While the importance of the intestinal microbiome has been realised for a number of years, the significance of the phrase microbiota–gut–brain axis is only just beginning to be fully appreciated. The microbiome is discussed as if it were a single entity, modifying the expression of the genetic inheritance of the individual by the generation of interkingdom signalling molecules, semiochemicals, such as dopamine. The purpose of the microbiome is to convey information about the microbial environment of the mother so as to calibrate the immune system of the new-born, giving it the ability to distinguish harmful pathogens from the harmless antigens of pollen, for example, or to help distinguish self from non-self. In turn, this requires the partition of nutrition between the adult and its microbiome to ensure that both entities remain viable until the process of reproduction. Accordingly, the failure of a degraded microbiome to interact with the developing gut of the neonate leads to failure of this partition in the adult: to low faecal energy excretion, excessive fat storage, and concomitant problems with the immune system. Similarly, a weakened gut–brain axis distorts interoceptive input to the brain, increasing the risk of psychiatric diseases such as autism.
- immune system
- autism
- microbiome
1. Introduction: Non-Communicable Disease, the Microbiome, and Symbiosis
2. Temporary Stimulation of the Gut–Brain Axis
The concept of the placebo has had a long and distinguished history; the hope being that it could allay the symptoms of disease long enough for the underlying condition to right itself. However, the development of the double-blind, placebo controlled, randomised clinical trial let the genie firmly out of the bottle [27]. Subsequent attempts to unravel exactly why the placebo is so effective merely illustrated the difficulties posed by onion skin-like layers of bias [28]. Interestingly, of course, the gut–brain axis has evolved to transfer information faster than can be achieved by conscious thought and, as such, it can easily be confused. As an example, the well-known phenomenon of traveller’s diarrhoea is an evolved response to unknown but essentially harmless microbes [29], presumably constituting a temporary stimulation of both the immune system and the gut–brain axis, albeit with a necessarily negative effect—making you feel bad for a good reason. In a similar fashion, in certain circumstances a subconscious “placebo-like” effect can have a surprisingly negative outcome. Essentially, this nocebo effect is the antithesis of the placebo effect, and may have fatal consequences, at least in hospitalised, vulnerable populations [30]. In addition, it potentially accounts for some of the non-specific side effects of medication [31]. A potential explanation of the value of the placebo effect in the context of a malfunctioning gut–brain axis is illustrated in Figure 1. The principle here is that a strengthening of this axis from either terminus will affect the other, presumably via the intermediacy of the vagus nerve, which is also known to be involved in inflammatory disorders and a variety of psychiatric conditions [32]. Indeed, it may be that raised production of semiochemicals has subtle disease-modifying effects, for example, by increased production of dopamine in the periphery [33]. In the context of a clinical trial, it is possible that discussion with the clinician, followed by the action of taking a mock medicine, stimulates hormone levels in the brain and hence semiochemical levels in the gut. In this context, it is important to note that the placebo effect does not require the recipient to be deceived [34].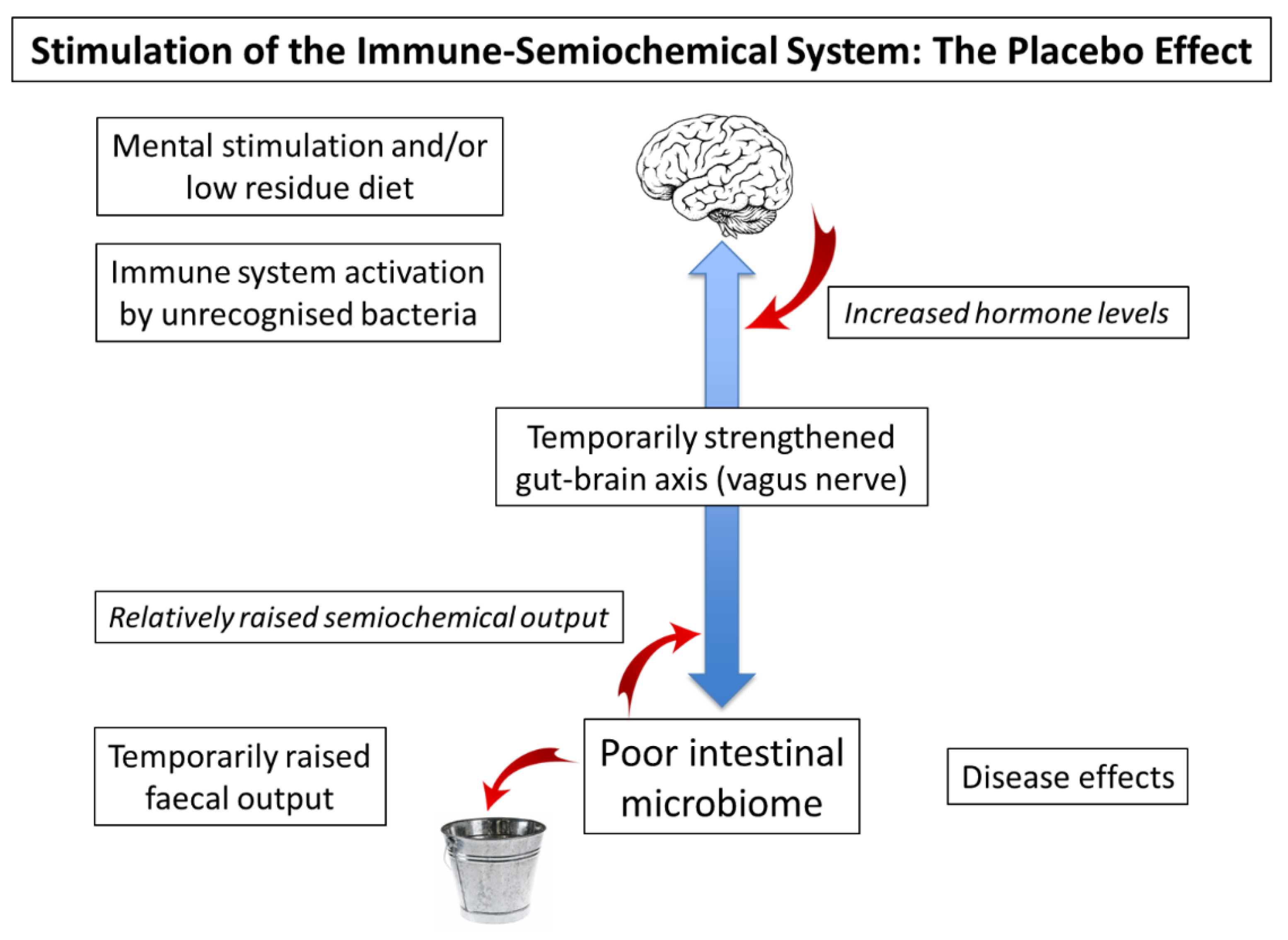
3. The Many Variations of Developmental Brain Disease
3.1. Autism Spectrum Disorder and Attention Deficit Hyperactivity Disorder
3.2. Dyslexia, Anxiety, Depression, and Other Conditions
3.3. Unexpected Strengths and the Problems of Definition in Psychology
4. Microbiome Measurement: Semiochemicals and Sentinel Cells
4.1. Semiochemical Measurement: An Ingestible Sensor
4.2. Microeukaryotes and Microbial Sentinel Cells
References
- Burkitt, D. A sarcoma involving the jaws in African children. Br. J. Surg. 1958, 46, 218–223.
- Burkitt, D.P. Some diseases characteristic of modern western civilization. Br. Med. J. 1973, 1, 274–278.
- LeBlanc, J.G.; Chain, F.; Martin, R.; Bermùndez-Humarán, L.G.; Courau, S.; Langella, P. Beneficial effects on host energy metabolism of short-chain fatty acids and vitamins produced by commensal and probiotic bacteria. Microb. Cell Factories 2017, 16, 79.
- O’Keefe, S.J. The association between dietary fibre deficiency and high-income lifestyle-associated diseases: Burkitt’s hypothesis revisited. Lancet Gastroenterol. Hepatol. 2019, 4, 984–996.
- Valdes, A.M.; Walter, J.; Segal, E.; Spector, T.D. Role of the gut microbiota in nutrition and health. Br. Med. J. 2018, 361, k2179.
- Sonnenburg, E.D.; Smits, S.A.; Tikhonov, M.; Higginbottom, S.A.; Wingreen, N.S.; Sonnenburg, J.L. Diet-induced Extinctions in the Gut Microbiota Compound over Generations. Nature 2016, 529, 212–215.
- Reese, E.D.; Chadaideh, K.S.; Diggins, C.E.; Schell, L.D.; Beckel, M.; Callahan, P.; Ryan, R.; Thompson, M.E.; Carmody, R.N. Effects of domestication on the gut microbiota parallel those of human industrialization. eLife 2021, 10, e60197.
- Marsella, R.; De Benedetto, A. Atopic dermatitis in animals and in people: An update and comparative review. Vet. Sci. 2017, 4, 37.
- Burberry, A.; Wells, M.F.; Limone, F.; Couto, A.; Smith, K.S.; Keaney, J.; Gillet, G.; van Gastel, N.; Wang, J.-Y.; Pietilainen, O.; et al. C9orf72 suppresses systemic and neural inflammation induced by gut bacteria. Nature 2020, 582, 89–94.
- Ganci, M.; Suleyman, E.; Butt, H.; Ball, M. The role of the brain-gut-microbiota in psychology: The importance of considering gut microbiota in the development, perpetuation, and treatment of psychological disorders. Brain Behav. 2019, 9, e01408.
- Brüssow, H. Problems with the concept of gut microbiota dysbiosis. Microb. Biotechnol. 2019, 13, 423–434.
- Sudo, N. Biogenic amines: Signals between commensal microbiota and gut physiology. Front. Endocrinol. 2019, 10, 504.
- Jheeta, S.; Smith, D. Seeing the wood for the trees: A new way to view the human intestinal microbiome and its connection with non-communicable disease. Med. Hypotheses 2019, 125, 70–74.
- Kaelberer, M.M.; Rupprecht, L.E.; Liu, W.W.; Weng, P.; Bohórquez, D.V. Neuropod cells: Emerging biology of the gut-brain sensory transduction. Annu. Rev. Neurosci. 2020, 43, 337–353.
- Hata, T.; Asano, Y.; Yoshihara, K.; Kimura-Todani, T.; Miyata, N.; Zhang, X.-T.; Takakura, S.; Aiba, Y.; Koga, Y.; Sudo, N. Regulation of gut luminal serotonin by commensal microbiota in mice. PLoS ONE 2017, 12, e0180745.
- Reigstad, C.S.; Salmonson, C.E.; Rainey, J.F., III; Szurszewski, J.H.; Linden, D.R.; Sonnenburg, J.L.; Farrugia, G.; Kashyap, P.C. Gut microbes promote colonic serotonin production through an effect of short-chain fatty acids on enterochromaffin cells. FASEB J. 2005, 29, 1395–1403.
- O’Mahony, S.M.; Clarke, G.; Borre, Y.E.; Dinan, T.G.; Cryan, J.F. Serotonin, tryptophan metabolism and the brain-gut-microbiome axis. Behav. Brain Res. 2015, 277, 32–48.
- Smith, P.M.; Howitt, M.R.; Panikov, N.; Michaud, M.; Gallini, C.A.; Bohlooly-Y, M.; Glickman, J.N.; Garrett, W.S. The microbial metabolites, short-chain fatty acids, regulate colonic Treg cell homeostasis. Science 2013, 341, 569–573.
- Laforest-Lapointe, I.; Arrieta, M.-C. Microbial eukaryotes: A missing link in gut microbiome studies. mSystems 2018, 3, e00201–e00217.
- Scanlan, P.D.; Stensvold, C.R.; Rajilic-Stojanovic, M.; Heilig, H.G.H.J.; De Vos, W.M.; O’Toole, P.W.; Cotter, P.D. The microbial eukaryote Blastocystis is a prevalent and diverse member of the healthy human gut microbiota. FEMS Microbiol. Ecol. 2014, 90, 326–330.
- Ward, T.L.; Dominguez-Bello, M.G.; Heisel, T.; Al-Ghalith, G.; Knights, D.; Gale, C.A. Development of the human mycobiome over the first month of life and across body sites. mSystems 2018, 3, e00140.
- Lagier, J.-C.; Million, M.; Hugon, P.; Armougom, F.; Raoult, D. Human gut microbiota: Repertoire and variations. Front. Cell. Infect. Microbiol. 2012, 2, 136.
- Barr, J.J.; Auro, R.; Furlan, M.; Whiteson, K.L.; Erb, M.L.; Pogliano, J.; Stotland, A.; Wolkowicz, R.; Cutting, A.S.; Doran, K.S.; et al. Bacteriophage adhering to mucus provide a non–host-derived immunity. Proc. Natl. Acad. Sci. USA 2013, 110, 10771–10776.
- Cryan, J.F.; O’Riordan, K.J.; Cowan, C.S.M.; Sandhu, K.V.; Bastiaanssen, T.F.S.; Boehme, M.; Codagnone, M.G.; Cussotto, S.; Fulling, C.; Golubeva, A.V.; et al. The microbiota-gut-brain axis. Physiol. Rev. 2019, 99, 1877–2013.
- Smith, D.; Palacios-Pérez, M.; Jheeta, S. The enclosed intestinal microbiome: Semiochemical signals from the Precambrian and their disruption by heavy metal pollution. Life 2022, 12, 287.
- Banchereau, J.; Briere, F.; Caux, C.; Davoust, J.; Lebecque, S.; Liu, Y.-J.; Pulendran, B.; Palucka, K. Immunobiology of dendritic cells. Annu. Rev. Immunol. 2000, 18, 767–811.
- de Craen, A.J.; Kaptchuck, T.J.; Tijssen, J.G.; Kleijnen, J. Placebos and placebo effects in medicine: Historical overview. J. R. Soc. Med. 1999, 92, 511–515.
- Hrobjartsson, A.; Kaptchuk, T.J.; Miller, F.G. Placebo effect studies are susceptible to response bias and other types of biases. J. Clin. Epidemiol. 2011, 64, 1223–1229.
- Giddings, S.L.; Stevens, A.M.; Leung, D.T. Traveler’s diarrhea. Med. Clin. N. Am. 2016, 100, 317–330.
- Cohen, S.I. Psychosomatic death: Voodoo death in a modern perspective. Integr. Psychiatry 1985, 3, 46–51.
- Barsky, A.J.; Saintfort, R.; Rogers, M.P.; Borus, J.F. Nonspecific medication side effects and the nocebo phenomenon. JAMA 2002, 87, 622–627.
- Breit, S.; Kupferberg, A.; Rogler, G.; Hasler, G. Vagus nerve as modulator of the brain-gut axis in psychiatric and inflammatory disorders. Front. Psychiatry 2018, 9, 44.
- Xue, R.; Zhang, H.; Pan, J.; Du, Z.; Zhou, W.; Zhang, Z.; Tian, Z.; Zhou, R.; Bai, L. Peripheral dopamine controlled by gut microbes inhibits invariant natural killer T cell-mediated hepatitis. Front. Immunol. 2018, 9, 2398.
- Charlesworth, J.E.; Petkovic, G.; Kelley, J.M.; Hunter, M.; Onakpoya, I.; Roberts, N.; Miller, F.G.; Howick, J. Effects of placebos without deception compared with no treatment: A systematic review and meta-analysis. J. Evid.-Based Med. 2017, 10, 97–107.
- Smith, D.; Jheeta, S.; Fuentes, H.V.; Palacios-Pérez, M. Feeding our microbiota: Stimulation of the immune/semiochemical system and the potential amelioration of non-communicable diseases. Life 2022, 12, 1197.
- Zhang, Y.; Yang, H.; Li, S.; Li, W.-D.; Wang, Y. Consumption of coffee and tea and risk of developing stroke, dementia, and poststroke dementia: A cohort study in the UK Biobank. PLoS Med. 2021, 18, e1003830.
- Ma, G.; Chen, Y. Polyphenol supplementation benefits human health via gut microbiota: A systematic review via meta-analysis. J. Funct. Foods 2020, 66, 103829.
- Zhou, L.; Foster, J.A. Psychobiotics and the gut-brain axis: In the pursuit of happiness. Neuropsychiatr. Dis. Treat. 2015, 11, 715–723.
- Sarkar, A.; Lehto, S.M.; Harty, S.; Dinan, T.G.; Cryan, J.F.; Burnet, P.W.J. Psychobiotics and the manipulation of bacteria-gut-brain signals. Trends Neurosci. 2016, 39, 763–781.
- Smith, D.; Palacios-Pérez, M.; Jheeta, S. Microbiome-gut dissociation in the neonate: Obesity and coeliac disease as examples of microbiome-function deficiency disorder. Gastrointest. Disord. 2022, 4, 108–128.
- Kang, D.-W.; Adams, J.B.; Gregory, A.C.; Borody, T.; Chittick, L.; Fasano, A.; Khoruts, A.; Geis, E.; Maldonado, J.; McDonough-Means, S.; et al. Microbiota transfer therapy alters gut ecosystem and improves gastrointestinal and autism symptoms: An open-label study. Microbiome 2017, 5, 10.
- Kang, D.-W.; Adams, J.B.; Coleman, D.M.; Pollard, E.L.; Maldonado, J.; McDonough-Means, S.; Caporaso, J.G.; Krajmalnik-Brown, R. Long-term benefit of microbiota transfer therapy on autism symptoms and gut microbiota. Sci. Rep. 2019, 9, 5821.
- Martínez-González, A.E.; Andreo-Martínez, P. Prebiotics, probiotics and fecal microbiota transplantation in autism: A systematic review. Rev. De Psiquiatr. Y Salud Ment. (Engl. Ed.) 2020, 13, 150–164.
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Publishing: Arlington, VA, USA, 2019.
- Pickersgill, M.D. Debating DSM-5: Diagnosis and the sociology of critique. J. Med. Ethics 2014, 40, 521–525.
- Caspi, V.; Houts, R.M.; Belsky, D.W.; Goldman-Mellor, S.J.; Harrington, H.; Israel, S.; Meier, M.H.; Ramraker, S.; Shalev, I.; Poulton, R.; et al. The p factor: One general psychopathology factor in the structure of psychiatric disorders? Clin. Psychol. Sci. 2014, 2, 119–137.
- Garcia-Gutierrez, E.; Narbad, A.; Rodriguez, J.M. Autism spectrum disorder associated with gut microbiota at immune, metabolomic, and neuroactive level. Front. Neurosci. 2020, 14, 578666.
- Chiarotti, F.; Venerosi, A. Epidemiology of autism spectrum disorders: A review of worldwide prevalence since 2014. Brain Sci. 2020, 10, 274.
- Pulikkan, J.; Mazumder, A.; Grace, T. Role of the gut microbiome in autism spectrum disorders. Adv. Exp. Med. Biol. 2019, 1118, 253–269.
- Bundgaard-Nielson, C.; Knudson, J.; Leutscher, P.D.C.; Lauritsen, M.B.; Nyegaard, M.; Hagstrøm, S.; Sørensen, S. Gut microbiota profiles of autism spectrum disorder and attention deficit/hyperactivity disorder: A systematic literature review. Gut Microbes 2020, 11, 1172–1187.
- Wu, D.; José, J.V.; Nurnberger, J.I.; Torres, E.B. A biomarker characterizing neurodevelopment with applications in autism. Sci. Rep. 2018, 8, 614.
- Sexton, C.C.; Gelhorn, H.L.; Bell, J.A.; Classi, P.M. The co-occurrence of reading disorder and ADHD: Epidemiology, treatment, psychosocial impact, and economic burden. J. Learn. Disabil. 2012, 45, 538–564.
- Taylor, H.; Vestergaard, M.D. Developmental dyslexia: Disorder or specialization in exploration? Front. Psychol. 2022, 13, 889245.
- Krishnan, S.; Cler, G.J.; Smith, H.J.; Willis, H.E.; Asaridou, S.S.; Healy, M.P.; Papp, D.; Watkins, K.E. Quantitative MRI revearls differences in striatal myelin in children with DLD. eLife 2022, 11, e74242.
- Espie, J.; Eisler, I. Focus on anorexia nervosa: Modern psychological treatment and guidelines for the adolescent patient. Adolesc. Health Med. Ther. 2015, 6, 9–16.
- Thibaut, F. Anxiety disorders: A review of the current literature. Dialogues Clin. Neurosci. 2017, 19, 87–88.
- Bear, T.L.; Dalziel, J.E.; Coad, J.; Roy, N.C.; Butts, C.; Gopal, P.K. The role of the gut microbiota in dietary interventions for depression and anxiety. Adv. Nutr. 2020, 11, 890–907.
- Miraglia, F.; Colla, E. Microbiome, Parkinson’s disease and Molecular mimicry. Cells 2019, 8, 222.
- Schuster, B.A.; Sowden, S.; Rybicki, A.J.; Fraser, D.S.; Press, C.; Holland, P.; Cook, J.L. Dopaminergic modulation of dynamic emotion perception. J. Neurosci. 2022, 42, 4394–4400.
- Shah, P.; Hall, R.; Catmur, C.; Bird, G. Alexithymia, not autism, is associated with impaired interoception. Cortex 2016, 81, 215–220.
- Remmers, C.; Michalak, J. Losing your gut feelings. Intuition in depression. Front. Psychol. 2016, 7, 1291.
- Bowe, W.P.; Logan, A.C. Acne vulgaris, probiotics and the gut-brain-skin axis—Back to the future? Gut Pathog. 2011, 3, 1.
- Postema, M.C.; van Rooij, D.; Anagnostou, E.; Arango, C.; Auzias, G.; Behrmann, M.; Filho, G.B.; Calderoni, S.; Calvo, R.; Daly, E.; et al. Altered structural brain asymmetry in autism spectrum disorder in a study of 54 datasets. Nat. Communcations 2019, 10, 4958.
- Sherkatghanad, Z.; Akhondzadeh, M.; Salari, S.; Zomorodi-Moghadam, M.; Abdar, M.; Acharya, U.R.; Khosrowabadi, R.; Salari, V. Automated detection of autism spectrum disorder using a convolutional neural network. Front. Neurosci. 2020, 13, 1325.
- Miller, L.K. The savant syndrome: Intellectual impairment and exceptional skill. Psychol. Bull. 1999, 125, 31–46.
- Jaarsma, P.; Welin, S. Autism as a natural human variation: Reflections on the claims of the neurodiversity movement. Health Care Anal. 2012, 20, 20–30.
- Zwilling, M.; Levy, B.R. How well environmental design is and can be suited to people with autism spectrum disorder (ASD): A natural language processing analysis. Int. J. Environ. Res. Public Health 2022, 19, 5037.
- Dietrich, A. The mythconception of the mad genius. Front. Psychol. 2014, 5, 79.
- Thys, E.; Sabbe, B.; De Hert, M. Creativity and psychopathology: A systematic review. Psychopathology 2014, 47, 141–147.
- Gritz, E.C.; Bhandari, V. The human neonatal gut microbiome: A brief review. Front. Pediatrics 2015, 3, 17.
- Smith, D.; Jheeta, S. The epidemiology of the dysfunctional microbiome in animals and in humans: The propensity for the development of non-communicable disease. EC Gastroenterol. Dig. Syst. 2020, 7, 83–93.
- Smits, S.A.; Leach, J.; Sonnenburg, E.D.; Gonzalez, C.G.; Lichtman, J.S.; Reid, G.; Knight, R.; Manjurano, A.; Changalucha, J.; Elias, J.E.; et al. Seasonal cycling in the gut microbiome of the Hadza hunter-gatherers of Tanzania. Science 2017, 357, 802–806.
- Irimia, A.; Chaudhari, N.N.; Robles, D.J.; Rostowsky, K.A.; Maher, A.S.; Chowdhury, N.F.; Calvillo, N.F.; Ngo, V.; Gatz, M.; Mack, W.J.; et al. The indigenous South American Tsimane exhibit relatively modest decrease in brain volume with age despite high systemic inflammation. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2021, 76, 2147–2155.
- Ryan, C.R. Towards an ethics of reciprocity: Ethnobotanical knowledge and medicinal plants as cancer therapies. Humanities 2014, 3, 624–644.
- Smith, D.; Jheeta, S. Measuring microbiome effectiveness: A role for ingestible sensors. Gastrointest. Disord. 2020, 2, 3–11.
- Beardslee, L.A.; Banis, G.E.; Chu, S.; Liu, S.; Chapin, A.A.; Stine, J.M.; Pasricha, P.J.; Ghodossi, R. Ingestible sensors and sensing systems for minimally invasive diagnosis and monitoring: The next frontier in minimally invasive screening. ACS Sens. 2020, 5, 891–910.
- Tito, R.Y.; Knights, D.; Metcalf, J.; Obregon-Tito, A.J.; Cleeland, L.; Najar, F.; Roe, B.; Reinhard, K.; Sobolik, K.; Belknap, S.; et al. Insights from characterizing extinct human gut microbiomes. PLoS ONE 2012, 7, e51146.
- McConnell, J.R.; Wilson, A.I.; Stohl, I.; Arienzo, M.M.; Chellman, N.J.; Eckhardt, S.; Thompson, E.M.; Pollard, A.M.; Steffensen, J.P. Lead pollution recorded in Greenland ice indicates European emissions tracked plagues, wars, and imperial expansion during antiquity. Proc. Natl. Acad. Sci. USA 2018, 115, 5726–5731.
- Thompson, R.C.; Allam, A.H.; Lombardi, G.G.; Wann, L.S.; Sutherland, M.L.; Sutherland, J.D.; Soliman, M.A.; Frohlich, B.; Mininberg, D.T.; Monge, J.M.; et al. Atherosclerosis across 4000 years of human history: The Horus study of four ancient populations. Lancet 2013, 381, 1211–1222.
- Gasbarrini, G.; Rickards, O.; Martínez-Labarga, C.; Pacciani, E.; Chilleri, F.; Laterza, L.; Marangi, G.; Scaldaferri, F.; Gasbarrini, A. Origin of celiac disease: How old are predisposing haplotypes? World J. Gastroenterol. 2012, 18, 5300–5304.
