are the result of cavitational collapse.2.5. Production of GO-TiO
2
2.5.1. Hydrothermal Method
Hydrothermal synthesis indicates a high-temperature and pressure technique for growing crystals from an aqueous solution in an autoclave. The characteristic of water is a solvent with a low boiling point, which allows it to be used under high pressure. Solvents with a high boiling point, such as dimethyl sulfoxide (DMSO), can be expensive and pose potential hazards, thus making water a very attractive option. Fine crystals of the desired nanocomposites are created using increased temperature. Hydrothermal synthesis enables the composition and consistency of the nanocrystals produced to be controlled. The key drawbacks associated with this process, however, are the inability to control material crystal growth (in the autoclave) and the cost of the equipment [31][107]. The hydrothermal reaction can also be used to partially reduce GO to graphene.2.5.2. Solvothermal Method
Solvothermal synthesis, analogous to the hydrothermal method, involves a process for fabricating crystals from non-aqueous organics using an autoclave at high temperatures and pressure [32][109]. Compared to the hydrothermal method, the solvothermal method typically has a greater effect on the size, shape, distribution, and crystallinity of the prepared nanocomposites.2.5.3. Mechanical Mixing
The simplicity and manipulations of the conditions of the reaction have made this strategy gradually gain popularity. This process involves mixing pristine or functionalized TiO2 with GO dispersions, likely accompanied by sonication and stirring to increase the optimum interaction between the precursors of the nanocomposite.3. Heterojunction of GO/TiO2 Photocatalyst
GO acts as one of the superior support materials for different semiconductors and metals. The photoelectrons generated at the TiO2 conduction band are rapidly transferred to the graphene layer by the TiO2 nanoparticle-decorated GO, which promotes the rate of organic dye pollutant degradation. In addition, during photocatalytic reactions, the high surface area of GO provides more surface adsorption sites for contaminants to greatly promote surface photocatalytic reactions, thus improving the catalytic activity. To summarize, graphene oxide has contributed in three ways to improving the photocatalytic degradation of pollutants: (1) improvement of the surface area of TiO2 due to its interaction with the two-dimensional matt structure of GO; (2) improvement of the adsorption of aromatic pollutants due to their strong π-π interactions with the aromatic network of GO; and (3) reduction of the recombination rate between the positive holes and the photogenerated electrons due to the significant electronic conductivity of GO, which functions as an electron sink for photogenerated electrons on the TiO2 surface [33][34][110,111].4. Advantages of GO/TiO2 Composite Structure
Research carried out by [35][113] showed that the surface area of GO/TiO2 composite (78.12 m2/g) is larger than that of bare TiO2 (57.01 m2/g) through the nitrogen adsorption/desorption measurement using the Brunauer–Emmett–Teller (BET) equation. The researchers showed that increase in the surface area enhanced the pollutant adsorption ability of GO/TiO2 composite to approximately 37% compared to that of the produced bare TiO2. The contact area between the produced photocatalyst and pollutants is enhanced [36][114]. Thus, the photodegradation performance of GO/TiO2 composite is better than that of bare TiO2. A large surface area provides more active sites for redox reaction [37][115] including separation and transfer of photogenerated electron-hole pairs [38][116], thus enhancing the photodegradation performance. Besides, large surface area also improves the utilization of light [38][116].
Band gap absorption edge of bare TiO2 is 440 nm, which mostly adsorbs UV light to initiate the photodegradation process. GO has great absorption in the visible light range [36][114]. Thus, when TiO2 combines with GO, the absorption range of GO/TiO2 composite can be expanded up to a wavelength range of 800 nm [35][113]. Compared with GO and TiO2, the light absorption characteristics of GO/TiO2 composite are enhanced. The generated Ti3+ enhances the visible light absorption range [36][114]. Research by [35][113] showed that prepared bare TiO2 had a bandgap reading of 3.20 eV and the bandgap reading of GO/TiO2 composite was 2.80 eV using a Tauc plot. This outcome indicates GO/TiO2 composite can absorb visible light effectively and therefore, enhance the photodegradation performance under visible light irradiation.
GO/TiO2 composite possess large surface area and a low photogenerated electron-hole pairs recombination rate to degrade the pollutant. Other than that, the visible light absorption range of the GO/TiO2 composite can also be enhanced due to the response of graphene to visible light [36][114]. Electrochemical impedance spectroscopy (EIS) and photocurrent study can be carried out using a three-electrode system consisting of FTO covered with a sample as a working electrode, a platinum plate as counter electrode, and Ag/AgCl as a reference electrode.
5. GO-TiO2-Related Nanocomposites in Wastewater Treatment and Their Findings
The GO-TiO2 heterostructure is one of the most significant and frequently studied semiconductor photocatalysts for the decomposition of organic pollutants [39][119]. Factors such as high surface area, excellent conductivity, and chemical stability allow carbonaceous materials such as graphene oxide to be a compactible surface modification combination with titanium dioxide. Hence, a plethora of studies have investigated and reported on graphene oxide-coupled TiO2, including other semiconductor materials for enhanced photocatalytic performance over organic pollutant degradation. Lin et al. reported on synthesized reduced graphene oxide decorated with titanium dioxide nanocomposites via a combination of ultrasonication and hydrothermal reaction which involved titanium tetrachloride (TiCl3) and graphene oxide (GO) as precursors [40][120]. As a result of combining the two components, the adsorption and degradation of methyl orange (MO) were significantly increased. The prepared sample was able to remove up to almost 90% of the dye, which was nearly threefold the rates of bare TiO2 and GO under ultraviolet lamp irradiation for 4 h. The process having lower degradation efficiency in sunlight than using ultraviolet illumination is due to the excitation of electrons in the conduction band being made easier by the high energy of ultraviolet light. The merit of a hybrid allows a greater surface area which contributes to the adsorption of MO onto GO surfaces via π-π interaction, followed by degradation of MO by active groups generated by TiO2, feats that cannot be accomplished by bare TiO2 and GO alone.
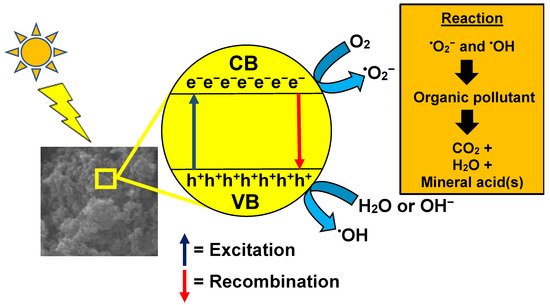
6. GO/TiO2 Photocatalytic Mechanism
The application of GO/TiO2 for the removal of aromatic pollutants such as dyes (methylene blue and methyl orange) reflects the efficiency of GO/TiO2 nanocomposites. Equations (1)–(8) describe the route taken by the photogenerated electrons followed by the decomposition of aromatic pollutants by GO/TiO2. The proposed mechanism of formation of radicals for the photodegradation of pollutants is in agreement with [41][126]. Lin et al. [40][120] also stated that dye molecules were initially adsorbed onto the surface of GO/TiO2 under the influence of π-π bonds of a graphene sheet. The irradiation source then initiates the photogeneration of electron-hole pairs on the surf ace of photocatalyst. The resultant hole will break apart water molecules into hydrogen ions and hydroxyl radicals (OH•). The resultant electrons in the valence band of TiO2 are transferred to the conduction band of GO to convert oxygen molecules into hydrogen peroxide and subsequently OH•. This whole process repeats as long as the irradiation source still remains active.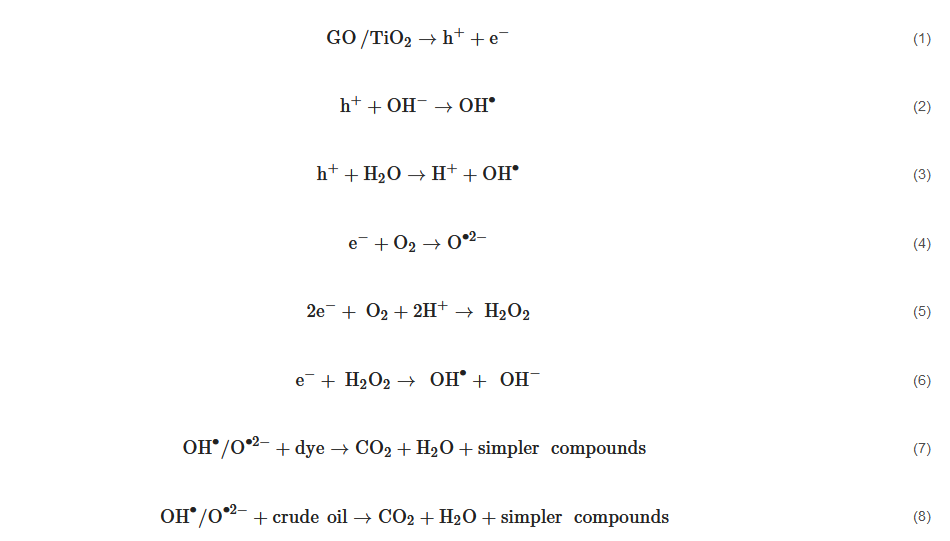 Band edge positions of the photocatalysts forming the GO/TiO2 composite have been determined for better estimation of the photogenerated charge carrier separation mechanism. The conduction band (CB) and valance band (VB) potentials of the photocatalysts represented by ECB and EVB can be calculated using Equations (9) and (10) [42][144] as below:
Band edge positions of the photocatalysts forming the GO/TiO2 composite have been determined for better estimation of the photogenerated charge carrier separation mechanism. The conduction band (CB) and valance band (VB) potentials of the photocatalysts represented by ECB and EVB can be calculated using Equations (9) and (10) [42][144] as below:
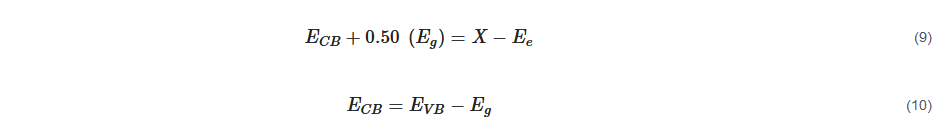 where Eg represents bandgap energy which can be obtained through the Tauc plot, X represents absolute electronegativity, and Ee represents energy of free electrons on the hydrogen scale which has an approximate reading of 4.50 eV.
On the other hand, Mott-Schottky (MS) can be utilized to determine the rationality of the ECB and EVB readings calculated through the previous equations. By using the MS plot, scholarswe are able to determine the type of semiconductor or metal oxide. Based on the literature, GO is a p-type semiconductor whereas TiO2 is an n-type semiconductor [43][145].
The flat band potential can be converted to normal hydrogen scale using Equation (11) [43][145] below:
where Eg represents bandgap energy which can be obtained through the Tauc plot, X represents absolute electronegativity, and Ee represents energy of free electrons on the hydrogen scale which has an approximate reading of 4.50 eV.
On the other hand, Mott-Schottky (MS) can be utilized to determine the rationality of the ECB and EVB readings calculated through the previous equations. By using the MS plot, scholarswe are able to determine the type of semiconductor or metal oxide. Based on the literature, GO is a p-type semiconductor whereas TiO2 is an n-type semiconductor [43][145].
The flat band potential can be converted to normal hydrogen scale using Equation (11) [43][145] below:
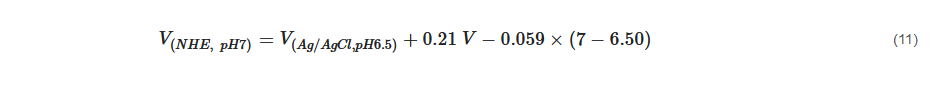
7. Factors Affecting the Degradation of Organic Pollutants
Due to variations in the lattice structure, morphology, surface area, particle size, as well as impurities on the catalyst surface, different photocatalysts will have different photocatalytic activities, thus, influencing the adsorption of pollutants at the surface of the photocatalyst, and the rate of recombination of electron-hole pairs. One of the important variables that will affect the performance of dye degradation is pH as it influences the dye reactions in several ways. Adsorption of pollutants onto the surface of the photocatalyst can contribute to the efficiency of the photocatalyst in the removal of pollutants. Pollutant adsorption depends on the initial pollutant concentration in the solution. The initial concentrations of the pollutant can thus influence the process of photodegradation. The pollutants that are adsorbed on the surface and not the bulk of the solution are involved in the process. High concentrations of pollutants will increase the turbidity of water and cover more active sites. Consequently, fewer photons will reach the surface; fewer OH2 species will therefore be produced, leading to a decrease in the efficiency of degradation.
The optimum amount of photocatalyst needed usually depends on the concentration of the pollutant and the volume of the solution in which the pollutants are mixed. As the concentration of photocatalyst is below the optimum level, the rate of photocatalysis will gradually increase along with the increase in photocatalyst loading. This can be explained due to the increase in available active sites on the photocatalyst surface, and the number of radicals generated from the photocatalyst to degrade and mineralize the pollutant. However, when the photocatalyst is further added beyond the optimum level, the rate of photodegradation gradually decreases. At high concentrations, the photocatalyst will likely agglomerate and thus lead to unfavorable scattering of light and fewer photons can reach the surface of the photocatalyst [44][151].
