Iron oxides are chemical compounds which havedifferent polymorphic forms, including γ-Fe2O3 (maghemite), Fe3O4(magnetite), and FeO (wustite). Among them, the most studiedare γ-Fe2O3 and Fe3O4,as they possess extraordinary properties at the nanoscale (such as superparamagnetism, high specific surface area, biocompatible etc.),because at this size scale, the quantum effectsaffect matter behavior and optical, electrical and magnetic properties.Therefore, in the nanoscale, these materials become ideal for surface functionalization and modification in various applications such as separation techniques, magnetic sorting (cells and other biomolecules etc.), drug delivery, cancer hyperthermia, sensing etc., and also for increased surface area-to-volume ratio, which allows for excellent dispersibility in the solution form.The current methods used are partially and passively mixed reactants, and,thus, every reaction has a different proportion of all factors which causes further difficulties in reproducibility.Direct active and complete mixing and automated approaches could be solutions to thissize- and shape-controlled synthesis, playing a key role in its exploitation for scientific or technological purposes. An ideal synthesis method should be able to allow reliable adjustment of parameters andcontrol over the following: fluctuation in temperature;pH, stirring rate;particle distribution; size control;concentration; and control over nanoparticle shape andcomposition i.e., crystallinity, purity, and rapid screening. Iron oxide nanoparticle (IONP)-based available clinical applications are RNA/DNAextraction and detection of infectious bacteria andviruses. Such technologies are important at POC (point of care) diagnosis.IONPs can play a key role in these perspectives.Although there are various methods for synthesis of IONPs,one of the most crucial goals is to control size and properties with high reproducibility to accomplish successful applications.Using multiple characterization techniques to identify and confirm the oxide phase of iron can provide better characterization capability. It is very important to understand the in-depth IONP formation mechanism, enabling better control over parameters and overall reaction and, by extension, properties of IONPs.This workprovides an in-depth overview ofdifferent properties, synthesis methods, and mechanisms of iron oxide nanoparticles (IONPs) formation, and the diverse range of theirapplications. Differentcharacterization factors and strategiesto confirm phase purity in the IONP synthesis field are reviewed.First, properties of IONPs and various synthesis routes with their merits and demerits are described. We also describe different synthesis strategies and formation mechanisms for IONPs such as for: wustite (FeO), hematite(α-Fe2O3), maghemite(ɤ-Fe2O3) and magnetite(Fe3O4). We also describe characterization of these nanoparticles and various applications in detail. In conclusion, we present a detailed overview on the properties, size-controlled synthesis, formation mechanisms and applications of IONPs.
- iron oxide nanoparticles(IONPs)
- formation mechanisms
- reproducible
- biomedical
1. Introduction
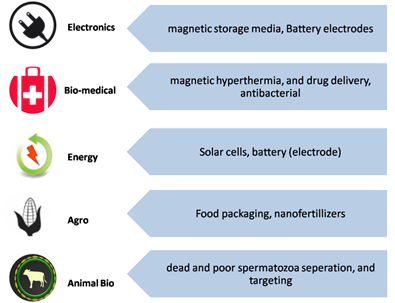
Iron oxides nanoparticle (IONP)-based technologies are catalyzing rapid developments in nanotechnology. Due to technological importance, extensive research has been carried out on the development of various synthetic routes to yield IONPs with desired properties[1]. Aare chemical compounds which have different polymorphic formong IONPs, mainly Fe3O4 aincludindg γ-Fe2O3 (marghe extensively studied[2]. In generalmite), iron oxidFes are classified into different phases 3O4 (magnetite), hematite, maghemite, and FeO (wustite). In the nano form, a material possesses interesting optical, magnetic, and electrical properties which cannot be found in their bulk form. This phenomenon can be described as thAmong them, the most studied are γ-Fe2O3 "quantum sizd Fe effect"[3][4][5]. In the nanometer range of I3ONPs4, the quantum effect dominates the behavior-affecting magnetic, electric, and optical properties of the matter. In the nanoscale, there is an impact of specific individual atoms or molecules, while in the bulk form, s they possess extraordinary property is attributed to the average of all the quantum forces that affect all of the atoms. For example, magnetic Fe3O4 nies at the nanoscale (such anoparticles are supersuper paramagnetic below the size of 20 nsm[6]., As the nanoparticle size decreases, this property tends towards paramagnetic or superparamagnetic magnetization. Therefore, a decrease in nanoparticle size will enhance superparamagnetic behavior and decrease ferromagnetic behavior. As the size of nanoparticles decreasesigh specific surface area, biocompatible etc.), because at this size scale, the relative oxygen concentration decreases; therefore, a slight reduction in the iron valance state occurs. Because of this ferrous ion content increase, an increase in magnetization should also be observed[7]. Squantum effects affect matter behavimilaorly, γ-Fe2O3 n and oparticles have gained technological importance due to their tical, electrical and magnetic and catalytic properties. High magnetization and hysteretic heating make them potential candidates in separation and biomedical areas, and the semiconducting property and chemically activeTherefore, in the nanoscale, these materials become ideal for surface allow catalytic activities such as photocatalytic ability[8][9]. Irfunctionalization and modification oxide nanoparticles (IONPs) have a broad range of significantn various applications in electronicsuch as[10][11], biomsedicine[12][13][14], enepargy[15][16], agrticulture[11][18], aond animal biotechnology[19][20], as shown technin Figqure 1. In a small size of about 10–20 nm, the superparaes, magnetic properties of Fe3O4 sorting (cells and γ-Fe2O3 nanoparticles become apparent, therefore, better performance can be achieved for the above-mentioned applications. Additionally, due to theher biomolecules etc.), drug delivery, cancer hyperthermia, sensing etc., and also for increased surfacee area-to-volume ratio, they showwhich allows for excellent dispensarsibility in the solutions[21].
Figure 1. Various applications of iron oxide nanoparticles (IONPs).
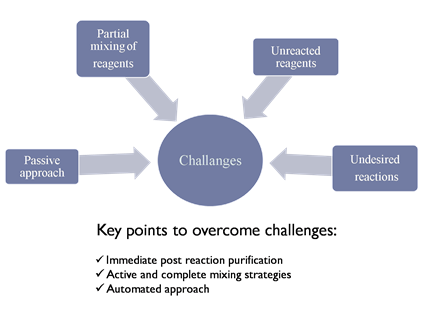
However, reproducible synthesis of IONPs with desired properties is still a probleorm[22]. Thise is because existing synthesiscurrent methods show a passive approach towards synthesis reaction. The main challenges and key points to overcome them are explained in Figure 2. In existing methods,used are partially and passively mixed reactants are mixed partially and passively. Unreacted components therefore effect the final product when undesired reactions takes place, as the p, and, thus, every reaction has a different proportion of all these factors is different in every reaction, making it which causes further difficult to achieve reproducibility in the desired properties[23]. Immeies in reprodiate purification of nanoparticles after reaction becomes necessary to minimize errorcibility. Direct active and complete mixing of reactants anand automated approaches could solve this issue. Researchers are mainly focused on be solutions to this size- and shape-controlled synthesis, as size determines the surface area, which playsplaying a key role in its exploitation for scientific or technological purposes[24].
Figure 2. Challenges and key points in reproducible synthesis of nanoparticles.
2. Manipulation of reaction parameters
Ma An ipulation of reaction parameters is necessary to obtain controlled nanoparticles in terms of size, shape, purity, crystallinity, and morphology. A synthetic route should enabledeal synthesis method should be able to allow reliable adjustment of parameters and control over reaction parameters: temperature; concentration;the following: fluctuation in temperature; pH;, stirring rate; particle distribution; size control; controcentration; and control over shape; nanoparticle e shape and composition and structure, which includes ci.e., crystallinity, purity, and rapid screening, and reliable adjustment of parameters[22][25][26][27].
I. Iron oxide nanoparticle (IONP)-based available clinical our opinion, the established synthetic routes of iron oxide nanoparticles have difficulty in controlling the particle size, shape, and properties. Many of the reportedapplications are RNA/DNA extraction and detection of infectious bacteria and viruses. Such technologies are important at POC (point of care) diagnosis. IONPs can play a key role in these perspectives. Although there are various methods have their own pros and cons, as described in Table 1. It is necessary to develop a new synthetic route for IONPs that yields nanoparticles in afor synthesis of IONPs, one of the most crucial goals is to control size and properties with high reproducible manner with excellent size control. This review explains various dimensions associated with synthesis of IONPs and their applications, and different synthesis mechanisms are summarized. Figures S1–7 represented in supplementary materials corresponds to various IONPs synthesis methods graphically presented which also includes key points for each corresponding methodility to accomplish successful applications. Using multiple characterization techniques to identify and confirm the oxide phase of iron can provide better characterization capability. It is very important to understand the in-depth IONP formation mechanism, enabling better control over parameters and overall reaction and, by extension, properties of IONPs.
1. Introduction
Iron oxide nanoparticle (IONP)-based technologies are catalyzing rapid developments in nanotechnology. Due to technological importance, extensive research has been carried out on the development of various synthetic routes to yield IONPs with desired properties [1]. Among IONPs, mainly Fe3O4 and γ-Fe2O3 are extensively studied [2]. In general, iron oxides are classified into different phases (magnetite, hematite, maghemite, wustite). In the nano form, a material possesses interesting optical, magnetic, and electrical properties which cannot be found in their bulk form. This phenomenon can be described as the “quantum size effect” [3–5]. In the nanometer range of IONPs, the quantum effect dominates the behavior-affecting magnetic, electric, and optical properties of the matter. In the nanoscale, there is an impact of specific individual atoms or molecules, while in the bulk form, property is attributed to the average of all the quantum forces that affect all of the atoms. For example, magnetic Fe3O4 nanoparticles are superparamagnetic below the size of 20 nm [6]. As the nanoparticle size decreases, this property tends towards paramagnetic or superparamagnetic magnetization. Therefore, a decrease in nanoparticle size will enhance superparamagnetic behavior and decrease ferromagnetic behavior. As the size of nanoparticles decreases, the relative oxygen concentration decreases; therefore, a slight reduction in the iron valance state occurs. Because of this ferrous ion content increase, an increase in magnetization should also be observed [7]. Similarly, γ-Fe2O3 nanoparticles have gained technological importance due to their magnetic and catalytic properties. High magnetization and hysteretic heating make them potential candidates in separation and biomedical areas, and the semiconducting property and chemically active surface allow catalytic activities such as photocatalytic ability [8,9]. Iron oxide nanoparticles (IONPs) have a broad range of significant applications in electronics [10,11], biomedicine [12–14], energy [15,16], agriculture [17,18], and animal biotechnology [19,20], as shown in Figure 1. In a small size of about 10–20 nm, the superparamagnetic properties of Fe3O4 and γ-Fe2O3 nanoparticles become apparent, therefore, better performance can be achieved for the above-mentioned applications. Additionally, due to the increased surface-to-volume ratio, they show excellent dispensability in solutions [21].
Figure 1. Various applications of iron oxide nanoparticles (IONPs).
However, reproducible synthesis of IONPs with desired properties is still a problem [22]. This is because existing synthesis methods show a passive approach towards synthesis reaction. The main challenges and key points to overcome them are explained in Figure 2. In existing methods, reactants are mixed partially and passively. Unreacted components therefore effect the final product when undesired reactions takes place, as the proportion of all these factors is different in every reaction, making it difficult to achieve reproducibility in the desired properties [23]. Immediate purification of nanoparticles after reaction becomes necessary to minimize error. Direct active and complete mixing of reactants and automated approaches could solve this issue. Researchers are mainly focused on size- and shape-controlled synthesis, as size determines the surface area, which plays a key role in its exploitation for scientific or technological purposes [24].
Figure 2. Challenges and key points in reproducible synthesis of nanoparticles.
Manipulation of reaction parameters is necessary to obtain controlled nanoparticles in terms of size, shape, purity, crystallinity, and morphology. A synthetic route should enable control over reaction parameters: temperature; concentration; fluctuation in temperature; pH; stirring rate; particle distribution; size control; control over shape; nanoparticle composition and structure, which includes crystallinity, purity, rapid screening, and reliable adjustment of parameters [22,25–27].
In our opinion, the established synthetic routes of iron oxide nanoparticles have difficulty in controlling the particle size, shape, and properties. Many of the reported methods have their own pros and cons, as described in Table 1. It is necessary to develop a new synthetic route for IONPs that yields nanoparticles in a reproducible manner with excellent size control. This review explains various dimensions associated with synthesis of IONPs and their applications, and different synthesis mechanisms are summarized. Figures S1–7 represented in supplementary materials corresponds to various IONPs synthesis methods graphically presented which also includes key points for each corresponding method.
Table 1.
Merits and demerits of different IONP nanoparticle synthesis methods.
|
Type of Synthesis | |||||||||
Pros | |||||||||
Cons | |||||||||
Reference | |||||||||
|
Microwave | |||||||||
Short reaction time, higher yields, excellent reproducibility, easy handling | |||||||||
Expensive, unsuitable for scale-up and reaction monitoring | |||||||||
[ | ][29][28,29] | ||||||||
|
Spray pyrolysis | |||||||||
Finely dispersed particles of predictable size, shape and variable composition | |||||||||
Aggregated particles, expensive | |||||||||
[ | |||||||||
|
Laser pyrolysis | |||||||||
Small particle size, narrow particle size distribution, near absence of aggregation | |||||||||
Complicated, very expensive | |||||||||
[ | ][31][21,31] | ||||||||
|
Pulsed wire discharge method | |||||||||
Fast process, higher purity of NPs | |||||||||
Batch process, limited production, high vacuum systems, costly process, contaminations in product | |||||||||
[ | ][34][33,34] | ||||||||
|
Chemical vapor condensation | |||||||||
Suitable for preparing small quantities to demonstrate desired properties in the laboratory | |||||||||
Low production, difficult to control size and particle size distribution | |||||||||
[35] | |||||||||
|
Co-precipitation | |||||||||
Convenient method, simple and rapid preparative method, easy control of particle size and composition | |||||||||
Extensive agglomeration, poor morphology and particle size distribution | |||||||||
[ | |||||||||
|
Thermal decomposition | |||||||||
Producing highly monodispersed particles with a narrow size distribution | |||||||||
High cost, long-time synthesis reaction, high temperature | |||||||||
[ | |||||||||
|
Microemulsion | |||||||||
Monodispersed nanoparticles with various morphology can be produced | |||||||||
Not very efficient and difficult to scale up | |||||||||
[ | ][42][39,42] | ||||||||
|
Polyol | |||||||||
Uniform size particles can be prepared, easy to scaleup | |||||||||
Needs high temperature, long time | |||||||||
[ | ][31][22,31] | ||||||||
|
Sol–Gel | |||||||||
Low processing cost, energy efficiency, high production rate, and rapid productivity | |||||||||
Limited efficiency, high cost | |||||||||
[ | |||||||||
|
Sonochemical | |||||||||
Simple, low cost, safe, environment friendly, absence of many reactants | |||||||||
Very small concentration of prepared NPs, particle agglomeration is very narrow | |||||||||
[ | ][46][33,46] | ||||||||
Biological synthesis of nanoparticles using plants and bacteria | Selectivity and precision for nanoparticle formation, cost effective, eco friendly | Limited knowledge, difficulty in controlling size and properties |
[47,48] |