The issue of global warming calls for a greener energy production approach. To this end, bioenergy has significant greenhouse gas mitigation potential, since it makes use of biological products/wastes and can efficiently counter carbon dioxide emission. However, technologies for biomass processing remain limited due to the structure of biomass and difficulties such as high processing cost, development of harmful inhibitors and detoxification of produced inhibitors that hinder widespread usage. Additionally, cellulose pre-treatment is often required to be amenable for an enzymatic hydrolysis process. Nanotechnology (usage of nanomaterials, in this case) has been employed in recent years to improve bioenergy generation, especially in terms of catalyst and feedstock modification. The role of nanotechnology to assist in bioenergy generation is discussed, particularly from the aspects of enzyme immobilization, biogas production and biohydrogen production.
- nanomaterials
- enzyme
- biogas
1. Introduction

2. Enzyme Immobilization
| Bioenergy | MNPs | Feedstock | Enzyme | Summary | Ref. |
|---|---|---|---|---|---|
| Bioethanol | Fe3O4 | Aspergillus fumigatus AA001 | Cellulase | Thermal stability for 8 h at 70 °C | [18][23] |
| Fe3O4 | Potato peels | Amylase/Amyloglucosidase | Bioethanol yield: 93% | [75][24] | |
| Fe3O4 | Allamanda schottii L. | Cellulase | Bioethanol yield (free enzymes): 182 g/L; (immobilized enzymes): 252 g/L | [76][25] | |
| FeCl3 | Sesbania aculeata | Cellulase | Bioethanol yield: 5.31 g/L | [29][15] | |
| MnO2 | Aspergillus fumigatus JCF | Cellulase | Cellulase binding efficiency of 75% Bioethanol yield: 21.96 g/L |
[77][26] | |
| Biodiesel | Fe2O3 | Neochloris oleoabundans | Lipase | Maximum biodiesel yield: 81% | [78][27] |
| Fe3O4 | Soybean oil | Lipase | Biodiesel yield: 90% with 60% immobilized lipase | [79][28] | |
| Fe3O4 | Soybean oil | Lipase | Conversion rate: 47.60% after 24 h; >30% enzyme activity even after 10 cycles | [30][29] | |
| Fe3O4 | Soybean oil | Lipase | Conversion rate: 88% after 192 h and 75% after 240 h | [80][30] | |
| Fe3O4 | Microalgae (Chlorella vulgaris) | Lipase | Maximum biodiesel yield: 97.1% | [81][31] | |
| Fe3O4 | Jatropha curcas oil | Rhizomucor miehei lipase | Maximum biodiesel yield: 70% | [82][32] | |
| Polyporous magnetic cellulose beads (PMCBs) | Yellow horn seed oil | Candida antarctica lipase B | Maximum biodiesel yield: 92.3%; catalyst easily removed by magnet and can be recycled at least 5 times. | [83][33] |
3. Biogas Production
4. Biohydrogen Production
4.1. Dark Fermentative Biohydrogen Process
4.2. Photo-Fermentative Biohydrogen Production
-
Nanoparticles enhance production of carbohydrates, which results in increased growth of algal cells. For example, silica nanoparticles enhanced growth of microalgal cells (measured from chlorophyll concentration), because silica nanoparticles scattered light within a reactor to ensure uniform light distribution during the photosynthetic process, which in turn promoted growth of microalgal cells [101][51]. Similar findings were also reported using zero-valent iron (Fe0) [102][52] and TiO2 nanoparticles, which increased chlorophyll and carotenoid pigments [103][53].
-
Nanoparticles enhance activity of key enzymes for metabolism of microalgal species such as glutamate dehydrogenase, glutamate–pyruvate transaminase, glutamine synthase and nitrate reductase [104,105][54][55]. The capability of nanoparticles to maintain the pH of a medium and to promote the activity of hydrogenase enzymes and substrate hydrolysis may promote higher biohydrogen yield by enhancing biohydrogen-producing metabolic pathways such as acetate and butyrate reactions [32,103,106][53][56][57].
4.3. Photocatalytic Hydrogen Production
| NP | Feedstock | Microorganism | H2 Yield Rate | H2 Yield Increase (%) | Ref. |
|---|---|---|---|---|---|
| Au | Artificial wastewater | Clostridium butyricum | 4.48 mol H2/mol sucrose | 61.7 | [93][43] |
| Ag | Inorganic salts | Clostridium butyricum | 2.48 mol H2/mol glucose | 67.5 | [111][62] |
| Pd | Glucose | Enterobacter cloacae + mixed culture | 2.48 mol H2/mol glucose | 6.4 | [112][63] |
| Ni | Inorganic salts | Granular sludge | 2.54 mol H2/mol glucose | 22.7 | [113][64] |
| Cu | Glucose | Clostridium acetobutylicum | 1.74 mol H2/mol glucose | N/A | [114][65] |
| Cu | Glucose | Enterobacter clocae | 1.44 mol H2/mol glucose | N/A | [114][65] |
| Fe | Inorganic salts | Enterobacter clocae | 1.9 mol H2/mol glucose | 68.4 | [115][66] |
| Fe | Growth medium | Rhodobacter sphaeroides + Escherichia coli | 3.1 mol H2/mol malate | N/A | [116][67] |
| Fe2O3 | Casava starch | Enterobacter aerogenes | 192.4 mL H2/g casava starch | 17 | [117][68] |
| Fe2O3 | Distillery wastewater | Mixed culture | 44.28 mL H2/g COD | N/A | [118][69] |
| Fe2O3 | Growth medium | Clostridium acetobutylicum | 2.33 mol H2/mol glucose | 52 | [119][70] |
| FeO | Growth medium | Mixed culture | 1.92 mol H2/mol glucose | 7.9 | [120][71] |
| TiO2 | Growth medium | Rhodopseudomonas palustris | N/A | 46.1 | [121][72] |
| SiO2 | Air: CO2 (97:3) | Chlamydomonas reinhardtii CC124 | 3121.5 H2/L/h | 45.2 | [101][51] |
| Biochar | Municipal solid waste | Enterobacter aerogenes + Escherichia coli | 96.3 mL/g | N/A | [86][36] |
| Biochar | Food waste | N/A | 944.5 mL/L | 31 | [87][37] |
| Biochar + ZV iron NP | Grass biomass | N/A | 50.6 mL/g dry grass | 89.8 | [88][38] |
| Ni + graphene | Industrial wastewater | Mixed culture | 41.3 mL/gCOD | 105 | [98][48] |
| CNT | Glucose | Anaerobic sludge | 2.45 mol/mol substrate | N/A | [122][73] |
| Hematite + TiO2 | Glucose | Clostridium pasteurianum CH5 | 2.20 mol/mol substrate | 5 | [123][74] |
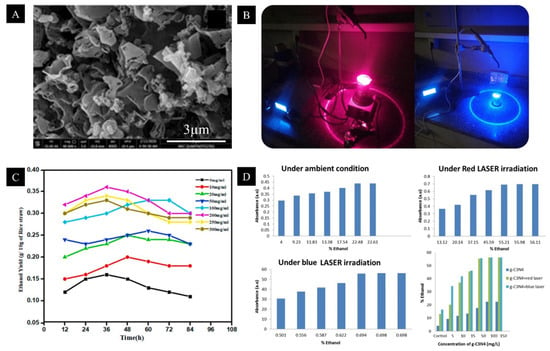
5. Bioethanol Production
References
- Erickson, P.; Lazarus, M.; Piggot, G. Limiting fossil fuel production as the next big step in climate policy. Nat. Clim. Chang. 2018, 8, 1037–1043.
- Johnsson, F.; Kjärstad, J.; Rootzén, J. The threat to climate change mitigation posed by the abundance of fossil fuels. Clim. Policy 2019, 19, 258–274.
- Antunes, F.A.F.; Gaikwad, S.; Ingle, A.P.; Pandit, R.; dos Santos, J.C.; Rai, M.; da Silva, S.S. Bioenergy and Biofuels: Nanotechnological Solutions for Sustainable Production; Springer: Berlin/Heidelberg, Germany, 2017; ISBN 9783319454597.
- Chai, W.S.; Bao, Y.; Jin, P.; Tang, G.; Zhou, L. A review on ammonia, ammonia-hydrogen and ammonia-methane fuels. Renew. Sustain. Energy Rev. 2021, 147, 111254.
- Du, H.; Shen, P.; Chai, W.S.; Nie, D.; Shan, C.; Zhou, L. Perspective and analysis of ammonia-based distributed energy system (DES) for achieving low carbon community in China. iScience 2022, 25, 105120.
- Elumalai, S.; Agarwal, B.; Runge, T.M.; Sangwan, R.S. Advances in Transformation of Lignocellulosic Biomass to Carbohydrate-Derived Fuel Precursors. In Biorefining of Biomass to Biofuels; Springer: Berlin/Heidelberg, Germany, 2018; pp. 87–116. ISBN 9783319676784.
- Scarlat, N.; Dallemand, J.-F.; Monforti-Ferrario, F.; Nita, V. The role of biomass and bioenergy in a future bioeconomy: Policies and facts. Environ. Dev. 2015, 15, 3–34.
- Den, W.; Sharma, V.K.; Lee, M.; Nadadur, G.; Varma, R.S. Lignocellulosic biomass transformations via greener oxidative pretreatment processes: Access to energy and value added chemicals. Front. Chem. 2018, 6, 141.
- Sindhu, R.; Binod, P.; Pandey, A. Biological pretreatment of lignocellulosic biomass—An overview. Bioresour. Technol. 2016, 199, 76–82.
- Singhvi, M.; Kim, B.S. Current developments in lignocellulosic biomass conversion into biofuels using nanobiotechology approach. Energies 2020, 13, 5300.
- Sankaran, R.; Markandan, K.; Khoo, K.S.; Cheng, C.K.; Ashokkumar, V.; Deepanraj, B.; Show, P.L. The Expansion of Lignocellulose Biomass Conversion Into Bioenergy via Nanobiotechnology. Front. Nanotechnol. 2021, 3, 96.
- Varma, R.S. Biomass-Derived Renewable Carbonaceous Materials for Sustainable Chemical and Environmental Applications. ACS Sustain. Chem. Eng. 2019, 7, 6458–6470.
- Rai, A.K.; Al Makishah, N.H.; Wen, Z.; Gupta, G.; Pandit, S.; Prasad, R. Recent Developments in Lignocellulosic Biofuels, a Renewable Source of Bioenergy. Fermentation 2022, 8, 161.
- Daystar, J.; Treasure, T.; Gonzalez, R.; Reeb, C.; Venditti, R.; Kelley, S. The NREL biochemical and thermochemical ethanol conversion processes: Financial and environmental analysis comparison. BioResources 2015, 10, 5083–5095.
- Baskar, G.; Naveen Kumar, R.; Heronimus Melvin, X.; Aiswarya, R.; Soumya, S. Sesbania aculeate biomass hydrolysis using magnetic nanobiocomposite of cellulase for bioethanol production. Renew. Energy 2016, 98, 23–28.
- Poorakbar, E.; Shafiee, A.; Saboury, A.A.; Rad, B.L.; Khoshnevisan, K.; Ma’mani, L.; Derakhshankhah, H.; Ganjali, M.R.; Hosseini, M. Synthesis of magnetic gold mesoporous silica nanoparticles core shell for cellulase enzyme immobilization: Improvement of enzymatic activity and thermal stability. Process Biochem. 2018, 71, 92–100.
- Misson, M.; Zhang, H.; Jin, B. Nanobiocatalyst advancements and bioprocessing applications. J. R. Soc. Interface 2015, 12, 20140891.
- Su, T.C.; Fang, Z.; Zhang, F.; Luo, J.; Li, X.K. Hydrolysis of Selected Tropical Plant Wastes Catalyzed by a Magnetic Carbonaceous Acid with Microwave. Sci. Rep. 2015, 5, 17538.
- Goh, W.J.; Makam, V.S.; Hu, J.; Kang, L.; Zheng, M.; Yoong, S.L.; Udalagama, C.N.B.; Pastorin, G. Iron oxide filled magnetic carbon nanotube-enzyme conjugates for recycling of amyloglucosidase: Toward useful applications in biofuel production process. Langmuir 2012, 28, 16864–16873.
- Verma, M.L.; Chaudhary, R.; Tsuzuki, T.; Barrow, C.J.; Puri, M. Immobilization of β-glucosidase on a magnetic nanoparticle improves thermostability: Application in cellobiose hydrolysis. Bioresour. Technol. 2013, 135, 2–6.
- Ren, Y.; Rivera, J.G.; He, L.; Kulkarni, H.; Lee, D.K.; Messersmith, P.B. Facile, high efficiency immobilization of lipase enzyme on magnetic iron oxide nanoparticles via a biomimetic coating. BMC Biotechnol. 2011, 11, 63.
- Hu, S.; Guan, Y.; Wang, Y.; Han, H. Nano-magnetic catalyst KF/CaO-Fe3O4 for biodiesel production. Appl. Energy 2011, 88, 2685–2690.
- Srivastava, N.; Srivastava, M.; Mishra, P.K.; Singh, P.; Ramteke, P.W. Application of Cellulases in Biofuels Industries: An Overview. J. Biofuels Bioenergy 2015, 1, 55.
- Sanusi, I.A.; Suinyuy, T.N.; Kana, G.E.B. Impact of nanoparticle inclusion on bioethanol production process kinetic and inhibitor profile. Biotechnol. Rep. 2021, 29, e00585.
- Vijayalakshmi, S.; Govindarajan, M.; Al-Mulahim, N.; Ahmed, Z.; Mahboob, S. Cellulase immobilized magnetic nanoparticles for green energy production from Allamanda schottii L: Sustainability research in waste recycling. Saudi J. Biol. Sci. 2021, 28, 901–910.
- Cherian, E.; Dharmendirakumar, M.; Baskar, G. Immobilization of cellulase onto MnO2 nanoparticles for bioethanol production by enhanced hydrolysis of agricultural waste. Cuihua Xuebao/Chin. J. Catal. 2015, 36, 1223–1229.
- Banerjee, S.; Rout, S.; Banerjee, S.; Atta, A.; Das, D. Fe2O3 nanocatalyst aided transesterification for biodiesel production from lipid-intact wet microalgal biomass: A biorefinery approach. Energy Convers. Manag. 2019, 195, 844–853.
- Xie, W.; Ma, N. Immobilized Lipase on Fe 3 O 4 Nanoparticles as Biocatalyst for Biodiesel Production. Energy Fuels 2009, 23, 1347–1353.
- Wang, X.; Dou, P.; Zhao, P.; Zhao, C.; Ding, Y.; Xu, P. Immobilization of lipases onto magnetic Fe3O4 nanoparticles for application in biodiesel production. ChemSusChem 2009, 2, 947–950.
- Wang, X.; Liu, X.; Zhao, C.; Ding, Y.; Xu, P. Biodiesel production in packed-bed reactors using lipase-nanoparticle biocomposite. Bioresour. Technol. 2011, 102, 6352–6355.
- Chiang, Y.D.; Dutta, S.; Chen, C.T.; Huang, Y.T.; Lin, K.S.; Wu, J.C.S.; Suzuki, N.; Yamauchi, Y.; Wu, K.C.W. Functionalized Fe3O4@Silica Core-Shell Nanoparticles as Microalgae Harvester and Catalyst for Biodiesel Production. ChemSusChem 2015, 8, 789–794.
- Zhou, Z.W.; Cai, C.X.; Xing, X.; Li, J.; Hu, Z.E.; Xie, Z.B.; Wang, N.; Yu, X.Q. Magnetic COFs as satisfied support for lipase immobilization and recovery to effectively achieve the production of biodiesel by great maintenance of enzyme activity. Biotechnol. Biofuels 2021, 14, 156.
- Zhang, H.; Liu, T.; Zhu, Y.; Hong, L.; Li, T.; Wang, X.; Fu, Y. Lipases immobilized on the modified polyporous magnetic cellulose support as an efficient and recyclable catalyst for biodiesel production from Yellow horn seed oil. Renew. Energy 2020, 145, 1246–1254.
- Abdelsalam, E.; Samer, M.; Attia, Y.A.; Abdel-Hadi, M.A.; Hassan, H.E.; Badr, Y. Influence of zero valent iron nanoparticles and magnetic iron oxide nanoparticles on biogas and methane production from anaerobic digestion of manure. Energy 2017, 120, 842–853.
- Velimirovic, M.; Schmid, D.; Wagner, S.; Micić, V.; von der Kammer, F.; Hofmann, T. Agar agar-stabilized milled zerovalent iron particles for in situ groundwater remediation. Sci. Total Environ. 2016, 563–564, 713–723.
- Sharma, P.; Melkania, U. Biochar-enhanced hydrogen production from organic fraction of municipal solid waste using co-culture of Enterobacter aerogenes and E. coli. Int. J. Hydrogen Energy 2017, 42, 18865–18874.
- Sunyoto, N.M.S.; Zhu, M.; Zhang, Z.; Zhang, D. Effect of biochar addition on hydrogen and methane production in two-phase anaerobic digestion of aqueous carbohydrates food waste. Bioresour. Technol. 2016, 219, 29–36.
- Yang, G.; Wang, J. Synergistic enhancement of biohydrogen production from grass fermentation using biochar combined with zero-valent iron nanoparticles. Fuel 2019, 251, 420–427.
- Su, L.; Shi, X.; Guo, G.; Zhao, A.; Zhao, Y. Stabilization of sewage sludge in the presence of nanoscale zero-valent iron (nZVI): Abatement of odor and improvement of biogas production. J. Mater. Cycles Waste Manag. 2013, 15, 461–468.
- Karri, S.; Sierra-Alvarez, R.; Field, J.A. Zero valent iron as an electron-donor for methanogenesis and sulfate reduction in anaerobic sludge. Biotechnol. Bioeng. 2005, 92, 810–819.
- Wang, T.; Zhang, D.; Dai, L.; Chen, Y.; Dai, X. Effects of metal nanoparticles on methane production from waste-activated sludge and microorganism community shift in anaerobic granular sludge. Sci. Rep. 2016, 6, 25857.
- Yang, Y.; Guo, J.; Hu, Z. Impact of nano zero valent iron (NZVI) on methanogenic activity and population dynamics in anaerobic digestion. Water Res. 2013, 47, 6790–6800.
- Zhang, Y.; Shen, J. Enhancement effect of gold nanoparticles on biohydrogen production from artificial wastewater. Int. J. Hydrogen Energy 2007, 32, 17–23.
- Ramsurn, H.; Gupta, R.B. Nanotechnology in solar and biofuels. ACS Sustain. Chem. Eng. 2013, 1, 779–797.
- Gordon, R.; Seckbach, J. (Eds.) The Science of Algal Fuels: Phycology, Geology, Biophotonics, Genomics and Nanotechnology; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012; ISBN 978-94-007-5110-1.
- Yang, G.; Wang, J. Improving mechanisms of biohydrogen production from grass using zero- valent iron nanoparticles. Bioresour. Technol. 2018, 266, 413–420.
- Beckers, L.; Hiligsmann, S.; Lambert, S.D.; Heinrichs, B.; Thonart, P. Improving effect of metal and oxide nanoparticles encapsulated in porous silica on fermentative biohydrogen production by Clostridium butyricum. Bioresour. Technol. 2013, 133, 109–117.
- Elreedy, A.; Ibrahim, E.; Hassan, N.; El-Dissouky, A.; Fujii, M.; Yoshimura, C.; Tawfik, A. Nickel-graphene nanocomposite as a novel supplement for enhancement of biohydrogen production from industrial wastewater containing mono-ethylene glycol. Energy Convers. Manag. 2017, 140, 133–144.
- Raliya, R.; Biswas, P.; Tarafdar, J.C. TiO2 nanoparticle biosynthesis and its physiological effect on mung bean (Vigna radiata L.). Biotechnol. Rep. 2015, 5, 22–26.
- Pádrová, K.; Lukavský, J.; Nedbalová, L.; Čejková, A.; Cajthaml, T.; Sigler, K.; Vítová, M.; Řezanka, T. Trace concentrations of iron nanoparticles cause overproduction of biomass and lipids during cultivation of cyanobacteria and microalgae. J. Appl. Phycol. 2015, 27, 1443–1451.
- Giannelli, L.; Torzillo, G. Hydrogen production with the microalga Chlamydomonas reinhardtii grown in a compact tubular photobioreactor immersed in a scattering light nanoparticle suspension. Int. J. Hydrogen Energy 2012, 37, 16951–16961.
- Eroglu, E.; Eggers, P.K.; Winslade, M.; Smith, S.M.; Raston, C.L. Enhanced accumulation of microalgal pigments using metal nanoparticle solutions as light filtering devices. Green Chem. 2013, 15, 3155–3159.
- Pandey, A.; Gupta, K.; Pandey, A. Effect of nanosized TiO2 on photofermentation by Rhodobacter sphaeroides NMBL-02. Biomass Bioenergy 2015, 72, 273–279.
- Yang, F.; Hong, F.; You, W.; Liu, C.; Gao, F.; Wu, C.; Yang, P. Influences of nano-anatase TiO2 on the nitrogen metabolism of growing spinach. Biol. Trace Elem. Res. 2006, 110, 179–190.
- Mishra, A.; Kumari, M.; Pandey, S.; Chaudhry, V.; Gupta, K.C.; Nautiyal, C.S. Biocatalytic and antimicrobial activities of gold nanoparticles synthesized by Trichoderma sp. Bioresour. Technol. 2014, 166, 235–242.
- Abraham, R.E.; Verma, M.L.; Barrow, C.J.; Puri, M. Suitability of magnetic nanoparticle immobilised cellulases in enhancing enzymatic saccharification of pretreated hemp biomass. Biotechnol. Biofuels 2014, 7, 90.
- Yates, M.D.; Cusick, R.D.; Logan, B.E. Extracellular palladium nanoparticle production using geobacter sulfurreducens. ACS Sustain. Chem. Eng. 2013, 1, 1165–1171.
- Hakamizadeh, M.; Afshar, S.; Tadjarodi, A.; Khajavian, R.; Fadaie, M.R.; Bozorgi, B. Improving hydrogen production via water splitting over Pt/TiO2/activated carbon nanocomposite. Int. J. Hydrogen Energy 2014, 39, 7262–7269.
- Markowska-Szczupak, A.; Wang, K.; Rokicka, P.; Endo, M.; Wei, Z.; Ohtani, B.; Morawski, A.W.; Kowalska, E. The effect of anatase and rutile crystallites isolated from titania P25 photocatalyst on growth of selected mould fungi. J. Photochem. Photobiol. B Biol. 2015, 151, 54–62.
- Cheng, P.; Yang, Z.; Wang, H.; Cheng, W.; Chen, M.; Shangguan, W.; Ding, G. TiO2-graphene nanocomposites for photocatalytic hydrogen production from splitting water. Int. J. Hydrogen Energy 2012, 37, 2224–2230.
- Sekoai, P.T.; Ouma, C.N.M.; du Preez, S.P.; Modisha, P.; Engelbrecht, N.; Bessarabov, D.G.; Ghimire, A. Application of nanoparticles in biofuels: An overview. Fuel 2019, 237, 380–397.
- Zhao, W.; Zhang, Y.; Du, B.; Wei, D.; Wei, Q.; Zhao, Y. Enhancement effect of silver nanoparticles on fermentative biohydrogen production using mixed bacteria. Bioresour. Technol. 2013, 142, 240–245.
- Mohanraj, S.; Anbalagan, K.; Kodhaiyolii, S.; Pugalenthi, V. Comparative evaluation of fermentative hydrogen production using Enterobacter cloacae and mixed culture: Effect of Pd (II) ion and phytogenic palladium nanoparticles. J. Biotechnol. 2014, 192, 87–95.
- Mullai, P.; Yogeswari, M.K.; Sridevi, K. Optimisation and enhancement of biohydrogen production using nickel nanoparticles—A novel approach. Bioresour. Technol. 2013, 141, 212–219.
- Mohanraj, S.; Anbalagan, K.; Rajaguru, P.; Pugalenthi, V. Effects of phytogenic copper nanoparticles on fermentative hydrogen production by Enterobacter cloacae and Clostridium acetobutylicum. Int. J. Hydrogen Energy 2016, 41, 10639–10645.
- Nath, D.; Manhar, A.K.; Gupta, K.; Saikia, D.; Das, S.K.; Mandal, M. Phytosynthesized iron nanoparticles: Effects on fermentative hydrogen production by Enterobacter cloacae DH-89. Bull. Mater. Sci. 2015, 38, 1533–1538.
- Dolly, S.; Pandey, A.; Pandey, B.K.; Gopal, R. Process parameter optimization and enhancement of photo-biohydrogen production by mixed culture of Rhodobacter sphaeroides NMBL-02 and Escherichia coli NMBL-04 using Fe-nanoparticle. Int. J. Hydrogen Energy 2015, 40, 16010–16020.
- Lin, R.; Cheng, J.; Ding, L.; Song, W.; Liu, M.; Zhou, J.; Cen, K. Enhanced dark hydrogen fermentation by addition of ferric oxide nanoparticles using Enterobacter aerogenes. Bioresour. Technol. 2016, 207, 213–219.
- Malik, S.N.; Pugalenthi, V.; Vaidya, A.N.; Ghosh, P.C.; Mudliar, S.N. Kinetics of nano-catalysed dark fermentative hydrogen production from distillery wastewater. Energy Procedia 2014, 54, 417–430.
- Mohanraj, S.; Kodhaiyolii, S.; Rengasamy, M.; Pugalenthi, V. Green synthesized iron oxide nanoparticles effect on fermentative hydrogen production by Clostridium acetobutylicum. Appl. Biochem. Biotechnol. 2014, 173, 318–331.
- Engliman, N.S.; Abdul, P.M.; Wu, S.Y.; Jahim, J.M. Influence of iron (II) oxide nanoparticle on biohydrogen production in thermophilic mixed fermentation. Int. J. Hydrogen Energy 2017, 42, 27482–27493.
- Zhao, Y.; Chen, Y. Nano-TiO2 enhanced photofermentative hydrogen produced from the dark fermentation liquid of waste activated sludge. Environ. Sci. Technol. 2011, 45, 8589–8595.
- Liu, Z.; Lv, F.; Zheng, H.; Zhang, C.; Wei, F.; Xing, X.H. Enhanced hydrogen production in a UASB reactor by retaining microbial consortium onto carbon nanotubes (CNTs). Int. J. Hydrogen Energy 2012, 37, 10619–10626.
- Hsieh, P.H.; Lai, Y.C.; Chen, K.Y.; Hung, C.H. Explore the possible effect of TiO2 and magnetic hematite nanoparticle addition on biohydrogen production by Clostridium pasteurianum based on gene expression measurements. Int. J. Hydrogen Energy 2016, 41, 21685–21691.
- Saeed, S.; Samer, M.; Mohamed, M.S.M.; Abdelsalam, E.; Mohamed, Y.M.A.; Abdel-Hafez, S.H.; Attia, Y.A. Implementation of graphitic carbon nitride nanomaterials and laser irradiation for increasing bioethanol production from potato processing wastes. Environ. Sci. Pollut. Res. 2022, 29, 34887–34897.
- Gupta, K.; Chundawat, T.S. Zinc oxide nanoparticles synthesized using Fusarium oxysporum to enhance bioethanol production from rice-straw. Biomass Bioenergy 2020, 143, 105840.
- Ivanova, V.; Petrova, P.; Hristov, J. Application in the Ethanol Fermentation of Immobilized Yeast Cells in Matrix of Alginate/Magnetic Nanoparticles, on Chitosan-Magnetite Microparticles and Cellulose-coated Magnetic Nanoparticles. Int. Rev. Chem. Eng. 2011, 3, 289–299.
- Kim, Y.K.; Park, S.E.; Lee, H.; Yun, J.Y. Enhancement of bioethanol production in syngas fermentation with Clostridium ljungdahlii using nanoparticles. Bioresour. Technol. 2014, 159, 446–450.
